Abstract
As life expectancy of patients with Duchenne muscular dystrophy (DMD) has increased to the 5th decade, in part due to improved ventilatory support, cardiomyopathy is projected to increase as a cause of death. International guidelines recommend an annual assessment of cardiac function and initiation of appropriate pharmacological treatment. We conducted an audit of the cardiac management in patients with DMD requiring ventilatory support and reported a case series of the collated cardiac investigations. Patients with DMD requiring ventilatory support were included in the study. The date of the last electrocardiogram (ECG), echocardiogram (ECHO), cardiology review and pharmacological management were retrieved from the medical records. If an annual cardiac assessment had not been performed this was requested and the latest ECGs and ECHO reports were collated. A total of 30 patients with DMD (29 males, mean (SD) age of 30 (7) years) met the inclusion criteria. Although there was ECG and ECHO documentation in 24 and 21 individuals, respectively, it was only recent in 10 and 6 individuals. In all, 60% of patients had been assessed by a cardiologist, but only 10% within the last year. Over half of the patients failed to attend their new appointments. From the available results, 18 of the 19 patients had an abnormal ECG, 11 of the 16 patients had left ventricular (LV) impairment and 55% of patients had a change in prescription following cardiac investigations. There is a need for a coordinated cardiorespiratory approach towards adult patients with DMD. Over a third of patients had normal LV function suggesting that cardiomyopathy is not inevitable in this group.
Keywords
Introduction
Duchenne muscular dystrophy (DMD) is an X-linked disease, characterized by progressive muscle weakness, affecting approximately 1 in 3500 male births. 1 The disease is caused by a mutation in the gene that codes for dystrophin, a cytoskeletal protein that is responsible for the structural integrity of the sarcolemmal membrane in myocytes and cardiomyocytes. 2 DMD is typically diagnosed in childhood and most require a wheelchair by 12 years of age. 3 Respiratory function declines in the 2nd decade of life and until recently, most patients died in their early 20s as a result of ventilatory failure. 3 However, with advances in care, including increased ventilatory support, patients are currently living into their 4th and 5th decades. 4 –6 As these patients live longer, the cardiac complications of DMD are emerging as a major cause of morbidity and mortality. 5
Cardiac involvement in DMD includes most commonly dilated cardiomyopathy (including a dilated left ventricle and a reduced ejection fraction on echocardiography) but also hypertrophic cardiomyopathy and conduction abnormalities. 7 The most common abnormalities found on electrocardiography include tachyarrhythmia, tall R waves or R/S ratio >1 in leads V1–V3, large Q waves in lateral leads, complete or incomplete left bundle branch block (LBBB) and complete right bundle branch block (RBBB). 8,9 Several studies have found that nearly all patients with DMD develop cardiomyopathy, although the age of onset varies considerably. 7,10,11 The structural evidence of cardiomyopathy precedes symptoms by years. 7 Furthermore, it is difficult to differentiate the signs and symptoms of heart failure, from respiratory complications of DMD compounded by the severe restriction in activity level. Pharmacological treatment, including angiotensin-converting enzyme (ACE) inhibitors with or without β blockers, for asymptomatic and symptomatic cardiomyopathy in DMD reduces both morbidity and mortality. 10,12 –14 ‘Failure to see a cardiac specialist until late in the disease, after clinical manifestations of cardiac dysfunction are evident, have led to late treatment and poor outcomes’. 15 Regular assessment is thus necessary for early detection of asymptomatic cardiomyopathy in patients with DMD. 16
Much of the existing data regarding prevalence and severity of cardiomyopathy was before ventilatory support was common for this group of patients, 7 and the literature regarding the pharmacological management of cardiomyopathy in DMD involves predominantly children and teenagers. 10,12 –14 Patients are now living into their 4th and 5th decades but to date there is little information about this group. One recent observational study, involving teenagers and young adults (mean age of 22 years), requiring ventilatory support reported a lower prevalence and progression of cardiomyopathy compared to other studies 17 raising a question regarding the common assumption that it was almost inevitable.
