Abstract
It has been well defined that obesity is strongly linked with several respiratory symptoms and diseases, but no convincing evidence has been provided for chronic obstructive pulmonary disease (COPD). In the current study, we aim to assess the possible prevalence of obesity in patients with COPD in a cross-sectional case–control study of individuals from the region of Stara Zagora, Bulgaria, and to explore whether the body mass has some effect on the lung function of COPD patients. The study included 158 patients with COPD (Global Initiative for Chronic Obstructive Lung Disease (GOLD) II, III, and IV stages) and 123 individuals unaffected by the disease (control). A higher frequency of obesity compared to the controls (20.3%) was observed in patients with COPD (29.1%, p = 0.093), especially in those with GOLD II stage (37.7%, p = 0.009). Prevalence of obesity was highest in COPD GOLD II, followed by GOLD III and IV stages (p = 0.068). When diabetes was considered as confounding factor, we found a significant prevalence of obesity in COPD patients than the controls with diabetes (p = 0.031). Interestingly, there was a statistically significant moderate positive correlation between the body mass index and forced expiratory volume in one second as a percentage of predicted value in the whole patients’ group (R = 0.295, p = 0.0002) as well as in the subgroups of GOLD II (R = 0.257, p = 0.024) and GOLD III COPD (R = 0.259, p = 0.031).The results of our study propose that the increased body mass, particularly obesity is frequent comorbidity to COPD, especially to less severe diseases. Moreover, the results suggest that the higher body weight may provide some protection against the impairment of lung functions in patients with stable COPD.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is a preventable and treatable disease, which is characterized by airway limitation that is not fully reversible. 1,2 COPD is a disease with systemic comorbid conditions including hypertension, diabetes mellitus, and cardiovascular diseases due to systemic inflammation. Chronic comorbidities affect health outcomes in patients with COPD, including mortality 3,4
In the recent decades, obesity has significantly increased in society reaching epidemic proportions. 5,6 Based on the criteria of the World Health Organization, body mass index (BMI—the individual’s body mass divided by the square of the height) can determines the following states: very severe underweight is indicated as less than 15.0 kg/m2, severe underweight is from 15.0 to 16.0 kg/m2, underweight is from 16.0 to 18.49 kg/m2, normal is from 18.5 to 24.99 kg/m2, overweight is from 25 to 29.99 kg/m2, and obese is over 30 kg/m2. 7
Obesity is a complex metabolic condition that influences many physiological systems. It has been well defined that there is a close relationship between obesity and cardiovascular and metabolic diseases, osteoarthritis, and several respiratory symptoms and diseases such as obstructive sleep apnea syndrome, obesity hypoventilation syndrome, asthma, and pulmonary embolism. 8,9 Since obese patients experience difficulty in breathing, the problem has long been the object of many studies. 2 Obesity prevents normal breathing, by squeezing the chest and thus reducing the volume of the lungs, and on the other hand, it interferes with the normal function of the respiratory muscles. 8
With regard to COPD, to date, some associations with obesity have been reported. 10,11 In a national survey in Korea, Park et al. reported a borderline significant association between COPD and abdominal obesity, 11 whereas Joo et al. in another population-based study for comorbidities of COPD reported that being underweight might be more prevalent in Koreans with COPD than without COPD. 10 In addition, there are some limited data that low BMI increases the risk of COPD in men, 12,13 however, there are also some studies with controversial conclusions. 13–15 On the other hand, patients in the early stages of COPD (Global Initiative for Chronic Obstructive lung Disease (GOLD) I and II stages) often are overweight, unlike those with GOLD III and IV stages, who had lower values of BMI. 9 Weight loss in more advance COPD stages may be due to different factors such as enhanced oxidative stress and increased production of proinflammatory cytokines like tumor necrosis factor-α, interleukin-6, and so on. 16
In the current study, we aim to assess the possible prevalence of obesity in patients with COPD in a cross-sectional case–control study of individuals from the region of Stara Zagora, Bulgaria, and to explore whether the body mass has some effect on the lung function of COPD patients.
