Abstract
Complex fractionated atrial electrograms (CFAEs) are related to atrial fibrosis, but their ablation has not yet shown superiority. The aim of the study was to compare, in terms of clinical outcome, two strategies of paroxysmal atrial fibrillation (AF) ablation in patients with type 1 diabetes mellitus (DM): pulmonary vein isolation (PVI) vs. PVI + CFAEs. Compared to an historical population of patient with paroxysmal AF and without DM, a higher percentage of patients with DM showed more than 25% of atrial area interested by CFAEs (study population, 58% vs historical group, 15%; p < 0.05). In PVI group, recurrences rate was similar in patients with HbA1c ⩽ 7.5% vs HbA1c > 7.5% (30% vs 22%; p = not significant), but a greater AF burden was observed in patients with HbA1c > 7.5% (6 ± 2 vs 1 ± 2; p < 0.05). In hazard ratios analysis PVI+CFAEs seems more effective than PVI alone in patients with HbA1c > 7.5% (hazard ratio, 1.28; p < 0.05), more than 25 years from DM diagnosis (hazard ratio, 1.25; p < 0.05) and more than five AF episodes/year (hazard ratio, 1.2; p < 0.05). Type 1 DM patients had complex atrial ‘substrate’, as documented by wider CFAEs areas. Despite this, 1-year follow-up recurrence rate was similar between two ablation approaches (PVI 27% vs. PVI+CFAEs 21%; p = not significant). In our study, only specific subgroups, like patients with disglycaemic state (HbA1c > 7.5%), long diabetes mellitus history and high AF burden, benefit from PVI+ CFAEs approach.
Keywords
Introduction
Diabetes mellitus (DM) has become a pandemic disease in the western world as well as in developing countries. DM may increase the incidence of atrial fibrillation (AF) and, when combined with other risk factors, the incidence of stroke and thromboembolism. The mechanisms of AF associated with DM are atrial electrical, structural and autonomic remodelling and insulin resistance. 1 Atrial structural remodelling remains the major substrate for DM-related AF. Kato et al. 2 demonstrated that the interatrial conduction disturbance resulting from the DM-related atrial interstitial fibrosis would be a major substrate for the initiation of AF. The polypeptide cell factor with kinds of biological activities, transforming growth factor (TGF)-β, is involved in a disproportionate increase in collagen and excessive extracellular matrix (ECM) deposition, and it is associated with atrial fibrosis, suggesting that the activation of TGF-β pathway may play an important role in DM-induced atrial structural remodelling. 3 Furthermore, diabetes produced myocardial stiffness before the development of myocardial fibrosis in association with increased formation of collagen-associated advanced glycation end products (AGEs) 4 and may change the expression and distribution of connexins resulting in atrial structure remodelling and the conduction abnormality common in AF. 5 It is well known that also other extra-cardiac diseases, like inflammatory bowel diseases, through inflammation factors expression could play a crucial role in the development and maintenance of AF. 6
Type 1 DM is more often associated with glycaemic fluctuations that increase frequency of AF.7,8 Hypoglycaemia induces sympathetic activation, leads to a catecholamine surge, increases cardiac workload and induces electrocardiographic changes; numerous case-reports demonstrate hypoglycaemia-associated AF. 9 Furthermore, hyperglycaemic state enhances the expression of inflammation markers (i.e. C-reactive protein and interleukin-6) that are linked to the initiation and perpetuation of AF.10,11 Fluid retention, which is associated with glycaemic fluctuations, is a recognized predisposing factor for AF via renin–angiotensin–aldosterone system activation and increasing atrial stretch. 12
AF ablation has become an established therapy for maintenances of sinus rhythm in patients with symptomatic paroxysmal AF and is more effective than antiarrhythmic drug (AAD) therapy with similar complication rate. 13 This is primarily achieved through pulmonary vein isolation (PVI), requiring complete isolation for full effectiveness. 14 In non-paroxysmal forms, more extensive ablations (i.e. substrate modification) may be required. Overall, such alternative ablation procedures seem to be more effective than PVI, even if their success rates appear extremely variable, and in many cases, the procedure is complex and time-consuming. In particular, studies analysing complex fractionated atrial electrograms (CFAEs) ablation alone and as an adjunct to left atrial linear lesions and/or PVI have demonstrated contrasting results.15–17
Moreover, in conditions with increased atrial remodelling like DM, it is known that anisotropy of conduction, anatomic barriers and interstitial fibrosis generate electrogram fractionation. 18
The aim of this randomized study was to assess clinical outcome comparing two strategies of catheter ablation (PVI vs PVI + CFAEs) for paroxysmal AF in type 1 DM patients.
Methods
Study population and design
In total, 64 patients with type 1 DM were referred to our institution for AF ablation: 32 were randomized to PVI and 32 to PVI + CFAEs ablation.
Inclusion criteria were as follows: type 1 DM and symptomatic paroxysmal AF refractory to at least 1 AAD; age >18 or <75 years; left ventricular ejection fraction (LVEF) > 30%; left atrial size < 55 mm; more than 1 year life expectance; no prior AF ablation; and no prior cardiac surgery.
Primary endpoint was defined as an electrocardiogram (ECG)-confirmed AF/AT recurrence lasting at least 30 s and occurred within 12 months from the ablation procedure after blanking period (90 days). As secondary endpoints, bleeding (> 2 g Hb loss), hospitalization rate and thromboembolic events (transient ischaemic attacks, strokes, pulmonary thromboembolisms and deep venous thrombosis) were analysed.
The study population was also compared to an historical population (100 patients) of non-DM patients undergone catheter ablation (PVI) for paroxysmal, symptomatic, drug-refractory AF in our centre. Study design is resumed in Figure 1.
