Abstract
Skeletal muscle quadriceps low-frequency fatigue (LFF) during exercise promotes improvements in exercise capacity with exercise training. In healthy subjects, eccentric muscle work induced by downhill walking (DW) generates higher muscular stress, whilst metabolic cost is lower compared to level walking (LW). We investigated quadriceps LFF and metabolic cost of DW in patients with chronic obstructive pulmonary disease. Ten participants (67 ± 7 years, FEV1 51 ± 15% predicted) performed DW, DW carrying a load (DWL) of 10% body weight via vest and LW, in random order. Quadriceps potentiated twitch force (TWqpot) was assessed before and after each walk, and muscle damage was assessed before and 24 hours after each walk via serum creatine kinase (CK) levels. Ventilation (VE) and oxygen consumption (VO2) were measured via breath-by-breath analysis during each walk. DW and DWL resulted in a greater decrease in TWqpot (−30 ± 14 N in DW, p < 0.05; and −22 ± 16 N in DWL, p < 0.05) compared to LW (−3 ± 21 N, p > 0.05). CK levels only increased 24 hours following DW and DWL (p < 0.05). DW and DWL showed lower VE and VO2 than LW (p < 0.05). DW is associated with enhanced quadriceps LFF and lower cardiorespiratory costs than LW. The addition of a chest load to DW does not seem to enhance these effects.
Introduction
Skeletal muscle weakness and exercise capacity impairment are common and important features of chronic obstructive pulmonary disease (COPD). 1 Skeletal muscle dysfunction is associated with poor exercise capacity and is a predictor of mortality. High-intensity exercise training is recommended to improve cardiovascular and skeletal muscle function, 1,2 however, treatment responses are variable. 3
The development of low-frequency fatigue (LFF) may be an important determinant of optimal response to exercise. LFF occurs when the muscle force response to low-frequency stimulation decreases in association with a slow (hours or days) recovery. 4 It is characterized by decreased intracellular calcium ion concentration 5,6 and muscle damage. 7 LFF can be effectively stimulated via eccentric muscle training due to its capacity to induce skeletal muscle damage. 8,9 The benefits of inducing LFF in people with COPD have been recently demonstrated in a study of exercise training by Burtin and colleagues. 10 In this study, the development of quadriceps LFF following a single exercise session (in approximately 30% of participants) was associated with superior improvements in functional exercise capacity and symptoms related to quality of life at the end of the 12-week high-intensity rehabilitation programme. 10,11 This highlights the potential importance of identifying treatment modalities capable of eliciting LFF.
Downhill walking (DW) is an exercise modality with a relatively high eccentric component embedded in a usual activity (walking). 12 Eccentric muscle work occurs due to the ‘braking’ action of quadriceps femoris required to slow the individual down during movement. Work may be further increased via the addition of a load to the chest (i.e. DW carrying load (DWL)). 12 DW is of particular interest for individuals with COPD as it offers a unique opportunity to efficiently induce skeletal muscle stress whilst simultaneously minimizing ventilatory demand during exercise. 13,14
The primary aim of this study was therefore to determine the extent to which DW and DWL induce quadriceps LFF compared to level walking (LW) in individuals with stable COPD. The secondary aims were to investigate the cardiorespiratory costs, dyspnoea and fatigue response and development of delayed onset muscle soreness following DW, DWL and LW.
Methods
Study population
Twelve individuals with COPD 15 were recruited from the outpatient clinic of University Hospital Gasthuisberg, Leuven, Belgium. All patients were familiar with treadmill walking and were free of exacerbations in the previous month before study commencement. Individuals were excluded if they had underlying disease or musculoskeletal limitations that would impair test procedures, if they required supplemental oxygen during exercise or could not walk at least 15 minutes continuously. Those with a body mass index (BMI) greater than 30 kg/m2 were also excluded due to the potential for musculoskeletal injury relating to the extra load requirements of the DWL intervention.