Patients and methods
Patients and control group
In the current cross-sectional case–control study, 158 patients with COPD and 123 control individuals were included. The patients were recruited in the Clinic of Internal Medicine, University Hospital, Medical Faculty, Trakia University, Stara Zagora, Bulgaria, by specialists in pulmonology. After a visit to the general practitioner (family doctors), patients with suspicion for chronic lung diseases are sent to the specialist in pulmonology, who set the diagnosis of the diseases, including the COPD. Few of the patients were determined as suspected for COPD during an initial screening program and were further examined and diagnosed by a specialist pulmonologist. The inclusion criteria for COPD were as follows: older than 40 years; forced expiratory volume in one second (FEV1) as a percentage of predicted value (FEV1% predicted) of < 80%; FEV1–forced vital capacity (FEV1/FVC) ratio of ≤ 0.70; FEV1 reversibility value of < 0.12 after inhalation of 400 µg Salbutamol.
The control group consisted of 123 individuals unaffected from COPD from the region of Stara Zagora, Bulgaria. Control group individuals are aged between 26 and 80 years (median of 57 years).
Based on the criteria of the World Health Organization, according to the BMI, all patients with COPD and controls were categorized mainly into the following four groups: (i) individuals with underweight, (ii) with normal weight, (iii) with overweight, and (iv) with obesity. 7
This work was approved by the Ethic Committee of Medical Faculty, Trakia University, Stara Zagora, Bulgaria. Informed consent was obtained from all patients and controls.
Spirometric analysis
Patients and controls underwent fast spirometry according to the method of Quanjer 17 with a spirometer (Schiller-SP1, Baar, Switzerland). Lung function values for each participant were compared with the predicted normal values of Quanjer et al. 18 Lung function was defined by an FEV1 as a percentage of predicted value and the ratio of FEV1to the forced vital capacity (FVC) (FEV1/FVC). Normal values were considered as those between 80 and 120% for FEV1% predicted and more than 0.70 for FEV1/FVC 17–19
Statistical analyses
Statistical analyses were performed using StatView v.4.53. (Abacus Concepts, Inc., Berkeley, CA, USA) and Statistical Package for the Social Sciences (SPSS) 16.0 for Windows (SPSS Inc., Chicago, IL, USA). Continuous variables were analyzed for normality by Kolmogorov–Smirnov and Shapiro–Wilk’s tests. The Student’s t test and analysis of variance test were applied for comparing the continuous variables with normal distribution in two or more independent groups, respectively, whereas the Mann–Whitney U test and Kruskal–Wallis test were applied for continuous variables with non-normal distribution. The frequencies of distribution of categorical variables in contingency tables were analyzed using χ 2 test and Fisher’s exact test. Correlations were performed by Pearson or Spearman’s test depending on the normality of the continuous variables. The prevalence ratio 20 and 95% confidence intervals were calculated via an interactive calculator for two-way contingency table analysis (http://statpages.org/ctab2x2.html) based on the statistics described by Rosner. 21 The results were confirmed by manual calculations according to the formulas described by Sullivan et al. 22
Results
The available demographic and clinical data of patients with COPD and of controls recruited in the current cross-sectional case–control study are presented in Table 1.
Demographic and clinical data of COPD patients and controls.
