Abstract
Introduction:
High-density lipoprotein cholesterol comprises a group of heterogeneous subfractions that might have differential effects on atherosclerosis. Moreover, prior investigations suggest that the presence of diabetes (T2D) modifies the impact of some subfractions on atherosclerosis. In this study, we aimed to evaluate the association between high-density lipoprotein cholesterol subfractions and carotid intima-media thickness in the baseline assessment of the Brazilian Longitudinal Study of Adult Health participants from the São Paulo investigation centre.
Methods:
We evaluated 3930 individuals between 35 and 74 years without previous cardiovascular disease not using lipid-lowering drugs. High-density lipoprotein cholesterol subfractions (HDL2-C and HDL3-C) were measured by vertical ultracentrifugation (vertical auto profile). The relationship between each high-density lipoprotein cholesterol subfraction and carotid intima-media thickness was analysed by multiple linear regression models.
Results:
Total high-density lipoprotein cholesterol, as well as HDL2-C and HDL3-C, was negatively associated with carotid intima-media thickness after adjustment for demographic data (all p < 0.001) and traditional risk factors (all p < 0.05). When stratified by T2D status, the HDL2-C/HDL3-C ratio showed a negative association with carotid intima-media thickness in participants with T2D (p = 0.032), even after fully controlling for confounding variables, including total high-density lipoprotein cholesterol.
Conclusion:
HDL2-C, HDL3-C and HDL2/HDL3-C ratio are inversely associated with carotid intima-media thickness after adjustment for traditional risk factors. Association of the HDL2-C/HDL3-C ratio is modified by the presence of diabetes, being more pronounced in diabetic individuals.
Keywords
Introduction
High-density lipoproteins (HDLs) comprise a group of heterogeneous particles, and high-density lipoprotein cholesterol (HDL-C) has a well-established inverse association with the risk of cardiovascular (CV) events in prospective longitudinal cohorts followed throughout the world.1,2 However, genetic polymorphisms that increase HDL-C do not correlate with CV risk when excluded variants additionally linked to low-density lipoprotein cholesterol (LDL-C) or triglycerides.3–5 Also, pharmacologic interventions with cholesteryl ester transfer protein (CETP) inhibitors to raise HDL-C levels did not reduce CV outcomes and even might be harmful.6–9 This lack of association has motivated studies of HDL-C subfractions to more comprehensively evaluate the mechanisms linking HDL-related pathways with atherogenesis. Among the methods used to separate those subfractions, the vertical auto profile (VAP) separates HDL-C into two subfractions: HDL2-C (larger and less dense) and HDL3-C (smaller and denser). 10
Carotid intima-media thickness (cIMT) is an important marker for subclinical vascular disease and is considered an atherosclerosis surrogate. 11 Increases in cIMT are accompanied by a higher incidence of CV morbidity including coronary artery disease, myocardial infarction12–14 and stroke. 15 Although low HDL-C has been associated with increased cIMT, 16 this relationship is not always observed, 17 suggesting that total HDL may not entirely explain HDL’s antiatherogenic properties. Also, the association of HDL-C subfractions and cIMT remains disputed since different results are obtained with the use of different separation methods.18–20 This relationship is even more intricate because diabetic individuals have lower HDL-C levels and a lower HDL2-C/HDL3-C ratio, which seems to modify the association of HDL-C with cIMT in comparison with the non-diabetic population. It is also well documented that the functionality of HDL species from patients with T2D is impaired and even dysfunctional.21,22
In this study, we evaluated the association between HDL-C subfractions and cIMT in a Brazilian sample without clinical manifestations of any cerebrovascular disease. In addition, we evaluated whether the presence of diabetes modifies this association.
Methods
Sample
The Brazilian Longitudinal Study of Adult Health (ELSA-Brasil) design has been previously described.23,24 Briefly, we enrolled 15,015 civil servants from six Brazilian cities, between August 2008 and December 2010. We included those who underwent HDL-C measurement by the VAP method and cIMT assessment – that were all participants from São Paulo centre. Exclusion criteria for this analysis were the cohort restrictions (pregnancy or recent childbirth; cognitive deficit or impaired communication; in retired servants, living outside of a study centre’s area) in addition to individuals with prior CV disease (myocardial infarction, stroke, heart failure and coronary revascularization) and participants using lipid-lowering drugs.