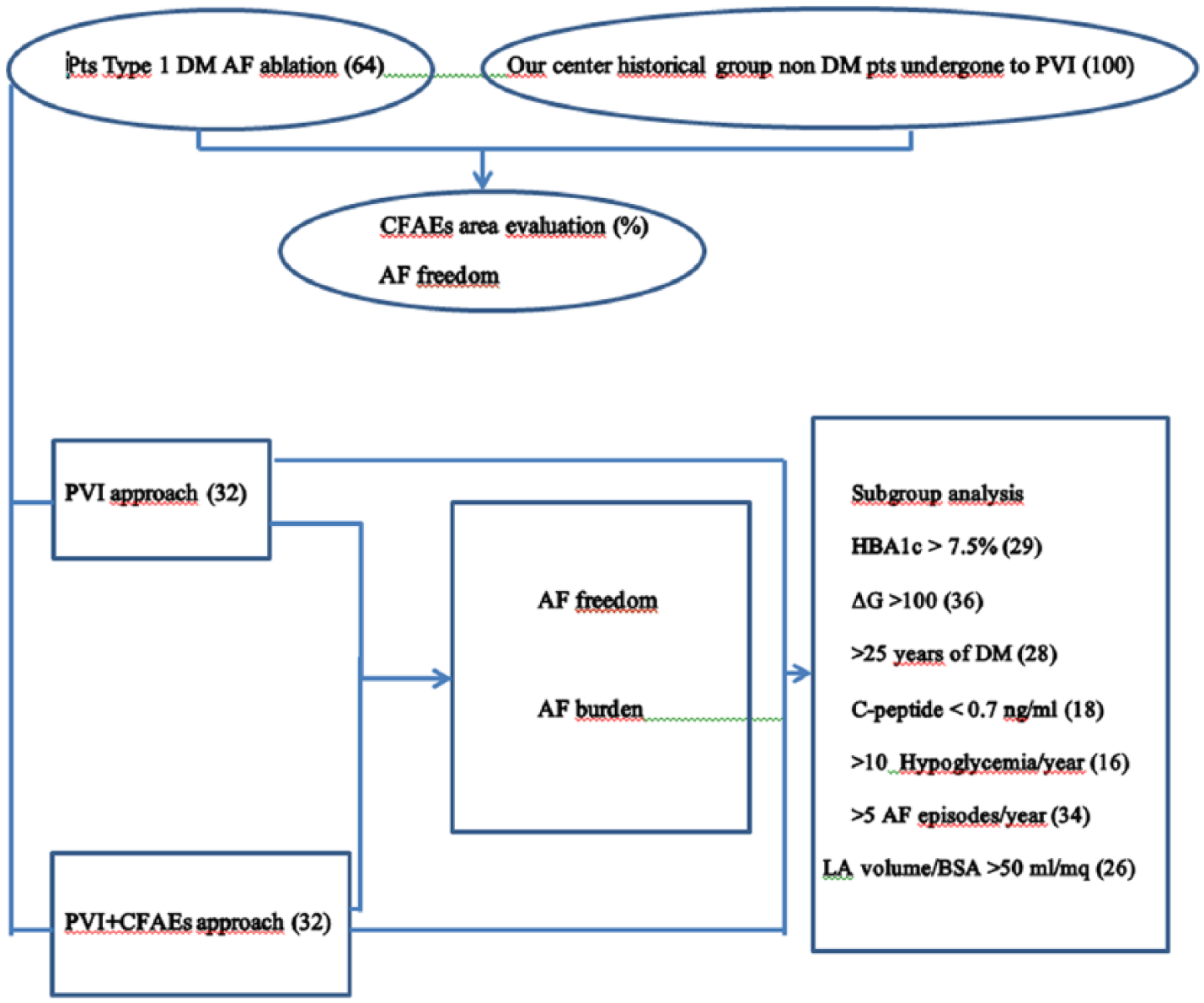
Graphic flowchart of study design.
Trans-thoracic echocardiography was performed within 1 month period preceding the procedure according to the recommendations of the American Society of Echocardiography. 19 Blood samples were obtained the morning of the procedure, at 6 h and at 18–24 h after the procedure. On September 2015, our internal review board (IRB) approved the research study. All patients provided written informed consent before the study.
EP study
All AADs were stopped at least five half-lives before ablation. Amiodarone was discontinued at least 1 month before the procedure. All patients underwent trans-oesophageal echocardiography before the procedure to exclude left atrial appendage thrombosis.
Procedure was performed in a fasting state using mild sedation. After trans-septal catheterization, a 3.5-mm open irrigated-tip catheter (THERMOCOOL or SMARTTOUCH, Biosense Webster, Inc., Irvine, CA, USA) was used for mapping and ablation. Real-time three-dimensional (3D) left atrial and right atrial maps were reconstructed using a non-fluoroscopic mapping system (CARTO, Biosense Webster, Inc., Irvine, CA, USA). Radiofrequency (RF) energy was delivered with a range of power of 20–30 W (reduced to 20–25 W when ablating on the posterior wall) and temperature was limited to 43°C.
Areas of fractionated atrial electrograms or rapid atrial activity were tagged using CARTO CFAEs analysis software. The software for CFAEs analysis can display, in addition to the local activation time, unipolar and bipolar voltage, propagation and isochronal map, three additional kinds of maps, according to the analysis of the intra-cardiac signal of each acquired point. These three new maps are as follows: (1) shortest complex interval (SCI) that displays the value of the shortest interval between two consecutive deflections, according to an arbitrary predefined setting of the voltage and the distance between consecutive peaks; (2) average complex interval (ACI) that displays the average value for all intervals considered for each point; (3) interval confidence level (ICL) that displays the total amount of intervals counted for each point, with the possibility to show two different colour tags for points with high (red) or medium (blue) level of fragmentation. The threshold of ICL that distinguishes points with high ICL [high fragmentation (HF)] and medium ICL [medium fragmentation (MF)] can be determined for each patient. For this study, we considered as high ICL the points with ⩾20 intervals and medium ICL the points with 10–20 intervals. These cutoffs are arbitrary and have been determined on the basis of a preliminary analysis of a bi-atrial mapping, in order to avoid the inclusion of points with few or transient CFAEs. 20 The CARTO map used to determine the presence of CFAEs was the ICL map. The setting used for this analysis was the best obtained in terms of sensitivity and specificity in the identification of CFAEs. Each point was automatically classified by the software into three categories: (1) HF; (2) MF; and (3) not fragmented. The setting used was low-voltage threshold of 0.05 mV, high-voltage threshold of 1 mV, minimum duration of 15 ms and maximum duration of 30 ms. Only points with ICL > 10 were considered in order to avoid the inclusion of points with few intervals measured. The mean value of the fragmented intervals (FF intervals) obtained from the points taken from each region of both atria was used for the analysis.
Glucose monitoring
HbA1c level was measured in each patient every 2 months for 12 months prior to the ablation. In addition, instant glucose levels were obtained by conventional glucometer and/or continuous glucose-monitoring (CGM) devices at least 8 times/day in the week before and after the ablation and every 30 min during the procedure. A dedicated endocrinologist performed evaluation of glucose control and any changes in insulin therapy. Episodes of hypoglycaemia were defined as a glucose level <70 mg/dL or as the occurrence of symptoms related to low blood glucose level (with documented low blood glucose at the time of symptoms) and symptom resumption after normalization of blood glucose values.
Follow-up
After the ablation, patients started anticoagulant and antiarrhythmic therapy. After 6 months, if AF recurrences were not documented, anticoagulant treatments were discontinued unless other major risk factors were present. The antiarrhythmic therapy was continued for at least 3 months. After this period, the decision to continue antiarrhythmic medications was based on AF occurrence or the presence of frequent and/or repetitive atrial ectopic beats. Every patient was included in an outpatient follow-up plan (1, 3, 6, 9 and 12 months post-ablation) with clinical assessment, ECGs and 24-h Holter ECGs. Additional follow-ups were planned in case of symptoms. Clinical success was defined as absence of documented arrhythmia recurrences (>30 s), withdrawal of AAD and no need of re-ablation after the blanking period (3 months). All patients were instructed to daily assess their pulse and to confirm on ECG any suspected recurrence of arrhythmia.
