Abstract
Chronic obstructive pulmonary disease (COPD) will be a major leading cause of death worldwide in the near future. Weakness and atrophy of the quadriceps are associated with a significantly poorer prognosis and increased mortality in COPD. Despite that skeletal muscle dysfunction may affect both respiratory and limb muscle groups in COPD, the latter are frequently more severely affected. Therefore, muscle dysfunction in COPD is a common systemic manifestation that should be evaluated on routine basis in clinical settings. In the present review, several aspects of COPD muscle dysfunction are being reviewed, with special emphasis on the underlying biological mechanisms. Figures on the prevalence of COPD muscle dysfunction and the most relevant etiologic contributors are also provided. Despite that ongoing research will shed light into the contribution of additional mechanisms to COPD muscle dysfunction, current knowledge points toward the involvement of a wide spectrum of cellular and molecular events that are differentially expressed in respiratory and limb muscles. Such mechanisms are thoroughly described in the article. The contribution of epigenetic events on COPD muscle dysfunction is also reviewed. We conclude that in view of the latest discoveries, from now, on new avenues of research should be designed to specifically target cellular mechanisms and pathways that impair muscle mass and function in COPD using pharmacological strategies and/or exercise training modalities.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) will be a major leading cause of death worldwide within the next few years. 1 –6 Muscle dysfunction is a major comorbidity in COPD that leads to poor exercise capacity in the patients and negatively impacts their quality of life. 7 Moreover, COPD patients usually suffer from other comorbidities or conditions such as chronic heart failure, malnutrition, and aging that may further deteriorate their underlying muscle dysfunction. Additionally, the number of exacerbations, reduced physical activity or immobilization leads to deconditioning, which further impairs muscle mass and function in patients with COPD. Despite that skeletal muscle dysfunction may affect both respiratory and limb muscle groups in COPD, the latter are frequently more severely affected. As the diaphragm needs to contract at an optimal resting length, the mechanical factors play a paramount role in the respiratory muscle dysfunction of COPD patients, while this is not the case in the dysfunction of the lower limb muscles (Figure 1). Furthermore, both weakness and atrophy of the lower limb muscles (quadriceps) were also associated with a significantly poorer prognosis and increased mortality in patients with COPD 8,9 (Figure 2). On the other hand, respiratory muscle dysfunction may also lead to hypercapnic respiratory failure, exercise limitation, increased risk for acute exacerbations, and even death in patients with COPD. 10 On this basis, the assessment of skeletal muscle dysfunction is of crucial importance and should be included in the conventional clinical evaluation of patients with COPD.

Schematic representation of the most important differences between respiratory and peripheral muscle dysfunction in COPD patients. The mechanical factors, mainly characterized by alterations in thorax geometry and inspiratory overloads, play a paramount role in the ventilatory muscle dysfunction of these patients. COPD: chronic obstructive pulmonary disease.

Schematic representation of the relevance of skeletal muscle dysfunction and exercise intolerance as factors that predict prognosis in patients with COPD. COPD: chronic obstructive pulmonary disease.
With the aim to draw the medical and scientific communities’ attention to the relevance and clinical implications of muscle dysfunction, the current review article focuses on the description of the magnitude of the problem and its prevalence in COPD. Moreover, the most significant biological mechanisms that have been shown to directly contribute to COPD muscle dysfunction are also being reviewed separately for the respiratory and the lower limb muscles in the current review article, with a special emphasis on epigenetic regulation of muscle mass and function. Nonetheless, guidance on how to assess muscle dysfunction in clinical settings is not described herein, as all these aspects have been adequately defined in previously published statements and guidelines. 11,12
Muscle dysfunction in COPD: Definitions
The function of a muscle depends to a great extent upon the physiological properties of its components, especially the fibers. Their speed of contraction and predominant metabolic profile, which determine their resistance to fatigue, are the most relevant physiological characteristics. Type I fibers are slow twitch, are composed by myosin heavy chain (MyHC) type I, have a predominantly oxidative metabolism, and are very resistant to fatigue. Type IIx are fast-twitch fibers, are composed by MyHC type IIx, have an anaerobic metabolism, and are low fatigue resistant. Type IIa fibers exhibit intermediate physiological and metabolic profiles and are composed by MyHC type IIa. In clinical settings, muscle function is evaluated through the “in vivo” measurements of skeletal muscle properties: (1) strength, defined as the ability of the muscle to develop maximal force and (2) endurance or the ability to sustain a submaximal force over time. In COPD, muscle dysfunction is characterized by a decline in either strength or endurance of the affected muscles. 7 While nutritional availability (glycogen stored in the muscle) is the main contributor to endurance, strength is determined by the size of the muscle. Moreover, other factors such as capillary density, oxygen delivery, and the oxidative capacity of the myofibers may also contribute to the outcome in muscle performance of COPD patients.