HDL-C and subfractions
Blood collection was performed in patients after nocturnal fasting. The samples were centrifuged at the sites and stored in tubes at −80°C. 25 The HDL-C, HDL2-C and HDL3-C subfractions were obtained by the VAP method through an apparatus, the vertical rotor of which holds the tube perpendicular to the base of the equipment. 10
cIMT measurement
The measurement protocol was published previously.26–28 cIMT measurements were performed using a Toshiba (Aplio XG™) with a 7.5-MHz linear transducer. Intima-media thickness (IMT) was measured in the outer wall of a predefined carotid segment of 1 cm in length from 1 cm below carotid bifurcation, during three cardiac cycles. All participating centres obtained the carotid images, and the images were forwarded to the centralized core reading centre in São Paulo. Images were classified as valid if the following was obtained for the left and right sides: (1) the anatomic guides for the common carotid arteries, (2) interfaces between the lumen and the far vessel wall and (3) interfaces between the media and the adventitia layers of the far vessel wall. We used MIA™ software to standardize the reading and interpretation of carotid scans.
Statistical analysis
We presented continuous variables as descriptive statistics with mean and standard deviation for normal distribution and, for non-normal distributions, with medians and quartiles. Categorical variables were shown in absolute and relative frequency. Baseline characteristics table was exposed according to total HDL-C quartiles to show different HDL-C subfraction levels across the total HDL-C groups. For correlation measurement, we used Pearson. The association between HDL-C subfractions and variables was determined by linear regression. We standardized HDL-C, HDL2-C, HDL3-C and HDL2-C/HDL3-C ratio, which were included as the main explanatory variables in separate multiple linear models. Models including HDL2-C, HDL3-C or HDL2-C/HDL3-C ratio were also further adjusted for total HDL-C (as exposed in Table 2 – model 4 and Table 3 – model 2).