Fasting glycaemic values of the 30 days prior follow-ups (measured either with CGM or conventional glucometer) were also analysed together with insulin therapy changes occurred.
Statistical analysis
The data were analysed using SPSS version 20.0 statistical software. The variables were expressed as mean values ± standard deviation (SD) or percentage. Comparisons between groups were performed by Student’s t-test for normally distributed variables, by Mann–Whitney U test for those not normally distributed and by Fisher test for categorical variables. Cox’s proportional hazards model was used to assess the association of variables with the considered endpoint (AF-free survival). Hazard ratios (HRs) were given with their 95% confidence intervals (CIs). The HR refers to a unit increase in the variable. The overall AF-free survival, estimated with the Kaplan–Meier method, was defined as the time from ablation until the date of first AF recurrence or the end of the observation. The comparisons were made by log-rank test.
Results
Baseline demographic, clinical and procedural characteristics are shown in Table 1. The two groups of patients were similar in terms of age, sex, underling heart disease, number of AADs tested and time from DM diagnosis. Percentage of PVs isolated was similar in the two groups.
Study population Vs Historical population comparison. Values expressed as n (%) and mean ± standard deviation.
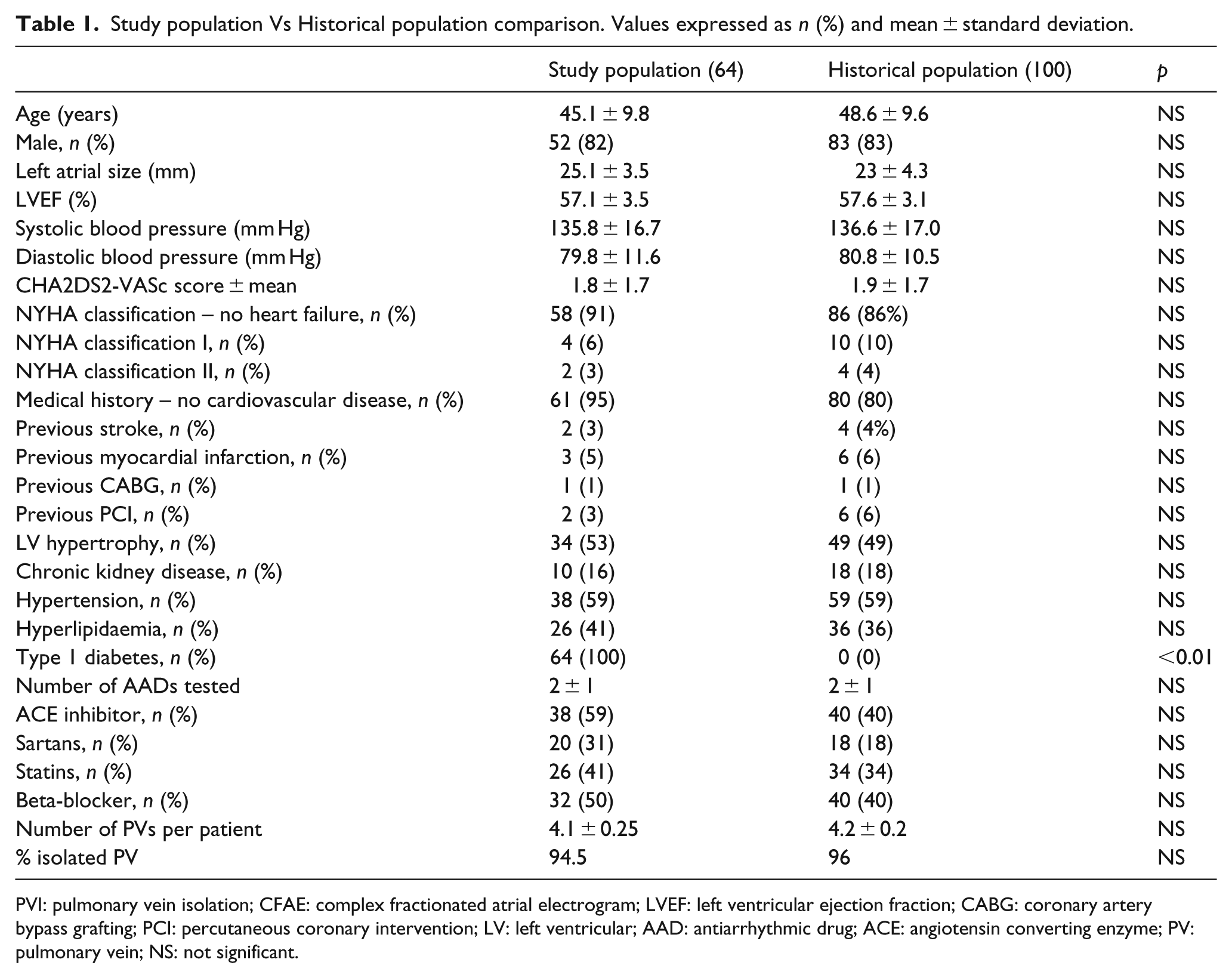
PVI: pulmonary vein isolation; CFAE: complex fractionated atrial electrogram; LVEF: left ventricular ejection fraction; CABG: coronary artery bypass grafting; PCI: percutaneous coronary intervention; LV: left ventricular; AAD: antiarrhythmic drug; ACE: angiotensin converting enzyme; PV: pulmonary vein; NS: not significant.
No major complication occurred. Two patients in the PVI group and three patients in the PVI + CFAEs group experienced a mild femoral haematoma that did not require any intervention. As expected, PVI + CFAEs ablation procedures, compared to PVI alone, were more time-consuming and required more RF applications.
In the study population (DM patients), compared to a historical population of non-DM patients undergone paroxysmal AF ablation (Table 2), a significant higher percentage of patients showed more than 25% of atrial area interested by CFAEs (study population, 58% vs historical group, 15%; p < 0.05) (Figure 2(a) and (b)). A wider CFAEs area was reported in DM patients with HbA1c constantly above 7.5% during the 12 months preceding ablation (HbA1c > 7.5%, 41% vs HbA1c ⩽ 7.5%, 24%; p < 0.05) (Figure 3).
Clinical and procedural characteristics of study population. Values expressed as n (%) and mean ± standard deviation.
