Abstract
Aim:
RhoA/Rho-associated kinase and arginase are implicated in vascular complications in diabetes. This study investigated whether RhoA/Rho-associated kinase and arginase inhibition protect from myocardial ischaemia–reperfusion injury in type 1 diabetes and the mechanisms behind these effects.
Methods:
Rats with streptozotocin-induced type 1 diabetes and non-diabetic rats were subjected to 30 min myocardial ischaemia and 2 h reperfusion after being randomized to treatment with (1) saline, (2) RhoA/Rho-associated kinase inhibitor hydroxyfasudil, (3) nitric oxide synthase inhibitor NG-monomethyl-
Results:
Myocardial arginase activity, arginase 2 expression and RhoA/Rho-associated kinase activity were increased in type 1 diabetes (p < 0.05). RhoA/Rho-associated kinase inhibition and arginase inhibition significantly reduced infarct size in diabetic and non-diabetic rats (p < 0.001). The cardioprotective effects of hydroxyfasudil and N-omega-hydroxy-nor-
Conclusion:
Inhibition of either RhoA/Rho-associated kinase or arginase protects from ischaemia–reperfusion injury in rats with type 1 diabetes via a nitric oxide synthase–dependent pathway. These results suggest that inhibition of RhoA/Rho-associated kinase and arginase constitutes a potential therapeutic strategy to protect the diabetic heart against ischaemia–reperfusion injury.
Introduction
It is well known that nitric oxide (NO) formed by endothelial nitric oxide synthase (NOS) plays a pivotal role in cardiovascular regulation.
1
Emerging evidence suggests that a decrease in NO bioavailability is a key factor behind cardiovascular disease including ischaemia–reperfusion (IR) injury associated with myocardial infarction
2
and vascular complication in diabetes.3,4 Although the signalling events leading to reduction in NO bioavailability remain to be fully clarified, recent data suggest that upregulation of RhoA/Rho-associated kinase (ROCK)
5
and arginase
6
is involved. Growing evidence suggests a pivotal role for ROCK in the pathophysiology of cardiovascular diseases associated with endothelial dysfunction and inflammation.
5
It has also been suggested that increased ROCK signalling events contribute to myocardial IR injury.
7
Interestingly, arginase which converts
Both the ROCK and arginase pathways are activated in diabetes mellitus – a disease associated with cardiovascular complications and poor outcome following acute myocardial infarction.4,16–18 Thus, ROCK and arginase activities are enhanced in the vasculature and have been proposed to be involved in the development of vascular dysfunction in diabetes.19–23 Considering that activated ROCK mediates upregulation of arginase that contributes to impaired vascular function in diabetes, 24 there seems to be an intriguing signalling pathway including ROCK-mediated activation of arginase leading to reduction in NO bioavailability and cardiovascular complications in diabetes. Moreover, it has been shown that the diabetic heart is more resistant to pharmacological and non-pharmacological (classical pre- and postconditioning) treatments aiming at reduction in IR injury. 25 In light of our recent observation showing that remote ischaemic perconditioning was ineffective in type 1 diabetic rats, 15 there is a need for identification of specific pharmacological strategies that protect from IR injury in diabetes.
Based on these observations, the aim of this study was to investigate whether inhibition of ROCK and arginase protects from IR injury in a rat model of type 1 diabetes and to identify the mechanisms behind these effects. We hypothesized that targeting ROCK has beneficial effects in the setting of myocardial IR in diabetes via a mechanism involving downregulation of arginase activity and increased NO bioavailability.
Methods
The study was approved by the regional Ethics Committee for laboratory animal experiments in Stockholm and conforms to the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996).
Induction of type 1 diabetes mellitus
Male Sprague–Dawley rats (Charles-River, Sulzfeld, Germany) of age 5–6 weeks were used in the study. Type 1 diabetes mellitus was induced by a single injection of streptozotocin (STZ, 55 mg kg−1) dissolved in sterile Dulbecco’s phosphate buffered saline (DPBS) in the tail vein under anaesthesia with isoflurane mixed with oxygen. 15 Only rats with blood glucose levels >15 mmol L−1 72 h after the STZ injection were considered to be diabetic and were included in the study. Healthy age-matched rats received an intravenous (iv) injection of vehicle (DPBS) and served as a non-diabetic control group. Blood glucose concentration was measured weekly and at the start of the of the IR experiment by FreeStyle Lite blood glucose monitoring system (Abbot Diabetes Care, Inc., CA, USA). The rats were used for further experiments 4–5 weeks after the injection of STZ or vehicle during which time they were given food and water ad libitum.
Experimental protocol
Rats were anaesthetized with pentobarbital (50 mg kg−1) intraperitoneal (ip) injection followed by an iv infusion of 5 mg kg−1 h−1 pentobarbital. Rectal temperature was maintained at 37.5°C–38.5°C by a heated operation table. Animals were tracheotomized and ventilated with air by a rodent pressure controlled ventilator (50–55 strokes min−1, 9 mL kg−1 tidal volume). The right carotid artery was cannulated and the catheter connected to a Statham blood pressure transducer for measurement of mean arterial pressure. Heart rate was determined from the arterial pressure curve. Haemodynamic parameters were continuously recorded on a personal computer equipped with PharmLab V5.0 (AstraZeneca R&D, Mölndal, Sweden). The left jugular vein was cannulated for administration of drugs. The heart was exposed via a left thoracotomy. A ligature was placed around the left coronary artery. After completion of the surgical preparation, the rats were allowed to stabilize for at least 15 min before being randomized into different groups (see below). Ischaemia was induced by tightening of the ligature around the left coronary artery. Successful occlusion was associated with cyanosis of the myocardial area at risk. Reperfusion was initiated after 30 min of ischaemia by removal of the snare and was maintained for 2 h. The reperfusion was associated with disappearance of the cyanotic colour of the myocardium.
In one part of the study aimed at investigating the role of ROCK, rats with and without diabetes were randomized to the following treatments given iv 15 min prior to coronary artery ligation: (1) saline (n = 5, non-diabetic rats; n = 5, diabetic rats); (2) ROCK inhibitor hydroxyfasudil (0.5 mg kg−1; n = 5, non-diabetic rats; n = 5, diabetic rats); (3) NOS inhibitor NG-monomethyl-
In an additional part of the study aimed at investigating the role of arginase, separate rats with and without diabetes were randomized to the following treatments given iv 15 min prior to coronary artery ligation: (1) saline (n = 7, non-diabetic rats; n = 14, diabetic rats); (2) arginase inhibitor N-omega-hydroxy-nor-
The doses were based on previous studies.