As we observed significant interaction terms between HDL2-C/HDL3-C ratio and T2D, we performed additional analyses stratified by diabetes status. The models were adjusted for race/ethnicity, age and sex, smoking, alcohol, physical activity, LDL-C, systolic blood pressure, waist circumference, fasting glucose, log-transformed triglycerides, estimated glomerular filtration rate and antihypertensive use. Statistical significance was defined as p < 0.05. Analyses were performed with Stata version 14.0 (StataCorp, USA).
Results
We included 3930 participants (1048 exclusions from São Paulo centre) with a mean age of 50.1 ± 8.4 years and 1793 (45.6%) males. The mean HDL-C was 1.42 ± 0.37 mmol/L and mean cIMT was 0.796 ± 0.195 mm (Table 1).
Demographic and clinical characteristics of the study sample according to HDL-C quartiles.
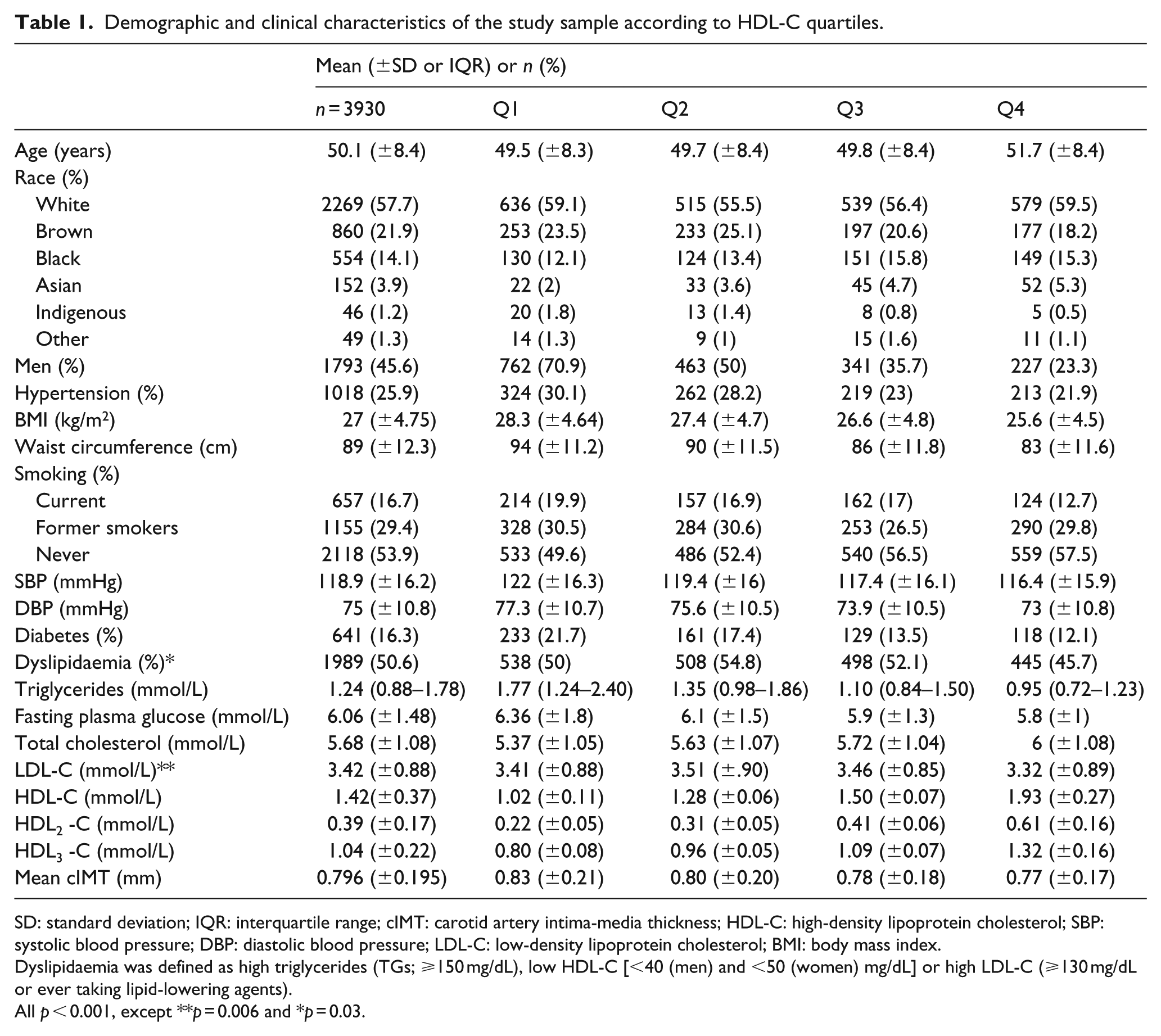
SD: standard deviation; IQR: interquartile range; cIMT: carotid artery intima-media thickness; HDL-C: high-density lipoprotein cholesterol; SBP: systolic blood pressure; DBP: diastolic blood pressure; LDL-C: low-density lipoprotein cholesterol; BMI: body mass index.
Dyslipidaemia was defined as high triglycerides (TGs; ⩾150 mg/dL), low HDL-C [<40 (men) and <50 (women) mg/dL] or high LDL-C (⩾130 mg/dL or ever taking lipid-lowering agents).
All p < 0.001, except **p = 0.006 and *p = 0.03.
As previous publications analysing HDL-C subfractions,18,29,30 we observed a high correlation between total HDL-C and both subfractions HDL2-C (p = 0.94) and HDL3-C (p = 0.96) and a lower correlation with the HDL2-C/HDL3-C ratio (p = 0.73). In crude models, an inverse association of HDL-C, HDL2-C, HDL3-C and HDL2-C/HDL3-C ratio with mean cIMT was noted. These results remained essentially unchanged after adjusting for demographic characteristics (model 1) and CV risk factors (except in HDL2-C/HDL3-C ratio – model 3). However, when subfractions were adjusted for HDL-C, they were no longer associated with lower mean cIMT (Table 2).
Association of total HDL-C, HDL2-C and HDL3-C subfractions and HDL2-C/HDL3-C ratio with cIMT.
