Abstract
Using a porcine model of diabetes mellitus and hypercholesterolaemia, we previously showed that diabetes mellitus and hypercholesterolaemia is associated with a chronic increase in blood–brain barrier permeability in the cerebral cortex, leading to selective binding of immunoglobulin G and deposition of amyloid-beta1-42 peptide in pyramidal neurons. Treatment with Darapladib (GlaxoSmithKline, SB480848), an inhibitor of lipoprotein-associated phospholipase-A2, alleviated these effects. Here, investigation of the effects of chronic diabetes mellitus and hypercholesterolaemia on the pig retina revealed a corresponding increased permeability of the blood–retina barrier coupled with a leak of plasma components into the retina, alterations in retinal architecture, selective IgG binding to neurons in the ganglion cell layer, thinning of retinal layers due to cell loss and increased glial fibrillary acidic protein expression in Müller cells, all of which were curtailed by treatment with Darapladib. These findings suggest that chronic diabetes mellitus and hypercholesterolaemia induces increased blood–retina barrier permeability that may be linked to altered expression of blood–retina barrier–associated tight junction proteins, claudin and occludin, leading to structural changes in the retina consistent with diabetic retinopathy. Additionally, results suggest that drugs with vascular anti-inflammatory properties, such as Darapladib, may have beneficial effects on eye diseases strongly linked to vascular abnormalities such as diabetic retinopathy and age-related macular degeneration.
Keywords
Introduction
Diabetic retinopathy is an early onset, long-term complication of diabetes mellitus (DM) that is regarded as a primary cause of partial or complete vision loss in these patients.1–5 The underlying reasons for its onset and the development of its pathology in DM patients are unknown. Previous studies have demonstrated chronic leak of plasma components via a compromised blood–retina barrier (BRB) in patients with DM, thus implicating DM-associated BRB compromise in the pathogenesis of diabetic retinopathy.1,2,4 Recently, we reported increased blood–brain barrier (BBB) permeability in a porcine model with chronic DM and hypercholesterolaemia (HC). 6 This increased BBB permeability in DMHC pigs was morphologically evident as perivascular, immunoglobulin G (IgG)-positive leak clouds and was accompanied by selective binding of IgG to pyramidal neurons in the cerebral cortex along with increased intracellular deposition of the amyloid-beta1-42 (Aβ42) peptide in these neurons. 6 Treatment of DMHC animals with Darapladib, a lipoprotein-associated phospholipase-A2 (LpPLA2) inhibitor with vascular anti-inflammatory properties developed by GlaxoSmithKline to treat atherosclerosis, led to reduced BBB permeability, IgG binding and Aβ42 deposition in cortical pyramidal neurons. 6
The BBB and BRB have many structural and functional features in common. 7 Both consist of a monolayer of vascular endothelial cells held together at their margins by tight junctions comprising proteins, including claudin-5 and occludin, that contribute to forming and maintaining an effective barrier that prevents the unwanted leak of plasma components into tissues that are fed by these blood vessels. In this study, we investigated the effects of a chronic DMHC on the permeability of the BRB and structural integrity of the retina using the DMHC pig model. Results revealed an increased permeability of the BRB coupled with alterations in retinal architecture including a leak of plasma components into the retina, selective IgG binding to neurons in the ganglion cell layer (GCL), thinning of some retinal layers due to cell loss, and increased glial fibrillary acidic protein (GFAP) expression in Müller cells, all of which were curtailed by treatment with Darapladib. These findings suggest that chronic DMHC induces increased BRB permeability that can lead to structural changes in the retina consistent with diabetic retinopathy. Furthermore, we show that the BRB and BBB respond similarly to chronic DMHC and Darapladib treatment. Additionally, DMHC pigs exhibited increased expression of the BBB-associated tight junction proteins, claudin-5 and occludin, in brain microvascular endothelial cells that may reflect a compensatory response to the constant challenge to BRB and BBB structural and functional integrity. Finally, our data suggest that drugs with vascular anti-inflammatory properties, such as Darapladib, may prove to be useful for alleviating the impact of chronic DMHC on the retina and brain.
Methods
Porcine model, tissue handling and processing
Details of the DMHC porcine model have been reported previously.6,8 Briefly, 6-month-old male Yorkshire domestic farm pigs (~25–35 kg) were selected as the animal model for this study. DM was induced in these animals by injecting a single dose of 125 mg/kg streptozotocin. After 3 days of DM induction, these animals were put on a hyperlipidaemic diet. Throughout the course of the study, blood levels of glucose and cholesterol were closely monitored and maintained at 350–400 mg/dL and 400–800 mg/dL, respectively. 8 One month after induction of DMHC, DMHC pigs were randomly separated into two groups: DMHC and Darapladib-DMHC. The Darapladib-DMHC group received an oral dose (10 mg/kg/day) of the LpPLA2 inhibitor, Darapladib (SB480848, GlaxoSmithKline). 8 Of the Yorkshire domestic farm pigs of the same age and sex, three were excluded from DM induction and the hyperlipidaemic diet. This group of pigs was used as age-matched normal controls. Darapladib-DMHC pigs received Darapladib treatment for 24 weeks. At the end of treatment period, animals from all three groups were euthanized. The brain and eyes were removed within 20 min of euthanizing and processed immediately. Animal use and experimental details of this study were approved by the IACUC of the University of Pennsylvania. 8
Determination of retinal thickness
The remarkably uniform and laminated histological architecture of the retina lends itself to direct measurement of the thickness of the various layers as shown in Figure 1. Using ImagePro 7.0 (Media Cybernetics, Silver Spring, MD), we measured the thickness of all retina layers. The thickness of each retina layer was measured in three separate sections obtained from each animal. In each section, measurements were made at two randomly selected locations in the middle part of the retina to minimize region-specific variability. Results were averaged for each pig. The subject group identity of specimens was blinded, and two independent investigators carried out this analysis. In addition, the percentage contribution of each retinal layer to the total thickness of the retina was calculated and compared among the different treatment groups.
