Abstract
Phenotyping of chronic obstructive pulmonary disease (COPD) with computed tomography (CT) is used to distinguish between emphysema- and airway-dominated type. The phenotype is reflected in correlations with lung function measures. Among these, the relative value of body plethysmography has not been quantified. We addressed this question using CT scans retrospectively collected from clinical routine in a large COPD cohort. Three hundred and thirty five patients with baseline data of the German COPD cohort COPD and Systemic Consequences-Comorbidities Network were included. CT scans were primarily evaluated using a qualitative binary emphysema score. The binary score was positive for emphysema in 52.5% of patients, and there were significant differences between the positive/negative groups regarding forced expiratory volume in 1 second (FEV1), FEV1/forced vital capacity (FVC), intrathoracic gas volume (ITGV), residual volume (RV), specific airway resistance (sRaw), transfer coefficient (KCO), transfer factor for carbon monoxide (TLCO), age, pack-years, and body mass index (BMI). Stepwise discriminant analyses revealed the combination of FEV1/FVC, RV, sRaw, and KCO to be significantly related to the binary emphysema score. The additional positive predictive value of body plethysmography, however, was only slightly higher than that of the conventional combination of spirometry and diffusing capacity, which if taken alone also achieved high predictive values, in contrast to body plethysmography. The additional information on the presence of CT-diagnosed emphysema as conferred by body plethysmography appeared to be minor compared to the well-known combination of spirometry and CO diffusing capacity.
Introduction
In patients with chronic obstructive pulmonary disease (COPD) the phenotyping into a predominantly emphysema- or airway-dominated type is generally considered as important. The diagnosis is usually based on chest computed tomography (CT) imaging. 1,2 In the case where no such CT data was available, several studies have evaluated how strongly lung function parameters correlate with CT findings. 3 A useful application of lung function would be monitoring of treatment, for example, in patients with alpha-1 antitrypsin deficiency, which otherwise would require repeated CT scans. 4 Spirometry is a marker for emphysema but in general its correlation with the degree of emphysema on CT is weak. 5 Transfer factor for carbon monoxide (TLCO) shows a closer relationship to the amount of emphysema 5 and is also established for treatment monitoring. 6 Lung hyperinflation as determined by, for example, body plethysmography also correlates with emphysema on CT scans, but the values may differ from CT-derived volume measures, 7 possibly due to differences in posture as body plethysmography is commonly performed in sitting position.
In clinical practice, CT scans are performed due to a variety of indications, possibly with intravenous application of iodinated contrast media. Thus, the degree of standardization is low which becomes relevant when CT scans are retrospectively compared. Moreover, scans may be available only in a proportion of patients with possible emphysema. In contrast, assessment of emphysema in prospective clinical studies can be based on CT scans with highly standardized acquisition parameters. 8 Therefore, in clinical practice, the question remains important to which extent the presence of emphysema can be predicted from other assessments than CT, especially lung function. Besides spirometry and CO diffusing capacity, body plethysmography could be informative. The method seems suited to quantify lung hyperinflation which is thought to be associated with emphysema but has not been studied in this respect.
To determine the additional value of body plethysmography regarding its predictive power for the presence or absence of emphysema, we compared this assessment with spirometry and CO diffusion capacity. Each of the lung function measurements was evaluated either alone or in combination with the other measurements, using a subset of data from the German COPD cohort COPD and Systemic Consequences-Comorbidities Network (COSYCONET) in which routine CT scans were available.