In 2010, the growing acknowledgement of the need for a coordinated multidisciplinary approach for these patients prompted an international group, the DMD Care Considerations Working Group, to develop standardized care recommendations for the comprehensive management of DMD. 15,18 For the cardiac management of DMD, it was recommended that a cardiac specialist be included in the team and there be an annual assessment of cardiac function including an electrocardiogram (ECG) and an echocardiogram (ECHO). 15 The guidelines for heart failure should be followed for pharmacological treatment. 15
The follow-up of adult patients with DMD varies considerably. Whilst it was not uncommon for patients to continue being supervised by their paediatric team as they became young adults, they are now often transitioned to a respiratory physician for monitoring and the initiation or maintenance of ventilatory support. As patients live longer and with improved management of respiratory failure, the presence of cardiac pathology is of increasing importance.
As part of a centre-wide best practice, quality assurance initiative, we conducted an audit of the cardiac management in patients with DMD under the care of a unit established for ventilator-assisted individuals and compared it against the recently published international guidelines. 15,18 We report the collated results of the latest annual cardiac assessments to estimate the prevalence and severity of cardiomyopathy in adults with DMD, many in their 4th to 5th decades who may not have been represented in previous studies.
Methods
Study design
An audit was conducted followed by a review of the cardiac investigations as a case series.
Participants
All ventilator-assisted individuals with DMD who had respiratory follow-up within the last 3 years at West Park Healthcare Centre (WPHC), Toronto, Ontario, Canada, and were alive at the time of the audit were included. They were identified by the respiratory therapist manager. The study was approved by the local research ethics board.
Audit
The gold standard for this audit was the standardized care recommendations for the comprehensive management of DMD developed by the international DMD Care Considerations Working Group. 15,18 The guidelines advise that a cardiac specialist is part of the team and an annual assessment of cardiac function after the age of 10 or the onset of cardiac symptoms 15 is performed. If the cardiac assessment reveals abnormalities of ventricular function, cardiac function should be assessed every 6 months and pharmacological treatment should be started. 15 The American Heart Association Guidelines for the management of heart failure recommend the use of ACE inhibitors and β blockers in all patients who have evidence of structural heart disease such as reduced left ventricular (LV) ejection fraction (LVEF), even if they are asymptomatic. 19
The medical records of patients that fulfilled the inclusion criteria were reviewed. Patient demographics (age, gender, age of diagnosis, age at which wheelchair was required, inpatient/outpatient status, duration of care at WPHC and co-morbid conditions) and ventilatory status (type of ventilation, type of interface, and duration of assisted ventilation) were recorded. Cardiac management included dates of ECGs, ECHOs and cardiology referrals. Current and previous use of cardiac medications such as ACE inhibitors, angiotensin receptor blockers (ARBs), β blockers, or spironolactone, and corticosteroids were recorded.
Protocol for cardiac management
A clinical management protocol was subsequently developed which recommends an annual cardiac assessment including an ECG and ECHO, for patients with DMD, in line with current guidelines. Patients identified from the initial audit whose cardiac management was not in accordance with the guidelines were scheduled for cardiac investigations after discussion with the attending respirologist.
Case series
The latest annual ECGs, ECHO reports and pharmacological management were reviewed and collated. ECGs were assessed for conduction abnormalities, arrhythmias, and criteria for left ventricular and right ventricular hypertrophy. Transthoracic ECHOs were interpreted by cardiologists experienced in echocardiography. LV and RV and functions were assessed according to standard methodologies. 20 Where possible, an estimate of LVEF or systolic functional grade was provided. 20 The estimated right ventricular systolic pressure was calculated when sufficient tricuspid regurgitation was present; right atrial pressure was assumed to be 10 mmHg. 21 ECHOs were examined specifically for findings of dilated cardiomyopathy including reduced LVEF, increased end diastolic LV volume and regional wall motion abnormalities. They were also examined for evidence of hypertrophy and diastolic dysfunction including increased LV wall thickness and reduced end diastolic LV volume. For the purposes of this study, normal LV function was defined as an LVEF >60%, mild LV dysfunction corresponded to an LVEF of 40–59%, moderate dysfunction with an LVEF of 20–39% and severe dysfunction with an LVEF of <20%.
Statistical analysis
Results are given as either frequency or mean ± standard deviation for continuous variables. Comparison of age and duration of ventilation between individuals with and without LV dysfunction was made using χ 2 test. The value of p < 0.05 was considered to be statistically significant. Statistical Package for Social Sciences version 20 (SPSS Inc., Chicago, Illinois, USA) software was used.