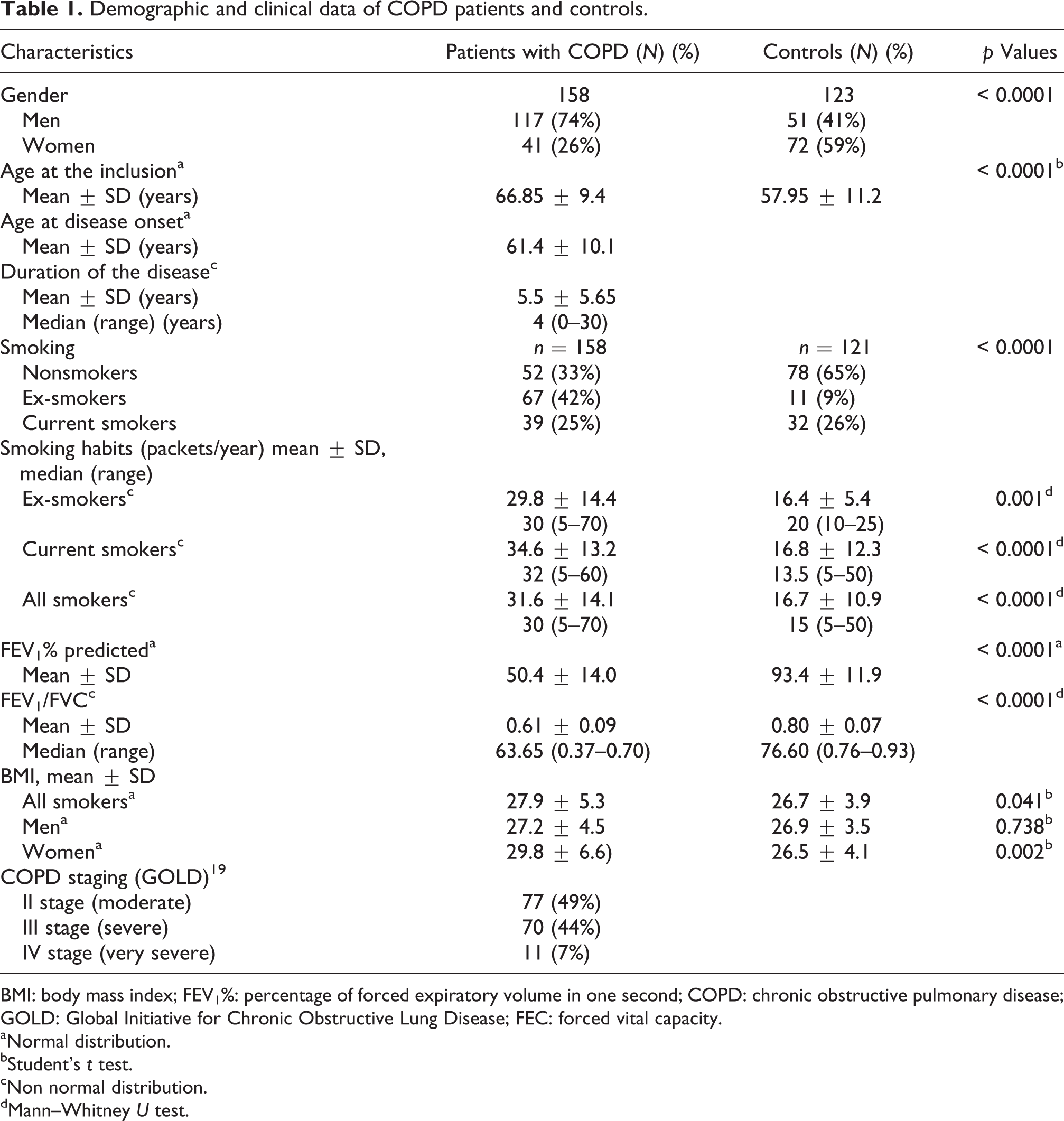
BMI: body mass index; FEV1%: percentage of forced expiratory volume in one second; COPD: chronic obstructive pulmonary disease; GOLD: Global Initiative for Chronic Obstructive Lung Disease; FEC: forced vital capacity.
aNormal distribution.
bStudent’s t test.
cNon normal distribution.
dMann–Whitney U test.