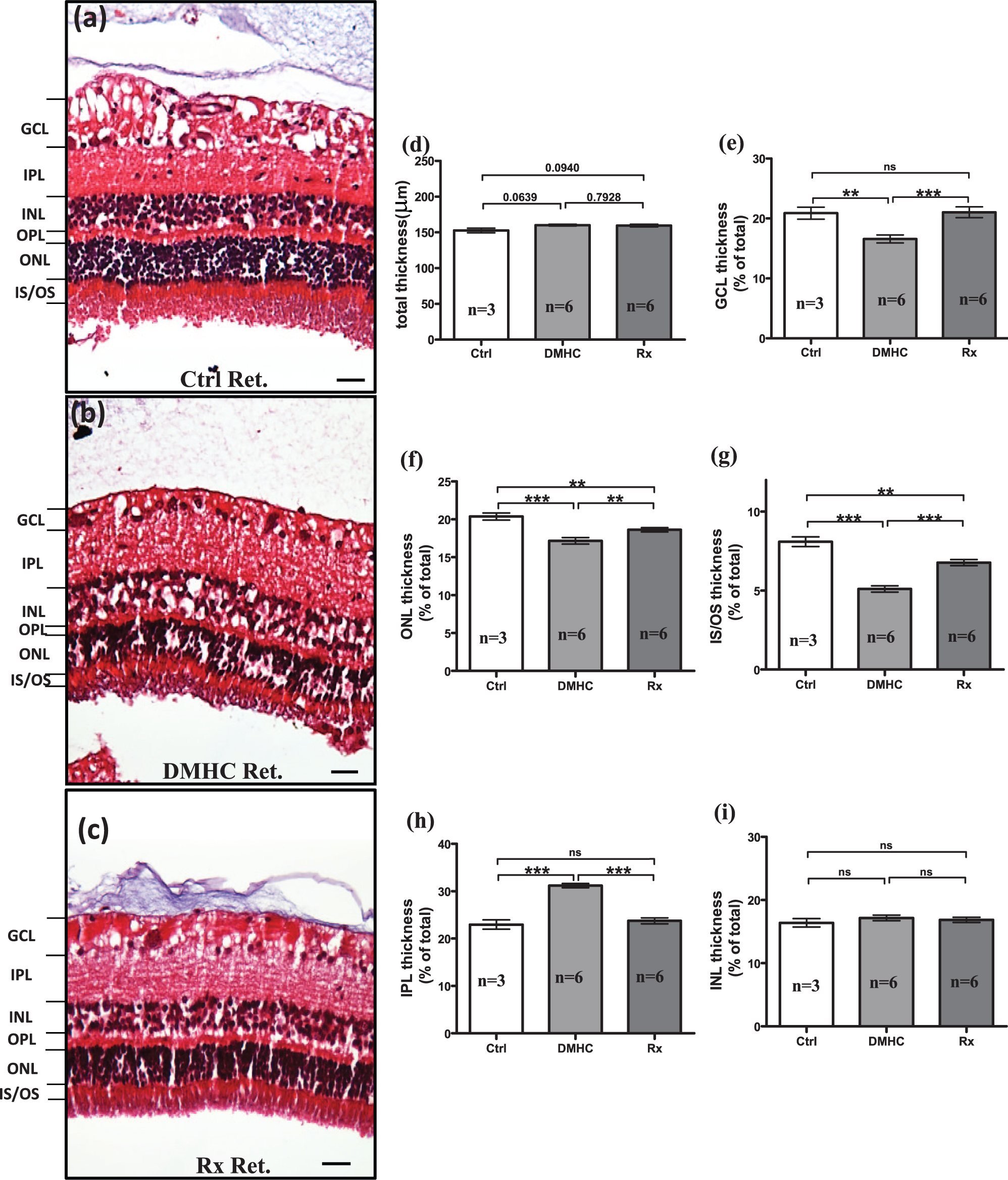
Chronic DMHC and Darapladib treatment alters the thickness of retinal layers. The effect of DMHC induction and Darapladib treatment on the porcine retina (Ret) was investigated by carrying out histological analyses of haematoxylin–eosin–stained retinal sections from all treatment groups – (a) control (Ctrl), (b) DMHC and (c) Darapladib-treated DMHC (Rx). The thickness of total and individual retinal layers was measured, and the percentage of individual retinal layer thickness in relation to total retinal thickness was calculated and used for comparison. (d) Total retinal thickness, (e) ganglion cell layer percentage, (f) outer nuclear layer percentage, (g) inner and outer segments (IS/OS) of photoreceptors percentage, (h) inner plexiform layer percentage and (i) inner nuclear layer percentage. Scale bar: 50 µm.
Immunohistochemistry and bright-field microscopy
Detection of the distribution of claudin-5 and occludin as well as IgG localization in the cerebral cortex and retina was carried out using immunohistochemistry (IHC) as described previously.6,9 Briefly, tissue sections were deparaffinized in xylene and rehydrated in decreasing concentrations of ethanol. The antigenicity of these tissues was enhanced by microwaving in citrate buffer. Sections were treated with 3% H2O2 for 10 min to quench endogenous peroxidase activity. Tissues were subsequently treated with blocking serum for 30 min at room temperature and then probed with anti-claudin-5 (Invitrogen, catalogue 35-2500; dilution 1:150) or anti-occludin (Invitrogen, catalogue 71-1500; dilution 1:800) antibodies for 1 h at room temperature. Extravasated IgG was detected using biotinylated anti-swine IgG antibody (Vector laboratories, Inc., catalogue BA-9020, Burlingame, CA, USA; dilution 1:100). The use of occludin antibody required pre-treatment with a protease (Sigma, catalogue P-5147, St. Louis, MO, USA; 1.5 mg/mL) for 10 min at 37°C. At the end of the primary antibody step, all tissue sections were thoroughly rinsed in phosphate-buffered saline (PBS) and then probed with biotin-labelled secondary antibodies [anti-mouse IgG (Vector laboratories, Inc., catalogue PK-6102; Dilution 1:100) for claudin-5 and anti-rabbit IgG (Vector laboratories, Inc., catalogue PK-6101; dilution 1:100) for occludin] for 30 min at room temperature. Sections were then rinsed in PBS, treated with avidin-peroxidase complex (Vectastain ABC Elite kit; Vector Laboratories, Inc., catalogue PK-6100), washed in PBS and visualized with 3-3-diaminobenzidine-4-HCL (DAB)/H2O2 (Imm-PACT-DAB; Dako, Code K3468). Nuclei were lightly counterstained with haematoxylin. Finally, tissues were dehydrated in increasing concentrations of ethanol, cleared in xylene and mounted in Permount (Fisher Scientific, Philadelphia, PA, USA). The specificity of the primary antibodies was tested by treating tissue sections with secondary antibody only or blocking sera only as negative controls. Slides were examined and photographed with a Nikon FXA microscope, and digital images were recorded using a Nikon DXM1200F digital camera and analysed using MATLAB (MathWorks, Natick, MA) and NIS-Elements software (Nikon, Melville, NY). The evaluator was blinded as to subject group identity of the samples during image analysis.