Methods
Study population
For this analysis we used visit 1 data of the COSYCONET cohort. 9 Only patients with spirometric global initiative for chronic obstructive lung disease (GOLD) grade 1–4 10 or COPD patients at risk (symptoms of chronic bronchitis and forced expiratory volume in 1 second (FEV1)/forced vital capacity (FVC) ≥ 0.7; see below) with CT scans of sufficient quality and plausible lung function data were included. This resulted in 335 of 2741 patients (Table 1). The COSYCONET study was approved by the ethical committees of all study centers, and all patients gave their written informed consent. 9
Baseline characteristics of the subgroups with positive and negative binary emphysema score according to CT.a
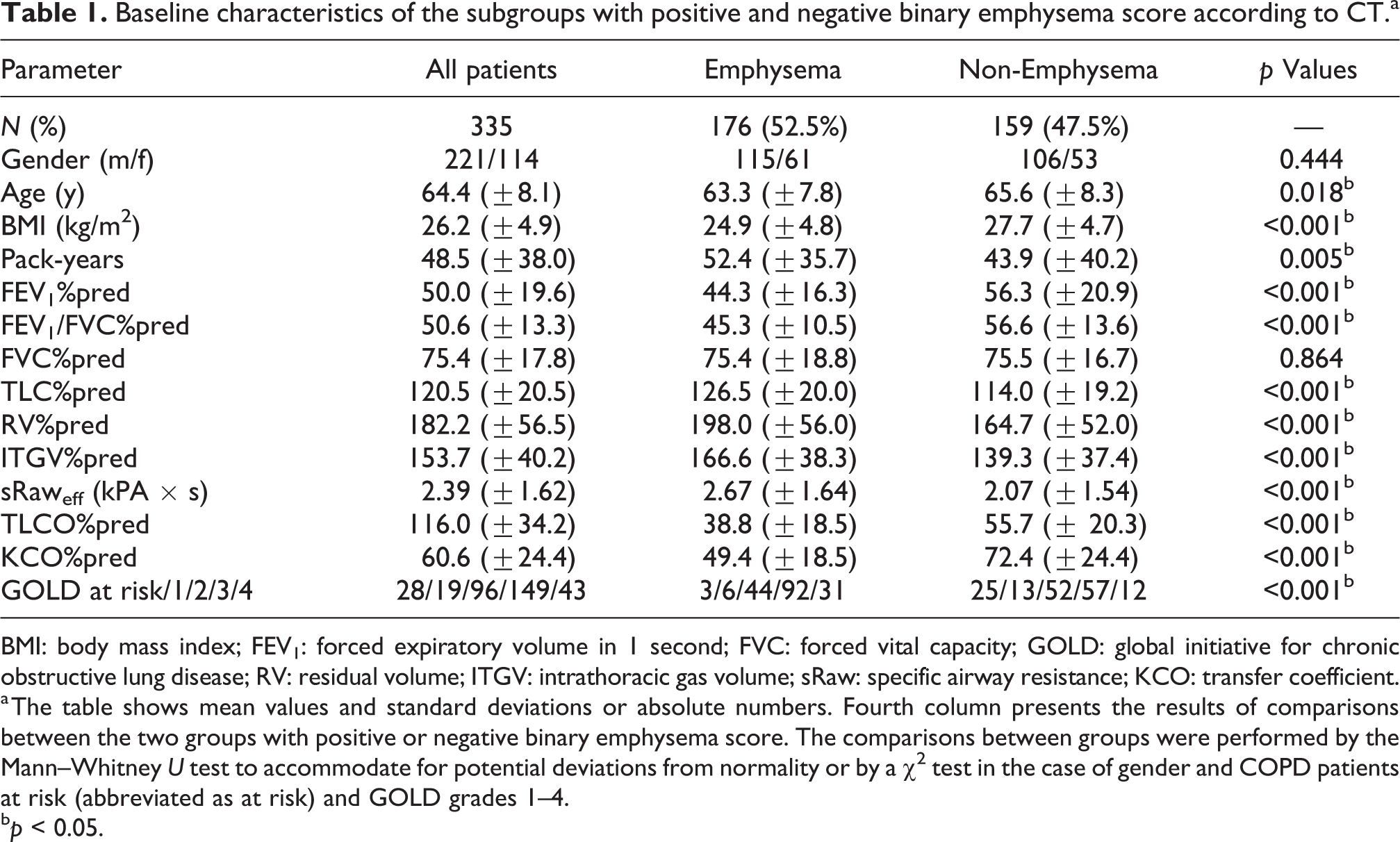
BMI: body mass index; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; GOLD: global initiative for chronic obstructive lung disease; RV: residual volume; ITGV: intrathoracic gas volume; sRaw: specific airway resistance; KCO: transfer coefficient.
a The table shows mean values and standard deviations or absolute numbers. Fourth column presents the results of comparisons between the two groups with positive or negative binary emphysema score. The comparisons between groups were performed by the Mann–Whitney U test to accommodate for potential deviations from normality or by a χ 2 test in the case of gender and COPD patients at risk (abbreviated as at risk) and GOLD grades 1–4.
b p < 0.05.