The particular figures and percentages of COPD patients and controls categorized mainly into four groups in accordance with their BMI are presented in Table 2. There was no statistically significant difference in the frequencies of each of the body mass groups between the controls and patients (p = 0.160). However, there was a marginal tendency for higher frequency of obesity in patients with COPD compared with controls (p = 0.059, Table 2). When controls and patients were dichotomized into obese and nonobese groups, that is, BMI over 30 kg/m2, we found that COPD patients were 1.22 times more likely to be obese patients than controls (p = 0.093, Table 2). The observed associations were highly significant (p = 0.007) when only patients with early COPD stage were analyzed (GOLD II COPD) (Table 3). Thus, patients with GOLD II COPD were 1.84 times more likely to be obese than controls (Table 3).
Frequency distribution of all COPD patients and controls according to WHO weight groups.
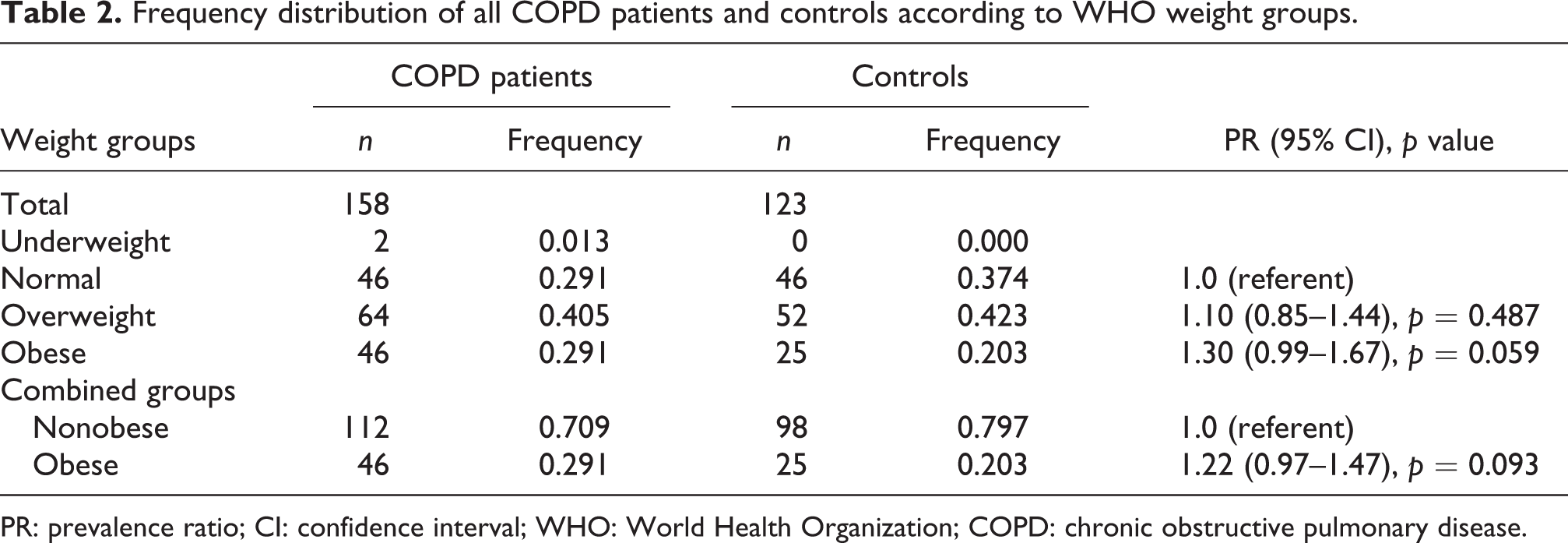
PR: prevalence ratio; CI: confidence interval; WHO: World Health Organization; COPD: chronic obstructive pulmonary disease.
Frequency distribution of COPD GOLD II patients and controls according to WHO weight groups.