Procedure
This study was approved by the ethics committee of UZ Gasthuisberg, Leuven, Belgium. Written informed consent was obtained from all patients. Participants attended clinic on four visits, each separated by 1 week. The first session comprised familiarization with DW and baseline measurement of spirometry, functional exercise tolerance (6-minute walking test (6MWT)), 16 cycle maximal exercise capacity, 17 handgrip force, 18 isometric quadriceps femoris strength, 19 usual dyspnoea (modified Medical Research Council dyspnoea scale) 20 and health status (COPD assessment test). 21 Thereafter, patients performed three walking protocols on a treadmill (DW, LW and DWL) on the same day and time of each week in random sequence.
Interventions
DW was performed using a modified treadmill set-up (Figure 1) at a negative inclination of 10%. This was based on previous research in healthy subjects demonstrating a nadir cardiopulmonary demand and heart rate response at −10% compared to other negative inclinations (range 0 to −18%) at the slower of the two walking speeds investigated (5.4 km/hour). 14 DWL was performed the same as DW with the addition of a vest, loaded with equal weight distribution around the chest equivalent to 10% of body weight. 22 LW consisted of conventional walking on a treadmill without any inclination. Participants were encouraged to walk as long as possible up to 20 minutes (minimum 15 minutes) for each modality. Velocity was kept constant for the three modalities (75% of the average walking velocity achieved during the initial 6MWT). 23 Participants were allowed to hold onto side hand bars during walking, if necessary.

Equipment set-up for DW. Above: Modified treadmill set-up for DW (portable metabolic monitor in situ); below: custom-made bracket to achieve 10% negative incline (approximate cost €200). DW: downhill walking.
Data collection
Markers of fatigue
The protocol used to verify the presence or absence of quadriceps femoris fatigue has been previously published by our research group. 11 Briefly, subjects sat in a recumbent chair with hips extended at 120°, knees flexed at 90° and arms crossed in front of the chest. Unpotentiated quadriceps twitch contraction (TWqunpot), maximal voluntary contraction (MVC) and potentiated quadriceps twitch contraction (TWqpot) were measured in the order described before, 15 and 40 minutes after each walk, and contractile force (in newtons) was recorded. At least five repetitions were made, and the average of the two best attempts (within 5% difference) was used for analysis. The femoral nerve was stimulated through a 45 mm figure-of-eight coil powered by a double Magstim stimulator (Magstim Co Ltd, Whitland, Dyed, Wales, UK). A regularly calibrated strain gauge force transducer (DS Europe, model 546QD, Milan, Italy) was used to register force. Signals were amplified (model 811A amplifiers; Hewlett-Packard, Palo Alto, California, USA) and stored on a computer. Consistent with previous research, a TWqpot decrease greater than 15% at 15′ was defined a priori as significant LFF. This threshold is equivalent to the upper limit of variability of two consecutive measurements of the TWqpot. 24
Markers of cardiorespiratory cost
Breath-by-breath measurement of gas exchange, oxyhaemoglobin saturation, ventilation (VE) and heart rate was performed during each walk via a portable device (Oxycon mobile, Carefusion, San Diego, California, USA) and averaged over 1 minute intervals. Blood levels of lactate were measured before and 2 minutes after each walk (colorimetric/enzymatic method, lactate oxidase/perioxidase, Hitachi (Japan)/Roche (Switzerland) – COBAS c702).
Other outcomes
Serum levels of creatine kinase (CK) were collected before and 24 hours after the walks as markers of muscle damage 25,26 (colorimetric/International Federation of Clinical Chemistry method, COBAS c702). Dyspnoea and leg discomfort were assessed with a modified Borg dyspnoea scale 27 every 2 minutes. Over the 7 days after test completion (at the same hour as per testing), participants documented perceived muscle soreness via a 15 cm visual analogue scale (VAS; higher scores = worse). 28
Analysis
Statistical analyses were performed with Statistical Analysis Software (SAS 9.3, SAS Institute Inc, Cary, North Carolina, USA). Data are described as mean (SD) or median (interquartile range) according to data distribution. Data normality was assessed by the Shapiro–Wilk test. One-way repeated measures analysis of variance was used to compare change over time within walks for TWqpot, TWqunpot and MVC. The magnitude of change in TWqpot force (N) from baseline to 15 minutes was compared between interventions via separate paired t-tests and reported as measures of effect size (Cohen’s d). Effect size classes were defined according to calculated d values (small (d ≤ 0.2), moderate (d = 0.5) or large (d ≥ 0.8)). 29 The incidence of quadriceps LFF was compared across the three interventions via χ2 test and between individual interventions via Fisher’s exact tests. Lactate and CK level changes were analysed via paired t-tests. Statistical significance was defined as p < 0.05 for all analyses.
