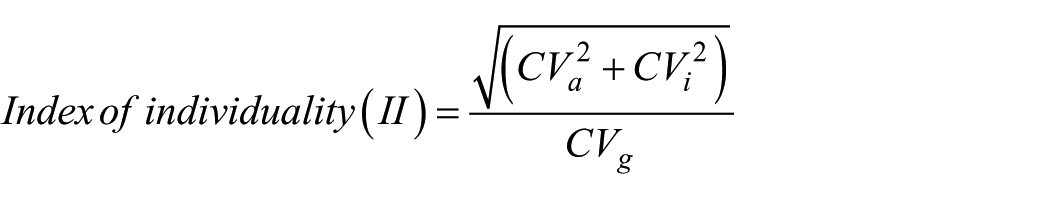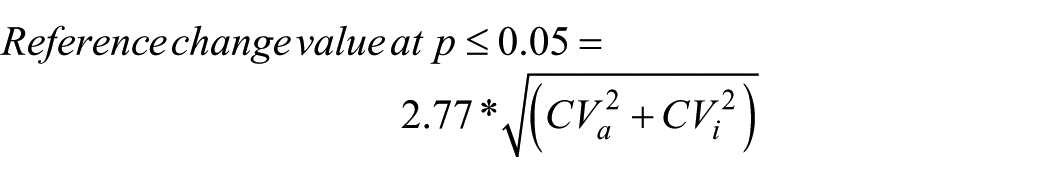Abstract
Introduction and objective:
Target lesion failure continues to limit the efficacy of percutaneous coronary intervention despite advancements in stent design and medical therapy. Identification of biomarkers to risk stratify patients after percutaneous coronary intervention has the potential to focus therapies on cohorts with increased benefits. Plasminogen activator inhibitor-1 has been identified as a candidate biomarker. Herein, we evaluate biological variables which impact plasminogen activator inhibitor-1 levels and analytical characteristics which impact its utility as a biomarker in humans.
Methods:
Plasma plasminogen activator inhibitor-1 was measured in 689 patients undergoing coronary angiography. Plasminogen activator inhibitor-1 levels were measured. Clinical and procedural characteristics were collected in a prospective registry.
Results:
Plasma plasminogen activator inhibitor-1 analytical (CVa = 4.1%), intra-individual (CVi = 44.0%) and inter-individual (CVg = 118.6%) variations with reference change value of 122.3% were calculated. Plasminogen activator inhibitor-1 levels were elevated in patients with cardiovascular risk factors, including type 2 diabetes, pre-diabetes, smokers, obesity, hypertension, and daytime variation in procedure and blood draw.
Conclusion:
Variation in plasma plasminogen activator inhibitor-1 levels is influenced by multiple biological and procedural characteristics. The performance of plasma plasminogen activator inhibitor-1 is consistent with biomarkers in clinical use (N-terminal pro-B-type natriuretic peptide and C-reactive protein) and its applicability is promising.
Keywords
Introduction
Plasminogen activator inhibitor-1 (PAI-1) is an anti-fibrinolytic protein involved in the regulation of fibrin clot formation. 1 By forming an inhibitory irreversible 1:1 complex with tissue-type plasminogen activator (tPA) and urokinase-type plasminogen activator (uPA), PAI-1 prevents fibrin clot degradation promoting hemostasis. 1 Although PAI-1 is expressed in multiple cell types, circulating plasma PAI-1 is primarily platelet-derived. 2 Mechanisms responsible for elevated plasma PAI-1 levels remain unknown; however, increased levels have been associated with type 2 diabetes, metabolic syndrome and coronary artery disease (CAD).3,4
Previous small studies have evaluated PAI-1 as a biomarker – quantifying plasma PAI-1 antigen as opposed to activity levels. While PAI-1 is linked to vascular risk factors, its function as a biomarker to predict cardiovascular adverse events has yielded mixed results due to heterogeneity in the populations studied and methodologies used. Moreover, PAI-1 levels may be influenced by technical factors such as time of blood draw and method of extraction.5,6 Finally, analytic characteristics of PAI-1 have yet to be reported. 7
Accordingly, we sought to determine the performance of PAI-1 following percutaneous coronary intervention (PCI). In particular, PAI-1 may play roles in thrombotic events and neointimal hyperplasia following revascularization. 1 Given the lack of robust data evaluating plasma PAI-1 as a biomarker in this population, we set out to determine which patient, clinical and procedural factors influence PAI-1 antigen levels to inform future outcome-based studies. Herein, we report biological and analytical considerations in PAI-1 measurement in patients undergoing coronary angiography and revascularization.