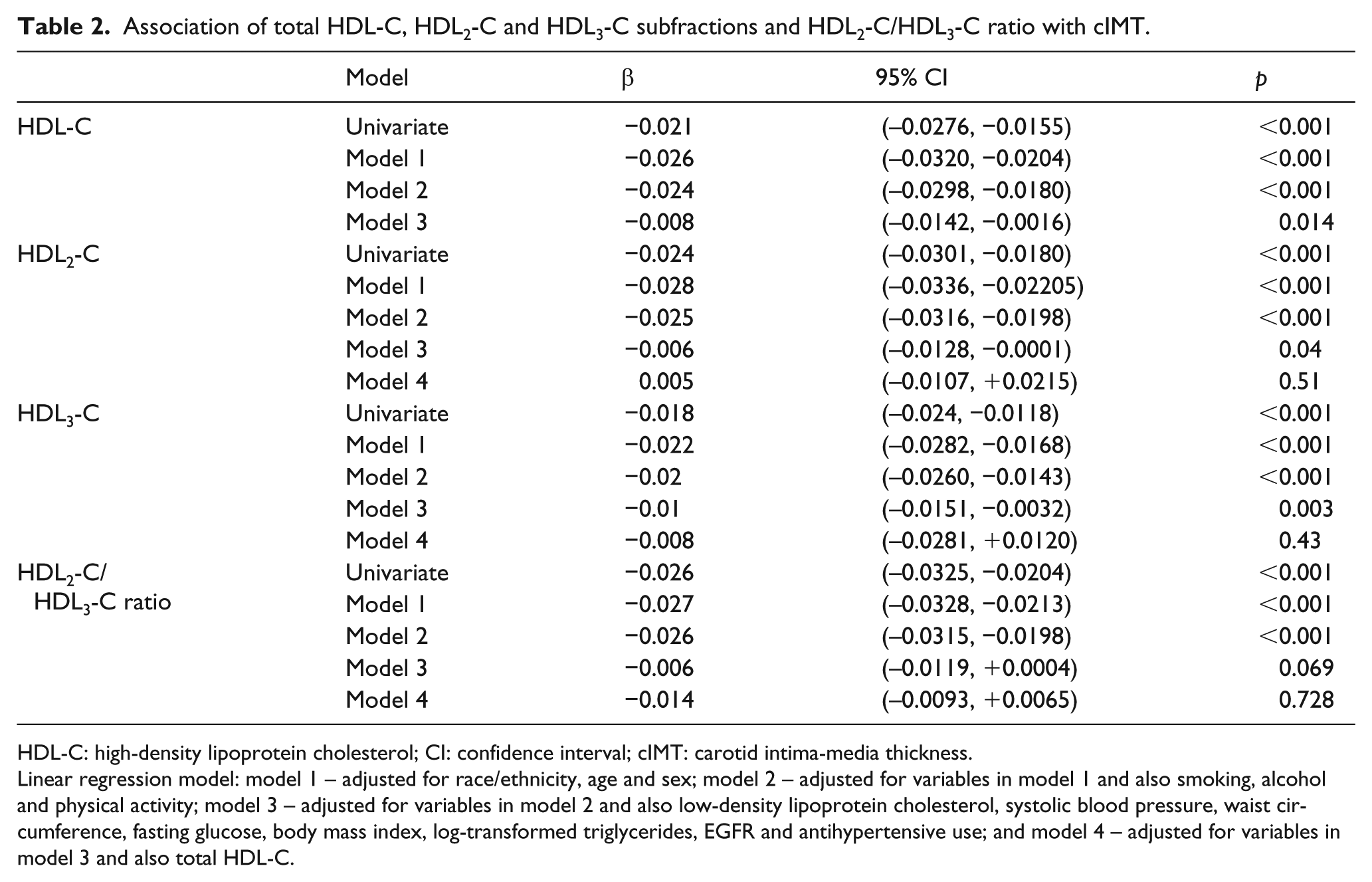
HDL-C: high-density lipoprotein cholesterol; CI: confidence interval; cIMT: carotid intima-media thickness.
Linear regression model: model 1 – adjusted for race/ethnicity, age and sex; model 2 – adjusted for variables in model 1 and also smoking, alcohol and physical activity; model 3 – adjusted for variables in model 2 and also low-density lipoprotein cholesterol, systolic blood pressure, waist circumference, fasting glucose, body mass index, log-transformed triglycerides, EGFR and antihypertensive use; and model 4 – adjusted for variables in model 3 and also total HDL-C.