Assessments
The patients’ clinical and functional state was assessed at the first study visit, using a broad panel of tests that were guided by standard operating procedures as defined by the study protocols of COSYCONET. 9 Lung function comprised spirometry, body plethysmography, and diffusing capacity for carbon monoxide (CO). 9 From spirometry, FEV1, FVC, and the FEV1/FVC ratio were chosen for analysis; from body plethysmography, intrathoracic gas volume (ITGV), total lung capacity (TLC), and effective specific airway resistance (sRaw); and from diffusing capacity, the transfer factor for CO (TLCO), and the transfer coefficient (KCO). Predicted values of FEV1, FEV1/FVC, and FVC were taken from global lung function initiative (GLI), 11 values of ITGV and TLC from European Coal and Steel Community, 12 and values of TLCO and KCO from van der Lee et al. 13 All lung function data were subject to defined quality control procedures.
Evaluation of CT scans
Chest CT scans acquired within 4 years prior to inclusion into the study were collected and submitted to the central image bank of COSYCONET. Scans with findings that could hamper the analysis, for example, lobar pneumonia, were excluded but those obtained for suspected acute pneumonia were included if normal with respect to this condition.
CT scans were subjected to a standardized visual assessment by a chest radiologist with 25 years of experience in the field (HUK). For this purpose, an algorithm based on modified guidelines of the COPD Gene CT Workshop Group 2 was applied. The extent of emphysema was evaluated semiquantitatively with a five-point scale (<5%, 5–25%, 26–50%, 51–75%, and >75%) along with the leading type of emphysema (centrilobular vs. panlobular emphysema) in a lobe-based approach. In addition, the presence of bronchiectases, bronchial wall thickening, centrilobular nodules, and mosaic attenuation was recorded. Moreover, the prevalence of paraseptal emphysema and bullae (each for right and left side) was documented, as well as abnormalities (i.e. collapse or stenosis) of the trachea or right or left main stem bronchi. On the basis of these findings, finally, the observer specified the predominant COPD phenotype (either emphysema-type or not) in a binary score. The final score used in the analysis thus comprised emphysema versus non-emphysema (airway-type or unspecified).
In addition to visual scoring, a software-based evaluation of the CT data was performed using an automated algorithm for the quantification of emphysema (YACTA (version 1.1.14.1)). 14 –16 This provided continuous metrics related to pulmonary emphysema: (1) the mean lung density (MLD) in Hounsfield Units (HU), (2) the 15th percentile of the lung density histogram (15TH), (3) the emphysema index (EI) defined as the volume fraction of emphysema (lung tissue dropping below a density threshold of −950 HU) in percent of total lung volume. In the present analysis, only the binary emphysema score and the three continuous scores (MLD, 15TH, and EI) were evaluated.
Statistical analysis
Mean values and standard deviations are given for all parameters. The values of the two groups were compared using the Mann–Whitney U test, or χ 2 statistics and contingency tables, as appropriate. In the multivariate analyses, some of the parameters were transformed to achieve a normal distribution or at least a distribution very close to normal. These transformations have to be understood as purely statistical tools, which allowed the application of powerful parametric methods. Accordingly, the relationship of the binary emphysema score (emphysema versus non-emphysema) to predictors was analyzed via linear discriminant analysis. For this purpose, comprehensive sets of lung function parameters from spirometry, body plethysmography, and diffusing capacity were chosen. We used a stepwise approach with forward selection. Body mass index (BMI) was kept in the initial set of predictors in all models since it might affect lung volumes. To check the consistency of the results, analyses were repeated in the form of logistic regression analyses. The three continuous scores (lung density, 15th percentile of lung density, and EI) were analyzed by multiple linear regression with forward selection. Moreover, the scores derived from the discriminant analyses were used for the construction and comparison of receiver operating characteristic curve (ROC curve). Statistical significance was assumed for p <0.05. Analyses were performed using the software packages SPSS Statistics 24 (IBM Corp., Armonk, New York, USA) and MedCalc (comparison of ROC curves; Version 17.9, MedCalc Statistical Software, Ostend, Belgium).
Results
Baseline characteristics
CT scans that fulfilled the selection criteria were available in 335 of 2741 patients (Table 1). The binary score was positive for emphysema in 52.5% of patients (10.7%, 31.6%, 45.8%, 61.7%, and 72.1% of COPD patients at risk and with GOLD grades 1–4). The distribution of the binary emphysema score was significantly different across grades 0–4 (p < 0.001). There were also significant differences between the two groups regarding FEV1, FEV1/FVC, ITGV, residual volume (RV), TLC, KCO, TLCO, each in terms of predicted values, moreover in sRaw, age, pack-years, and BMI (Table 1).