Muscle weakness of the lower limb muscles, which is frequent in COPD, is commonly defined as a decline in the ability to generate force. 13,14 In clinical and experimental settings, the force of a muscle group is typically quantified using a dynamometer, thus reductions in muscle quadriceps force may help predict weakness. 9,14 Additionally, prolonged and strong contractions may lead to muscle fatigue, which increases in direct proportions to the rate of nutritional depletion. Interventions such as strength exercise training and specific nutritional support administered to patients with COPD improve their loss of muscle force and susceptibility to fatigue. 15
Prevalence of muscle dysfunction in COPD
In COPD patients, impaired muscle function and mass loss are common systemic manifestations. Respiratory and limb muscles are usually affected in these patients, thus contributing to ventilatory constraints, poor exercise tolerance, and reduced quality of life. 16 As such, in a European-based study, it was estimated that in one-third of the COPD patients, even at very early stages of their disease, quadriceps muscle dysfunction was already an apparent manifestation, which inversely correlated with body mass index, airflow obstruction, dyspnea, exercise capacity and dyspnea scores, but not with the airway obstruction. 14 In another investigation conducted on a cohort of 161 COPD patients who were evenly distributed from Global Initiative for Chronic Obstructive Lung Disease stage I to stage IV, quadriceps strength and rectus femoris cross-sectional area were also significantly reduced (19–25%) compared to a population of age-matched healthy controls. 17 Peripheral muscle endurance also impaired in COPD patients, even in those with preserved physical activity and mild-to-moderate airflow obstruction in another study. 18 An association between an early onset of muscle fatigue and decreased endurance was demonstrated independently of the airflow limitation or quadriceps muscle weakness of the patients. 18
The prevalence of respiratory and diaphragm muscle dysfunction, although not fully elucidated in large cohort studies, was also reported to be significant in different cohorts of patients with advanced COPD (20–30% decrease) compared to age-matched healthy controls. 19 –23 Poor muscle mass and impaired limb muscle dysfunction, as assessed by mid-thigh cross-sectional area and quadriceps weakness, respectively, were also shown to be strongly related to exercise capacity and quality of life, 16,24,25 to increase the utilization of health-care resources, 26 and to predict survival and mortality in patients with COPD. 8,9
Peripheral and respiratory muscle dysfunction in COPD
Lower limb muscle dysfunction
The peripheral muscles of patients with COPD showed a greater susceptibility to fatigue. 18,27 –30 Interestingly, during normal walking, the gastrocnemius and tibialis anterior muscles were also shown to be more prone to fatigue in COPD patients. 31,32 As abovementioned, strength and endurance properties are impaired in the lower limb muscles of patients with COPD. Although strength is not the most sensitive measure to evaluate muscle function, it is a very useful measurement in clinical settings due to its accessibility, which enables the clinician to easily diagnose skeletal muscle dysfunction in COPD. Importantly, the outcome of measurements of muscle strength varies widely depending on the type of tests employed: 11,12 voluntary maneuvers, in which the patient’s cooperation is of crucial importance, as opposed to nonvoluntary maneuvers in which the patient’s collaboration is irrelevant. Moreover, quadriceps muscle strength may also impair as a result of exacerbations in COPD patients, 33 –36 which are known to negatively influence muscle mass and exercise tolerance even several weeks after hospital discharge. 33 Despite that the assessment of muscle endurance may be more sensitive to the design of tailored interventions in COPD patients, its use in clinical settings is limited, probably as a result of a lack of standardized protocols or reference values.