We found an interaction between T2D status and the HDL2-C/HDL3-C ratio. Furthermore, when stratified by the presence of T2D, the HDL2-C/HDL3-C ratio was inversely associated with cIMT in participants with diabetes in full models (Figure 1), but not in participants without diabetes. We also found that HDL3-C was negatively associated with cIMT in non-T2D individuals and not in participants with T2D. In this case, a p value of borderline significance (p = 0.051) was observed. After further adjustment for HDL-C levels, only the association between the HDL2-C/HDL3-C ratio and cIMT in individuals with diabetes remained significant (p = 0.032; Figure 2 and Table 3).
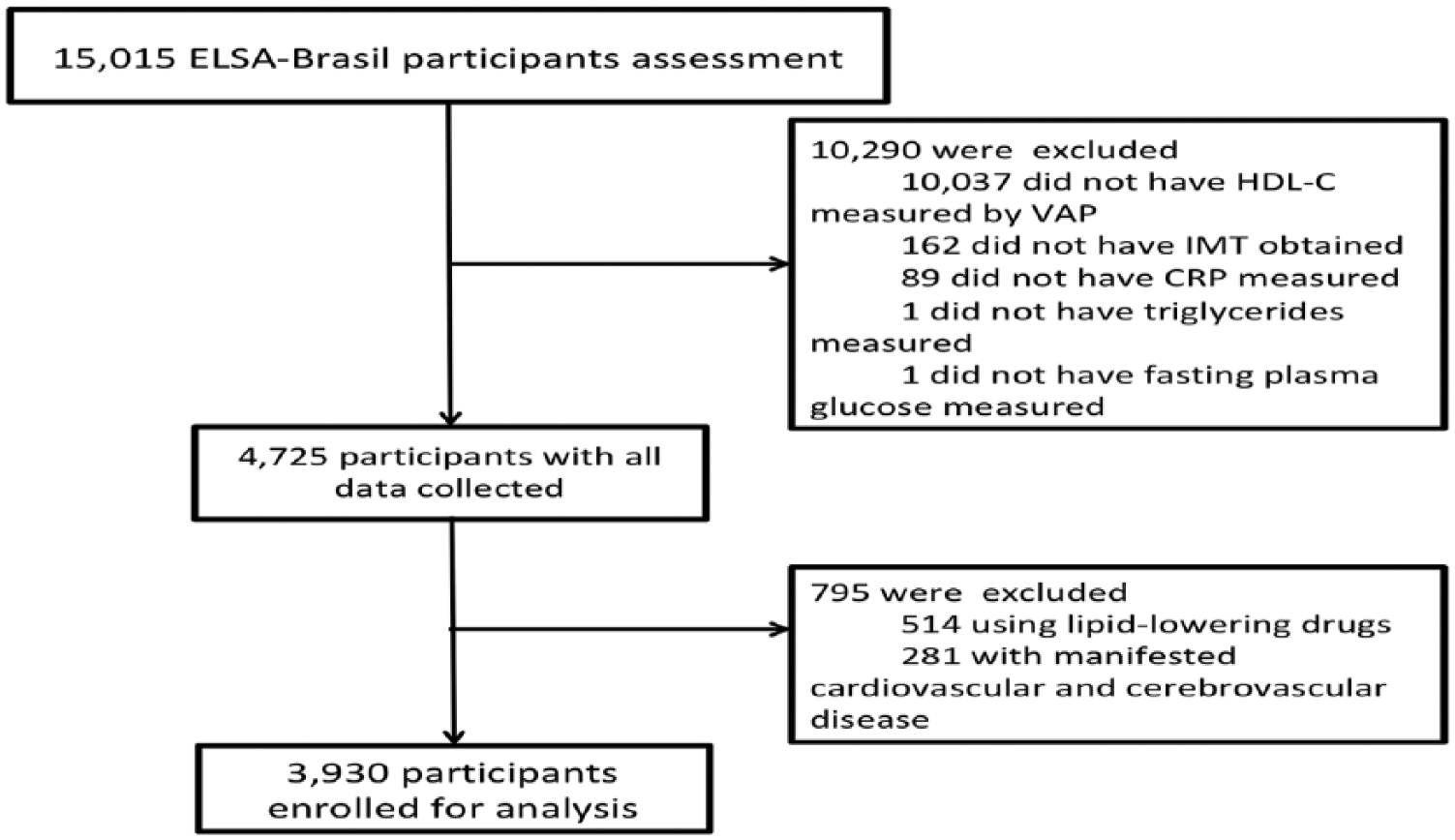
Detailed enrolment flow diagram.