CT scans
CT scans had been performed for a variety of purposes (among them 53.1% suspected emphysema, 5.1% COPD, 6.9% suspected tumor, 4.0% suspected pneumonia). Frequent values of slice thickness were 1.0 mm (33.6%), 3 mm (11.1%), and 5 mm (26.7%). The number of contrast-enhanced CT scans was equally distributed between patients with positive (40.9%) and negative (42.5%) emphysema score (p = 0.50).
Binary emphysema score
When discriminant analysis was started with the set of all lung function parameters, KCO%pred, FEV1/FVC%pred, sRaweff, and RV%pred remained as significant variables separating the two groups according to the binary emphysema score (Wilks’ Lambda 0.682, p < 0.001; specificity 71.9%; sensitivity 77.7%; positive predictive value 74.9%). All other lung function parameters and BMI were eliminated in the selection procedure irrespective of the fact that there were significant differences between groups (see Table 1). In these analyses FEV1/FVC%pred was evaluated as square root, and sRaw as logarithm in order to approximate a normal distribution; if the untransformed values were taken, RV was replaced by ITGV with virtually the same results of classification. The robustness of the discriminant analysis was checked by bootstrapping (1000-fold repetition). This yielded similar results as the initial analysis (sensitivity 77.7%, specificity 71.9%, positive predictive value 74.9%).
When omitting body plethysmography and using only spirometry and diffusing capacity, KCO%pred, FEV1/FVC%pred, and FVC%pred remained as significant predictors of the binary emphysema score (specificity 73.7%, sensitivity 76.3%, positive predictive value 75.0%).
When only the diffusing capacity parameters were used, KCO%pred remained (positive predictive value 71.2%). When only spirometric parameters were used, FEV1/FVC%pred and BMI remained (positive predictive value 69.8%). When only body plethysmographic parameters were used, ITGV%pred and BMI remained (positive predictive value 61.7%). The combination of body plethysmography and spirometry or body plethysmography and diffusing capacity yielded similar relationships as spirometry or diffusing capacity alone.
Logistic regression analyses were performed analogous to discriminant analyses and yielded similar results regarding the selection of parameters. Moreover, the binary emphysema score was not significantly related to the administration of intravenous contrast enhancement, in accordance with the results of cross-tabulation (see above).
To illustrate these results, ROC analyses were performed (Figure 1); the respective areas under the curve are given in Table 2. Using the DeLong method for the comparison of ROC curves, there was no significant difference between the triple combination of spirometry/diffusing capacity/body plethysmography, and the dual combination of spirometry/diffusing capacity (p = 0.624). The triple combination was significantly better than each of the single assessments (p < 0.05 each). Moreover, body plethysmography was inferior to both spirometry and diffusing capacity (p < 0.05 each).

Receiver operating characteristic (ROC) curves for different combinations of predictors. This figure shows the ROC curves for different combinations of predictors, either spirometry, body plethysmography, and diffusing capacity (blue); or spirometry and diffusing capacity (green); or spirometry (brown), diffusing capacity (yellow), and body plethysmography (purple) separately. The sets of parameters for the different combinations are given in the results section, and the corresponding AUC are given in Table 2. AUC: areas under the curve.
Corresponding areas under the curve for the different combinations of predictors.a

AUC: area under the curve.
a This table shows the corresponding areas under the curve for the different combinations of predictors, either spirometry, body plethysmography, and diffusing capacity; or spirometry and diffusing capacity; or spirometry, body plethysmography, and diffusing capacity separately. The sets of parameters for the different combinations are given in the results section.bNon-parametric assumption.
c Null hypothesis: AUC.
Continuous scores
Complete and quantitatively plausible data on MLD were available in 252 patients. Using all lung function parameters and BMI as predictors of MLD in stepwise multiple linear regression analyses, FEV1/FVC%pred, TLC%pred, and BMI (p ≤ 0.009 each) remained significant (R2 = 0.388). Predictors of the 15th percentile were FEV1/FVC%pred, KCO%pred, BMI (p ≤ 0.005 each), as well as ITGV%pred (p = 0.025; R2 = 0.415). Predictors of the emphysema index were ITGV%pred, FEV1/FVC%pred, TLCO%pred, and BMI (p < 0.012 each; R2 = 0.356). The relationship between the 15th percentile and the prediction score derived from multiple regression is illustrated in Figure 2. When introducing the presence of contrast enhancement as additional binary predictor into the regression analysis, this turned out to be significant (p < 0.05) for MLD, but not for EI and the 15th percentile of MLD.