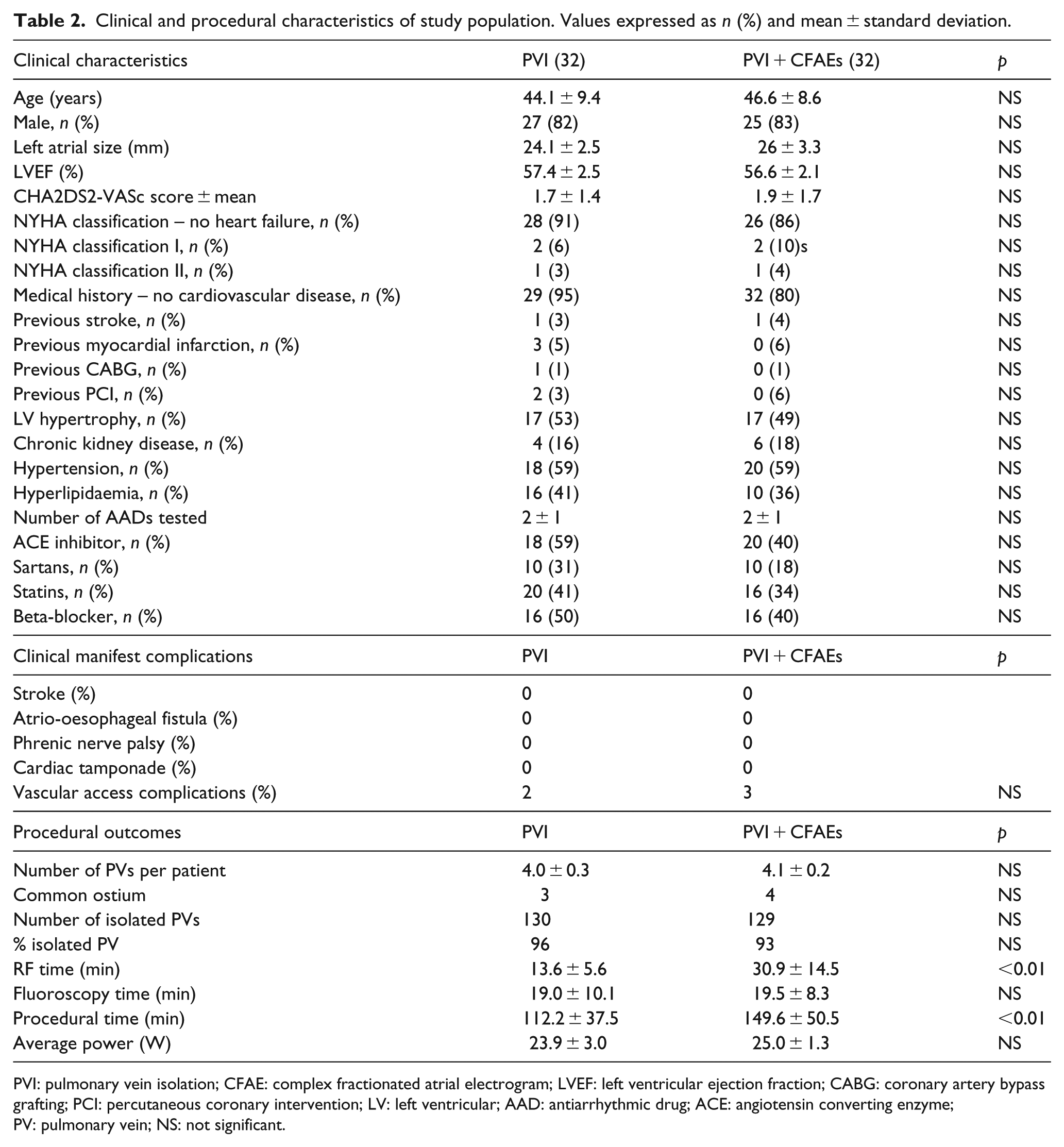
PVI: pulmonary vein isolation; CFAE: complex fractionated atrial electrogram; LVEF: left ventricular ejection fraction; CABG: coronary artery bypass grafting; PCI: percutaneous coronary intervention; LV: left ventricular; AAD: antiarrhythmic drug; ACE: angiotensin converting enzyme; PV: pulmonary vein; NS: not significant.
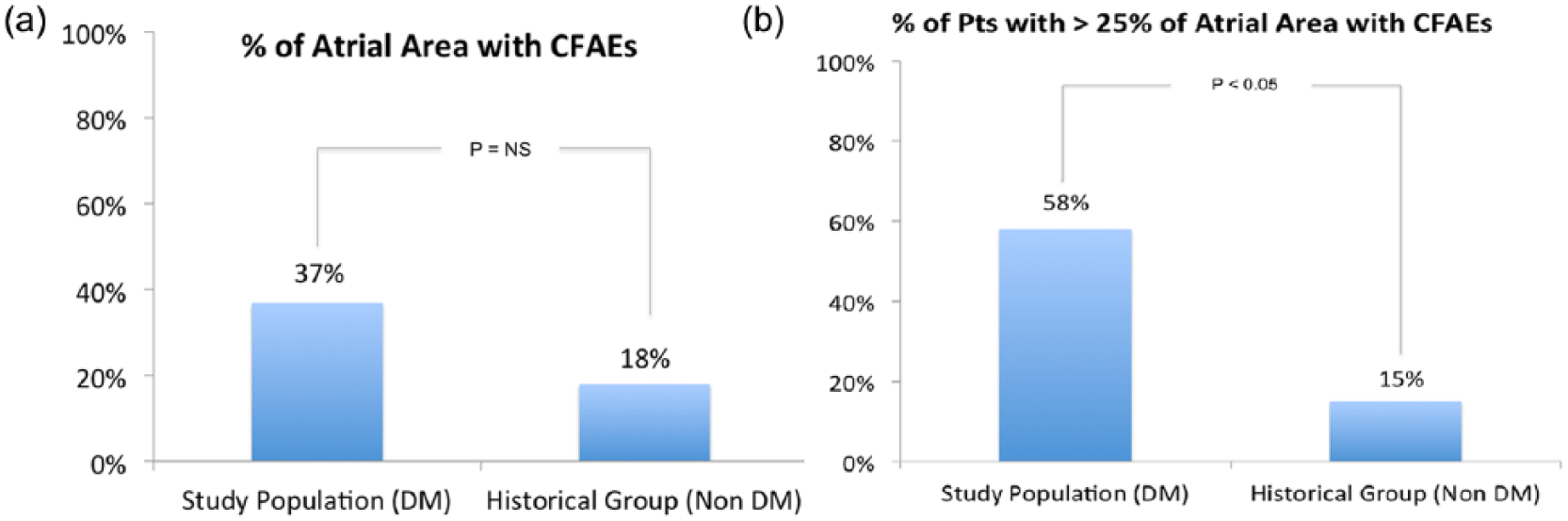
(a) In left panel, % of atrial area interested by CFAEs in the study population and in the historical group. (b) In right panel, % of patients with > 25% of atrial area interested by CFAEs (chosen as the median value between the two groups).