Immunofluorescence microscopy and image analysis of the retina
Retina sections were deparaffinized using xylene and rehydrated through a graded series of decreasing concentrations of ethanol. Antigen Unmasking Solution (Vector Laboratories, Inc.) was used according to the manufacturer’s instructions. Sections were then placed in 0.5% Triton X-100 in PBS for 5 min. After washing briefly with PBS, they were incubated overnight at 4°C with rabbit anti-GFAP antibody (dilution 1:200; Sigma, catalogue G3893), washed three times with PBS and then incubated with Alexa Fluor 488 goat anti-rabbit secondary antibodies (dilution 1:200; Molecular Probes; Life Technology, Carlsbad, CA, USA) for 1 h at room temperature. Sections were thoroughly washed in PBS and then mounted using Vectashield with 4′,6-diamidino-2-phenylindole (DAPI; Vector Laboratories, Inc.). Negative controls were treated with non-immune serum or without primary antibody. Images were taken at 20× magnification on a Nikon Eclipse E800 microscope (Nikon) equipped with a QImaging Retiga Exi digital camera (QImaging Burnham, Canada). Cells positive for GFAP were counted throughout the thickness of the retina and then quantified using ImagePro7.0 (Media Cybernetics, Silver Spring, MD). Briefly, a 300 × 300-pixel square area was randomly selected in each retinal section in three locations across the retina, and the positive GFAP area was determined per total area using a fixed threshold. For total cell counts, sections were mounted in Vectashield with DAPI (Vector Laboratories, Inc.). Cell number was determined by counting the DAPI-positive nuclei in a fixed area. The results of these measurements were then averaged for each section. The evaluator was blinded as to subject group identity of the samples during image analysis. Using the image analysis software ImagePro 7.0 (Media Cybernetics, Silver Spring, MD), the total number of IgG-positive ganglion cells and total cells in GCL was counted. The percentage of the IgG-labelled ganglion cells was then quantified out of total ganglion cells.
Localization and estimation of claudin-5 and occludin protein expression in the pig cerebral cortex
Although claudin-5 and occludin are well known to be key tight junction proteins associated with both BRB and BBB, the pig cerebral cortex was chosen for this quantification over the retina in DMHC pigs because of the greater tissue volume available for carrying out protein localization and expression studies, and the fact that the cerebral cortex has a more extensive plexus of blood vessels that is readily visualized and thus more accessible to quantification. Expression of the tight junction proteins, claudin-5 and occludin, in pig cerebral cortex was evaluated semi-quantitatively using IHC by taking images at five random locations as described previously. 6 Images were analysed using MATLAB (MathWorks). Optimal imaging parameters were first determined and then standardized for the visualization of claudin-5 or occludin. These conditions were maintained for all claudin-5 and occludin images as well as for controls. Using MATLAB, IHC images were first converted into black and white. Sites of claudin-5 and occludin localization were rendered white, whereas areas lacking these proteins appeared black. The percentage of total image area occupied by white and black was calculated by MATLAB. Using similar conditions for claudin-5 and occludin, the relative expression levels of these proteins were calculated.
Claudin-5 and occludin gene expression in the pig cerebral cortex
To parallel the relative expression of claudin-5 and occludin proteins, the relative quantification of claudin-5 and occludin gene expression was also investigated in the pig cerebral cortex using the reporter dye SYBR green and an Applied Biosystems 7500 real-time polymerase chain reaction (RT-PCR) system. The primers used for this study were generated using Applied Biosystems Primer Express 3.0. Relative quantification of the claudin-5 and occludin gene expression was conducted using beta-actin (β-actin), a housekeeping gene, as a standard. The amplification profiles of all samples were analysed using Applied Biosystems software. The primers used for this study are as follows: β-actin (forward) 5′-tccagaggcgctcttcca-3′, (reverse) 5′-cgcacttcatgatcgagttga-3′; claudin-5 (forward) 5′-cggcgactacgacaagaagaa-3′, (reverse) 5′-gccctccaaagcggagtt-3′; and occludin (forward) 5′-tgcacccagcaacgacatata-3′, (reverse) 5′-ggctgagaaagcattggtcga-3′.
Statistics
Student’s t test with Welch’s correction was used for calculating statistical significance. Variations within each treatment group were represented by either standard deviation or standard error.
Results
Using the chronic DMHC pig model, we previously reported increased BBB permeability in the cerebral cortex which resulted in IgG binding and Aβ42 deposition within cortical pyramidal neurons as well as neurodegenerative changes resembling early pathological features of Alzheimer’s disease. 6 We further showed that these changes were blocked by treatment with Darapladib, an LpPLA2 inhibitor with vascular anti-inflammatory properties. In view of the well-established link between DM, retinal pathology (particularly diabetic retinopathy), Alzheimer’s disease and the structural and functional similarities between BBB and BRB, in this study, we have investigated the effects of chronic DMHC and treatment with Darapladib on retinal structure and BRB function.