Respiratory muscle dysfunction
In COPD, despite that the respiratory muscles undergo a positive adaptation (training-like effect) that renders them more fatigue resistant, 12,21,37,38 maximal inspiratory and expiratory pressures (strength) and endurance of these muscles are frequently reduced in the patients. 19,21 –23,37,39,40 Respiratory muscle dysfunction imposes ventilatory constraints to the patients that may worsen the underlying respiratory failure frequently present in COPD patients, especially in advanced stages. Furthermore, respiratory muscle dysfunction constitutes an important factor accounting for an increased risk of hospital readmissions. 10 In addition, acute exacerbations also impair respiratory muscle function as they severely affect whole-body muscle mass. 33 –36 Changes in lung volume during exacerbations pose the diaphragm and the rib cage in a mechanical disadvantage, which may further deteriorate the function of other respiratory muscles such as the intercostal muscles. 36
Etiological factors of muscle dysfunction in COPD
Cigarette smoke, hypoxia, hypercapnia and acidosis, metabolic alterations of several types, malnutrition, genetics, systemic inflammation, aging, comorbidities, concomitant treatments, exacerbations, and inactivity are counted among the most relevant etiological factors that contribute to muscle dysfunction in COPD patients through the action of several biological mechanisms.
Limb muscle dysfunction
As aforementioned, muscles from the lower limbs are usually more severely affected than those of the upper limbs and the respiratory muscles. In patients with COPD, most of the studies have focused so far on the analyses of the vastus lateralis of the quadriceps. Whether the same findings could be observed in other muscles of the lower limbs remains unknown. However, as the quadriceps is a major locomotor muscle and is easily accessible, it has been chosen as the best option for the studies aimed at exploring the mechanisms involved in the limb muscle dysfunction and exercise intolerance of COPD patients. To date, several etiologic factors leading to a deconditioned phenotype of the lower limb muscles in COPD have been identified (Figure 3).

Schematic representation on how the identified etiological factors (dark red panels) contribute to lower limb muscle dysfunction in COPD through the action of biological mechanisms (black panels) that negatively impact muscle phenotype and function in the patients. COPD: chronic obstructive pulmonary disease.
The most relevant contributing factors are the following: cigarette smoke, genetics, hypoxia, hypercapnia and acidosis, metabolic derangements including vitamin D, and testosterone deficiencies, drugs (especially systemic corticosteroids), other comorbidities, exacerbations, systemic inflammation, nutritional abnormalities, reduced physical activity, and aging as highlighted in several reviews and statements 11,12,37 (Figure 3). Nonetheless, physical inactivity, 17 nutritional abnormalities, 41 repeated exacerbations, 34 –36,42 and corticosteroids 36,43 are the main contributors to muscle function and mass impairment, especially in advanced COPD.
Respiratory muscle dysfunction
In COPD, since the respiratory muscles are chronically exposed to the inspiratory loads and must remain active throughout the existence of the patients, they are less severely affected than the lower limb muscles. In the last years, respiratory muscles have been studied through the analyses of the costal diaphragm, with very restricted access, and only via thoracotomy performed for clinical reasons (mainly lung cancer and lung volume reduction surgery).
In COPD patients, modifications in ventilatory mechanics as a result of static pulmonary hyperinflation, which modifies thorax geometry and shortens the diaphragm length, displace the muscle away from its optimal length to generate the required forces. 37 Additionally, their respiratory muscles need to overcome the increased work of breathing resulting from the greater elastic, resistive, and threshold inspiratory loads imposed by airflow limitation. 37 These factors, which are inherent to the respiratory condition, mostly contribute to ventilatory muscle dysfunction in COPD (Figure 4). However, in these patients, the respiratory muscles also undergo a positive adaptation (training-like effect) that renders them, especially the diaphragm, more fatigue resistant compared to diaphragm forces developed by healthy subjects when exposed to identical lung volumes 37,38 (Figure 4).

Schematic representation on how the different etiological factors (dark red panels) contribute to respiratory muscle dysfunction in COPD through the action of several biological mechanisms (black panels) that modify muscle phenotype and function in the patients. Several etiological factors exert beneficial effects (training-like effect, green panel) on muscle mass and performance through the action of different biological mediators (adaptive muscle phenotype, right-hand side panels) that lead to a certain adaptation of the inspiratory muscles in COPD. These adaptive mechanisms partly counterbalance the deleterious effects of other factors and biological mechanisms (middle black panels). COPD: chronic obstructive pulmonary disease.