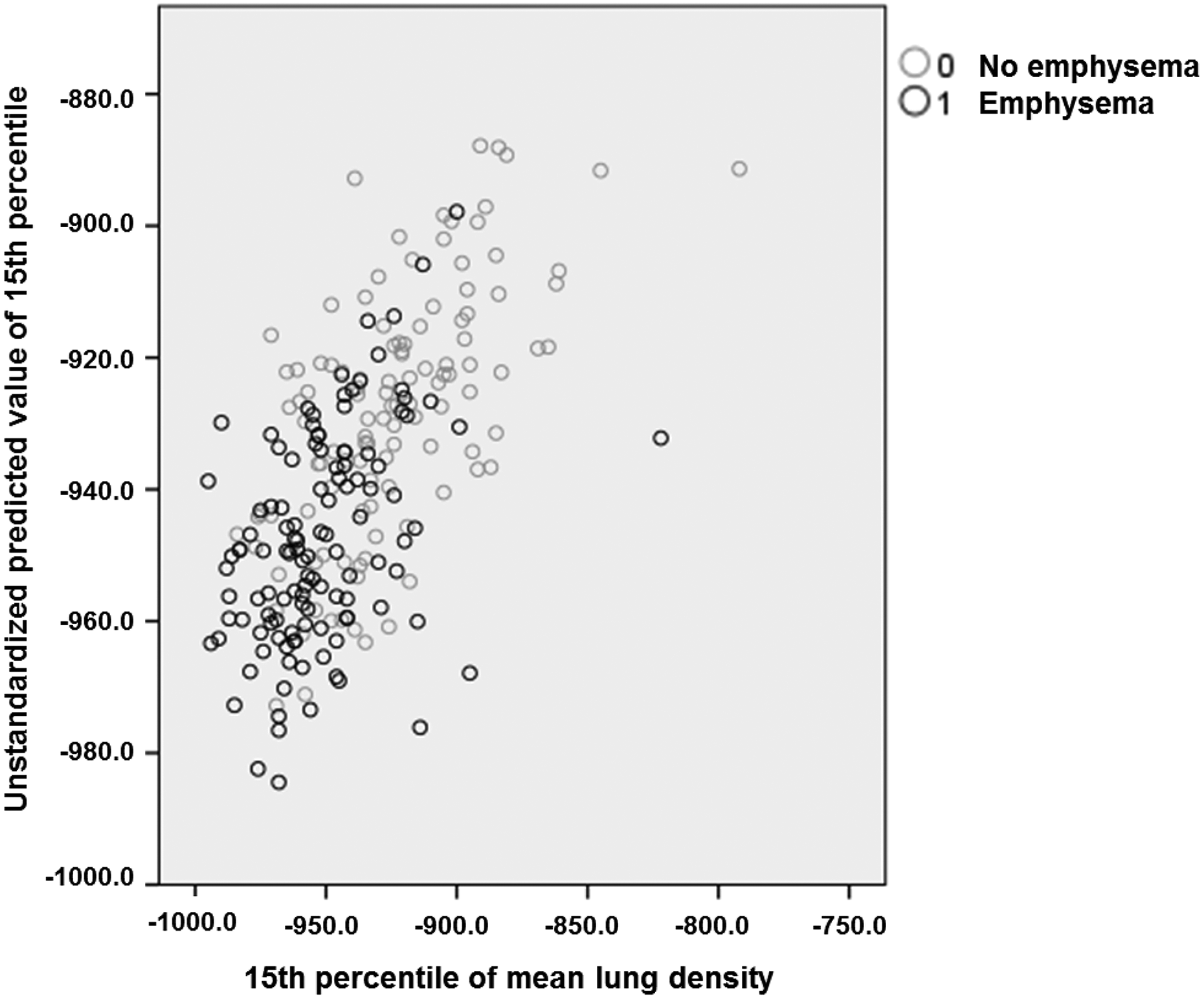
Relationship between the 15th percentile and the prediction score. The figure shows the predicted values from multivariate regression against the observed values of the 15th percentile of MLD. The regression equation comprised KCO%pred, ITGV%pred, FEV1/FVC%pred, and BMI (see text). The black circles indicate patients with positive binary emphysema score and the grey circles indicate patients with negative binary score. MLD: mean lung density; KCO: transfer coefficient; ITGV: intrathoracic gas volume; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; BMI: body mass index.