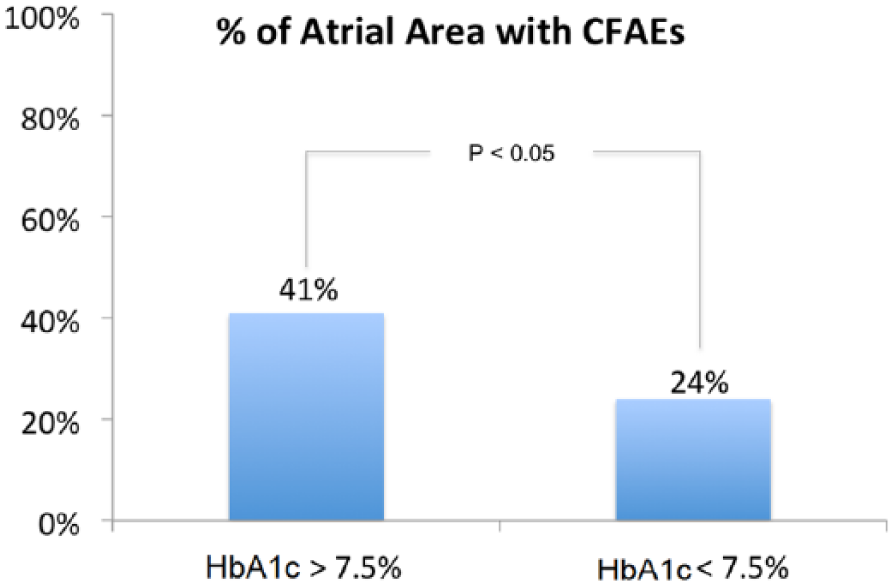
Percentage of atrial area interested by CFAEs in study population in patients with Hb1Ac constantly > 7.5% compared to those with HbA1c < 7.5% during the 12 months before ablation.
Success rate of catheter ablation in study population was similar to that one observed in an historical population of non-DM patients ablated at our centre [study population, 83% vs historical group, 85%; p = not significant (NS)] (Figure 4).
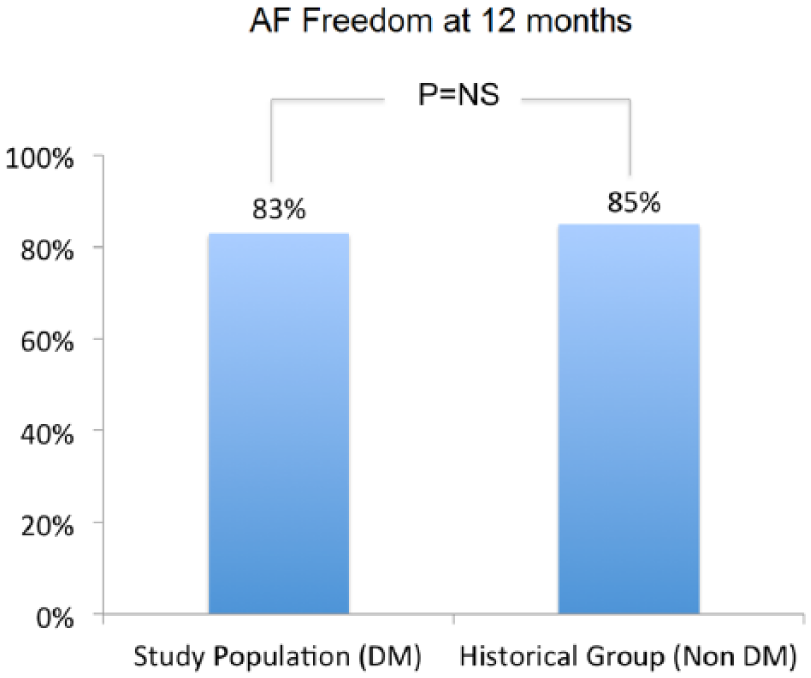
AF freedom during 12 months follow-up in study population and in historical group.
In study population, during follow-up, the recurrence rate was similar in the two groups (PVI, 27% vs PVI + CFAEs, 21%; p = NS) (Figure 5(a) and (b)). In patients with recurrences, AF burden expressed as number of AF episodes/patients was similar in the two groups (PVI, 4 ± 2 vs PVI + CFAEs, 3 ± 2; p = NS) (Figure 6).
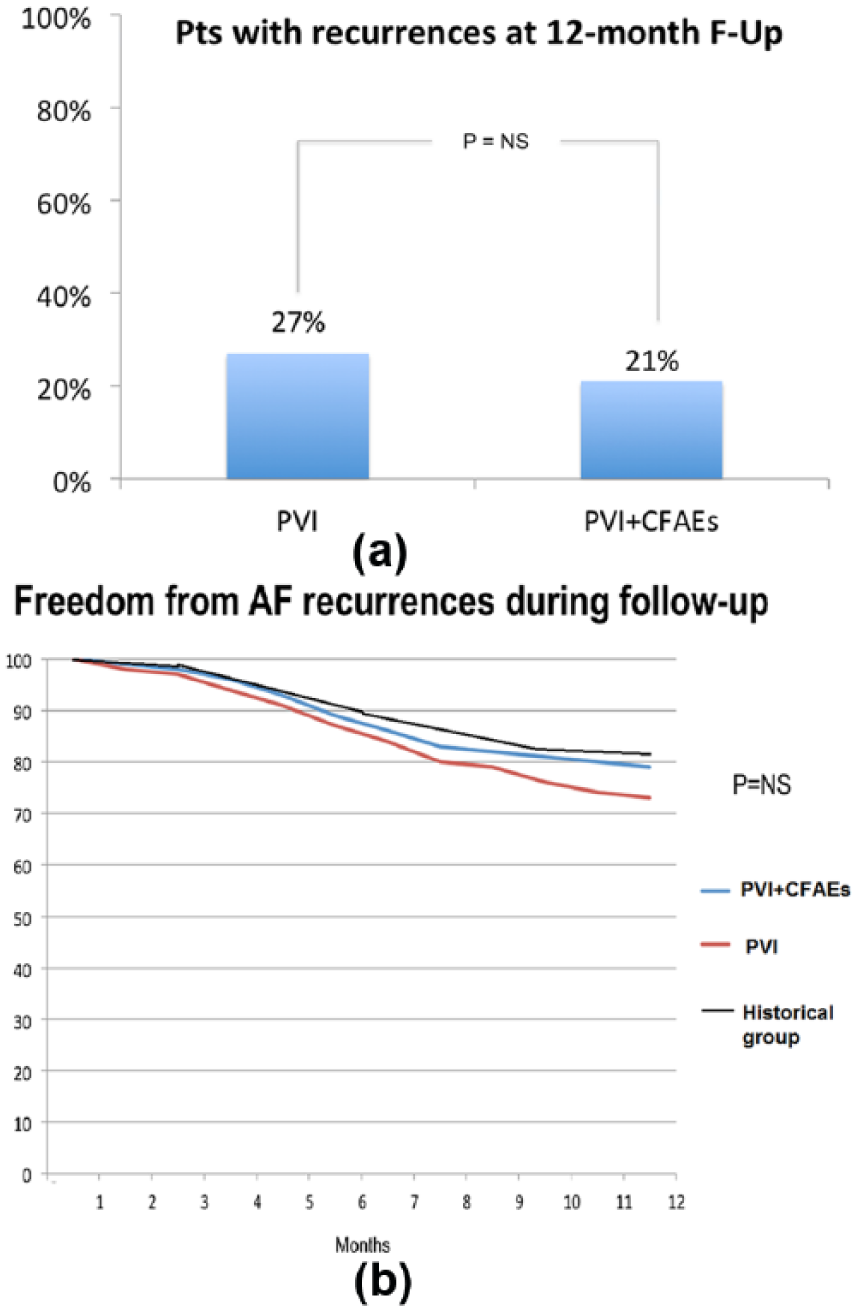
(a) In upper panel, AF recurrence rate during 12 months follow-up in study population. (b) In lower panel, AF-free survival curves (PVI + CFAEs group, PVI group and historical group).