Chronic DMHC altered the thickness of the individual retinal layers
The thickness of individual layers of the retina was measured using haematoxylin and eosin–stained transverse sections (Figure 1). Neither chronic DMHC nor Darapladib treatment of DMHC pigs led to significant changes in the total thickness of the retina (Figure 1(a) to (d)). However, in DMHC pigs, we found a significant decrease in the thickness of several individual layers of retina, namely, the GCL, the outer nuclear layer (ONL) and the inner-segment (IS)/outer-segment (OS) photoreceptor layer compared to controls (Figure 1(d) to (g)). By contrast, the inner plexiform layer (IPL) in DMHC animals showed a significant increase in thickness and was more disorganized compared to controls (Figure 1(h)), similar to oedematous changes seen in diabetic retinopathy cases. This explains the observation of similar total retinal thickness among treatment groups in spite of significant reduction in the GCL, ONL and IS/OS thickness. The inner nuclear layer (INL) retained a comparable thickness across all treatment groups (Figure 1(i)). Importantly, treatment with Darapladib blocked the effects of DM and HC on the thickness of GCL, ONL and IS/OS layer, as their thickness in the Darapladib-treated DMHC group was comparable to that of controls (Figure 1(e) to (g)). Darapladib treatment also reduced IPL thickness in DMHC animals to near control levels (Figure 1(h)).
DMHC reduced the number of cells in the GCL and INL of the retina
The thickness of any given cellular layer in the retina is directly proportional to the number of cells comprising that layer. To determine the effects of chronic DMHC and Darapladib treatment on cell number within the GCL and INL, we counted and compared the numbers of cells in these layers that appeared in tissue sections (Figure 2). Results showed that chronic DMHC was associated with a significant reduction in the number of cells within the GCL and INL compared to controls (Figure 2(d) and (e)). By contrast, the total number of cells in both GCL and INL was maintained at levels comparable to controls in Darapladib-treated DMHC pigs (Figure 2(d) and (e)). We suggest that Darapladib somehow protects these cells from the damaging consequences of chronic DMHC.
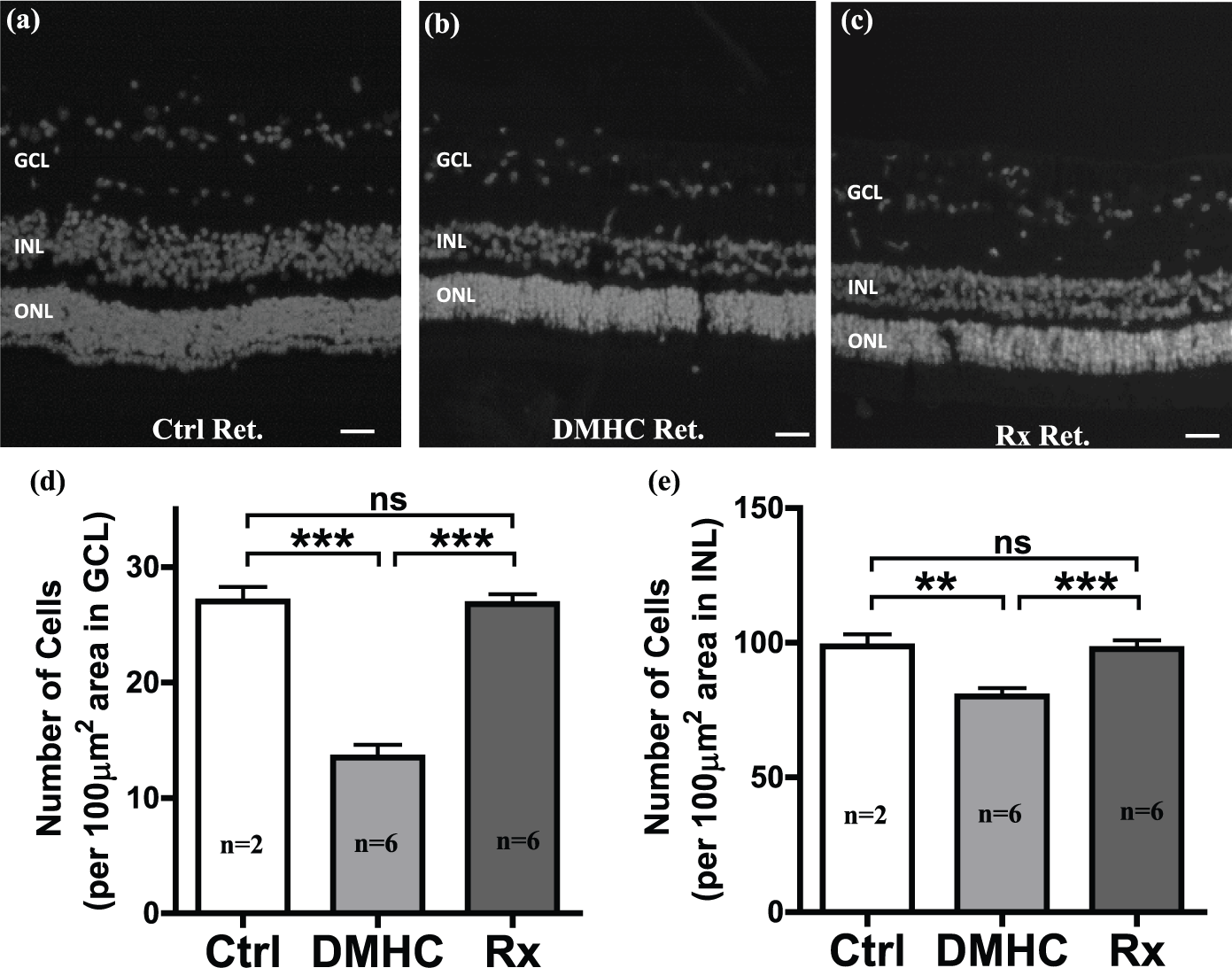
DMHC-induced cell loss in the GCL and INL was blocked by Darapladib treatment. Retinal (Ret) sections from pigs of all treatment groups were prepared with DAPI staining for histological assessment and nuclear count in the GCL and INL for (a) control (Ctrl), (b) DMHC and (c) Darapladib-treated DMHC (Rx). The number of cells in (d) GCL and (e) INL was quantified using image analysis. Scale bar: 50 µm.