Discussion
In the present study, we evaluated the relationship between lung function tests, especially body plethysmography and EIs derived from CT scans. Body plethysmography was compared with spirometry and CO diffusing capacity, the well-known parameters that have been investigated previously. 17,18 sRaw and RV were statistically significantly correlated with a binary emphysema score, in addition to KCO and FEV1/FVC. When omitting body plethysmography and including only spirometry and diffusing capacity, the same correlation was achieved but with additional inclusion of FVC from spirometry. When using only spirometry or diffusing capacity, the predictive power was lower, and it was lowest when using only body plethysmography. The results for three continuous EIs were similar to those of the binary score but the associations were weaker. These findings suggest that spirometry and CO diffusing capacity confer the majority of lung function-based information on the presence of CT-diagnosed emphysema, whereas body plethysmography plays only a secondary role.
When COSYCONET patients were recruited, it was not possible to perform CT scans prospectively under standardized conditions, as in other studies. 19 Therefore, we had to use non-standardized scans obtained from clinical routine, many of which were not optimized for the assessment of emphysema. Despite these limitations, it was possible to process the scans in a manner that a binary emphysema score (emphysema versus non-emphysema) and three continuous emphysema indices could be evaluated. Owing to the outstanding experience of the observer, the visual evaluation of the binary score was probably less hampered by this lack of standardization than the continuous indices, whereas the quantitative indices relied on evaluations of lung density and were therefore more prone to variation. Heussel et al. have demonstrated that the intravenous administration of iodinated contrast media results in an increase of lung density and a decrease of quantitative emphysema parameters. 20 In the present study, the application of contrast enhancement influenced some of the software-based quantitative EIs but was not related to the binary score. As a consequence, the quantitative indices yielded less conclusive results than the binary score.
It seems remarkable that despite the lack of standardization and the fact that up to 4 years occurred between CT scan and lung function assessment, the retrospectively collected CT scans still allowed to identify correlations with lung function that were similar to those reported previously for spirometry and CO diffusing capacity. 17,21
To our knowledge, the set of common body plethysmographic parameters has not been evaluated for this purpose before, except for two reports including the ratio RV/TLC. 17,22 In our study, TLC turned out to be irrelevant and the value of RV was similar to that of ITGV. RV/TLC, if tentatively included, was always eliminated from the set of predictors. Although sRaw and RV turned out to be significant predictors in addition to the KCO and FEV1/FVC, the gain in information was small, as illustrated in the ROC curves. We used the effective value of sRaw which is more robust than total resistance. 23 Moreover, in contrast to other lung function parameters that were evaluated in terms of % predicted, we used the absolute values, as in adults the dependence on anthropometric characteristics is absent or weak. Taken together, these findings suggest that body plethysmography confers little additional information on the presence of emphysema, if spirometry and diffusing capacity are already available.
Age and smoking history in terms of pack-years were always eliminated as predictors. When the binary score was taken as outcome, BMI was included as a predictor if spirometry or body plethysmography were evaluated separately. Probably this was reflected by the effect of body weight on the volume-related parameters that remained as predictors. Regarding the selection of predictors and the ranking of their value, the results of linear discriminant analysis and logistic regression analysis were in concordance, underlining the validity of our statistical findings.
