Abstract
Purpose
Combination of Rivaroxaban plus Aspirin improved cardiovascular outcome in patients with stable cardiovascular disease. The aim was to determine if Rivaroxaban and acetylsalicylic acid alone or in combination may protect mitochondrial mitophagy in human coronary artery endothelial cells (HCAEC) exposed to D-glucose.
Methods
HCAEC were incubated under different conditions: 5 mmol/L glucose D-glucose (control), 30 mmol/L D-Glucose with and without 50 nmol/L Rivaroxaban (Rivaroxaban), 0.33 mmol/L ASA (ASA) or Rivaroxaban (12.5 nmol/L)+ASA (0.33 mmol/L; (Riva+ASA).
Results
HCAEC incubated with D-glucose showed an increased Factor Xa expression. The mitochondrial content of Pink-1 and Parkin were significantly reduced in high glucose-incubated HCAEC compared to control. Rivaroxaban+ASA significantly increased the mitochondrial content of Pink-1 and Parkin, and the mitochondrial membrane potential compared to D-Glucose group. Both ASA alone and Riva+ASA reduced reactive oxygen species (ROS) and tissue factor production induced by high glucose exposure.
Conclusion
Under high glucose condition combining Rivaroxaban+ASA increased the mitochondrial content of Pink-1 and Parkin, restored mitochondria membrane potential and reduced ROS and tissue factor expression in HCAEC. It suggests potential effects induced by dual use of Rivaroxaban and ASA on the coronary endothelium subjected to high glucose condition.
Keywords
Introduction
Combination of anti-coagulant and anti-thrombotic treatments, based on the inhibition of both platelets and coagulation, reduces adverse cardiovascular events in diabetic patients. The COMPASS trial demonstrated that patients with stable atherosclerotic peripheral vascular disease undergoing combined treatment of an anti-thrombocoagulant regimen with Rivaroxaban (an oral selective inhibitor of factor Xa, FXa) plus daily low dose of Aspirin (ASA) showed better cardiovascular outcome than patients who were treated with Aspirin or Rivaroxaban alone. 1 However, cellular mechanism by which combining Rivaroxaban and ASA resulted in better clinical outcomes than monotherapy has not been elucidated.
Mitochondrial respiration is highly coupled to adenosine triphosphate production, which plays a central role in the vascular functionality. High glucose levels promote increased mitochondrial production of reactive oxygen species (ROS). ROS are closely associated with mitochondrial dysfunctionality and tissue damage. 2 Interestingly, our group recently reported positive effects of both ASA and Rivaroxaban on mitochondrial functionality and expression of mitochondria proteins related to mitochondria dynamic, including mitophagy.3,4
The aim of the present work was to determine if Rivaroxaban alone or in combination with ASA may influence mitochondrial functionality in human coronary arterial endothelial cells exposed to mimicked diabetic hyperglycaemia conditions.
Material and methods
Experimental design and human coronary artery endothelial cells culture
The human coronary artery endothelial cell (HCAEC) line, (Ref. 350–05a, Merck KGaA, Germany) was incubated under the following experimental conditions: HCAEC incubated with physiologic D-glucose concentration (5 mmol/L, control group), HCAEC incubated with 30 mmol/L D-Glucose to mimic an hyperglycemic condition (+Glucose group), HCAEC incubated with 30 mmol/L D-Glucose+50 nmol/L Rivaroxaban (Bay 59–7939, Rivaroxaban group), HCAEC incubated with 30 mmol/L D-Glucose+0.33 mmol/L acetylsalicylic acid (ASA group) and 30 mmol/L D-glucose incubated HCAEC with Rivaroxaban (12.5 nmol/L) +ASA (0.33 mmol/L) (Riva+ASA group).
Content of thrombo-coagulant proteins and mitochondrial proteins in HCAEC
For protein electrophoresis, HCAEC homogenate were loaded and running onto denaturing SDS-PAGE polyacrylamide gels and blotted onto nitrocellulose membranes. Nitrocellulose membranes were incubated with polyclonal antibody against FXa (1:800; AssayPro, St Charles, MO, USA) and monoclonal antibody against tissue factor (TF) (dilution 1:1000; SC-393657, Santa Cruz Biotechnology. Dallas, USA). Homogenate of isolated mitochondria from HCAEC (mitochondria isolation kit. catalog#8987, Thermoscientific, IL, USA) was loaded onto a nitrocellulose membrane and incubated with an anti-Pink-1 polyclonal antibody (1:1000; ab23707. Abcam. Cambridge UK) or anti-Parkin polyclonal antibody (1:1000; ab15954, Abcam. Cambridge, UK). Anti-rabbit IgG (1:2000 for FXa, Pink-1 and Parkin) and anti-mouse IgG (1:2500 for TF) were used as secondary antibodies. The protein was detected using chemiluminescence reagents (ECL; GE Healthcare, Little Chalfont Buckinghamshire, UK) and densitometry was used to measure signal intensity (Ibright FL100, Thermofisher Scientific. Massachussetts, USA).