IgG binds selectively to ganglion cells in the GCL of the retina
In our previous study which examined the effects of chronic DMHC on the cerebral cortex in these same pigs, we revealed a greater degree of IgG binding to cortical pyramidal neurons in DMHC animals. This selective IgG binding to neurons was not only indicative of a compromised BBB but also provided further confirmation of the widespread presence of brain-reactive auto-antibodies (neuron-binding) in the blood. 6 In this study, we show that extravasated IgG also exhibited selective affinity for neurons in the GCL of the retina. Image analysis revealed an increased presence of IgG-positive ganglion cells in the retina of DMHC pigs over controls, with IgG detected at both the cell surface and in the cytoplasm of the DMHC group (Figure 3(a) to (c)). Cells in other retinal layers lacked detectable IgG-positive cells. We calculated the percentage of IgG-positive ganglion cells relative to total ganglion cells in the GCL as an indicator of BRB integrity. Among the treatment groups, DMHC animals had the highest percentage of IgG-positive ganglion cells followed by controls and finally the DMHC-Darapladib group (Figure 3(d)).
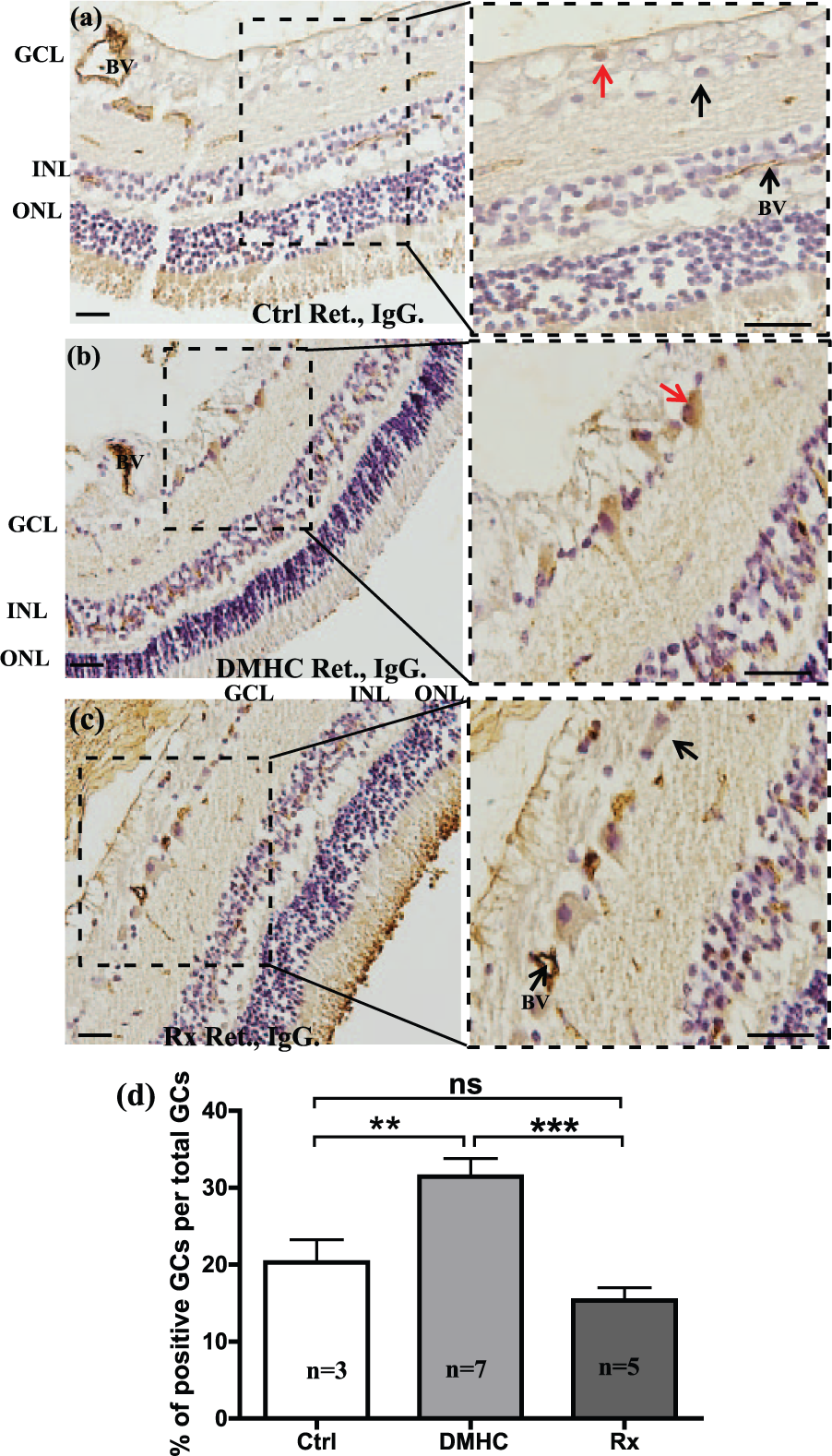
DMHC pigs have the highest incidence of IgG-labelled ganglion cells. Retinal (Ret) sections from pigs of (a) control (Ctrl), (b) DMHC and (c) Darapladib treatment (Rx) groups were probed with anti-swine IgG antibodies. DMHC pigs demonstrated the highest number of IgG-labelled ganglion cells (red arrow). Ganglion cells in the other treatment groups failed to show similar extent of IgG labelling (black arrowhead). (d) The percentage of IgG-labelled ganglion cells in the GCL for each treatment group was quantified using ImageJ software and IHC. Scale bar: 50 µm.
DMHC triggers reactive gliosis in the retina
The effects of chronic DMHC with or without Darapladib treatment on the activity of retinal glial cells were also investigated. The level of expression of GFAP in the retina was used to access the activity of glial cells and their response to the condition of chronic DMHC (Figure 4). Results showed that GFAP expression was markedly increased in the DMHC retina compared to controls and Darapladib-treated DMHC pigs (Figure 4(a) to (d)). In the DMHC group, the GFAP-positive fluorescence signal extended from the inner- to outer-limiting membranes, confirming that GFAP expression was predominantly associated with Müller cells (Figure 4(b)). The proximal and distal ends of Müller cells positioned within the GCL exhibited the most intense GFAP expression in the DMHC group (Figure 4(b)). By contrast, in both control and DMHC-Darapladib animals, GFAP immunoreactivity was generally reduced and confined to the GCL (Figure 4(a) to (c)). We also measured and compared the intensity of GFAP-positive immunostaining (Figure 4(d)). An eightfold increase in GFAP-positive signal was observed in the DMHC group compared to controls. Additionally, although Darapladib treatment significantly reduced GFAP expression in DMHC animals by nearly threefold, GFAP expression was still significantly elevated in DMHC-Darapladib pigs compared to controls (Figure 4(d)).