Results
Two participants were excluded from the study after recruitment. One withdrew consent, whilst the other was unable to walk the minimum required distance (15 minutes) during LW. Ten participants completed the study protocol and were included in the analyses. Demographic characteristics are detailed in Table 1.
Participant characteristics (n = 10).a
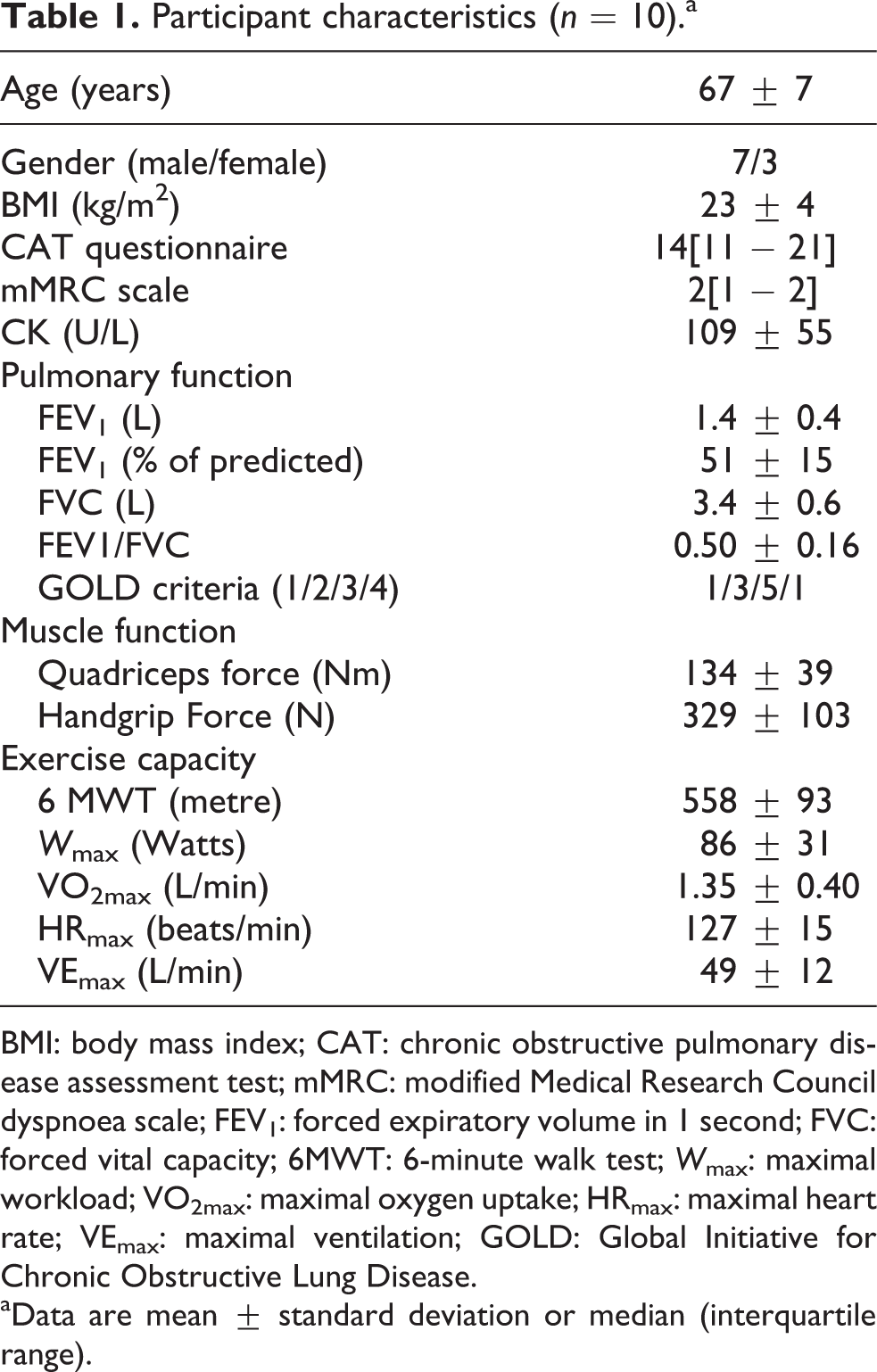
BMI: body mass index; CAT: chronic obstructive pulmonary disease assessment test; mMRC: modified Medical Research Council dyspnoea scale; FEV1: forced expiratory volume in 1 second; FVC: forced vital capacity; 6MWT: 6-minute walk test; W max: maximal workload; VO2max: maximal oxygen uptake; HRmax: maximal heart rate; VEmax: maximal ventilation; GOLD: Global Initiative for Chronic Obstructive Lung Disease.