In addition, the same etiological factors that affect the lower limb muscles, may also affect, to a different extent, the respiratory muscles in COPD: cigarette smoke, hypoxia, hypercapnia and acidosis, metabolic derangements, malnutrition, genetics, systemic inflammation, aging, comorbidities, concomitant treatments, exacerbations, and reduced physical activity 11,12,37 (Figure 4). Nevertheless, static hyperinflation, the number of exacerbations, nutritional abnormalities, and aging are probably the most relevant contributing factors to respiratory muscle dysfunction in COPD patients. 12,21,37,41 Furthermore, respiratory muscle dysfunction worsens in COPD patients during exercise or exacerbations as a result of dynamic hyperinflation. 37
Molecular and cellular mechanisms in COPD muscle dysfunction
The specific contribution of biological events and pathways to both respiratory and limb muscle dysfunction has been extensively discussed in different comprehensive reports 11,12,21,37,44 as well as in more specific reviews that were mainly focused on the discussion of structural abnormalities, 45 muscle remodeling, 21 oxidative stress and muscle wasting, 46 epigenetic regulation of muscle mass and function, 47,48 autophagy, 49 and metabolic derangements 50 together with the influence of exercise training 51 and exacerbations. 36 In the sections below, the contribution of the most relevant biological mechanisms to both peripheral and respiratory muscle dysfunction in COPD patients is being thoroughly described.
Lower limb muscles
In the lower limb muscles of COPD patients, several molecular and cellular events mediate the effects of the different etiological factors known to cause dysfunction of these muscles as well as modifications in their phenotype. Studies that have demonstrated the contribution of each biological event in the quadriceps muscle dysfunction of COPD patients are summarized below and in Figure 3.
Structural abnormalities
A fiber-type switch toward a less fatigue resistant (phenotype from slow-twitch to fast-twitch fibers) is a major feature in the vastus lateralis of patients with advanced COPD. 13,52,53 Furthermore, fast-twitch fibers are also of smaller size in patients with advanced COPD and poor muscle mass 13 thus contributing to muscle weakness (Figure 5). Additionally, factors such as aging, 54 malnutrition, 55 and systemic corticosteroid treatment 56 may worsen the fast-twitch fiber atrophy observed in the patients. Besides, a reduction in the capillary numbers and contacts 57,58 that could eventually impair oxygen delivery to the myofibers 59 –61 is another contributing biological mechanism. Other structural abnormalities such as muscle cell membrane and sarcomere damage may also negatively influence muscle function. 13,45,62 In fact, greater levels of sarcomere disruption were recently shown in the vastus lateralis of patients with COPD cachexia 63 (Figure 6). Mitochondrial derangements 50 of several types including decreased density as a result of fewer proportions of slow-twitch fibers 13,50,64 were also shown to alter muscle function in COPD 65 –67 (Figure 3).

Immunohistochemical staining using a specific antibody of type II fibers (dark brown) in the vastus lateralis of a COPD patient and a healthy control subject. Note that type II fibers were of smaller size in the muscles of the patients (for review see Puig-Vilanova et al., 2015). COPD: chronic obstructive pulmonary disease.

Ultrastructural images of sarcomerae in the vastus lateralis of a COPD patient and a healthy control subject. Note that sarcomerae were significantly more disrupted in the muscles of the COPD (for review see Puig-Vilanova et al., 2015). COPD: chronic obstructive pulmonary disease.
Redox balance
Oxidative stress is defined as the imbalance between oxidants and antioxidants in favor of the former. Increased levels of oxidative stress have been consistently demonstrated in the vastus lateralis of patients with COPD by our group and others. 13,68 –74 The most widely studied markers of oxidative stress are the oxidation of proteins such as protein carbonylation taking place within the skeletal muscles of patients with COPD, for review see references. 75,76 Importantly, oxidative stress markers were also shown to inversely correlate with several clinical and physiological parameters such as exercise capacity, body composition, and quadriceps strength of the patients. 13,69 Furthermore, an improvement in quadriceps muscle endurance was also seen in response to decreased systemic oxidative stress levels in severe COPD patients. 72 A rise in levels of systemic oxidative stress has also been shown in patients with severe COPD, 63,74 especially in those with muscle wasting, which was partly reversed after exercise training. 74
Although oxidative stress and inflammation have been proposed to be potentially associated, evidence shows that local inflammatory events are not prominently expressed in muscles of COPD patients, 13,68,70,77 even in atrophying muscles or during exacerbations. 36,42 Therefore, local oxidative stress and inflammation are not interrelated in COPD muscles. The content and activity of antioxidant enzymes, especially of mitochondrial superoxide dismutase 2, were greater in the quadriceps of patients with severe COPD than in the control subjects, whereas no differences were observed in levels of catalase enzyme. 13,63,69
Chronic hypoxia induces a reduction in muscle mass probably as a result of the interaction of several molecular mediators such as inflammation, 78 hypoxia inducible factor-1 signaling pathway, 79 oxidative stress, 73,80 and reduced oxidative enzyme capacity and capillary numbers. 81 Hypercapnia, which may worsen during exacerbations, may also play a role in COPD muscle dysfunction through acidosis as it enhances ubiquitin–proteasome proteolytic system activity 82,83 and/or through a reduction in protein anabolism 82,84 (Figure 3).