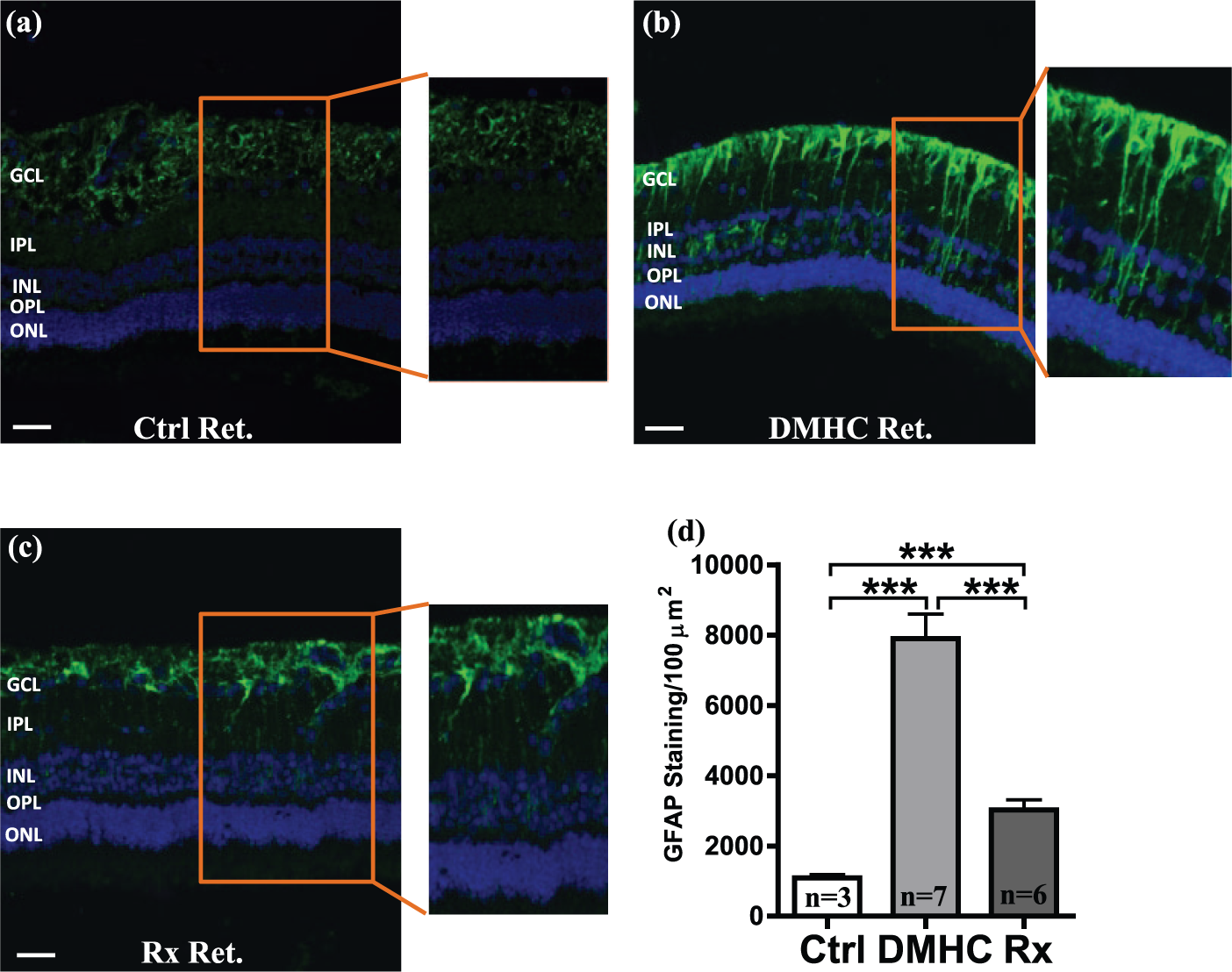
DMHC induction and Darapladib treatment alter GFAP expression. Retinal (Ret) sections from (a) control (Ctrl), (b) DMHC and (c) Darapladib-treated pigs (Rx) were probed with anti-GFAP antibody to study its expression by Müller cells. (d) Using ImagePro software and IHC, GFAP expression by pigs from all treatment groups were quantified. Scale bar: 50 µm.
The functional status of the BRB and BBB is correlated
Given the comparable architecture and overlapping functional responsibilities of the BRB and BBB, we next explored what sort of correlation there is, if any, between the functional status of the BRB and BBB. In an attempt to compare the functional integrities of the BRB and BBB, we compared reformulated data obtained from our previous study 6 and juxtaposed it to new data obtained here from measurements of the retinal microvasculature. Results show that there is a significant correlation between the permeability status of the BBB and BRB in each of the three subject groups (Figure 5).
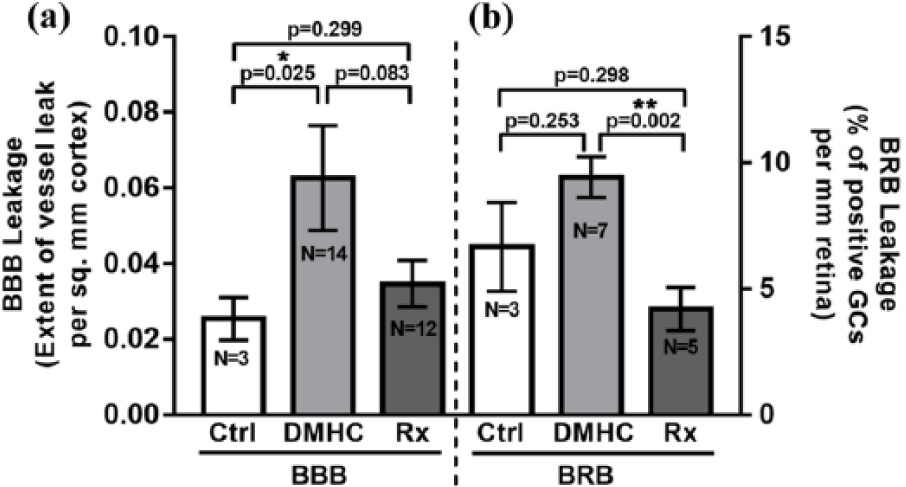
(a) Response profiles of the BRB and BBB are comparable to DMHC and Darapladib. We compared averaged values of BBB leakage in DMHC and Darapladib-treated DMHC pigs with the values for BRB leakage in the same cohorts. Both the BBB and BRB underwent similar changes in each treatment group, suggesting that the status of the BRB can be used as an indicator of the status of the BBB and perhaps the presence of associated BBB pathology. (b) The data from individual pigs were compiled and used for comparison of BBB and BRB permeability in the same animals. The semiquantitative data was juxtaposed to compare extent of leakiness in the BRB and BBB. Individual values were obtained as GCs per millimetre retina (for BRB) or plasma leak cloud area per square millimetre cerebral cortex (for BBB).