To assess possible effects of hydroxyfasudil and nor-NOHA on blood glucose levels, nine diabetic rats were anaesthetized by pentobarbital as above and randomized into three groups not subjected to myocardial ischaemia and reperfusion: (1) saline (n = 3), (2) hydroxyfasudil (0.5 mg kg−1; n = 3) or (3) nor-NOHA (100 mg kg−1; n = 3) given iv. Blood glucose was measured before anaesthesia, before the iv drug administration and 15, 30 and 45 min after drug administration. These time points correspond to before ischaemia, during ischaemia and onset of reperfusion in the IR protocols.
Determination of infarct size and collection of myocardial samples
Infarct size was measured as previously described. 11 After 2 h of reperfusion, the coronary artery was reoccluded and 1.5 mL of 2% Evans blue solution was injected into the left jugular vein to stain non-ischaemic myocardium for the determination of the area at risk. The rats were euthanized by exsanguination, and the heart was extracted. The right atrium, the right ventricle and the left atrium were removed; the remaining left ventricle was frozen and cut into seven to nine slices perpendicular to the base-apex axis. The third slice (counting from the apex) was weighed; the ischaemic and non-ischaemic parts were separated, frozen on dry ice and stored at −80°C until analysed. All other slices were weighed, scanned from both sides for the determination of the area at risk and put in 1% triphenyltetrazolium chloride solution for 15 min at 37°C to distinguish viable myocardium from necrotic. After 24 h of incubation in 4% formaldehyde, slices were scanned again from both sides, and the extent of myocardial necrosis and the area at risk were determined by planimetry of computer images (Adobe Photoshop 6.0, Adobe Photoshop CS 2; Adobe Systems, San Jose, CA, USA).
Determination of arginase and ROCK activity
Arginase activity was determined using a colorimetric assay previously described.
15
The assay measures the urea production using α-isonitrosopropiophenone. Following homogenization and centrifugation of tissue samples, 50 µL of the supernatant was added to 75 µL of Tris–HCl (50 mmol L−1, pH 7.5) containing 10 mmol L−1 MnCl2. The mixture was activated by heating for 10 min at 56°C. Each sample was then incubated at 37°C for 1 h with
Activity of ROCK was determined as described previously by analysing the extent of phosphorylation of ezrin (Thr567) by immunoblotting. 15
Immunoblotting
Left ventricular tissue samples were extracted in ice-cold radioimmunoprecipitation assay (RIPA) buffer. Total protein content of the extracts was quantified using bicinchoninic acid protein assay kit (Pierce Biotechnology, Rockford, IL, USA). The proteins were separated on a 7.5% sodium dodecyl sulphate (SDS) gel (40 µg per lane) and transferred onto nitrocellulose membranes (Amersham Biosciences, Piscataway, NJ, USA). Membranes were blocked for 1 h at room temperature and incubated overnight at 4°C with primary antibodies against arginase 1 and arginase 2 (1:1000; Sigma–Aldrich, St Louis, MO, USA) or phosphorylated ezrin (p-ezrin; anti-phosphorylated Thr567, 1:1000; BD Pharmingen, San Diego, CA, USA). IRDye 800–conjugated goat anti-rabbit and IRDye 800–conjugated goat anti-mouse IgG (1:15,000; LI-COR Biosciences, Lincoln, NE, USA) were used as secondary antibodies and bands were visualized using infrared fluorescence scanner (IR-Odyssey; LI-COR Biosciences). Equal loading was confirmed by expression of glyceraldehyde 3-phosphate dehydrogenase (GAPDH) (1:5000; Sigma–Aldrich, St Louis, MO, USA). Band densities were analysed with Image Studio Lite Version 3.1 (LI-COR Biosciences).
Chemicals
Pentobarbital was purchased from Apoteksbolaget (Stockholm, Sweden); hydroxyfasudil from Tocris Bioscience (Bristol, UK); nor-NOHA from Bachem (Bubendorf, Switzerland);
Statistical analysis
Data are presented as mean ± standard error of mean (SEM). Repeated-measures two-way analysis of variance (ANOVA) with Bonferroni post hoc test was used for multiple comparisons of haemodynamic parameters and blood glucose levels following ROCK and arginase inhibition in diabetic rats. One-way ANOVA followed by Bonferroni post hoc test was used for comparisons of protein expression, enzyme activity and infarct size. Paired and unpaired t-tests were used for comparisons of two groups. A p-value <0.05 was considered to be significant. All analyses were performed using Prism™ 5 software (GraphPad, Inc., San Diego, CA, USA).
Results
Matching of diabetic and non-diabetic rats
Body weight was significantly lower, and blood glucose level was significantly higher in all groups of diabetic animals. There were no significant differences in blood glucose or body weight between the experimental groups within diabetic and non-diabetic animals in the first part (Table 1) and in the second part of the study (Table 2).
Body weight and blood glucose in diabetic and in non-diabetic rats, part 1.
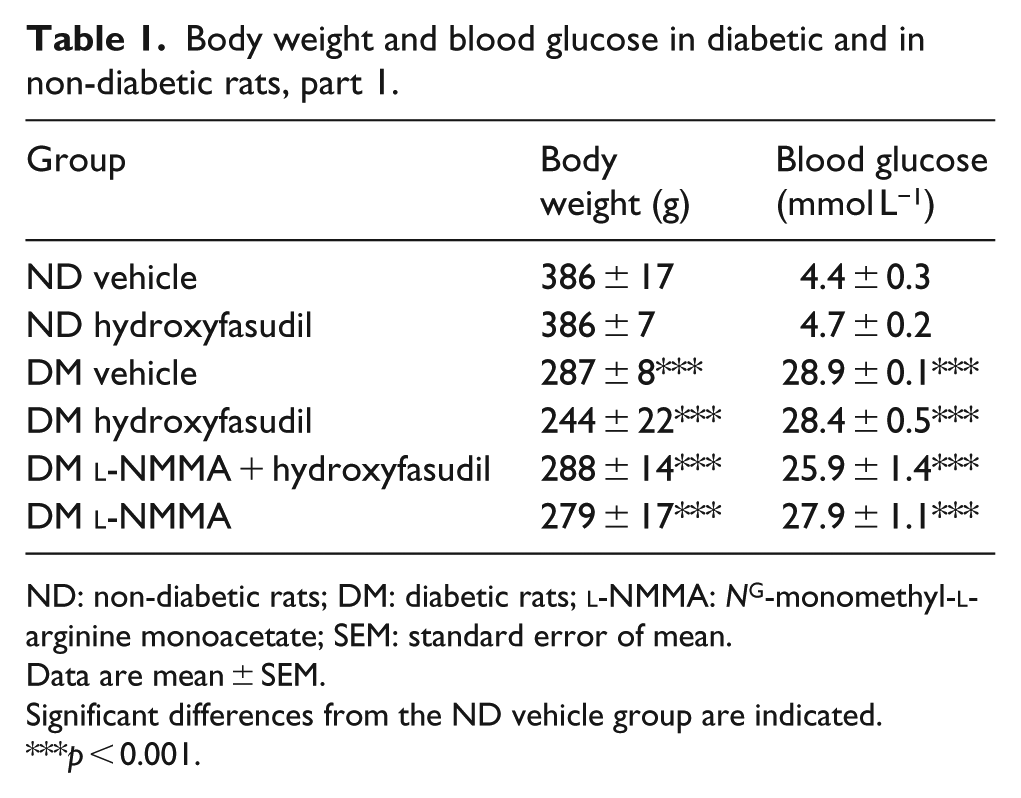
ND: non-diabetic rats; DM: diabetic rats;
Data are mean ± SEM.