Methods
Ethics statement
This study was approved by the Ottawa Health Science Network Research Ethics Board (OHSN-REB) (Protocol #: 20160516-01H), and written informed consent was obtained from all patients. The study conforms with the 1975 Declaration of Helsinki for the use of human blood.
Study design and patient data collection
The University of Ottawa Heart Institute is a quaternary care centre servicing a catchment of 1.2 million people with all procedures prospectively indexed in a registry. From October 2016 to August 2018, blood samples were collected on consecutive eligible patients referred for PCI. Arterial blood samples were collected in 3.2% sodium citrate tubes (Becton Dickinson, Franklin Lakes, NJ, USA) immediately after the completion of the procedure and processed within 2 h after collection. Samples were centrifuged at 1200×g for 10 min at 4°C without brakes. Next, platelet-rich plasma was spun at 3200×g for 10 min at 4°C to remove contaminants and the plasma were stored at −80°C until further processing.
Patient data were obtained from the Cardiovascular and percutaneous clinical trials (CAPITAL) revascularization registry which indexes over 1200 clinical data points on patients undergoing coronary angiography at the institute. CAD confirmed by coronary angiography was defined as stenosis ⩾50%. Time of procedure was collected and dichotomized to morning (08:00–12:00 h) or afternoon (12:00–16:00 h). Diabetes status was defined as haemoglobin A1c (HbA1c) levels ⩾6.5% at presentation. Pre-diabetes was defined as having HbA1c levels between 6.0% and 6.4% according to the Canadian Diabetes Association guidelines. Medication status was indexed at the time of the procedure.
PAI-1 quantification
Plasma samples from 689 patients were thawed and PAI-1 antigen levels were determined by a commercially available enzyme-linked immunosorbent assay (ELISA) according to the manufacturers’ recommendations (KHC3071) (ThermoScientific, Waltham, MA, USA). Laboratory assessments of PAI-1 antigen levels from patient samples were performed within 6 months of storage by the same research personnel to minimize analytical variations.
Statistical analysis
Continuous variables were reported with mean (±standard deviation) or median (interquartile range) as appropriate. Categorical variables were reported as proportions (%). Plasma PAI-1 levels were first examined for normality using Shapiro–Wilks test, and subsequently, standard t-test or Mann–Whitney U-test was performed as appropriate. Coefficient of variation (CV) was determined for analytical CV (CVa), inter-subject CV in our population (CVg) and intra-subject CV (CVi).7,8 Index of individuality (II) allows to determine whether monitoring or reference range is required for a biomarker.
Univariate linear regression was performed with all factors with a predetermined p value <0.20 to identify variables for inclusion prior to a multivariable linear regression analysis. All statistical analyses were performed using SAS v9.4 (SAS Institute, Inc.). The value of p < 0.05 was considered significant. Figures were created using GraphPad Prism v7 (GraphPad Software, La Jolla, CA, USA).
Result and discussion
PAI-1 antigen levels were measured in 689 patients. The mean age of our cohort at baseline was 67.3 ± 11.2 years, of which 66.9% were male (Supplemental Table 1). Overall, our cohort was overweight with an average body mass index of 29.5 ± 6.2 kg/m2. Cardiac risk factors were prevalent, including pre-diabetes (9%), type 2 diabetes (37%) of which 29.5% were insulin-managed, active smoker (34%), dyslipidaemia (63%) and hypertension (65%). Patients were managed with contemporary medical therapy with most patients receiving statins (78%), aspirin (ASA, 89%) and P2Y12 inhibitors (89%). A high proportion of patients had obstructive CAD (79%). 46% of patients underwent PCI, with 89% performed via transradial access and 86% having received unfractionated heparin during the procedure.