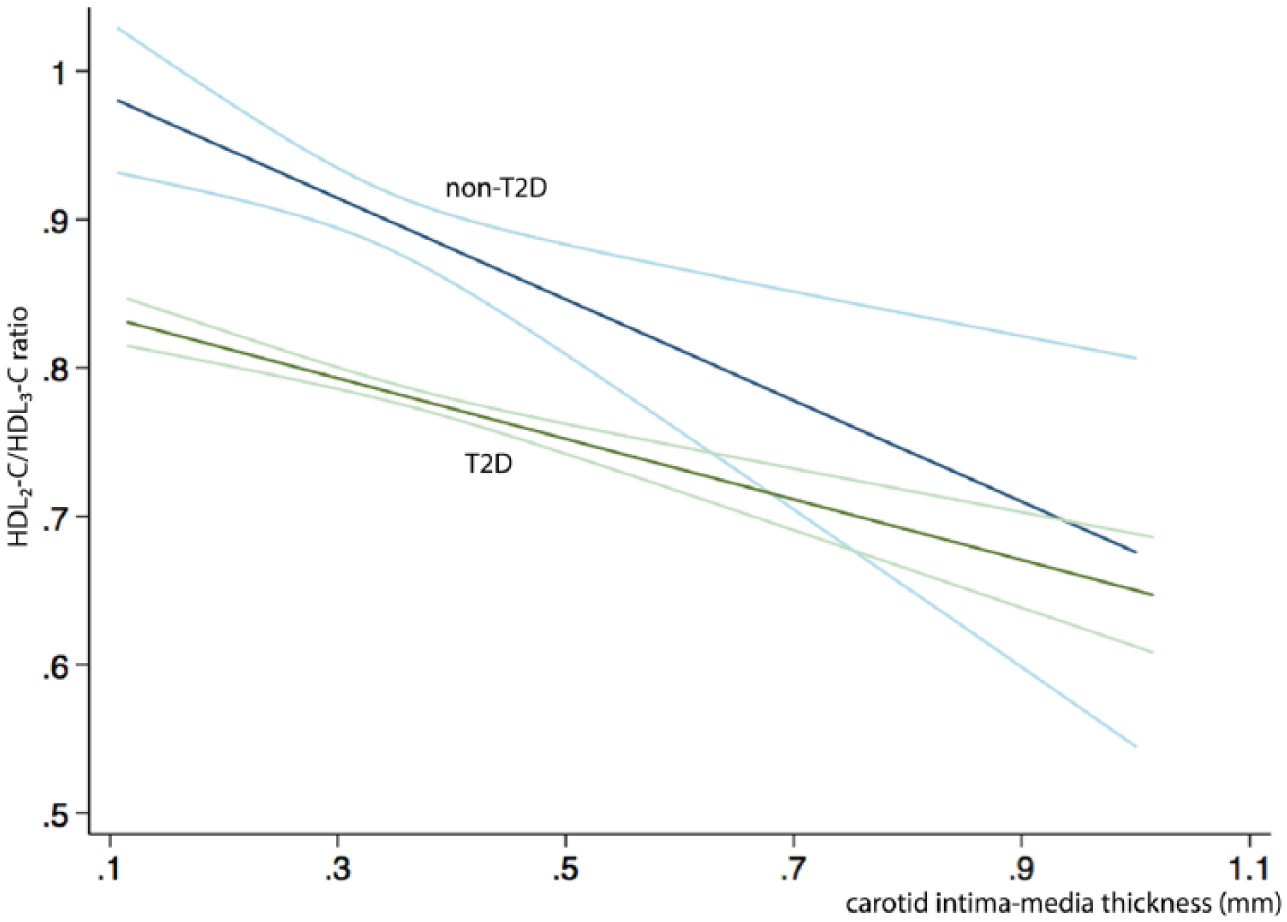
Association between the HDL2-C/HDL3-C ratio and cIMT in univariate analysis.
Association of total HDL-C, HDL2-C and HDL3-C subfractions and the HDL2-C/HDL3-C ratio with cIMT when stratified by diabetes mellitus status.