Significant differences from the ND vehicle group are indicated.
p < 0.001.
Body weight and blood glucose in diabetic and in non-diabetic rats, part 2.
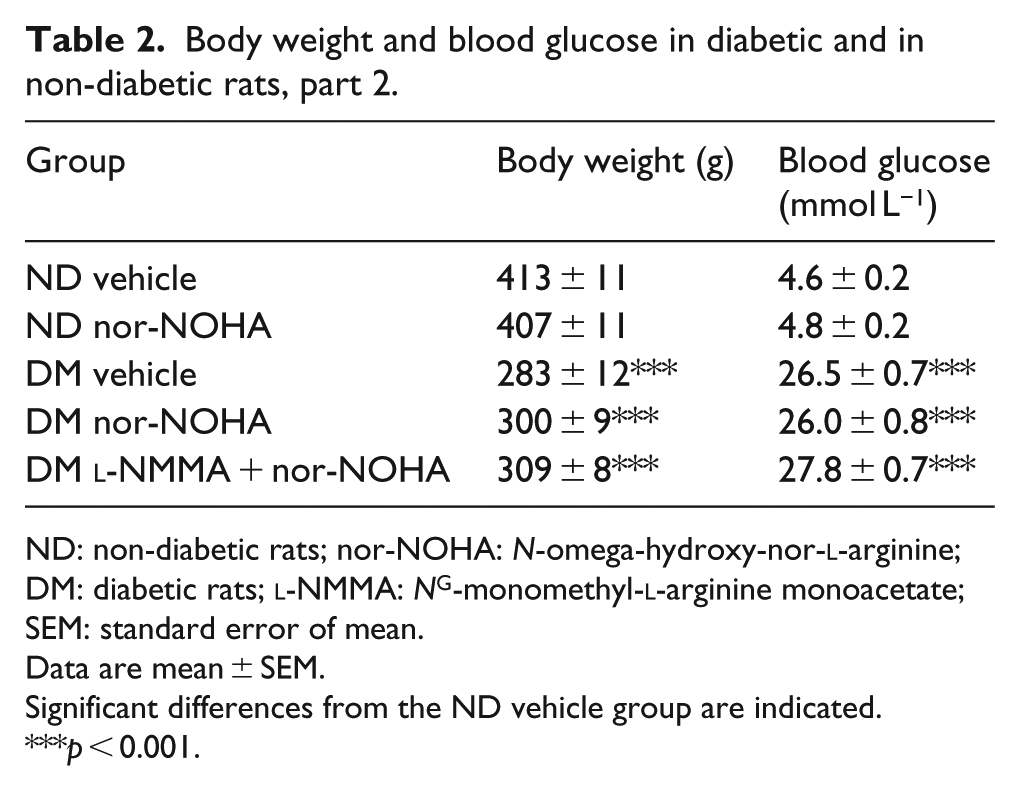
ND: non-diabetic rats; nor-NOHA: N-omega-hydroxy-nor-
Data are mean ± SEM.
Significant differences from the ND vehicle group are indicated.
p < 0.001.
Effects of ROCK inhibition and arginase inhibition on blood glucose level in diabetic rats
Blood glucose levels were not affected by either the ROCK inhibitor hydroxyfasudil or the arginase inhibitor nor-NOHA in comparison to saline in diabetic rats (Figure 1).
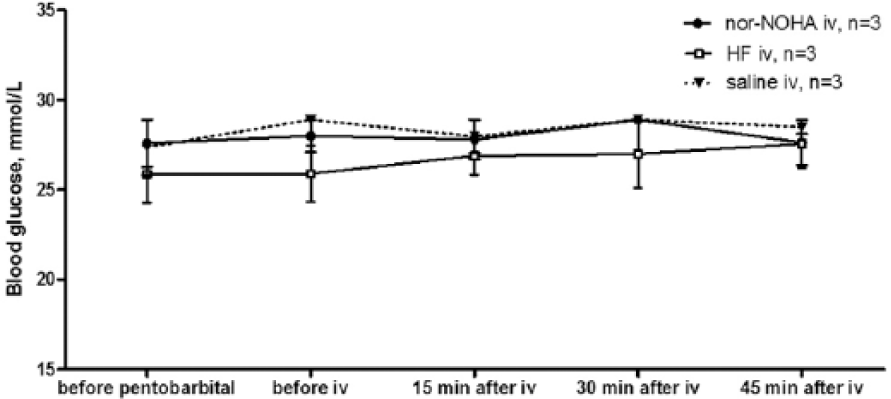
The effects of ROCK inhibitor hydroxyfasudil and arginase inhibitor nor-NOHA on blood glucose levels of diabetic rats. The diabetic rats were given saline (control, n = 3), ROCK inhibitor hydroxyfasudil (HF; n = 3) or arginase inhibitor nor-NOHA (n = 3). iv: intravenous injection. Data are presented as mean ± SEM.
Haemodynamics in rats subjected to myocardial IR
Haemodynamic parameters of the rats included in the first part and the second part of the study are presented in Tables 3 and 4, respectively. In comparison with the non-diabetic animals, heart rate was significantly lower in rats with type 1 diabetes (p < 0.05). There were no significant differences in heart rate or mean arterial pressure among non-diabetic or diabetic rats between the two parts of the study.