Cigarette smoke may also contribute to limb muscle dysfunction in COPD through several biological mechanisms such as decreased type I fiber sizes and proportions and reduced mitochondrial activity, while concomitantly inducing a rise in oxidative stress levels in healthy smokers and experimental animal models of chronic exposure to cigarette smoke 70,85 (Figure 3).
Enhanced muscle proteolysis
Several markers of the ubiquitin–proteasome pathway, especially atrogin-1 and total protein ubiquitination levels, have been consistently increased in the vastus lateralis of patients with severe COPD, characteristically in those with muscle wasting and/or cachexia. 13,33,63,86,87 Moreover, several redox signaling cellular pathways such as nuclear factor and forkhead box (FoxO)1 and FoxO3 were shown to mediate the loss of muscle mass in COPD cachectic patients. 13,63 Importantly, levels of several specific muscle proteins such as creatine kinase and MyHC were shown to be significantly decreased in the vastus lateralis of patients with severe COPD and muscle wasting. 13,63 Moreover, levels of carbonylation of those proteins were also increased in the same muscles, suggesting that oxidation may render those proteins more prone to degradation 13,63,76 (Figure 3).
Myostatin
Myostatin, which is a member of the transforming growth factor-β superfamily, is almost exclusively expressed in skeletal muscles and is a potent negative regulator of muscle mass. Myostatin expression was increased in the vastus lateralis 87,88 and diaphragm 89 of severe COPD patients. It has also been suggested that resistance training reduces myostatin levels in the limb muscles of non-wasted COPD patients, 90,91 eventually contributing to enhanced muscle mass in these patients. Nevertheless, in another investigation, 13 muscle protein levels of myostatin did not significantly differ between severe COPD patients and healthy controls (Figure 3).
Apoptosis
Severe COPD patients with normal weight exhibited increased levels of terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling (TUNEL)-positive nuclei in their vastus lateralis compared to control subjects. 92 Importantly, in that investigation, severe COPD patients with low body mass index exhibited even much greater levels of DNA fragmentation through the TUNEL assay in their vastus lateralis compared to healthy controls and severe COPD patients with normal weight, and those levels inversely correlated with the patients’ exercise capacity. 92 In another investigation conducted by our group, 77 levels of TUNEL-positive nuclei were also significantly greater in the vastus lateralis muscles of COPD patients with normal body composition than in control subjects. Figure 7 illustrates TUNEL positively stained nuclei in vastus lateralis of COPD patients. In the same investigation, 77 the vastus lateralis muscles exhibited extremely few fibers positively stained for active caspase-3 in both severe COPD patients and control individuals. Also, the ratio of cleaved caspase-3 to procaspase-3 was low in the limb muscles of both patients and controls. 77 These findings are in complete agreement with those reported in a previous study, 93 in which no cleaved caspase-3 immunohistochemical localization was found in the limb muscles of either COPD patients or control subjects.

A representative example of TUNEL-positive nuclei (dark brown staining, black arrow) together with non-stained nuclei (blue, red arrow) in the vastus lateralis of a COPD patient. COPD: chronic obstructive pulmonary disease; TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling.
Ultrastructural evaluation of cells is the gold standard for the diagnosis of apoptosis as it confirms the diagnosis made by other indirect methodologies (immunohistochemical procedures on light microscopy). Furthermore, early phases of apoptosis can only be identified using electron microscopy, whereas late phases of this phenomenon are seen on both light and electron microscopy. However, electron microscopy is not widely used for this purpose. Previously we also reported data on the ultrastructural diagnosis of apoptotic nuclei in both respiratory and limb muscles of patients with severe COPD. 77 The lower limb muscles exhibited very low and similar levels of either early or advanced ultrastructural nuclear apoptosis in both severe COPD patients and control subjects. 77 In Figure 8, examples of early and late nuclear apoptosis are shown in vastus lateralis muscles of a severe COPD patient.