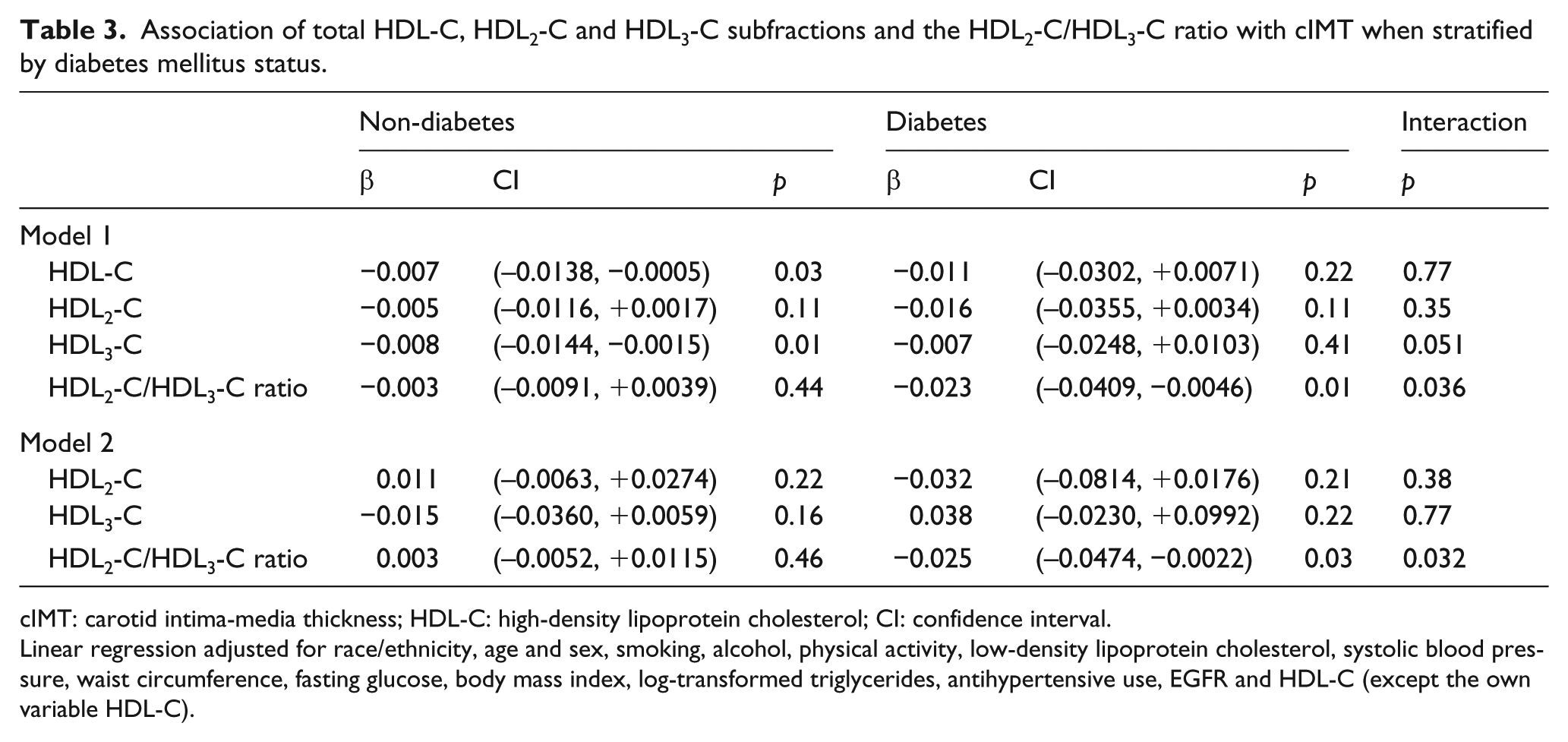
cIMT: carotid intima-media thickness; HDL-C: high-density lipoprotein cholesterol; CI: confidence interval.
Linear regression adjusted for race/ethnicity, age and sex, smoking, alcohol, physical activity, low-density lipoprotein cholesterol, systolic blood pressure, waist circumference, fasting glucose, body mass index, log-transformed triglycerides, antihypertensive use, EGFR and HDL-C (except the own variable HDL-C).