Haemodynamic data, part 1.
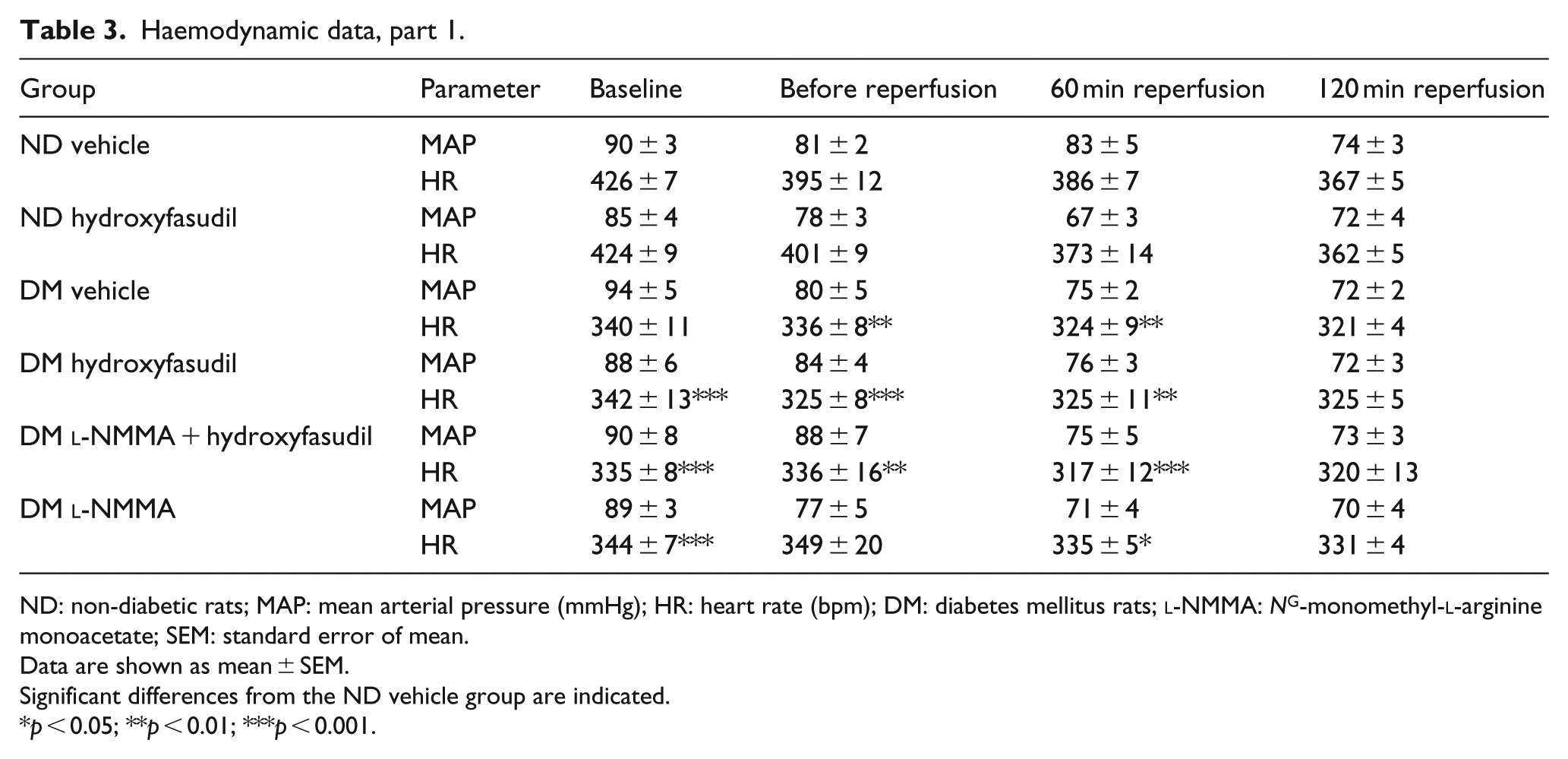
ND: non-diabetic rats; MAP: mean arterial pressure (mmHg); HR: heart rate (bpm); DM: diabetes mellitus rats;
Data are shown as mean ± SEM.
Significant differences from the ND vehicle group are indicated.
p < 0.05; **p < 0.01; ***p < 0.001.
Haemodynamic data, part 2.
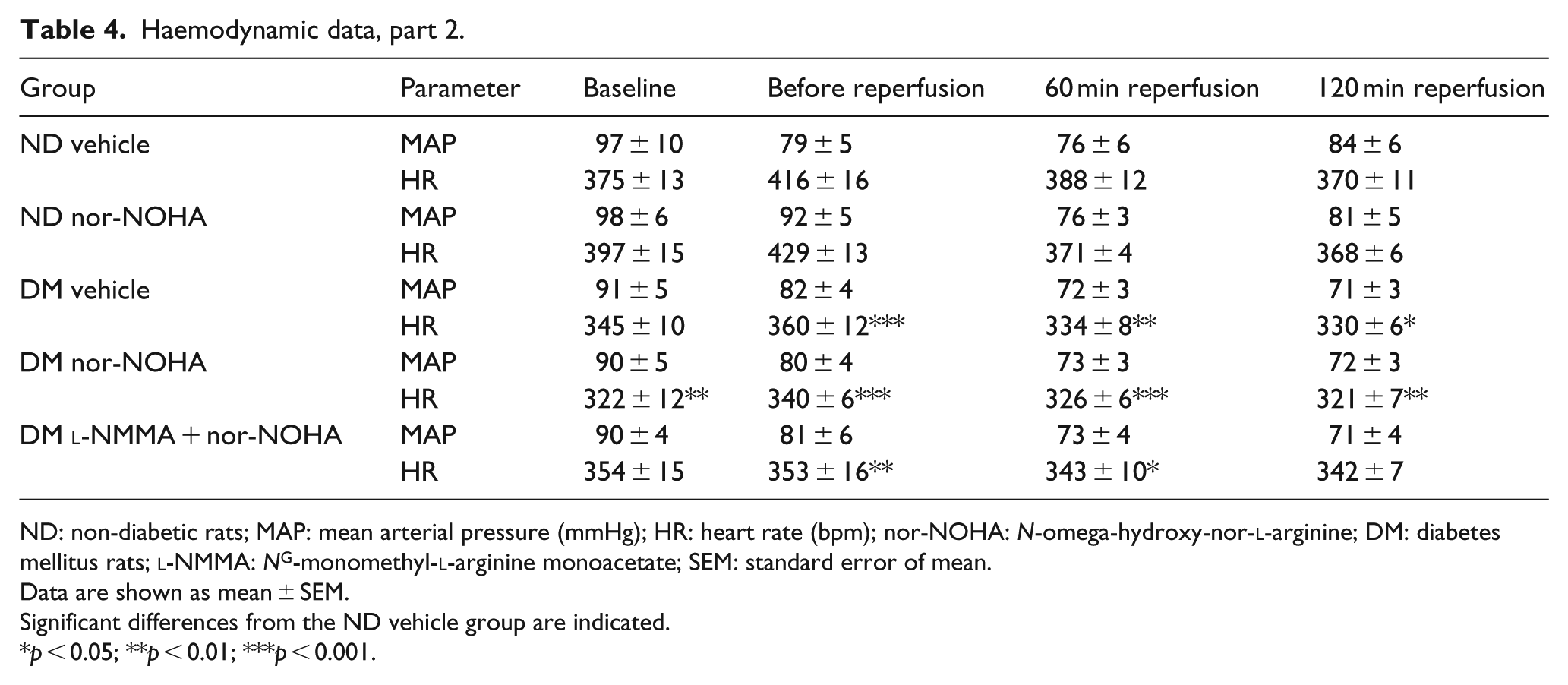
ND: non-diabetic rats; MAP: mean arterial pressure (mmHg); HR: heart rate (bpm); nor-NOHA: N-omega-hydroxy-nor-
Data are shown as mean ± SEM.