To assess biomarker performance, we assessed CVs in our population of interest. Since plasma PAI-1 levels were not normally distributed, CVs are reported for both baseline and log-transformed data. The CVa for the assay was low (n = 80) at 4.1%. Variability within subjects (CVi) of those with a repeat sample in 1 year was moderate at 44.0% (n = 40). Variability between subjects (CVg) in our entire cohort shows wide variation, CVg = 118.6% (n = 689). Finally, the II and reference change value (RCV) of PAI-1 were 0.4 and 122.3%, respectively. The CVa, CVi and CVg of the log transformed antigen levels were 0.7%, 24.8% and 12.9%, respectively. The performance of plasma PAI-1 antigen parallel established markers for CAD such as C-reactive protein (CRP, CVa = 5.2%, CVi = 42.2%, CVg = 92.5%, II = 0.5) and for heart failure such as N-terminal pro-B-type natriuretic peptide (NT-proBNP, CVa = 3.3%, CVi = 25.1%, CVg = 54.0%, II = 0.2).9,10
Next, we sought to identify patient comorbidities, medications and biochemical parameters which may have impacted PAI-1 levels. We first performed a univariate regression model and included all associated factors with a p value <0.20 in a multivariable linear regression to identify associated variables (Supplemental Table 2). We identified body mass index, active smoking, diabetes, pre-diabetes and hypertension to be positively associated with changes in plasma PAI-1 levels (Figure 1(a) to (c)). Notably, confirmed presence