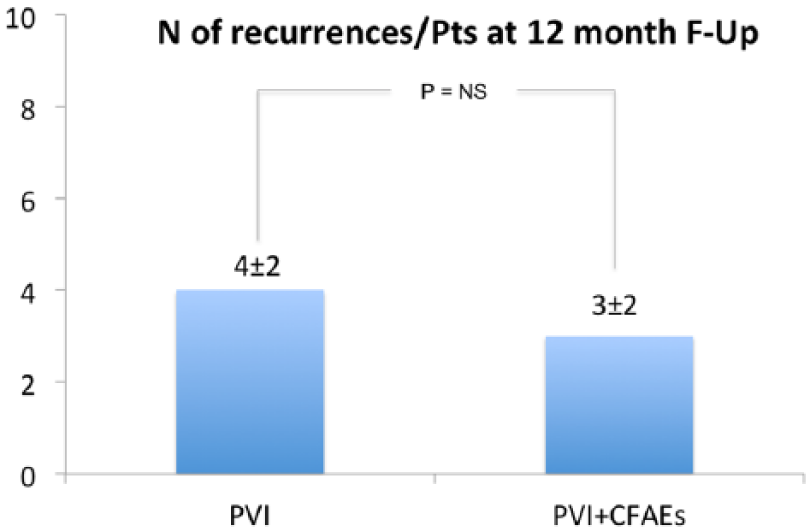
AF burden in study population with recurrences (number of atrial fibrillation, atrial flutter and atrial tachycardia >30 s recurrences).
In the PVI group, recurrences occurred with similar rate in patients with HbA1c ⩽ 7.5% compared to those with HbA1c > 7.5% (HbA1c > 7.5%, 30% vs HbA1c ⩽ 7.5%, 22%; p = NS), but a greater AF burden was observed in those with HbA1c > 7.5% (6 ± 2, HbA1c > 7.5% vs 1 ± 2, HbA1c ⩽ 7.5%; p < 0.05) (Figure 7(a) and (b)). This was not the case for PVI + CFAEs group (Figure 8(a) and (b)).
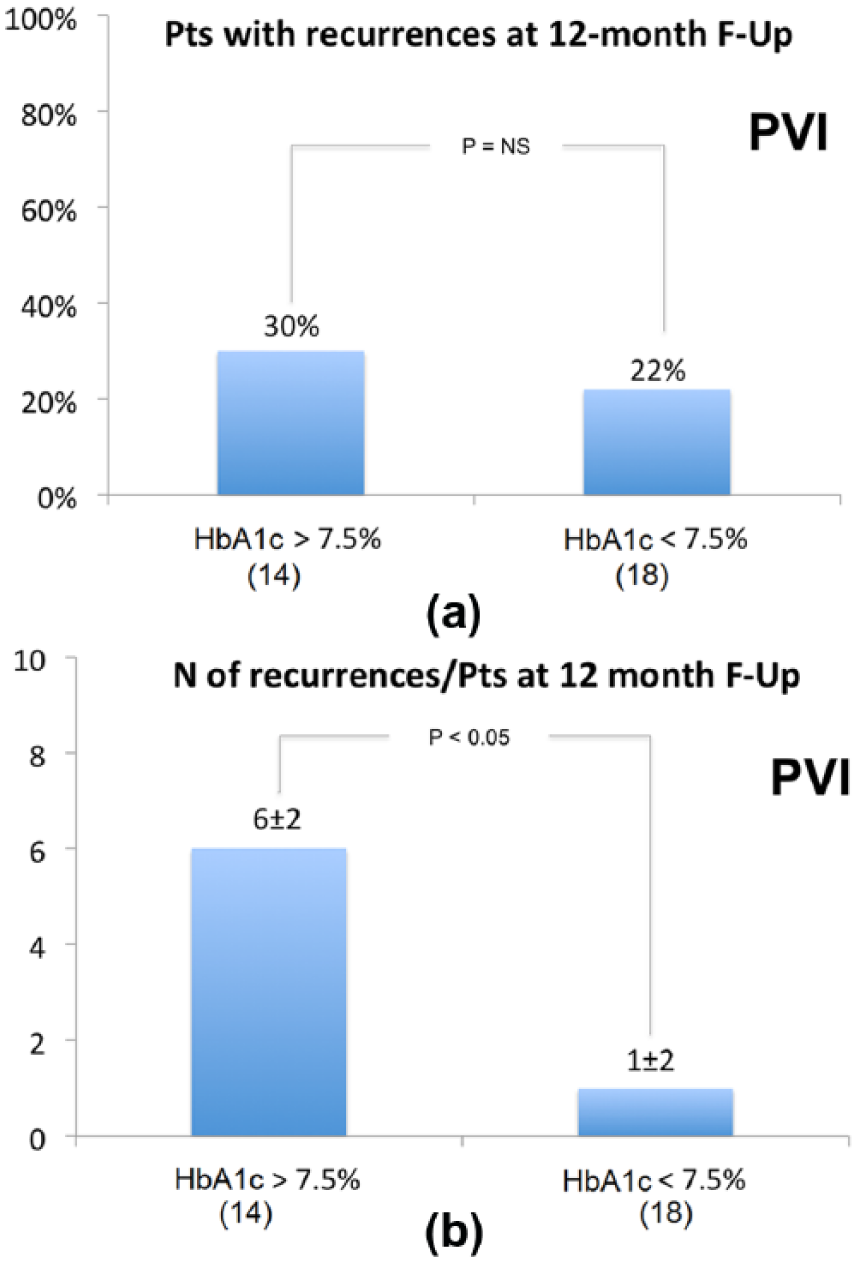
(a) In upper panel, AF recurrence rate during 12 months follow-up in PVI group according to HbA1c values. (b) In lower panel, AF burden in PVI patients with recurrences (number of atrial fibrillation, atrial flutter and atrial tachycardia >30 s recurrences) according to HbA1c values.