aData are mean ± standard deviation or median (interquartile range).
Participants were mostly males (70%), with normal BMI (23 ± 4kg/m2) and decreased exercise capacity (88 ± 10% predicted 6MWT; 64 ± 20% predicted maximal workload during cardiopulmonary exercise capacity). Mean walking speed across all interventions was 4.2 ± 0.7 km/hour. Eight participants achieved the maximum target of 20 minutes treadmill walking (one walked 16′10′′ in DWL whilst the other 16′ in LW).
Contractile muscle fatigue
Both the DW and DWL interventions were associated with statistically significant decreases in TWqpot, TWqunpot and MVC over time. These changes were not observed following LW (Table 2). A decrease in TWqpot greater than 15% (a priori definition of LFF) was observed at 15 and 40 minutes in 9/10 participants following DW, 6/10 participants following DWL and only 3/10 participants following LW (χ 2 = 7.5, p = 0.02). This incidence of LFF was significantly greater for both DW and DWL compared to LW but did not differ between DW and DWL. Although the cut point of 15% is somewhat arbitrarily chosen and could be criticized, the present findings do not depend on this threshold. When approached as a continuous variable, contractile muscle fatigue was observed more evidently in DW. The mean decrease in TWqpot was −22 ± 6% at 15 min and −24 ± 7% at 40 min for DW (p < 0.0001), −16 ± 12% at 15 min and −16 ± 10% at 40 min for DWL (p < 0.0001) and −5 ± 9% at 15 min and −7 ± 10% at 40 min for LW (p = 0.62) (Figure 2). In comparison with LW, these reductions from baseline to 15 minutes in TWqpot force were significantly greater for both DW (p = 0.0008; Cohen’s d = 1.9) and DWL (p = 0.008; Cohen’s d = 1.01) but not significantly different between DW and DWL (p = 0.18; Cohen’s d = 0.5). Similar patterns of change across the three interventions were observed for TWqunpot and MVC (Table 2). Serum CK levels were significantly greater following DW (+49 ± 53%) and DWL (+50 ± 60%) but not LW (+14 ± 24%) after 24 hours.

Change in potentiated quadriceps twitch muscle force. (a) level walking; (b) downhill walking; (c) downhill walking with load. Data are represented as percentage of initial values. Bars represent mean change. Lines show individual responses. Dotted line represents the threshold of fatigue (15% of decrease in muscle force from the initial values). PRE: values before the walk; 15 min: potentiated twitch force 15 minutes after walking; 40 min: potentiated twitch force 40 minutes after walking. *p < 0.05: versus PRE.