Chronic DMHC alters claudin-5 and occludin protein expression in the cerebral cortex
We next sought to determine the effects of chronic DMHC with or without Darapladib treatment on the expression and localization of claudin-5 and occludin proteins in brain vascular endothelial cells (BVECs) using IHC and image analysis (Figure 6). To address this, we focused on the blood vessels in the cerebral cortex instead of the highly laminated retina for three reasons: (1) claudin-5 and occludin are expressed in BVECs that provide the key structural and functional components of the BRB and BBB, (2) the cerebral cortex provides a greater tissue volume for carrying out more accurate protein localization and expression studies and (3) the cerebral cortex has an extensive and more randomized plexus of blood vessels that is readily visualized and thus more amenable to protein localization and expression studies than are comparable vessels in the retina. As expected, these proteins are expressed solely in BVECs and were localized primarily to the BBB-associated tight junctions in all treatment groups (Figure 6(a) to (f)). Neurons and astrocytes lacked claudin-5 and occludin expression (Figure 6). The specificity of the anti-claudin-5 and anti-occludin immunostaining was verified by controls that included probing with secondary antibody only (Figure 6(g)) or blocking sera only (data not shown).
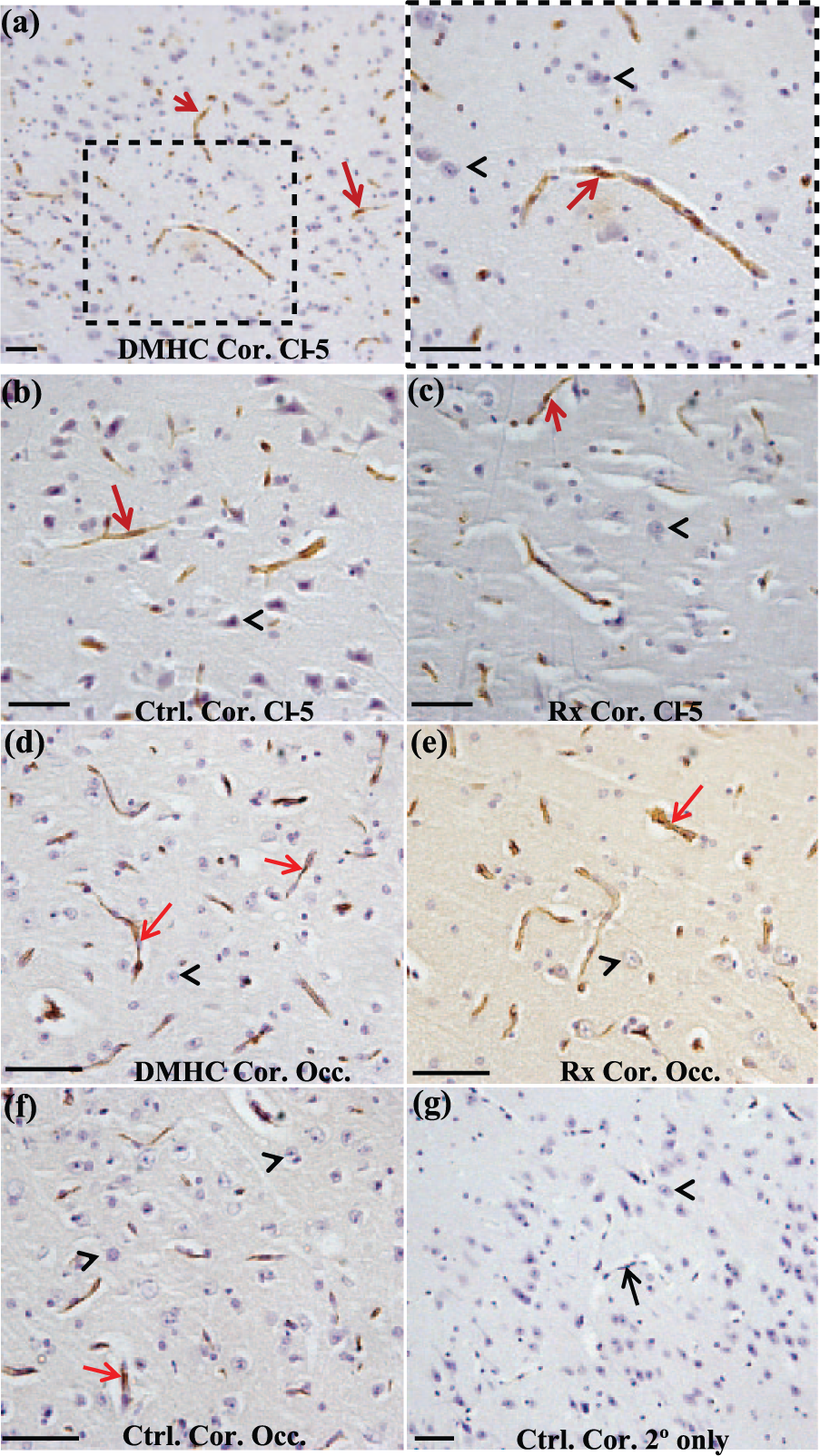
Selective expression of claudin-5 and occludin by BVECs in the cerebral cortex groups. Cortical sections of (a) DMHC, (b) age-matched control (Ctrl) and (c) drug treated (Rx) were probed with anti-claudin-5 antibodies (Cl-5) to study the localization of claudin-5. For better visualization, a small region of DMHC cortex is also magnified, inset (a). Similarly, cortical sections from (d) DMHC, (e) age-matched control and (f) drug treated were probed with anti-occludin antibodies (Occ). Claudin-5 and occludin localization in the cerebral cortex present as punctate spots on the BVECs of cerebral blood vessels (red arrows). Cortical pyramidal neurons were devoid of both claudin-5 and occludin (black arrowheads). (g) The specificity of anti-claudin-5 and anti-occludin antibodies was tested by probing cortical tissue with secondary antibody only. Scale bar: 50 µm.