Significant differences from the ND vehicle group are indicated.
p < 0.05; **p < 0.01; ***p < 0.001.
Infarct size
There were no significant differences in the area at risk between any of the groups (Figures 2(a) and 3(a)). Infarct size was comparable in the two parts of the study and it did not differ between diabetic control rats and non-diabetic controls (Figures 2(b) and 3(b)). The ROCK inhibitor hydroxyfasudil significantly reduced infarct size by comparable magnitudes in diabetic and non-diabetic rats from 66% ± 2% and 66% ± 2% to 48% ± 4% and 46% ± 2%, respectively (p < 0.001, Figure 2(b)). Administration of the NOS inhibitor
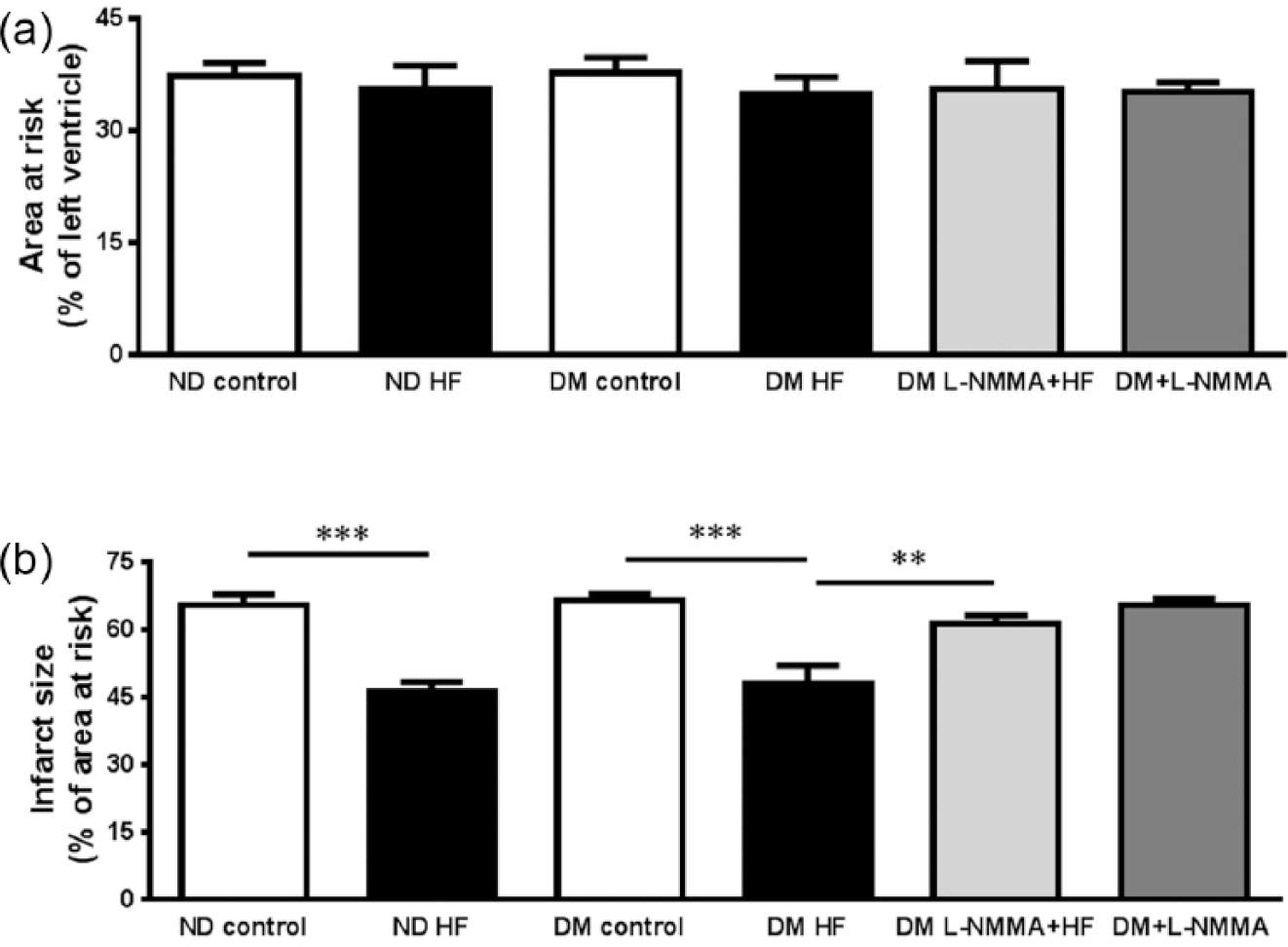
(a) Area at risk and (b) infarct size after 30 min of ischaemia and 2 h of reperfusion. The diabetic (DM) and non-diabetic (ND) rats were given saline (control, n = 5, DM; n = 5, ND), ROCK inhibitor hydroxyfasudil (HF; n = 5, DM; n = 5, ND), NO synthase inhibitor
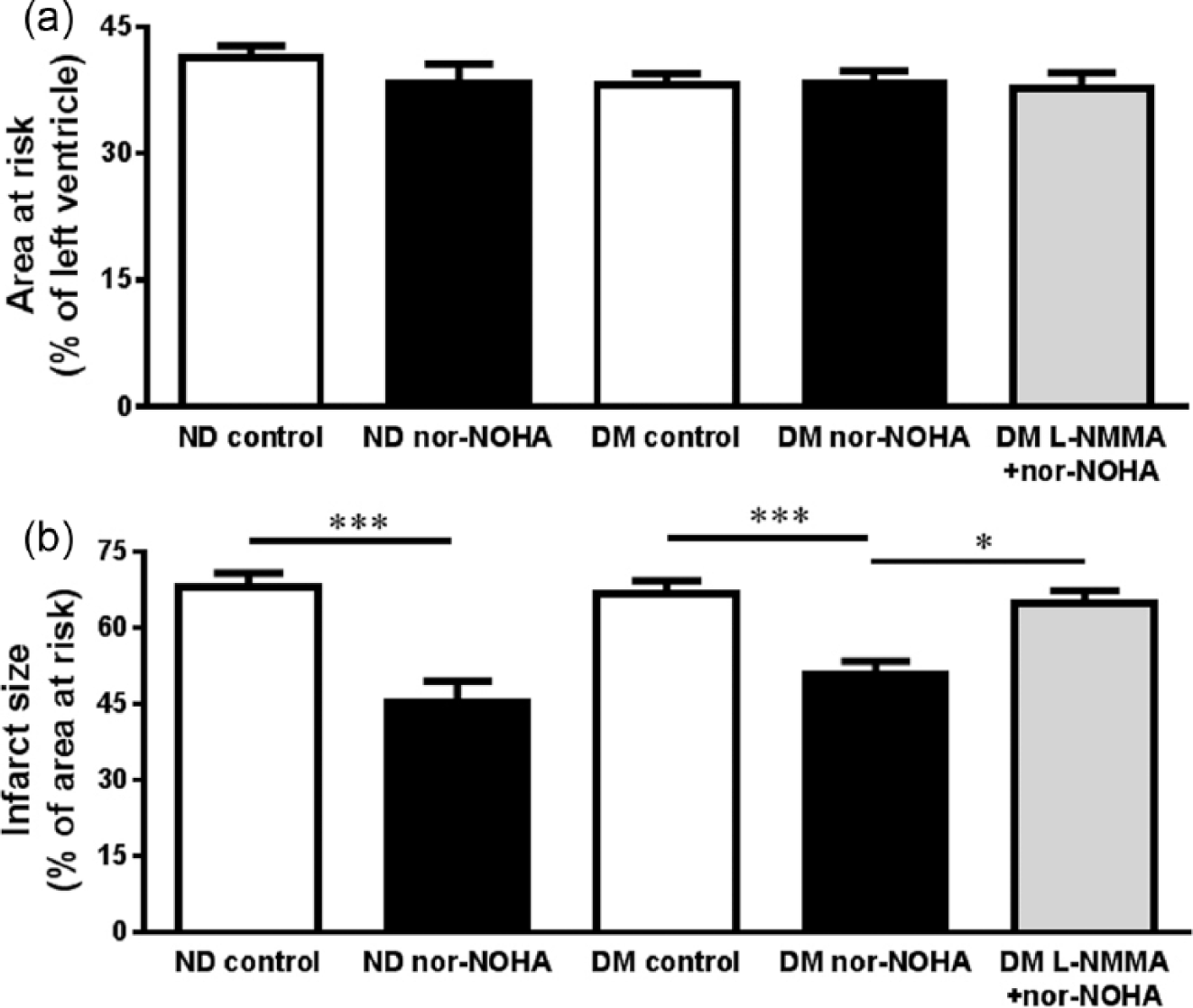
(a) Area at risk and (b) infarct size after 30 min of ischaemia and 2 h of reperfusion. The diabetic (DM) and non-diabetic (ND) rats were given saline (control, n = 14, DM; n = 7, ND), arginase inhibitor nor-NOHA (n = 11, DM; n = 8, ND) or NO synthase inhibitor
Administration of nor-NOHA significantly reduced infarct size both in diabetic and non-diabetic rats from 67% ± 3% and 68% ± 3% to 51% ± 3% and 45% ± 4%, respectively (p < 0.001, Figure 3(b)). The NOS inhibitor
Effect of diabetes on ROCK activity, arginase expression and activity
ROCK activity and arginase activity in the non-ischaemic myocardium were significantly elevated in rats with diabetes in comparison with non-diabetic animals (Figure 4(a) and (c)). However, ROCK activity in ischaemic-reperfused myocardium was not significantly different between diabetic and non-diabetic animals (Figure 4(b)). Arginase activity in ischaemic-reperfused myocardium was significantly higher in diabetic rats than in non-diabetic animals (Figure 4(d)). Furthermore, arginase activity was significantly higher in ischaemic-reperfused myocardium than in non-ischaemic myocardium of both non-diabetic and diabetic rats (p < 0.05). There was no difference in arginase 1 expression in non-ischaemic myocardium from rats with and without diabetes (Figure 4(e)), whereas myocardial expression of arginase 2 was significantly increased in diabetic rats (Figure 4(f)).