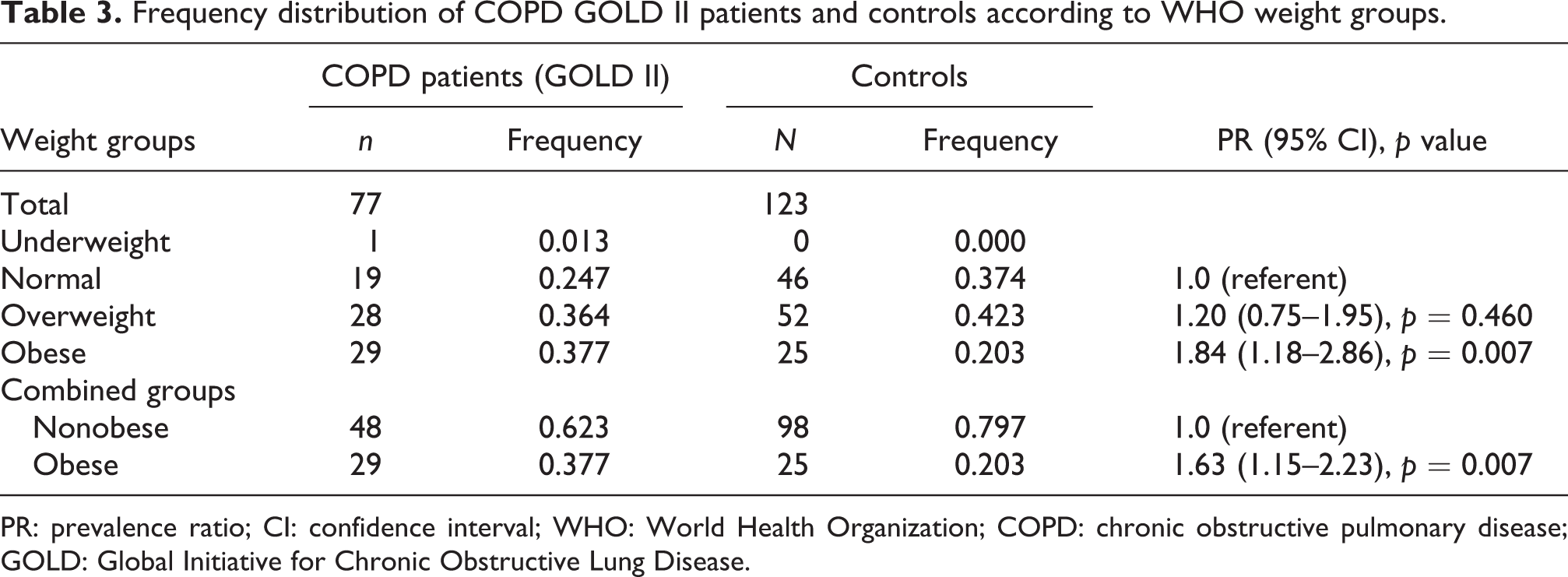
PR: prevalence ratio; CI: confidence interval; WHO: World Health Organization; COPD: chronic obstructive pulmonary disease; GOLD: Global Initiative for Chronic Obstructive Lung Disease.
Considering that diabetes is frequently associated with obesity and often exists as a comorbidity condition to COPD, we analyzed the proportion of obesity in control and patient groups according to the presence of diabetes. Of all the participants, 25 patients (16%) and 47 controls (38%) reported to have diabetes. We found that patients with diabetes were more often obese than controls with diabetes (p = 0.031, Fisher’s exact test) (Figure 1), whereas there was no statistically significant difference in the frequency of obesity in the subgroups without that disease (p = 0.284, χ 2 test; Figure 1).

Proportion of obesity in controls and COPD patients with and without diabetes. COPD: chronic obstructive pulmonary disease.