Results
Audit
The demographics of 30 patients who met the audit inclusion criteria are shown in Table 1.
Patient demographics.
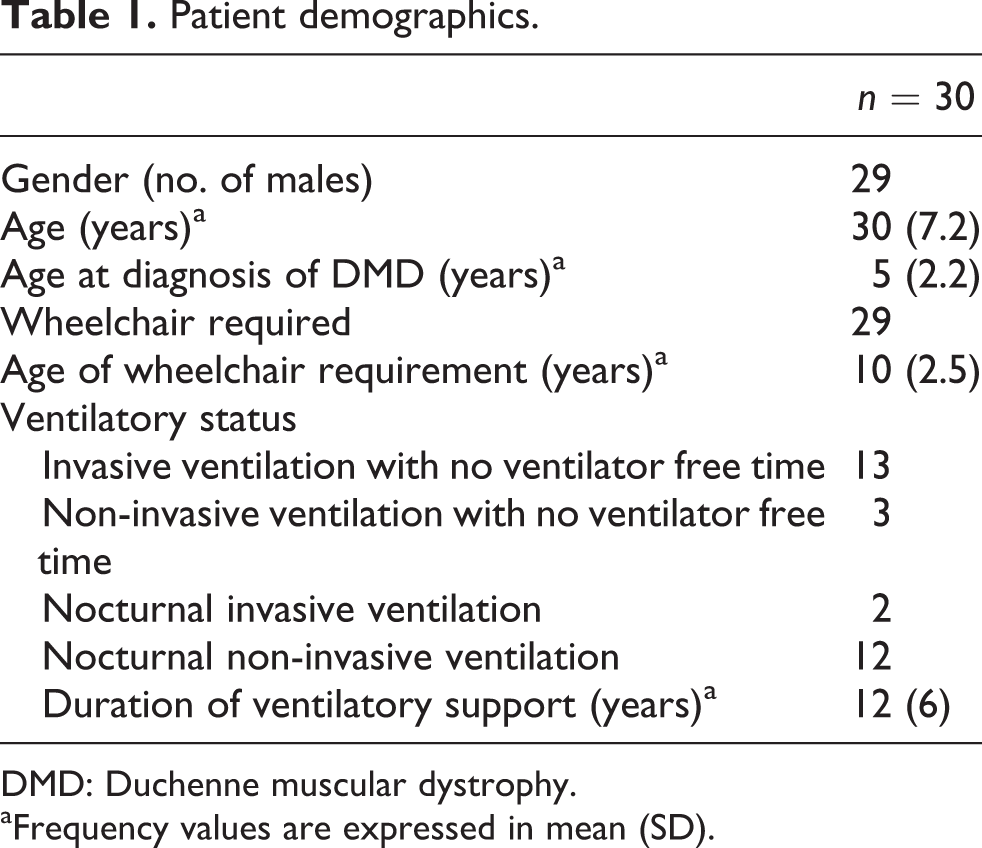
DMD: Duchenne muscular dystrophy.
aFrequency values are expressed in mean (SD).
In 24 of the 30 and 21 of the 30 patients, there was previous documentation of an ECG and ECHO, respectively. However, only 10 of the 30 and 6 of the 30 patients had a documented ECG and ECHO, respectively, within the year preceding the time of the audit. Of the 30 individuals, 18 had previously been assessed by a cardiologist; 16 of the 30 patients were receiving pharmacological treatment, at the time of the audit, for cardiomyopathy; of these 16 patients, 8 were on both an ACE inhibitor and β blocker, 6 were on an ACE inhibitor only, 1 was on a β blocker only and 1 was on spironolactone. Of the remaining 14/30 patients not receiving pharmacological treatment at the time of the audit, 2 had received pharmacological treatment in the past, but the reason for cessation was unclear. Eight of the 30 patients were currently on Deflazacort.
Case series
Of the initial 30 patients identified in the audit, 20 and 24 patients needed an updated ECG and ECHO, respectively, as they had not had these investigations within the last year. Cardiology appointments were organized for all the patients but there was a low uptake despite a verbal explanation by a respirologist. Updated ECGs were obtained for 9 of the 20 patients and updated ECHOs were obtained for 10 of the 24 patients, see Figure 1 for details. Between the audit and the development of the protocol, three patients died (all three had had recent ECGs and two had had recent ECHOs) and one transferred care.