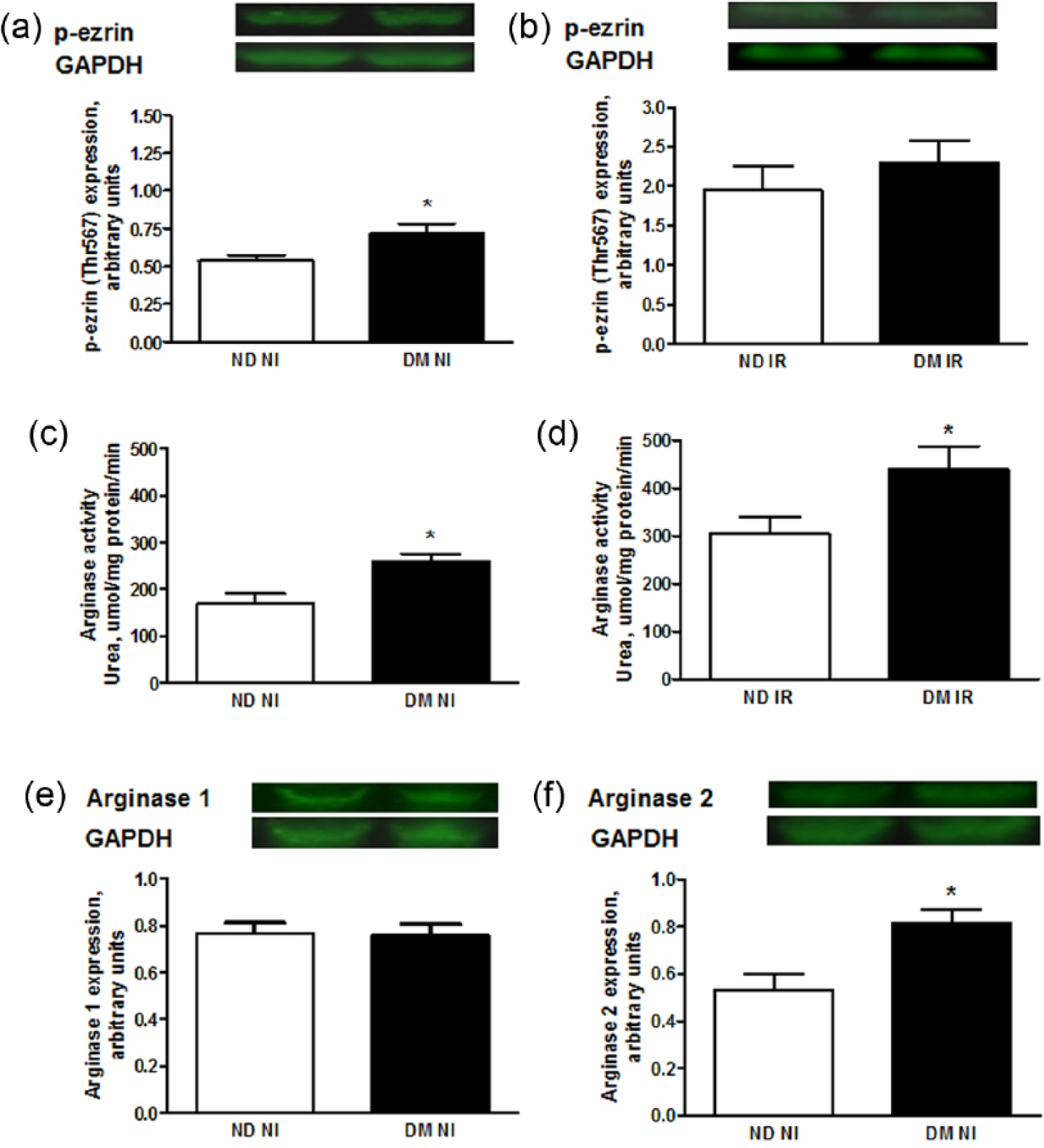
The effect of diabetes on activity of ROCK and arginase and the expression of arginase isoforms. Expression of phosphorylated ezrin (p-ezrin) as a marker of ROCK activity in (a) non-ischaemic and (b) ischaemic myocardium of non-diabetic (ND) and diabetic rats (DM), arginase activity in (c) non-ischaemic and (d) ischaemic myocardium of ND and DM rats, expression of (e) arginase 1 and (f) arginase 2 in non-ischaemic ventricular tissue from non-diabetic (ND) and diabetic rats (DM). Representative immunoblots are included. Data are presented as mean ± SEM; n = 6–10; *p < 0.05; **p < 0.01.
Effect of IR on myocardial arginase activity in non-diabetic and diabetic rats
Myocardial IR significantly increased arginase activity in ischaemic-reperfused myocardium in comparison to non-ischaemic myocardium by comparable magnitudes in non-diabetic and diabetic animals, respectively (Figure 5(a) and (b)).
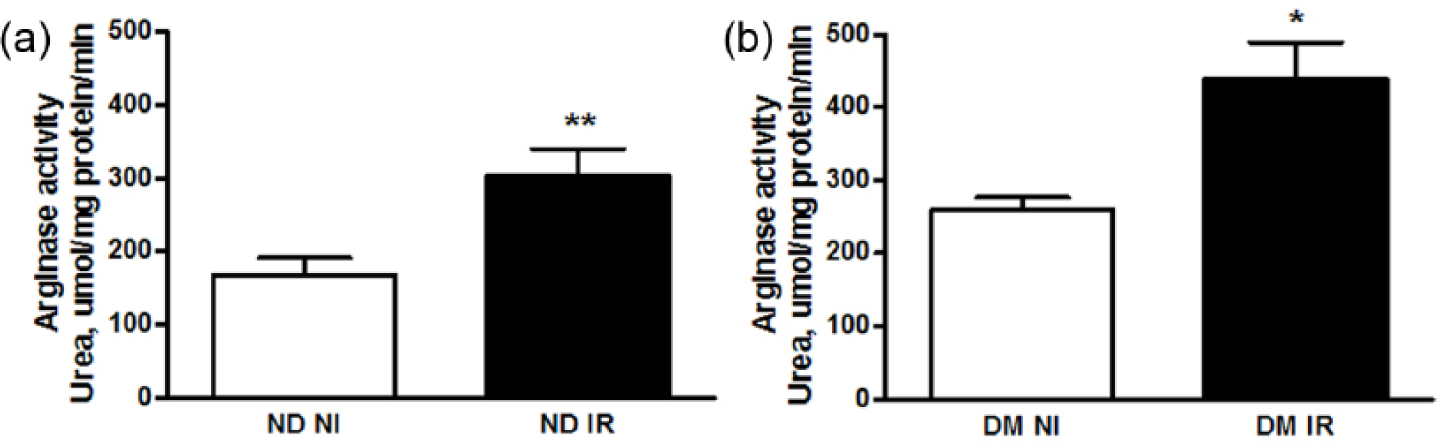
The effect of ischaemia–reperfusion on myocardial arginase activity in non-diabetic and diabetic rats. Arginase activity in non-ischaemic (NI) and ischaemic-reperfused (IR) myocardium of (a) non-diabetic (ND) and (b) diabetic (DM) rats. Data are presented as mean ± SEM; n = 6–7; *p < 0.05; **p < 0.01.
Effect of ROCK inhibition on arginase activity following IR
ROCK inhibition significantly attenuated myocardial arginase activity in diabetic rats following IR. The effect of ROCK inhibition on arginase activity was reversed by NOS inhibition (Figure 6).