Reactive oxygen species production by HCAEC
ROS production was determined by a fluorescence (fluorescein Ex/Em = 490/525 nm) microplate assay commercial kit (ab139476, Abcam, Cambridge, UK) following the manufacturer´s instructions. ROS production was measured after stimulation of HCAEC with a submaximal concentration of thrombin (0.025 U/mL) during 10 min. ROS were measured using a microplate fluorescence reader (iBright Imaging System).
Changes in mitochondria membrane potential (ΔΨm) in HCAEC
HCAEC were incubated for 24 h under different experimental conditions. To improve the measurement of changes in ΔΨm, experiments were performed adding a submaximal concentration of thrombin (0.025 U/mL) for 10 min ΔΨm was determined using a JC-10 fluorometric assay kit (MAK159 Sigma-Aldrich. St Louis. USA). JC-10 fluorescent probe was added to HCAEC and incubated for 45 min, at 37°C according to manufacturer instruction. Fluorescence intensity was detected using the iBright Imaging System.
Statistical analysis
Values are expressed as mean ± standard error of mean (S.E.M). The expression of proteins as well as ROS production were compared using the non-parametric Mann-Whitney test, with SPSS 25.0 software. p value < 0.05 was considered as statistically significant.
Results
Content of thrombo-coagulant and mitochondrial proteins
FXa expression was significantly increased in HCAEC incubated with high glucose concentration (30 mmol/L) compared to those incubated under physiological glucose concentration (5 mmol/L) (Figure 1(a)). TF expression was also significantly increased in experiments performed under high levels of glucose compared to control (Figure 1(b)). Addition of Rivaroxaban had no significant effect on TF expression (p = 0.07) compared to high glucose alone. However, addition of either ASA alone or combined with Rivaroxaban significantly reduced TF expression compared to HCAEC incubated with high glucose alone, reaching lower level than control (Figure 1(b)). Panel a.- Representative Western-blot of the Factor Xa (FXA) expression in human coronary artery endothelial cells (HCAEC) incubated with normal D-glucose concentration (5mmol/L, control) and in the presence of 30 mmol/L D-Glucose (+Glucose). Panel b.