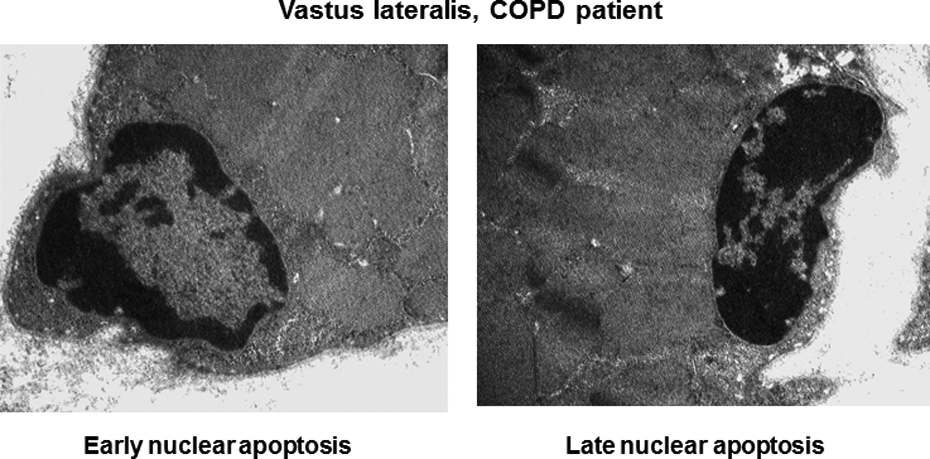
Representative electron micrographs of early and late apoptotic nuclei as identified using electron microscopy in the vastus lateralis of a COPD patient. COPD: chronic obstructive pulmonary disease.
Autophagy
Autophagy is the process whereby cellular components are degraded and recycled within the cells. A rise in autophagy events has been demonstrated in two recent studies, in which several molecular markers of autophagy and the number of autophagosomes were increased in the vastus lateralis of patients with advanced COPD 94,95 (Figures 3 and 9).

Representative electron micrographs of autophagosomes (white arrows) as identified using electron microscopy in the vastus lateralis of a COPD patient and a healthy control subject. Note that the number of autophagosomes was greater in the muscles of the patient (for review see Puig-Vilanova et al., 2015). COPD: chronic obstructive pulmonary disease.
Epigenetic events
Epigenetic control of cells, defined as the process whereby gene expression is regulated by heritable mechanisms that do not affect DNA sequence, has also emerged as a potential biological mechanism that may regulate muscle function and mass in COPD. 96,97 The epigenetic modifications identified so far in cells are as follows: (1) DNA methylation, (2) histone acetylation, (3) histone methylation, and (4) non-coding RNAs such as microRNAs (Table 1). DNA methylation at the 5 position of cytosine specifically reduces gene expression. Acetylation is a transient, enzymatically controlled biochemical process and the commonest posttranslational modification of histones. Acetylation, a process mediated by histone acetyltransferases results in a rather open chromatin structure that is transcriptionally active, while deacetylation through the action of histone deacetylases (HDAC) blocks transcription (Table 1). Additionally, methylation of histones may activate or repress gene transcription depending on the proteins recruited to the chromatin (Table 1).
Epigenetic modifications in cells.

DNA, deoxyribonucleic acid; RNA, ribonucleic acid.