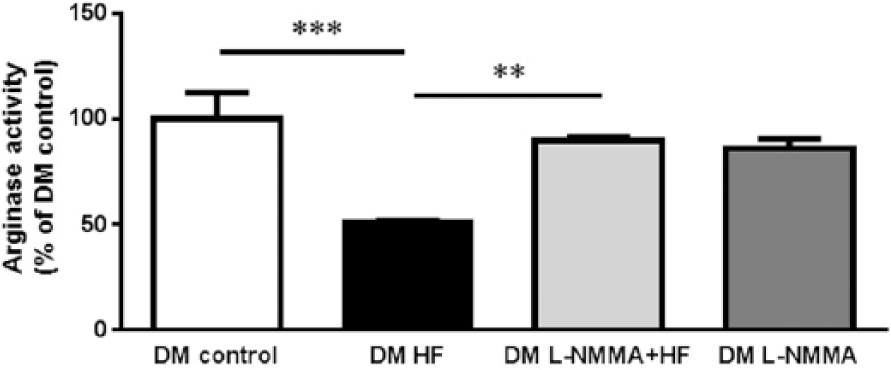
The effect of ROCK inhibition on arginase activity following ischaemia–reperfusion. Myocardial arginase activity in rats with diabetes mellitus subjected to control ischaemia–reperfusion (DM control), ROCK inhibition by hydroxyfasudil (DM HF), the combination of NOS inhibition and ROCK inhibition (DM
Discussion and conclusion
In this study, we show that ROCK activity, arginase activity and arginase 2 protein expression are upregulated in the myocardium of type 1 diabetic rats. Inhibition of ROCK by hydroxyfasudil protects the diabetic heart from IR injury. The cardioprotective effect of ROCK inhibition is associated with significant decrease in arginase activity in ischaemic-reperfused myocardium. In turn, inhibition of arginase by nor-NOHA protects the diabetic heart from IR injury emphasizing the importance of arginase for the deleterious consequences of IR injury in diabetes. The protective effects of both ROCK and arginase inhibition are abolished by the NOS inhibitor
It is well established that attenuation of NO bioavailability is a dominant mechanism contributing to IR injury 27 and cardiovascular complications associated with diabetes. 28 Although the underlying mechanisms are not fully understood, recent data suggest that upregulation of ROCK 5 and arginase 6 is involved in signalling events leading to reduction in NO bioavailability. We therefore hypothesized that these signalling pathways are of importance for the development of IR injury in diabetes and targeting them would result in reduction in infarct size in a model of diabetes. Furthermore, there seems to be an intriguing link between ROCK and arginase whereby activation of arginase involves activation of ROCK.13,29 Whether this is of functional importance in the setting of IR has previously not been investigated.
Based on the involvement of ROCK in the signalling events contributing to IR injury 7 and its key function in the development of diabetic vascular complications, we first investigated whether targeting ROCK had beneficial effects in the setting of myocardial IR in diabetes. Accordingly, we found that myocardial ROCK activity was upregulated in non-ischaemic myocardium from rats with diabetes. Of further importance, administration of the ROCK inhibitor hydroxyfasudil attenuated infarct size in rats with type 1 diabetes. This cardioprotective effect of ROCK inhibition was abolished by NOS inhibition supporting the notion that cardioprotection from ROCK inhibition is mediated via a NOS-dependent mechanism in diabetes.
Given the previous data suggesting that ROCK may stimulate arginase activity and that arginase may be involved in the development of IR injury, we next investigated the effect of ROCK inhibition on arginase activity and the efficacy of arginase inhibition to reduce IR. Recent studies have been shown that diabetes is associated with elevation of arginase activity and impairment of vascular endothelial function characterized by low availability of NO.17,21 Furthermore, ROCK is reported as a trigger for increased arginase activity in diabetes.
24
It was found that the ROCK inhibitor hydroxyfasudil markedly attenuated the increase in arginase activity induced by IR demonstrating that arginase activity induced by IR is mediated via a signalling pathway involving ROCK. This observation in rats with type 1 diabetes is in line with that obtained in non-diabetic rats
15
and supports the notion that arginase activity is influenced by ROCK activity. Administration of the NOS inhibitor
Cardioprotective treatment strategies aimed at limiting IR injury have been described to be attenuated or absent in animal models of diabetes. Thus, it has been shown that the diabetic heart is more resistant to pharmacological and non-pharmacological (classical pre- and postconditioning) treatments aiming at reducing IR injury. 25 In addition, we have recently shown that remote ischaemic perconditioning which markedly reduced infarct size in non-diabetic rats failed to induce cardioprotective effect in type 1 diabetic rats. 15 It is therefore an unmet need to develop treatment strategies that protect the myocardium in the presence of diabetes. This study provides evidence that pharmacological inhibition of ROCK and arginase effectively reduces infarct size by magnitudes comparable to that obtained in non-diabetic animals. This opens up for new possibilities for treatment of diabetic disorders.
Certain limitations of the study need to be acknowledged. These data are obtained in a model of type 1 diabetes and it is not known whether similar results are valid for other models of diabetes. There was no significant difference in the vulnerability to IR in type 1 diabetic hearts in comparison with non-diabetic hearts. This is in agreement with previous studies, although some studies have described both smaller and larger infarct size in various animal models of diabetes. 25 Of importance, the purpose of this study was to investigate the efficacy of the cardioprotective interventions, not to compare the extent of IR injury per se between diabetes and non-diabetes. The presently used model is known to be resistant to cardioprotection induced by remote ischaemic perconditioning, demonstrating a marked difference from non-diabetic rats and thereby the suitability of the model for this type of study. 15 These experiments were performed using an isoform non-selective arginase inhibitor. The isoform of arginase involved in IR can therefore not be identified. Future studies using arginase knock-out models of diabetes may clarify the arginase isoform mediating this effect.
In conclusion, this study demonstrates that myocardial ROCK activity, arginase activity and arginase 2 expression are increased in type 1 diabetic animals. Both ROCK and arginase inhibition reduce infarct size via a NOS-dependent mechanism. Furthermore, the cardioprotective effect of ROCK inhibition involves downregulation of arginase. These findings suggest that ROCK and arginase are promising therapeutic targets for limitation of myocardial injury in diabetes.
Footnotes
Acknowledgements
The authors thank Marita Wallin for excellent technical assistance.
Author contributions
Y.H. and A.K. contributed equally to the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Swedish Heart and Lung Foundation, the Research Council Medicine (10857), Torsten Söderberg Foundation, the Stockholm County Council (ALF), Karolinska Institutet/Stockholm County Council Strategic Cardiovascular Programme, Diabetes Research and Wellness Foundation and Novo Nordisk Foundation.