Change in quadriceps muscle force and CK.a

TWqpot: potentiated quadriceps twitch contraction; TWqunpot: unpotentiated quadriceps twitch contraction; CK: creatine kinase; Pre: values collected prior the walks; 15 min: values collected 15 minutes after the end of the walks; 40 min: values collected 40 minutes after the end of the walks; 24 h: values collected 24 hours after the end of the walks; DW: downhill walking; DWL: downhill walking carrying load; LW: level walking.
aData are mean ± standard deviation.
bSignificant between-group difference in magnitude of change from baseline compared to LW (p < 0.05).
cSignificant within-group effect over time (analysis of variance) for corresponding intervention (p < 0.05).
dSignificant within-group change from baseline (p < 0.05).
The inducement of LFF did not appear related to the onset of muscle soreness in the week following the test protocols. VAS scores were generally low (median 1/15, range 0–6 for all interventions) without considerable variability within or between individuals. No clinically or statistically significant differences in VAS scores were observed between the three modalities on any day after the interventions (p > 0.05). There was a small, statistically significant decrease (improvement) in VAS scores from day 1 to day 7 (only) following DWL.
Cardiorespiratory responses during the walking modalities
A detailed overview of the cardiorespiratory responses to the three walking modalities is provided in Table 3. No differences in resting oxygen consumption (VO2), VE and heart rate were observed between DW, DWL and LW. Steady-state conditions, defined as a lack of variation greater than 10% during the final 5 minutes of measurement, were reached in all patients in all modalities. DW and DWL were associated with significantly lower VO2 and VE during steady state than LW. During LW, participants reached 74 ± 14% of their individual VO2 peak and 74 ± 22% of VE peak. These values were significantly lower during DW (64 ± 16% of VO2 peak; 65 ± 20% of VE peak) and DWL (68 ± 15% of VO2 peak; 68 ± 16% of VE peak; Figure 3). No significant differences in heart rate were observed during steady state between the three modalities. Lactate levels were generally low and did not change from resting values for any modality. There were no significant differences in perceived dyspnoea and fatigue between the three modalities in the final minute of the walks (p > 0.05); however, small, statistically significant increases were observed from baseline to test completion for all modalities (Table 3).

Oxygen uptake and V′E in the three modalities. (a) Oxygen consumption during walking; (b) Ventilation during walking. Data are mean ± standard deviation. Dotted line represents the maximal value achieved during maximal cycling exercise test. VO2 = oxygen uptake; V'E = minute ventilation; LW = level walking; DW = downhill walking; DWL = downhill walking carrying load. # = p < 0.05 DW versus LW at iso-time; † = p < 0.05 DWL versus LW at iso-time.
Responses in physiological measurements during the walks.a

VO2: oxygen uptake; VE: ventilation; MVV: maximal voluntary ventilation; Borg: perceived effort; SpO2: oxyhaemoglobin saturation; Rest: immediately before the walks; Steady: values achieved during steady state walking; Pre: values collected prior the walks; Post: values collected immediately after the end of the walks; LW: level walking; DW: downhill walking; DWL: downhill walking carrying load; VCO2: carbon dioxide elimination; HR: heart rate.
aData are mean ± standard deviation or median [interquartile range].
bSignificant within-group difference, steady state versus rest (p < 0.05).
cSignificant between-group difference at steady state compared to LW (p < 0.05).
Discussion
Contractile muscle fatigue
This study demonstrates that DW induces greater quadriceps LFF than LW in individuals with COPD, as measured by both twitch contraction and a serum biomarker. The addition of an extra load around the chest does not appear to affect contractile muscle fatigue or markers of muscle damage. Conventional (flat) walking was associated with virtually no LFF. This is consistent with previous findings in this patient group. 30,31 Importantly, walking downhill proved to be both a reliable and an efficient means of inducing quadriceps fatigue, with 9 of 10 participants having detectable quadriceps LFF following a single 20-minute exercise session.
The observed difference in LFF between DW and LW may have been affected by gait pattern alterations in response to the negative incline. This study did not, however, seek to investigate biomechanical adaptations in people with COPD. DW is known to elicit greater eccentric quadriceps muscle contraction than walking on a flat surface in healthy individuals. 12,22,32 The repetitive eccentric contractions inherent in DW therefore predisposes to more muscle damage than concentric contractions. 8,9 This was indirectly confirmed in our study via elevated serum CK levels. 25,33 Our finding that LFF and elevated CK were observed during both DW conditions but not LW supports the hypothesis that a higher eccentric: concentric muscle contraction ratio occurs during DW in patients with COPD.