- Representative Western-blot of tissue factor (TF) expression in HCAEC incubated with normal D-glucose concentration (5 mmol/L, control) or with high glucose, 30 mmol/L D-Glucose (+Glucose). Experiments adding 50 nmol/L Rivaroxaban, 0.33 mmol/L ASA and 12.5 nmol/L Rivaroxaban +0.33 mmol/L ASA to high D-glucose-incubated HCAEC are also represented. Panel c.- Representative dot-blots to measure mitochondrial content of Pink-1 and Parkin proteins HCAEC incubated with 5 mmol/L D- (control) or 30 mmol/L D-Glucose (+Glucose). Experiments adding 50 nmol/L Rivaroxaban, 0.33 mmol/L ASA and 12.5 nmol/L Rivaroxaban +0.33 mmol/L ASA to high D-glucose-incubated HCAEC are also represented. Panel d.- Reactive oxygen species (ROS) production and changes in mitochondria membrane potential (ΔΨm) in HCAEC incubated with 5 mmol/L D- (control) or 30 mmol/L D-Glucose (+Glucose). Experiments adding 50 nmol/L Rivaroxaban, 0.33 mmol/L ASA and 12.5 nmol/L Rivaroxaban +0.33 mmol/L ASA to high D-glucose-incubated HCAEC are also represented. All experiments were also performed in presence of submaximal thrombin concentration (0.025 U/mL). Bar graphs of the four panels show the densitometric analysis represented in arbitrary units (A.U.). Results are represented as mean ± SEM of six different experiments. * p < 0.05 compared to the experiments performed normal glucose (5 mmol/L, control). # p < 0.05 compared to the experiments performed with 30 mmol/L glucose (+Glucose).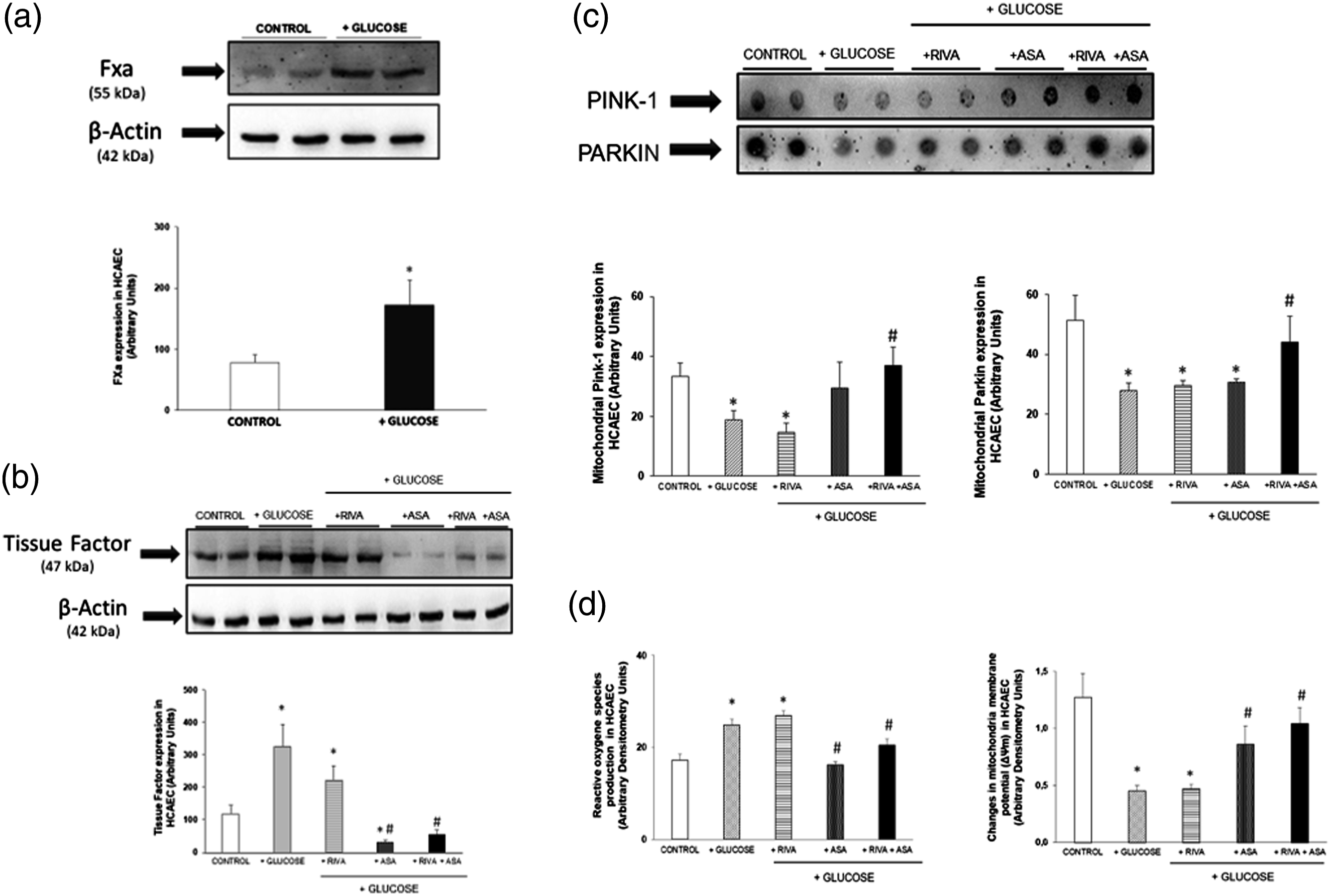
Mitochondrial content of both Parkin and Pink-1 proteins was found to be significantly reduced in HCAEC incubated under 30 mmol/L glucose concentration compared to control group. In 30 mmol/L-incubated HCAEC, mitochondrial content of Pink-1 and Parkin proteins was not modified by addition of Rivaroxaban. However, Rivaroxaban+ASA combination significantly increased mitochondrial content of both Parkin and Pink-1 proteins compared to those found in HCAEC incubated with 30 mmol/L glucose alone (Figure 1(c)).
ROS production and modifications of ΔΨm
After thrombin stimulation, ROS production was significantly increased in 30 mmol/L glucose-incubated HCAEC compared to control group. Addition of Rivaroxaban alone did not revert increased ROS production in HCAEC under high glucose levels. However, addition of ASA alone and ASA+Rivaroxaban prevented the increase in ROS production observed in 30 mmol/L glucose-incubated HCAEC (Figure 1(d)).