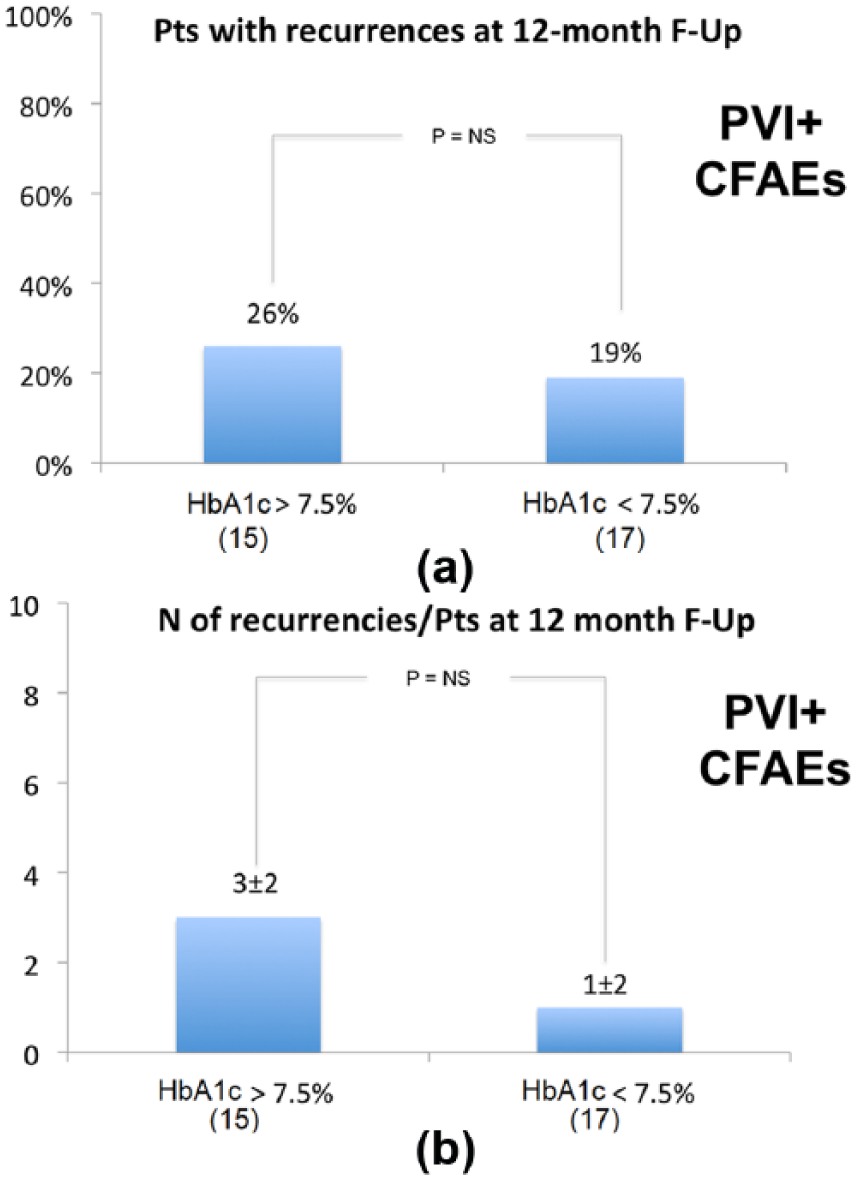
(a) In upper panel, AF recurrence rate during 12 months follow-up in PVI + CFAEs group according to HbA1c values. (b) In lower panel, AF burden in PVI + CFAEs patients with recurrences (number of atrial fibrillation, atrial flutter and atrial tachycardia >30 s recurrences) according to HbA1c values.
Hazard ratios for primary endpoint are shown in Figure 9. A significant benefit in terms of outcome was observed with PVI + CFAEs in patients with HbA1c > 7.5% (HR, 1.28; CI, 1.11–1.45; p < 0.05), more than 25 years from DM diagnose (HR, 1.25; CI, 1.09–1.50; p < 0.05) and more than five AF episodes/year (HR, 1.2; CI, 1.03–1.55; p < 0.05). In patient with >100 mg/dL glucose level fluctuations (Δ > 100 mg in Figure 8), documented by conventional glucometer monitoring or CGM during the week before and after the ablation and every 30 min during the procedure, and no significant difference between PVI and PVI + CFAEs group was observed.
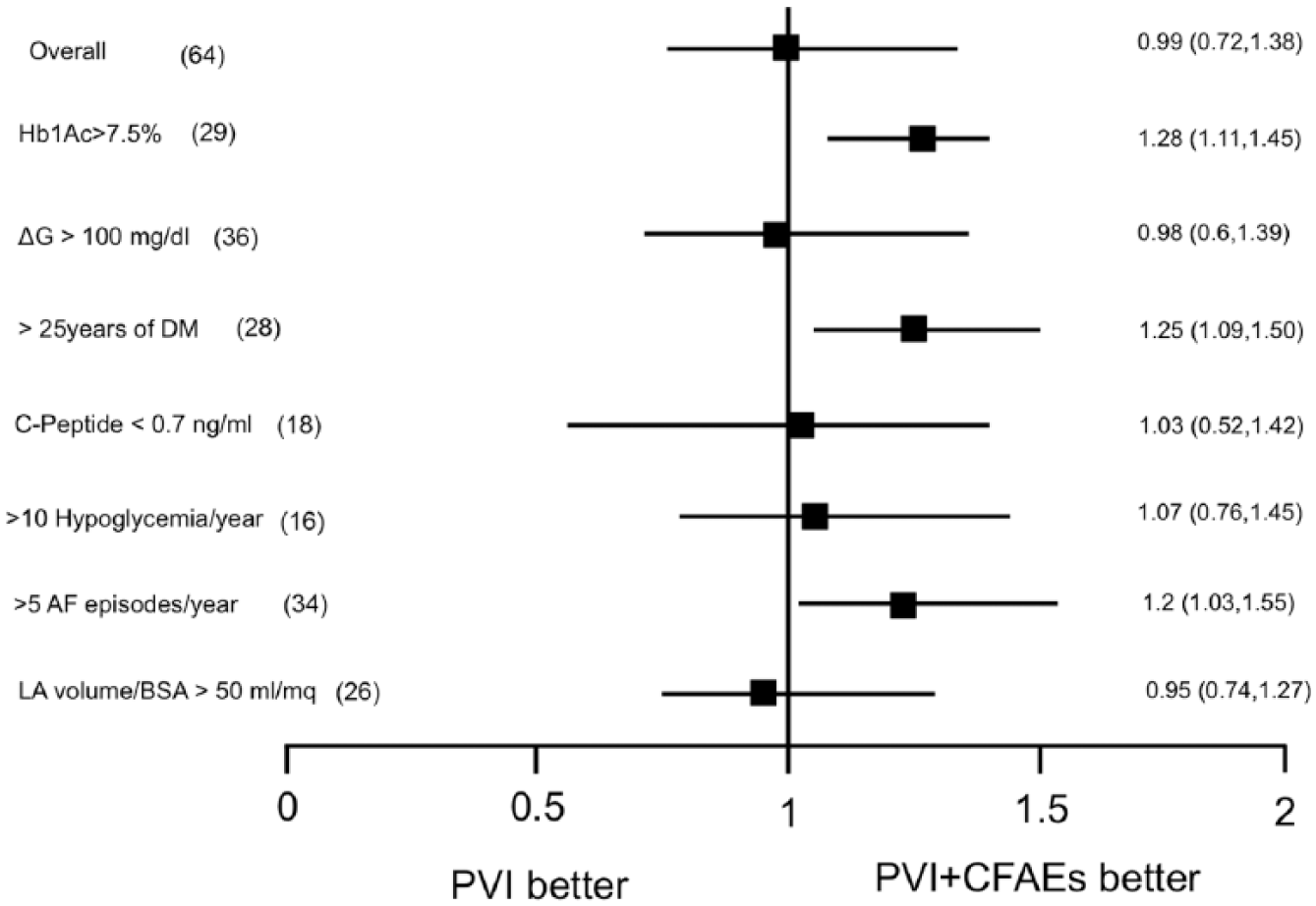
Hazard ratios for primary endpoint in specific subgroups.