of CAD did not influence PAI-1 antigen levels. New procedural factors were identified which potentially affect measured plasma PAI-1 levels such as the use of nitroglycerin and daytime variation in coronary angiography (Figure 1(d) and (e)). Finally, females appear to have higher circulating levels of PAI-1 (33.7% change) compared to their male counterparts (Figure 1(f)).
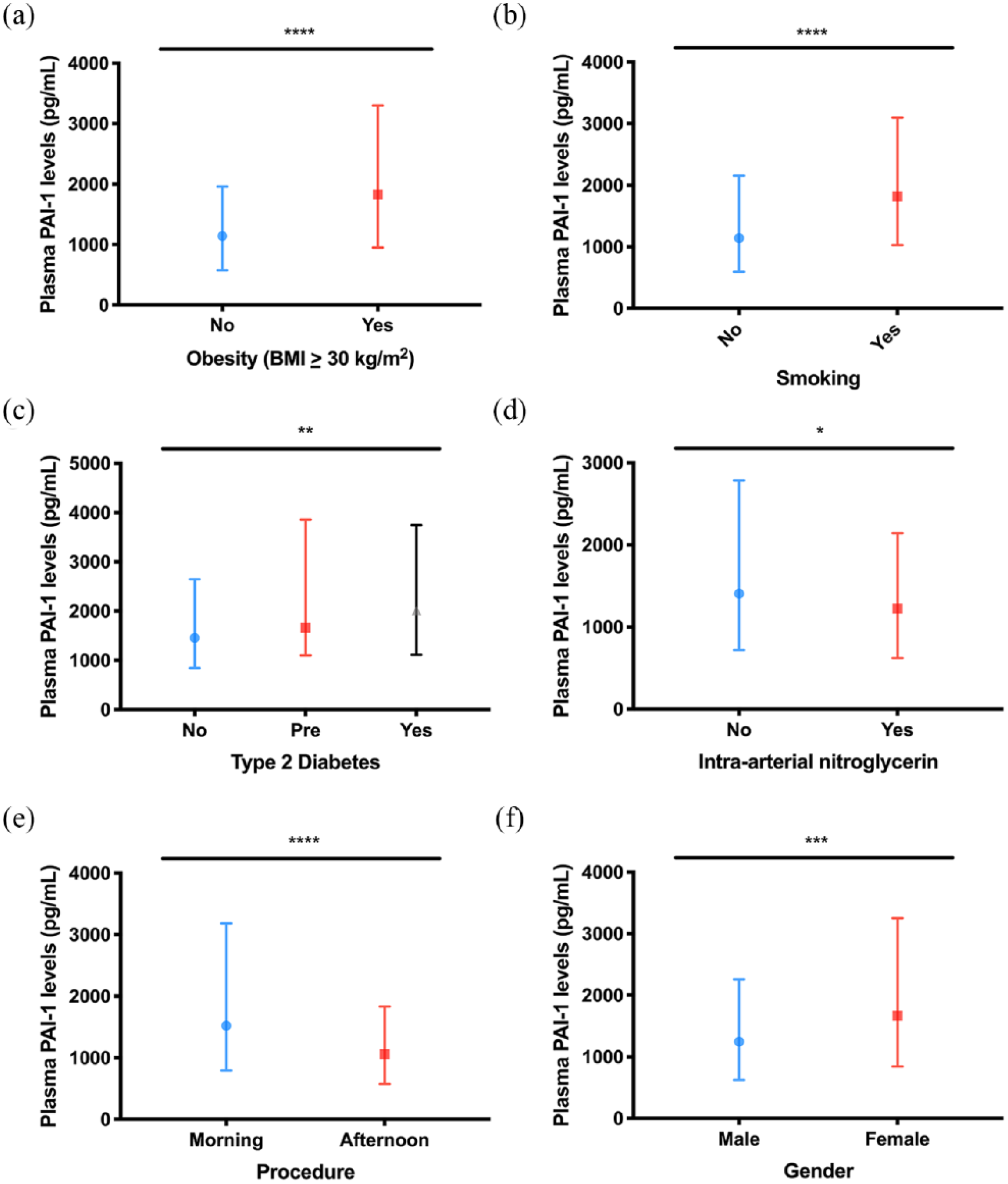
Influence of plasma PAI-1 levels (pg/mL) by biological and procedural characteristics: (a) PAI-1 levels are elevated with obesity (BMI ⩾ 30 kg/m2) (1828.0 [951.6–3301.0] pg/mL (n = 272) vs 1140.0 [577.0–1961.0] pg/mL (n = 417), p < 0.0001), (b) PAI-1 levels are elevated with active smoking (1822.0 [1028.0–3100.0] pg/mL (n = 236) vs 1143.0 [596.6–2156.0] pg/mL (n = 453), p < 0.0001), (c) PAI-1 levels are elevated with type 2 diabetes mellitus (2014.0 [1115.0–3743.0] pg/mL (n = 210) vs 1661.0 [1103.0–3861.0] pg/mL (n = 62) vs 1455.0 [842.3–2647.0] pg/mL (n = 181), p < 0.01), (d) PAI-1 levels are decreased with intra-arterial administration of nitroglycerin (1227.0 [620.9–2144.0] pg/mL (n = 267) vs 1407.0 [719.2–2785.0] pg/mL (n = 413), p = 0.03), (e) PAI-1 levels are decreased in procedures performed in the afternoon (1063.0 [576.6–1832.0] pg/mL (n = 270) vs 1521.0 [796.5–3183.0] pg/mL (n = 406), p < 0.0001), (f) PAI-1 levels are elevated in females (1665.0 [846.6–3252.0] pg/mL (n = 225) vs 1249.0 [624.0–2258.0] pg/mL (n = 460), p < 0.0001).