MicroRNAs, encoded by eukaryotic nuclear DNA, are non-coding single-stranded RNA molecules (18–24 nucleotides) that function in the posttranscriptional regulation of gene expression (Table 1). They exert their action via base pairing with complementary sequences in mRNA molecules that result in gene silencing via translational repression or target degradation (Table 1). MicroRNAs may have different mRNA targets, and in a similar manner, a given mRNA may also be targeted by multiple microRNAs. MicroRNAs regulate many physiological cellular processes and may play a significant role in the pathogenesis of several lung diseases including COPD. 98
Specifically, within the muscle fibers, muscle-enriched microRNAs regulate several events including muscle repair and myogenesis and adaptation to environmental factors such as overloading and deconditioning. For instance, miR-1 promotes muscle cell differentiation and innervation, whereas miR-133 favors myoblast proliferation by repression of the serum response factor, which inhibits myotube formation. Moreover, miR-206 also stimulates innervation and myotube formation by targeting the p180 subunit of DNA polymerase α which leads to DNA synthesis inhibition and cell cycle withdrawal. 99 –101 Other microRNAs such as miR-181, miR-27, and miR-486, which exert their actions in several tissues, are also abundantly expressed in muscles and may regulate skeletal muscle development and phenotype. 47,48
In COPD patients with preserved body composition, a pioneering study 97 demonstrated that miR-1 levels were reduced, while those of HDAC4 were increased in their vastus lateralis. In another study, 53 levels of the transcription factor Yin Yang 1, which modifies histones, inversely correlated with the size of slow- and fast-twitch fibers in the limb muscles of COPD patients with normal body composition. Furthermore, plasma levels of muscle-specific microRNAs were also shown to be increased in patients with severe COPD. 96 Recently, in the vastus lateralis of patients with mild COPD, miR-1 expression was shown to increase, while also positively correlated with both forced expiratory volume in 1 second and quadriceps force. 102 Furthermore, in another study, 95 the expression of miR-1, miR-206, and miR-27a; levels of lysine-acetylated proteins and histones; and acetylated histone 3 were increased in the quadriceps of COPD patients, especially in those with muscle weakness, while expression of HDAC3, HDAC4, and sirtuin-1 were decreased. Collectively, these findings suggest that several epigenetic events are differentially expressed in the limb muscles of COPD patients, probably as an attempt to counterbalance the underlying mechanisms that alter muscle function and mass (Figure 3).
Respiratory muscles
Several molecular and cellular events that are involved in the pathophysiology of peripheral muscle dysfunction in COPD patients also contribute to their respiratory muscle dysfunction. The most widely studied mechanisms are described below and summarized in Figure 4.
Structural adaptations
Adaptive cellular events take place in the respiratory muscles, especially the diaphragm of COPD patients. Such mechanisms exert beneficial effects on the diaphragm fibers, rendering them more resistant to fatigue, thus neutralizing the deleterious effects of other mechanisms. The most relevant adaptive phenotypic features identified so far in the diaphragm of COPD patients are the following: shorter sarcomere length, 103 larger proportions of slow-twitch fibers (Figure 10), greater myoglobin content, 20,104 increased capillary contacts per fiber, 104 higher mitochondrial density and activity, 103,105 and enhanced aerobic enzyme capacity 105,106 (Figure 4). Furthermore, in COPD patients, similar adaptive cellular events were also reported to take place in the external 107 –109 and parasternal 110 intercostal muscles, in which a fast-to-slow transformation in both fiber-type and MyHC proportions were consistently observed. Furthermore, in patients with advanced COPD, biological mechanisms similar to those involved in the peripheral muscle dysfunction also contribute to the pathophysiology of the dysfunction of their respiratory muscles. 12 Such mechanisms are described below.

Immunohistochemical staining using a specific antibody of type I fibers (dark brown) in the diaphragm of a COPD patient and a healthy control subject. COPD: chronic obstructive pulmonary disease.
Structural abnormalities
Sarcomere disruption, as a fingerprint of muscle injury, is common in the diaphragm of patients with severe COPD and was significantly increased after a period of respiratory loading. 111 Moreover, contractile function of isolated fibers was also impaired in the diaphragm of patients with mild COPD. 112 Alterations in structural titin were also shown in the diaphragm of COPD patients in another study. 113 Nevertheless, other authors have reported no alterations in the expression of costameric proteins or muscle damage in the diaphragm of COPD patients compared to the controls. 114
Apoptosis
The levels of TUNEL-positive nuclei were also shown to be increased in the diaphragm of COPD patients as demonstrated in a previous investigation from our group 77 (Figure 4). Nonetheless, in the same muscles, levels of early or advanced ultrastructural nuclear apoptosis and caspase-3 were similar between COPD patients and the control subjects. 77 We concluded that the rise in TUNEL-positive nuclei detected in the diaphragm of the patients could rather be the result of increased DNA repair activity than apoptosis, 77 as this event does not seem to occur in COPD beyond the physiological levels detected in the muscles of the control subjects. Figure 11 illustrates TUNEL-positively stained nuclei in diaphragm of COPD patients.