Flow chart of the cardiac investigations requested. The shaded boxes represent the investigations that were collated for the case series.
ECG and ECHO findings
Abnormal ECGs were reported in 18 of the 19 patients (Table 2).Of the16 ECHO results available, 5 showed no LV dysfunction, 5 mild LV dysfunction, 5 moderate LV dysfunction and 1 severe LV dysfunction (Table 3). Additionally, 10 of the 16 ECHOs were noted to be technically difficult. There was no difference in mean (SD) age (27 [7] vs. 32 [9] years, p = 0.25) or duration of assisted ventilation (9.8 [6.4] vs. 10.9 [6.9] years, p = 0.32) between 5 patients with normal LV function and 11 patients with reduced LV function. One of the five patients with normal LV function was female.
Frequency of abnormalities detected by electrocardiography.

LVH: left ventricular hypertrophy; RVH: right ventricular hypertrophy; RBBB: right bundle branch block; LAFB: left anterior fascicular block; IVCD: interventricular conduction delay; ECG: electrocardiograph.
Frequency of abnormalities detected by echocardiography.

LVH: left ventricular hypertrophy; LV: left ventricular; RV: right ventricle.
Pharmacological management
Table 4 shows the originally prescribed medication for the 16 patients who had a recent ECHO according to severity of LV function. Of the 11 patients, 6 with abnormal LV function were prescribed new medication and 3 had their medication increased as a result of the updated cardiac investigations after the audit.
Pharmacotherapy according to severity of LV dysfunction.

ACE: angiotensin-converting enzyme; ARB: angiotensin receptor blocker; LV: left ventricular.
Discussion
In this study, the cardiac management of patients with DMD under the care of a local respiratory unit specializing in mechanical ventilation was audited against recent international guidelines, 15,18 and a new policy of annual cardiac investigations was implemented. Although undertaken as part of a centre-wide quality assurance initiative in best practice, we believe that the results have general clinical messages of importance to a wider audience. The mean age (30 years) and duration of ventilatory support (12 years) in the current study highlights the improvement in life expectancy of DMD patients with appropriate respiratory care, supporting the need to focus more closely on other co-morbidities such as cardiomyopathy. Several studies have demonstrated the importance of initiating pharmacotherapy for cardiomyopathy prior to the onset of symptoms, in order to improve the morbidity and mortality of DMD. 10,12 –14 We found that although cardiac assessment had been performed in over two-thirds of patients with DMD, few were currently undergoing annual assessments and the treatment regime could be improved in the majority, in line with the new international guidelines. 15
The ECG abnormalities identified, such as conduction defects and signs consistent with hypertrophic or dilated cardiomyopathy, were consistent with the existing literature. 7,9 The most common ECG abnormalities previously reported include rather than included tachyarrhythmia, tall R waves or R/S ratio >1 in leads V1–V3, large Q waves in lateral leads, complete or incomplete LBBB and complete RBBB. 8,9 The observation in our series was that that 42% of patients had dominant R waves in V1 and that lateral or inferior Q waves were particularly common representing ‘scarring’ consistent with the description of Spurney who noted frequent scarring in the posterobasal and lateral sections of the left ventricle in the dystrophic myocardium. 8 This is thought to be due to the absence of dystrophin ‘causing a pathological cycle of infiltrating inflammatory cells and fibroblasts causing myocardial cell death and fibrosis’. Only one of the ECGs (5%) was normal in our population.