Herein, we demonstrate that plasma PAI-1 has similar analytical performance characteristics to clinically used biomarkers such as CRP and NT-proBNP which have high CVg and CVi. Due to a low II of PAI-1 (II < 0.6), changes from population-based reference values in an individual have limited value as a diagnostic test to detect pathologies, but may be useful as a monitoring test in patients undergoing coronary angiography.7,11 The measured critical difference (at p ⩽ 0.05) between two serial measurements of PAI-1 was determined to be 122%, similar in performance to CRP (118%). 9 Thus, use of PAI-1 will be of little benefit in screening healthy populations for early stages of CAD, but may be useful when serial monitoring is performed to examine response to therapy or ascertaining risk. 12
To date, our report is derived from the largest cohort of patients undergoing coronary angiography and the first to report the analytical performance of PAI-1 as a biomarker. Our study confirms known factors impacting PAI-1 levels while also highlighting factors including circadian variation, female sex and pre-diabetes – factors which should be incorporated into future studies. 13 Whether plasma PAI-1 levels predict outcomes following coronary revascularization remains to be established in larger cohorts.
Similar levels of plasma PAI-1 levels were observed in those with pre-diabetes and type 2 diabetes in those undergoing coronary angiography. This likely is due to two reasons contributing to the pro-inflammatory state observed in diabetes: (1) pre-diabetes is often not found exclusively in patients presenting for a coronary angiogram and is often found in concert with other cardiovascular risk factors including hypertension, dyslipidaemia and obesity forming the foundation of the diagnostic criteria for metabolic syndrome14,15 and (2) platelet hyper-reactivity is observed in pre-diabetes exhibited by increase in platelet aggregation and potentially, subsequent release of PAI-1 from α-granules.2,16,17
Our study is not without limitations. First, variation of circulating PAI-1 levels due to circadian rhythm cannot be fully captured by accounting for the daytime variation of coronary angiogram since each individual has a variable diurnal pattern. 6 Second, this remains a retrospective analysis although the registry data are collected prospectively and independent of PAI-1 samples to minimize bias. Finally, this study remains underpowered to link PAI-1 levels with clinical outcomes, and although a large sample size was achieved given the observed CVi and CVg, future studies will need to enrol significantly larger cohorts to assess this relationship.
In summary, plasma PAI-1 antigen levels demonstrate similar biomarker performance characteristics when compared to established clinically utilized biomarkers. Pre-diabetes, diabetes, sex and smoking status are positively associated with PAI-1 levels. In contrast, age, procedural administration of intra-arterial nitroglycerin and daytime variation in coronary angiogram appear to be inversely associated with PAI-1 levels. Careful consideration of biological and analytical variations should be incorporated into future studies.
Supplemental Material
Supplemental_Tables_for_PAI-1_RTR – Supplemental material for Performance of plasminogen activator inhibitor-1 as a biomarker in patients undergoing coronary angiography: Analytical and biological considerations
Supplemental material, Supplemental_Tables_for_PAI-1_RTR for Performance of plasminogen activator inhibitor-1 as a biomarker in patients undergoing coronary angiography: Analytical and biological considerations by Richard G Jung, Trevor Simard, Pietro Di Santo, Alisha Labinaz, Robert Moreland, Anne-Claire Duchez, Kamran Majeed, Pouya Motazedian, Rebecca Rochman, Young Jung and Benjamin Hibbert in Diabetes & Vascular Disease Research
Footnotes
Author contributions
R.G.J. and T.S. have equally contributed to this work. R.G.J., T.S. and B.H. developed the idea for the study. R.G.J., P.D.S., A.L., R.R., P.M. and K.M. consented patients and processed blood samples. R.G.J. and A.C.D. analysed plasma samples for PAI-1 levels. R.G.J., Y.J. and B.H. performed the statistical analysis. R.G.J. and B.H. wrote the manuscript. T.S., P.D.S., Y.J. and B.H. critically reviewed the final version of the manuscript. All authors were involved in the critical revision of the manuscript and approved the final version to be published.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorships and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was supported by the Innovation Fund of the Alternative Funding Plan for the Academic Health Sciences Centres of Ontario and Canadian Foundation of Innovation. R.G.J. was funded by the Vanier CIHR Canada Graduate Scholarship for his graduate studies. A.C.D. was supported by the Cardiac Endowment Fund at the University of Ottawa Heart Institute. The authors would like to thank the CAPITAL Research Group for all their assistance in the completion of the project.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.