A representative example of TUNEL-positive nuclei (dark brown staining, black arrow) together with non-stained nuclei (blue, red arrow) in the diaphragm of a COPD patient. COPD: chronic obstructive pulmonary disease; TUNEL: terminal deoxynucleotidyl transferase-mediated dUTP nick-end labeling.
Oxidative stress
Increased oxidative stress as measured by protein carbonylation was seen in the diaphragm of patients with severe COPD. 19 Moreover, protein oxidation levels inversely correlated with the degree of the airway obstruction among the patients. 19 In another study, 22 the most significantly carbonylated proteins were identified in the diaphragm of patients with COPD: actin, creatine kinase, and carbonic anhydrase. Carbonylation levels of these proteins were greater in the diaphragm of the patients than in the controls. 22 Importantly, creatine kinase activity levels and MyHC protein content were significantly lower in the diaphragm of COPD patients than in the healthy subjects. 22 These findings suggest that oxidation may render the target proteins more susceptible to be degraded, as also shown in another investigation, 115 in which MyHC content also decreased in the diaphragm of COPD patients. On the other hand, oxidation of specific residues may also impair the activity of specific enzymes that play key roles in muscle metabolism 115 (Figure 4).
Enhanced proteolysis
As abovementioned oxidative stress may be a major trigger to increased muscle proteolysis in COPD. 76 In a seminal study, 115 several markers of proteolysis were shown to be upregulated in the diaphragm of patients with COPD. A substantial rise in several markers such as 20S proteasome activity, ubiquitin–proteasome ligases, and caspase-3 activity was observed in the diaphragm of patients with mild-to-moderate COPD. 115 In another relevant study, 89 several mechanisms of proteolysis such as the ubiquitin–proteasome pathway and myostatin were upregulated in the diaphragm of COPD patients, while MyoD expression was reduced, suggesting that these events may influence diaphragm remodeling in COPD (Figure 4).
Epigenetic events
Abnormalities in the expression of several epigenetic markers have been recently reported. 116 In this regard, muscle-specific microRNA expression was downregulated, while that of HDAC4 and myocyte enhancer factor 2C protein levels were higher in the diaphragm of the COPD patients. 116 However, levels of DNA methylation levels and muscle fiber types and sizes did not differ between patients and controls. 116 We concluded that these epigenetic events act as biological adaptive mechanisms to better overcome the continuous inspiratory loads of the respiratory system in COPD patients 116 (Figure 4).
Other factors
In stable COPD, a fragile balance between adaptive and deleterious biological mechanisms exists in the respiratory muscles. Nevertheless, the predominance of factors such as exacerbations, nutritional abnormalities, and aging may rapidly skew the balance toward a rather negative phenotype of the diaphragm, thus leading to ventilatory failure and eventual death of the patients. 21,36,37
Concluding remarks
Muscle dysfunction in COPD is a common systemic manifestation that should be evaluated on routine basis in clinical settings. Despite that muscle dysfunction with and without mass loss takes place in both respiratory and limb muscle groups, the latter are usually more severely affected, with the resulting implications on the patients’ quality of life. Impairment of muscle strength is the most relevant feature observed in both diaphragm and quadriceps muscle dysfunction in COPD. Most of the investigations have been conducted on the basis of a reduction in force generation by muscles of COPD patients. In the last two decades, much progress has been achieved on the cellular and molecular mechanisms that lead to respiratory and peripheral muscle dysfunction and wasting in patients with COPD, especially in those with a more severe disease. Despite that ongoing research will shed light into the potential contribution of additional mechanisms to COPD muscle dysfunction, current knowledge points toward the involvement of a wide spectrum of cellular and molecular events that are differentially expressed in respiratory and limb muscles. Moreover, several so-called systemic and local factors also play a paramount role in the development of respiratory and peripheral muscle dysfunction in COPD patients. In view of the latest discoveries, new avenues of research should be designed in the near future to specifically target cellular mechanisms and pathways that impair muscle mass and function in COPD using pharmacological strategies and/or different exercise training modalities.
Footnotes
Authors’ Note
Dr EB was a recipient of the ERS COPD Research Award 2008.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: CIBERES, FIS 11/02029 (FEDER), FIS 14/00713 (FEDER), SAF-2011-26908, SAF-2014-54371-R, SEPAR 2010, SEPAR 2014, FUCAP 2009, FUCAP 2011, FUCAP 2012, and Marató TV3 (MTV3-07-1010) (Spain) have contributed to support part of the research described in this review.