Discussion
CFAEs areas as well as sites showing rapid, regular signals [so-called dominant frequency (DF) sites or ‘rotors’] have been proposed as markers of ‘substrate’, and clinical results have shown some promises when these targets are ablated.21,22 Yet, we still understand remarkably little about what these targets mechanistically represent, especially in the human situation. In animal isolated preparation AF models, rotor sites have been demonstrated using optical mapping techniques, 23 but existence of rotors in human model has never been definitively proven due to the limited resolution of our mapping techniques. However, CFAEs have been shown to exist immediately adjacent to rotor sites, as result of collision of wavefronts emerging from the driver sites. 24 Seminal work by Konings et al. 25 has shown in human-induced AF that CFAEs occurred in regions of slow conduction or at pivot points of wavefront perpetuation. This is supported by further evidence that CFAEs may be associated with sites of structural remodelling and microscopic fibrosis. 26 However, none of these observations directly answer the question whether CFAEs sites are direct contributors or passive bystanders in AF process. Human AF mapping, for example, has shown that DF sites occur predominantly in the pulmonary vein (PV) antra in paroxysmal AF, while non-PV locations are much more common in persistent AF, mirroring our knowledge of the location of AF triggering foci. 27 However, which of these CFAEs and DF sites need to be targeted and to what extent remain to be defined. Also unclear is the CFAEs role in patients with atrial fibrosis observed at high-density magnetic resonance imaging. Even Jadidi et al. 28 have described an inverse relationship between fractionated electrograms and atrial fibrosis in persistent AF.
An important consideration of Nademanee for successfully eliminating AF with CFAEs ablation has resulted in much subsequent clinical investigation before mechanistic questions have been answered.27,29 Studies by other investigators, however, have shown that CFAEs ablation alone is not effective for AF. 30 The results of our study are in line with the ones reported by other groups who perform regularly CFAEs ablation.17,21,29 However, it is important to underline that most of the studies published so far on CFAEs ablation included patient with persistent AF and type 2 DM, in which the more complex substrate could be explained by the longer duration of arrhythmia itself (‘AF begets AF’) and the presence of co-morbidities together with a delay in the type 2 DM diagnosis. It is not surprising that CFAEs ablation in addition to PVI did not provide a higher benefit in terms of AF recurrences, even in our study population. This reflects the recent observation of the STAR-AF II trial that in persistent AF patients treated with PVI the additional ablation of CFAEs did not reduce the recurrence rate. 31
Although, in our study, we include patients with paroxysmal forms of AF, it has to be noted that patients with DM1 have a more complex atrial substrate, usually associated with persistent AF forms, as demonstrated by the presence of a wider CFAEs area compared to the historical group [>25%, chosen as the median value between the two groups, in order to identify indirectly a significant fibrotic atrial cardiomyopathy (FACM)]. 32
Even if CFAEs ablation is not contemplated in current AF ablation guidelines, according to the results of our study, patients with long-lasting DM, with more than five AF episodes/year and with HbA1c > 7.5% may benefit from longer and more complex procedure, with the same risk of complications. These findings are the most important results of our study. The role of an uncontrolled glycaemic state in myocardial substrate alteration is emphasized, and substrate modification with CFAEs ablation approach could play an important role in AF free survival.
It is well known that elevated HbA1c values have been associated with endothelial dysfunction, and a reduction of its values improves vascular properties such as aortic compliance and flow-mediated dilation. 33 Alterations of endothelial function predispose to AF by increasing atrial stretch, activation of inflammatory markers and favouring atrial fibrosis and AF development. 34 In this study, patients with HbA1c > 7.5%, treated with PVI only approach, had a wider CFAEs area and a higher recurrences burden compared to patients with HbA1c < 7.5%. That kind of observation was not confirmed in patients experiencing acute glucose fluctuations (>100 mg/dL), in which the two different ablation strategies had a similar outcome. This is different from what has been observed in acute events such as ischaemic stroke, in which instantaneous blood glucose values have an important prognostic value. 35
Two previous studies linked glycaemic indexes to incident AF in patients with DM.36,37 In a population-based case-control study, Dublin et al. 36 found a 14% increased risk of incident AF for every 1% increase in HbA1c. In the Atherosclerosis Risk in Communities (ARIC) cohort, Huxley et al. 37 found HbA1c levels to be positively and independently correlated to AF incidence in patients with and without DM. These results, combined with our study observations, suggest that catheter ablation combined with optimal glycaemic control may further increase the rate of maintenance of sinus rhythm and reduce the need for re-ablation. Maintenance of ‘eu-glycemic state’ in accordance with guidelines may decrease the incidence of AF. 1
These observations underline the important role of the ‘dis-glycemic state’ along with reactive oxygen species, interleukins and intra-cellular transducing signal proteins 38 in the pathogenesis of AF in DM patients and open new research perspectives both in the field of the ‘upstream therapies’ (i.e. rosveratrol or probucol39,40) able to prevent atrial remodelling and of more complex AF ablation procedures (like CFAEs ablation) to change atrial myocardium substrate.
Limitations
The small sample size is definitely a limitation of the study. Further multicentre studies are needed to confirm these encouraging data. The control group (historical group) analysis was carried out retrospectively to ensure an adequate number of controls. However, experienced operators have carried out all procedures, so learning curve does not influence the study.
To evaluate recurrences, patients were instructed to daily assess their pulse and to access to the clinic for ECG confirmation: no continuous rhythm-monitoring devices (i.e. implantable loop recorder) were used.
Conclusion
This is the first randomized study that investigated atrial remodelling in type 1 DM humans using electroanatomical mapping system. Type 1 DM patients had a more complex atrial ‘substrate’ than non-DM patients, and it is documented by wider CFAEs areas. Despite this, in study population at 1-year follow-up, the recurrence rate was similar between CFAE-treated patients and PVI-only-treated patients. In our study, only specific subgroups of type 1 DM patients, like patients with documented disglycaemic state (HbA1c > 7.5%, long DM history) benefit from more complex ablations.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this study, we collected ethical approval from our internal review board.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Informed consent was obtained from all individual participants included in the study. All data have been anonymized before data collection.