DWL had less of an effect on LFF than DW observed by a lack of statistical difference and moderate effect size when comparing the decrease in 15′ Twqpot between the two modalities. This result was not expected. Running on a decline carrying a load has been demonstrated to increase the eccentric: concentric muscle contraction ratio in young healthy subjects; 12 however, one cannot assume this applies equally to an elderly COPD population. For example, the self-selected walking speed, cardiopulmonary responses and muscle composition of young healthy subjects would differ considerably to older adults living with chronic health conditions. It is plausible that the effect observed following DW could be a potential ‘ceiling’ upon which further improvement may not be likely to occur. It is also possible that the addition of a chest load resulted in alterations to gait pattern (e.g. cadence and stride length) and/or muscle activation to reduce the quadriceps braking demand. Neither of these factors were monitored in this study. It is, however, useful for clinicians to know that LFF can be elicited during walking without the necessity of adding extra weight during a walking activity in individuals with COPD.
In order to verify the clinical feasibility of using DW as an exercise training modality, we investigated skeletal muscle soreness in the week after the session. Despite knowledge that high-intensity eccentric exercise may enhance delayed onset of muscle soreness, 25,26 we found no significant differences in the perception of muscle soreness and very low ratings of discomfort overall.
Cardiorespiratory cost of DW
Both DW and DWL elicited lower cardiorespiratory demands (VO2, VE and VE/maximum voluntary ventilation (MVV) than LW in this patient group. This finding is consistent with previous data in young and old healthy adults. 13,34,35 Our data confirm, in a small sample, that this phenomenon applies equally to individuals who are chronically ill and ventilatory limited. The mild reduction in cardiorespiratory cost of activity associated with DW and DWL is appealing for individuals with COPD as many are symptom (i.e. breathlessness) limited on exertion. 2,31 The clinical significance of the difference in absolute reduction of VE between the DW and LW interventions (approximately 5 L/min) is difficult to ascertain. The average difference of VE/MVV between the DW and LW interventions was, however, 10 ± 9%. One could speculate, for example, that individuals with more severe airways disease might derive greater benefit from DW than others.
Our data also compare favourably with previous research of the metabolic cost of exercise training modalities in COPD by Probst and colleagues. 23 In this study, the mean VO2 levels achieved during treadmill walking (set at 75% of mean 6MWT speed) were 0.98 ± 0.32 L/min. Interestingly, mean VO2 data from the DW and DWL interventions in our study compare similarly to the lower energy demands observed during cycling (0.92 ± 0.25 L/min) by Probst and colleagues. It is important to note that exercise intensities above 40% of VO2 reserve are recommended to develop or maintain physical fitness. 36 Our data showed that DW and DWL achieved 50% and 58% of VO2 reserve (60 and 64% of VO2 peak), respectively. Thus, despite eliciting a lower VO2 than LW, it would be appropriate to consider DW a suitable modality to achieve exercise of a moderate intensity.
One could speculate the metabolic work could have been larger in the downhill modalities as they were associated with higher CK and greater contractile muscle fatigue. This, however, would be difficult to couple with the lower cardiopulmonary stress and the fact that the mechanical work of DW compared to LW is clearly less. It is possible that higher CK levels and incidence of contractile muscle fatigue were due to the eccentric nature of muscle contraction. Finally, the lack of differences observed in the heart rate between the DW and LW corroborates with previous investigation. 37 Some mechanisms such as the decreased mechanical work required during eccentric contractions for a given oxygen uptake 38 and/or increased sympathetic modulation and fibre activation during eccentric contraction 39 may explain this uncoupling.