Discussion
Our results demonstrate that both HDL2-C and HDL3-C are inversely associated with cIMT even after controlling for demographic and CV risk factors, but they are not independent predictors. This association is particularly robust with HDL2-C/HDL3-C ratio for diabetics, whereas such findings were noted in non-diabetics only with HDL3-C.
Previous studies have observed controversial results in the relationship between HDL-C subfractions and cIMT. While some studies have found no association of HDL2-C or HDL3-C with cIMT,19,20 a recent publication has shown that, in addition to HDL-C, the HDL2-C subfraction is associated with a lower cIMT, suggesting that this subfraction is atheroprotective. 18 In contrast to these data, a Finnish study 31 demonstrated that HDL-C, HDL2-C and HDL3-C subfractions and also the HDL2-C/HDL3-C ratio are negatively associated with cIMT, a finding that was reproduced in our study. These discordant results might be explained by ethnic differences in the population, differences in baseline characteristics (younger, more obese people), and participants not being treated with lipid-lowering therapies.
The lipid panel is known to be altered in about 50% of the individuals with diabetes, either due to high triglycerides or LDL-C levels or by low HDL-C. 32 Diabetic dyslipidaemia is related to the impact of insulin resistance on metabolic pathways and systemic changes related to diabetes, such as renal disease. HDL-C was the only lipid parameter to be independently associated with carotid IMT in a cohort of young adults with type 2 diabetes 33 and, in another trial, low-HDL-C elevation secondary to statin use was related to the IMT decrease.34,35
Diabetes leads to impaired reverse cholesterol transport through both reduced HDL concentrations and HDL dysfunction. Thus, a more pronounced inverse association of the HDL2-C/HDL3-C ratio with cIMT could be explained in some pathways: inhibition of lipoprotein lipase, where less apolipoprotein A-I (apo A-I) is liberated from chylomicrons, decreasing HDL formation in serum; ABCA1 down-regulation on the surface of visceral adipocytes, also reducing HDL formation; 36 lecithin–cholesterol acyltransferase (LCAT) dysfunction, 37 which lowers cholesterol esterification – decreases the maturation of HDL3 to HDL2; and finally, increases in CETP activity, which promote triglyceride enrichment of HDL particles, rendering them better substrates for lipolysis and catabolism by hepatic lipase.
Indeed, studies have shown that, in patients with diabetes, raising HDL-C levels by drugs other than statin is not accompanied by improvement or reverse cholesterol transport. 38
Thus, considering that the infusion of reconstituted HDL in patients with coronary heart disease (CHD) led to changes in inflammatory markers, cholesterol efflux and plaque conformation, 39 perhaps this fact calls attention to need to clinically evaluate the functionality of HDL instead of its level individually, especially in the diabetic milieu. Moreover, although CETP inhibitors are known to increase asymmetrically HDL-C subfractions, 40 as well as statin use, which alters the HDL composition and HDL3-C levels, 41 it is unclear whether the variability of the HDL2-C/HDL3-C ratio may result in CV benefit.
Strengths of this study include a large cohort, a method of direct subfraction measurement and the use of a computer-aided protocol for cIMT measurements that provided reliable and accurate cIMT data. Our study also has some limitations. As a cross-sectional study, the described associations do not confirm causation. Also, cIMT was used instead of plaque burden, and total plaque area may be more representative of atherosclerosis than cIMT. However, the cIMT may be more reversible, which may explain the weaker association with CV disease events than the plaque. 28 We are also unable to evaluate the antiatherogenic effects of HDL and its subfractions directly mechanistically.
In conclusion, in a unicentric, cross-sectional study of a Brazilian sample, HDL-C and its subfractions HDL2-C and HDL3-C are inversely associated with cIMT. In participants with diabetes, the negative association is limited to the HDL2-C/HDL3-C ratio, which has a statistically significant association even when adjusted for HDL-C.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The ELSA-Brasil baseline study was supported by the Brazilian Ministry of Health (Science and Technology Department) and the Brazilian Ministry of Science and Technology (Financiadora de Estudos e Projetos and CNPq National Research Council; Grant Nos 01 06 0010.00 RS, 01 06 0212.00 BA, 01 06 0300.00 ES, 01 06 0278.00 MG, 01 06 0115.00 SP and 01 06 0071.00 RJ).