Although over two-thirds of our patients had echocardiographical evidence of cardiomyopathy, a substantial proportion were not receiving optimal pharmacological management. However, most of them were receiving a single agent. It has recently been established that pharmacotherapy is beneficial in children and teenagers with LV systolic dysfunction. 10,12 –14 A randomized controlled trial of children with DMD between the ages of 9 and 13 demonstrated that early administration of an ACE inhibitor, prior to the onset of LVEF decline, reduced the onset and progression of LV dysfunction at 5 years. 14 Ten-year follow-up showed a mortality benefit of early ACE inhibitor treatment. 12 In an observational study, Viollet et al. found that in patients with DMD, ranging in age from 7–27 years LV function declined without treatment during 6–52 months of surveillance and both ACE inhibitors alone or in combination with β blockers improved LV function. 10 This is consistent with an observational study of teenagers and young adults (mean age 22 years) receiving ventilatory support 17 in whom no deterioration in LV function over 3 years was observed amongst patients with reduced LV function on pharmacological treatment (ACE inhibitors or ARB). 17
In contrast to clinical reports in which nearly all adult patients with DMD develop cardiomyopathy, 7,10,22 31% (5 of the 16) of our DMD patients were found to have normal LV function (LVEF >60%) despite their being older (mean age 30 (7) years). This is supported by the data from Kwon el al. who noted amongst 15 of the 31 young adults (mean age 22 [5] years) receiving ventilatory support that the LVEF was >50%. 17 It is therefore conceivable that ventilatory support delays the progression of cardiomyopathy especially since respiratory failure contributes to the cardiac morbidity and mortality in this population. 23,24 Our data add to this emerging literature showing that cardiomyopathy, previously described as inevitable, 10 may not be universal even in a population with a mean age in the 4th decade. Steroid therapy is also associated with a reduction in mortality and the onset and progression of cardiomyopathy. 25 Only eight patients in our study were currently on corticosteroids.
Chest wall deformities such as scoliosis can make echocardiography more difficult 8 and were noted by the technician in over half of our studies performed. More sensitive measurement techniques such as cardiac magnetic resonance (CMR) imaging have been used in children with DMD, 8 but in addition to the greater cost, this technique may not be practical for ventilated patients who would be required to lie flat. However, despite the clinical challenges, CMR imaging has been shown to be sensitive for early findings of cardiac disease prior to systolic dysfunction including reduced myocardial circumferential strain and areas of fibrosis identified with the use of late gadolinium enhancement. 26 –28 CMR has also been used to characterize the pattern of remodelling and changes in LV geometry that occur with cardiac disease in DMD as a unique process not fully explained by the traditional thinking of progressive dilated cardiomyopathy. 29 CMR is not currently included in clinical guidelines for DMD, but it is a promising area of research to further understand the development and progression of cardiac disease in this population. Other measures, such as multigated cardiac radionuclide ventriculography, have been used and compared with a combination of echocardiography and brain natriuretic peptide, but detected rates of LV dysfunction were similar. 30
There are multi-professional guidelines to be considered for patients with DMD, and this audit highlights the gap between the multidisciplinary approach that occurs in paediatric centres and the situation for patients who have transitioned to the care of an adult respirologist. Respiratory physicians need to be aware of the importance of cardiomyopathy in this population and the new international guidelines developed to address it 15,18 especially if they are the only specialist involved.
Limitations
We report an audit from a single centre, with modest patient numbers. In the absence of a cardiac protocol there is no reason to think that the same findings would not occur in larger centres. Our patient population included only patients who required ventilator support; however, cardiomyopathy should be considered independently of the need for ventilation. The compliance rate with the offered cardiac assessments was low reducing the numbers in the case series further. However, this highlighted the difficulty this patient population has in attending health care appointments. These assessments were made outside of their usual respiratory appointment and over half failed to attend despite a verbal discussion with a respiratory physician explaining the purpose of the appointment. A ‘one-stop’ multidisciplinary annual clinic may be more desirable for patients and their carers.
Conclusion
This report highlights the need for the implementation of international guidelines in clinical care settings. Many individuals with DMD now survive to adulthood so new strategies are needed to reflect this. As care of the paediatric population transitions to specialist care of an adult population, supervised primarily by a respiratory physician, inclusion of a cardiologist preferably at the time of a clinic respiratory follow-up would hopefully promote improved cardiac care. This study adds to the emerging evidence that in patients with long-standing DMD, who have adequate ventilation, cardiac dysfunction is not universal. Large studies are needed to understand the natural history of cardiac involvement in the ventilated DMD population.
Footnotes
Funding
RE was supported by a Post-Doctoral Fellowship for Exercise Rehabilitation by West Park Foundation and a National Institute for Health Research (NIHR) Clinical Lectureship in Respiratory Medicine, UK. RG was supported by the National Sanatorium Chair for Pulmonary Rehabilitation and by West Park Foundation