ΔΨm was significantly lower in 30 mmol/L glucose incubated HCAEC than in control group. In high glucose-incubated HCAEC, Rivaroxaban alone did not modify ΔΨm, but ASA significantly increased ΔΨm compared to 30 mmol/L glucose-incubated HCAEC. Moreover, in 30 mmol/L glucose-incubated HCAEC, ASA+Rivaroxaban restored ΔΨm to similar levels found in control HCAEC (Figure 1(d)).
Discussion
It is widely established that endothelial cells contribute to activation of FX and fibrin clot formation through surface proteins. Our results have revealed that HCAEC has a weak ability to express FXa under physiological glucose conditions. However, FXa expression is increased when HCAEC are exposed to high glucose levels. Interestingly, it was also reported that human endothelial cells produce clotting factors necessary for FX activation via the intrinsic (FVIII-FIX) and extrinsic (tissue factor [TF]-FVII) coagulation pathways. 5 TF is involved in inflammatory and thrombo-coagulating complications associated with diabetes mellitus. 6 Accordingly, increased expression of TF was found in HCAEC exposed to high glucose levels. Only addition of ASA alone or combined with Rivaroxaban achieved to decrease TF expression in HCAEC incubated with 30 mmol/L glucose. It suggests that ASA has higher ability to inhibit TF synthesis compared to that exerted by FXa inhibition, via Rivaroxaban. Interestingly, it was previously reported that ASA reduces TF synthesis in human monocytes. 7
Hyperglycaemia increases oxidative stress and seems to exert adverse effects on mitochondria functionality. 2 Mitophagy is the mechanism by which dysfunctional mitochondria are removed. 8 The mitochondrial proteins Parkin and Pink play a crucial role in mitophagy. 8 Parkin is selectively translocated from cytosol to damaged mitochondria and bound to Pink-1 with the aim of promoting mitophagy. 8 Interestingly, exposure of HCAEC to high-glucose decreased the mitochondrial content of both Pink-1 and Parkin proteins, suggesting a decrease in mitophagy. Accordingly, it was recently reported that protein expression of Pink-1 and Parkin was significantly reduced in hearts of diabetic animals. 9 Previous studies in human abdominal aneurysms demonstrated effects of Rivaroxaban on the ratio of Pink-1/Parkin mitochondrial content. 3 However, in high glucose-incubated HCAEC only co-administration of Rivaroxaban+ASA induced a significant increase in the mitochondrial content of both Pink-1 and Parkin proteins. It may support that in HCAEC exposed to high glucose, Rivaroxaban+ASA have an apparent more effective ability to promote mitophagy than ASA and Rivaroxaban alone. On the other hand, dysfunctional mitochondria generate ROS accumulation. Interestingly, it has been reported a relationship between mitophagy stimulation and oxidative stress reduction in diabetic patients. 10 Rivaroxaban+ASA combination achieved the lowest ROS production in HCAECs incubated with high glucose. It should be pointed out that ASA alone also reduced ROS production in HCAEC incubated with high glucose levels. Under high glucose condition, ASA alone also tended to increase the mitochondrial content of Pink-1 protein, although mitochondrial content of Parkin protein was not modified. Interestingly, studies in Pink-1−/− cells have suggested that changes in Pink-1 expression could be enough to control mitophagy and ROS production. 11 In this line of evidence, reduction of ΔΨm has been closely associated with decreased mitochondria function and elevated ROS production, 12 being observed in the present study that addition of Rivaroxaban+ASA restored ΔΨm values similar to those found in the control group.
As conclusion, in high glucose-exposed HCAEC, incubated with a combined anti-thrombo-coagulant regimen (Rivaroxaban+ASA), the mitochondrial content of the mitophagy-related proteins Pink-1 and Parkin was increased, being also increased the mitochondrial membrane potential and reducing both ROS and TF production. The present study is merely descriptive, future experiments are warranted to determine mechanistic pathways.
Footnotes
Acknowledgements
We thank GenObIA-CM, B2017/BMD-3773 and FIS (Fondo de Investigaciones Sanitarias. Instituto de Salud Carlos III) and the European Union through the Fondo Europeo de Desarrollo Regional (FEDER, “Una manera de hacer Europa”): grant no. PI17/01408.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Bayer Pharmaceuticals.
Ethics approval
The present work was performed using a commercial human cell line, in line with the ethical standards as laid down in the 1964 Declaration of Helsinki.