When body weight was analyzed in different stages of COPD, we found a tendency (p = 0.068) for smaller proportion of obesity in very severe COPD (GOLD IV: 2 of 11, 18.2%) than in severe (GOLD III: 15 of 70, 21.4%) and especially than in the moderate COPD (GOLD II: 29 of 77, 37.7%; Figure 2). The difference in the frequency of obesity between stages II and IV was relatively high (19.5%), but possibly due to the small number of patients with stage GOLD IV (n = 11), there was no statistical significance. Statistical significance was detected only for the difference of the frequency of obesity (16.3%) between stages II and III (p = 0.032).
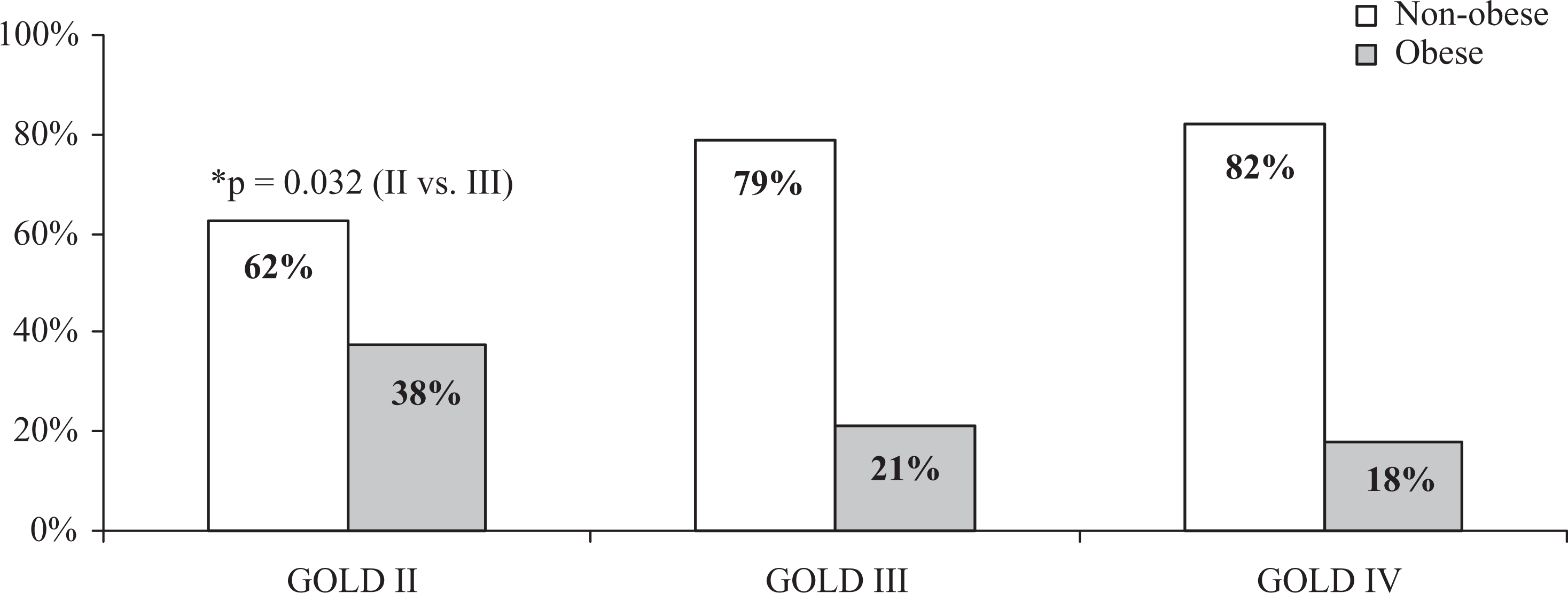
Proportion of obesity in different stages of COPD. COPD: chronic obstructive pulmonary disease.
Interestingly, there was moderate but highly statistically significant positive correlation between the BMI and FEV1% predicted in all COPD patients (R = 0.295, p = 0.0002, Pearson’s correlation; Figure 3(a)). The same types of moderate, but significantly positive correlations were observed in the subgroups of patients with GOLD II (R = 0.257, p = 0.024, Pearson’s correlation), and GOLD III COPD (R = 0.259, p = 0.031, Pearson’s correlation) (Figure 3(b) and (c)). Only in the subgroup of GOLD IV COPD patients, the statistical significance was lost, possibly due to the small number of patients of this subgroup (Figure 3(d)).