Safety considerations
To the best of the authors’ knowledge, this is the first study to investigate DW in individuals with COPD. Little is known about the potential side effects of DW apart from investigations of its effect on muscle damage and delayed onset of muscle soreness. 25,26,35,40 We had no reports of serious adverse events but tolerable discomfort was reported from participants in the hip (n = 1) and knee (n = 2) during DWL. One participant was excluded due to dyspnoea and oxygen desaturation after 5 minutes of the LW intervention. Another two participants required rests due to dyspnoea (one subject walked 16′10′′ of DWL; the other 16′ of LW), but both were able to complete 20 minutes of DW.
Limitations
Some methodological limitations may affect the implications arising from this study. The modest sample size should be considered when applying the findings to the broader COPD population, whilst the lack of a healthy, age-matched control group mean results cannot be extrapolated beyond a COPD context. However, as a large premise for investigating DW in COPD is the preservation of ventilatory function on exertion, this may be less important for a ‘normal’ population. Interestingly, our observed changes in metabolic data (e.g. VO2 and VE) were fundamentally similar to those of Gault and colleagues who investigated healthy subjects of similar age to our cohort. 35
We considered the increase in serum CK levels 24 hours after DW and DWL reflective of induced muscle damage (and LFF). This response following eccentric muscle contractions is well supported in the medical literature. 8,9,25,33 In the absence of a truly confirmatory muscle biopsy analysis, however, this biomarker may be more accurately considered a surrogate biomarker of LFF.
The walking protocols were implemented at one relative set speed, despite the potential for variability in cardiorespiratory and fatigue responses according to treadmill inclination, speed and intensity. 13,37 The responses to the protocols at different speeds would certainly be of some clinical value but was beyond the scope of this study. Importantly, our study design was able to verify LFF and cardiorespiratory demands in all three modalities. It is also possible that, despite a fixed speed across walking conditions, training load may have differed between test conditions. The recruitment and activation of different muscle groups may therefore have contributed to changes observed in the downhill conditions compared to LW. Whilst we did not measure muscle activity during the protocol, the combined increase in serum CK levels and reductions in quadriceps force (Twqpot) and cardiopulmonary responses suggest a true quadriceps fatiguing effect of DW.
Finally, it is possible that recurrent eccentric braking contractions, inherent in DW, may become a fatiguing stimulus that negatively impacts on skeletal muscle in COPD. From a LFF perspective, recovery can take hours to days, 4 meaning that inadequate recovery may be possible when performing repeated exercise training sessions over short time periods. This is not, however, supported by recent data from Burtin and colleagues 10 who demonstrated that individuals with COPD who developed fatigue during exercise training and completed three sessions per week improved more upon completion of the training programme than those who did not fatigue.
Future perspectives and clinical application
In individuals with COPD, the development of LFF after a single exercise session relates to significant improvements in exercise capacity and health-related quality of life following a physical exercise training programme. 10 This study demonstrates that DW elicits LFF more than conventional LW. These results therefore provide a clear rationale for further exploration into the potential effectiveness of rehabilitation incorporating a DW exercise modality. The intervention is simple to adapt into existing rehabilitation models and may prove important for individuals who do not usually fatigue during training. The effect of inducing skeletal muscle fatigue on exercise compliance must also be considered.
Conclusion
Individuals with COPD develop more quadriceps LFF during DW compared to LW and have lower cardiorespiratory costs. It is important for future investigations to evaluate the effectiveness of DW as part of a comprehensive rehabilitation programme on broader, important health outcomes.
Footnotes
Acknowledgements
The authors would like to thank physiotherapists Ilse Muylaert, Iris Coosemans, Veronica Barbier and the whole staff of the Respiratory Rehabilitation Department and the Pulmonary Function Department at the University Hospitals Gasthuisberg, Leuven, Belgium, for helping with the logistics and execution of the procedures in this study. We also would like to thank Patricia Besem, Rob Verlinden and Carthy L Aguillon for helping with the data collection. Finally, we thank Maarten Spruyt and Willem Dewit for the help with blood samples.
Funding
This work was supported by the Flemish Research Foundation (G.0871.13) and PROactive IMI-JU (115011). CAC is funded by The National Council for Scientific and Technological Development (CNPq), Brazil (202425/2011-8). CO is the recipient of a European Respiratory Society Fellowship (LTRF 2014 – 3132).