For the continuous CT indices, the results resembled those obtained for the binary score, but were more diverse. For the 15th percentile of lung density which showed the best results in terms of goodness of fit, FEV1/FVC%pred, ITGV%pred, and KCO%pred again appeared as significant predictors, in addition to BMI. Regarding MLD, various results have been published, including data indicating that the 15th percentile is less sensitive to changes in lung volume and more robust in longitudinal studies. 24,25 The inclusion of BMI probably reflected the dependence of lung density on body mass. 26 Due to these weaknesses, we focused on the binary emphysema score that was based on the visual inspection of the CT scan by an experienced radiologist and less dependent on technical variability than the continuous scores. Indeed it is known that visual assessment of CT scans of COPD patients can provide reproducible, substantial information on emphysema and airway type. 27,28
Limitations
Our study is inter alia limited by the fact that subjects who are most likely to have severe emphysema and hyperinflation, for example, lung transplantation or lung volume reduction patients, were excluded from the study due to the exclusion criteria of “having undergone major lung surgery.” In these patients, however, the question of predicting emphysema solely from lung function data might be of minor relevance. We also included only 12% of the total study cohort in the present analysis, thus our findings primarily apply to this subgroup of patients. Probably the selection bias associated with this has not weakened the associations identified in this study, since the sub-cohort studied showed a slightly greater impairment of lung function than the total COSYCONET cohort. The major limitation of our study is the retrospective collection of CT scans obtained for various clinical indications. It is reasonable to expect that this contributed to additional variability in the qualitative and quantitative evaluation which should have resulted in a weakening of correlations with lung function parameters. Moreover, up to 4 years between the CT scan and the study visit were allowed, which also could have weakened the association with lung function. This may have introduced some discrepancies between pulmonary function and CT assessment of emphysema. Irrespective of these limitations, the correlations found in our analysis were comparable to those reported in previous studies with prospective CT scans. We therefore believe that our conclusions regarding the additional value of body plethysmography are valid. Importantly, the binary emphysema score referred to the presence or absence of emphysema, thus the absence of emphysema did not necessarily mean the presence of an airway-dominated type.
Conclusions
RV, ITGV, and sRaw derived from body plethysmography conferred information on the presence of CT-diagnosed emphysema but less than spirometry and CO diffusing capacity. In particular, the additional information appeared to be minor compared to the well-known combination of spirometry and CO diffusing capacity. The maximum positive predictive value was about 75%. Thus, regarding the prediction of the presence of CT-diagnosed emphysema, the additional information as conferred by body plethysmography appeared to be minor compared to the well-known combination of spirometry and CO diffusing capacity. Irrespective of this, information on lung volumes is still important for therapeutic decisions in emphysema treatment, for example, regarding lung volume reduction.
Footnotes
Acknowledgment
The authors thank all patients of COPD and Systemic Consequences-Comorbidities Network (COSYCONET) for their kind cooperation and all study centers for their excellent work. Moreover, the authors are grateful to the Scientific Advisory Board of COSYCONET for their continuing support and helpful recommendations. The members of the board are Edwin JR van Beek (Clinical Research Imaging Centre (CRIC), The Queen’s Medical Research Institute, University of Edinburgh, UK), Klaus Friedrich Rabe (LungenClinic Grosshansdorf, Zentrum für Pneumologie und Thoraxchirurgie, Grosshansdorf, Germany), Joseph M Antó (Universitat Pompeu Fabra, Barcelona, Spain), Norbert Krug (Frauenhofer Institut für Toxikologie und experimentelle Medizin, Hannover, Germany), Michael Kiehntopf (Universitätsklinikum Jena, Institut für klinische Chemie und Laboratoriumsmedizin, Jena, Germany), Jørgen Vestbo (University of Manchester and South Manchester University Hospital NHS Foundation Trust, Manchester, UK), and Emiel F Wouters (Maastricht University Medical Center, Maastricht, The Netherlands). Furthermore, the authors thank the study coordinators Sandra Söhler and Inge Kokot (Philipps-University, Marburg, Germany).
Authors’ contribution
KK was involved in the conception of the study, analyzing and interpreting the data, statistical analysis, conceptualizing and drafting of the manuscript. BJ was involved in the interpretation of the data from this analysis and drafting of the manuscript. FB, JB, RMH, JB, and PAG were involved in the interpretation of the data from this analysis, took part in the discussion and critical revision of this manuscript. HW was involved in the interpretation of the data from this analysis and drafting of the manuscript. PA was involved in the analysis and interpretation of the data and also took part in the discussion and critical revision of this manuscript. CFV contributed to the overall design of COSYCONET, interpretation of data from this analysis, and the development and critical revision of the manuscript. HUK performed an assessment of all CT scans. HUK and RAJ were involved in the design and setup of the study, as well as quality control, statistical analysis, and conceptualizing and drafting of the manuscript. All authors approved the final submitted version and agreed to be accountable for all aspects of the work.