Correlation between the BMI and FEV1% predicted in all patients with COPD (a) and in the subgroups of GODL II (b), GOLD III (c) and GOLD IV COPD (d). (Pearson correlation test). BMI: body mass index; FEV1%: percentage of forced expiratory volume in one second; COPD: chronic obstructive pulmonary disease; GOLD: Global Initiative for Chronic Obstructive Lung Disease.
Discussion
The results of our study showed that individuals with COPD were 1.30–1.84 times more likely to be obese than non-COPD individuals, especially when they also suffered from diabetes. From the results of our cross-sectional case–control study, we are not able to conclude about the role of obesity as risk factor for the development of COPD; however, we may suggest that obesity is an often comorbidity to COPD. Although several previous studies have reported that increased BMI is negatively associated with COPD, 12,13 at least two studies have obtained results similar to ours. Thus, in a large population-based cross-sectional study in France including 121,965 individuals, a statistical association has been found between the abdominal obesity and airway obstruction. 14 In addition, obesity was linked to respiratory symptoms as dyspnea on exertion and productive cough as well as with decreased functional capacity in adults with newly diagnosed COPD. 5
In our study, we also observed a prevalence of obesity in moderate (GOLD II) compared to severe (GOLD III) and very severe (GOLD IV) COPD patients. Similarly, Steuten et al. have also previously reported a prevalence of obesity in GOLD stages I and II and high frequency of the low body weight in GOLD stage IV. 23 Several retrospective and prospective studies in different COPD populations have provided evidence that the low body weight is associated with higher mortality. 23,24 Analogously, Lainscak et al. have also reported significant association of body weight with the survival of the patients with COPD, as the lowest mortality was found for patients with BMI between 25.09 and 29.05 kg/m2 (overweight), whereas the highest rate of mortality and shorter survival had patients with lowest BMI. 25 The same research group has also observed that the FEV1 (in milliliters) increased significantly along the increase in body weight. In our study, we also observed a highly statistical significant positive correlation between BMI and FEV1% independently on the stage of the disease. Similar findings have been reported by Lam et al. 15 They have observed significantly higher proportion of overweight (≥ 25.0 kg/m 2 ) in Chinese men and women with normal lung function (30.2 and 33.5%, respectively) as compared to men and women with airflow obstruction (15.6 and 27.7%, p < 0.001), whereas the underweight was more often among individuals with airflow obstruction (12.5% vs. 4.3% for men and 9.0% vs. 3.8% for women). 15 This might be explained with the process of loosing of respiratory muscle performance due to skeletal muscle weakness, which is one of the main systemic effects of COPD. 26,27 Skeletal muscle weakness is associated with slow but substantial loss of muscle mass that can be found during ageing. 26 The analyses performed in a variety of studies have shown that there are significant alterations of skeletal muscle function and structure in COPD patients, as reduction in skeletal muscle tissue, decrease in muscle fatty acid oxidation capacity, changes in muscle types from type 1 (slow twitch) to type 2 (fast twitch). 28 There are data that the skeletal muscle atrophy is related to respiratory function, exercise intolerance, health status, mortality, and health-care resource utilization. 26,29
In conclusion, the results of our study propose that the increased body mass, particularly obesity, is frequent comorbidity to COPD, especially to less severe diseases. Moreover, the results suggest that the higher body mass may provide some protection against the impairment of lung functions in patients with stable COPD. However, due to several limitations of our study as cross-sectional but not longitudinal, we are not able to draw confidant conclusions about the role of obesity as risk factor for the development of COPD and impairment of the lung functions. In this respect, a larger longitudinal study is warranted to be conducted.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