Declaration of conflicting interests
Kathrin Kahnert, Bertram Jobst, Frank Biertz, Jürgen Biederer, Henrik Watz, Rudolf M Huber, Jürgen Behr, Rudolf A Jörres declare no competing interests and have nothing to disclose. Claus F Vogelmeier gave presentations at symposia and/or served on scientific advisory boards sponsored by AstraZeneca, Boehringer Ingelheim, CSL Behring, Chiesi, GlaxoSmithKline, Grifols, Menarini, Mundipharma, Novartis, Teva, Cipla, Omniamed, and MedUpdate. Hans-Ulrich Kauczor declares no competing interests and has nothing to disclose with regard to the content of the article. Philippe A Grenier has nothing to disclose with regard to the content of the article.
Ethics approval and consent to participate
All assessments were approved by the central (Marburg (Ethikkommission FB Medizin Marburg)) and local (Bad Reichenhall (Ethikkommission bayerische Landesärztekammer)); Berlin (Ethikkommission Ärztekammer Berlin); Bochum (Ethikkommission Medizinische Fakultät der RUB); Borstel (Ethikkommission Universität Lübeck); Coswig (Ethikkommission TU Dresden); Donaustauf (Ethikkommission Universitätsklinikum Regensburg); Essen (Ethikkommission Medizinische Fakultät Duisburg-Essen); Gießen (Ethikkommission Fachbereich Medizin); Greifswald (Ethikkommission Universitätsmedizin Greifswald); Großhansdorf (Ethikkommission Ärztekammer Schleswig-Holstein); Hamburg (Ethikkommission Ärztekammer Hamburg); MHH Hannover/Coppenbrügge (MHH Ethikkommission); Heidelberg Thorax/Uniklinik (Ethikkommission Universität Heidelberg); Homburg (Ethikkommission Saarbrücken); Immenhausen (Ethikkommission Landesärztekammer Hessen); Kiel (Ethikkommission Christian-Albrechts-Universität zu Kiel); Leipzig (Ethikkommission Universität Leipzig); Löwenstein (Ethikkommission Landesärztekammer Baden-Württemberg); Mainz (Ethikkommission Landesärztekammer Rheinland-Pfalz); München LMU/Gauting (Ethikkommission Klinikum Universität München); Nürnberg (Ethikkommission Friedrich-Alexander-Universität Erlangen Nürnberg); Rostock (Ethikkommission Universität Rostock); Berchtesgadener Land (Ethikkommission Land Salzburg); Schmallenberg (Ethikkommission Ärztekammer Westfalen-Lippe); Solingen (Ethikkommission Universität Witten-Herdecke); Ulm (Ethikkommission Universität Ulm); and Würzburg (Ethikkommission Universität Würzburg)) ethical committees, and written informed consent was obtained from all patients.
Authors’ note
The study was conducted from September 2011 to December 2013 and comprised 2741 patients recruited within the COSYCONET framework (![]() , Identifier: NCT01245933).
, Identifier: NCT01245933).
For further information see, Karch A, Vogelmeier C, Welte T, Bals R, Kauczor HU, Biederer J, Heinrich J, Schulz H, Glaser S, Holle R et al., The German COPD cohort COSYCONET: Aims, methods, and descriptive analysis of the study population at baseline. Respir Med 2016, 114: 27–37.
The basic data are part of the German COPD cohort COSYCONET (![]() ) and available upon request. There is a detailed procedure for this on the website of this network. Specifically, the data can be obtained by submission of a proposal which is evaluated by the steering committee. All results to which the manuscript refers to are documented by appropriate texts, figures, or tables.
) and available upon request. There is a detailed procedure for this on the website of this network. Specifically, the data can be obtained by submission of a proposal which is evaluated by the steering committee. All results to which the manuscript refers to are documented by appropriate texts, figures, or tables.
Funding
The authors disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the German Federal Ministry of Education and Research (BMBF) Competence Network Asthma and COPD (ASCONET) and performed in collaboration with the German Centre for Lung Research (DZL). The project is funded by the BMBF with grant number 01 GI 0881, and is supported by unrestricted grants from AstraZeneca GmbH, Bayer Schering Pharma AG, Boehringer Ingelheim Pharma GmbH & Co. KG, Chiesi GmbH, GlaxoSmithKline, Grifols Deutschland GmbH, MSD Sharp & Dohme GmbH, Mundipharma GmbH, Novartis Deutschland GmbH, Pfizer Pharma GmbH, and Takeda Pharma Vertrieb GmbH & Co. KG for patient investigations and laboratory measurements.