Figure 7 shows a comparison of the relative levels of claudin-5 and occludin proteins in BVECs of the cerebral cortex in DMHC, DMHC-Darapladib and control pigs (Figure 7). Brains were divided anteroposteriorly into three regions: anterior, middle and posterior, and brain samples were taken from each region for analysis. MATLAB was used to evaluate claudin-5 and occludin protein expression in each of the three brain regions in an effort to account for potential region-specific variations in the density and type of vessels. In the anterior cortex, DMHC pigs showed the greatest expression of claudin-5, followed by the Darapladib-treated DMHC and age-matched control pigs (Figure 7(a)). Claudin-5 expression in the anterior cortex of DMHC and Darapladib-treated DMHC pigs was significantly higher than in controls (Figure 7(a)). The DMHC-Darapladib group showed a reduction in claudin-5 expression compared to the DMHC group, but this difference did not reach statistical significance. In the middle region of cortex, claudin-5 expression was similar to that of the anterior cortex, being significantly higher in the DMHC group compared to controls (Figure 7(b)). The reduction in claudin-5 expression in Darapladib-treated DMHC compared to DMHC-only animals approached statistical significance. Interestingly, treatment of DMHC animals with Darapladib resulted in a reduced expression of claudin-5 to levels not significantly different from controls. Finally, in the posterior cerebral cortex, the DMHC-Darapladib group showed the highest expression of claudin-5, followed by DMHC and controls, with the variations attaining statistical significance in all treatment groups (Figure 7(c)).
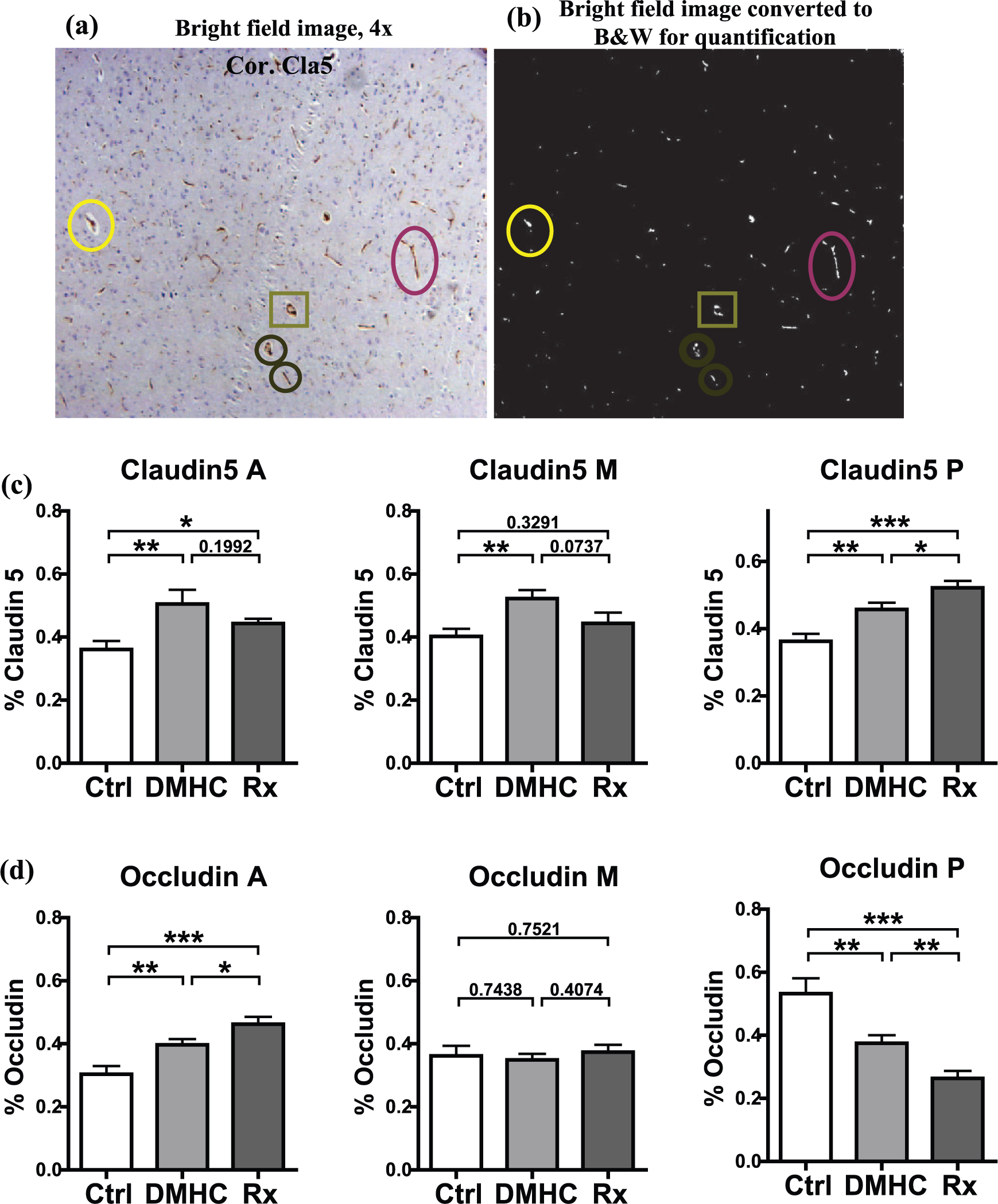
DMHC induction and Darapladib treatment influence the expression of claudin-5 and occludin protein in cortical vasculature. Cortical sections probed with claudin-5 and occludin antibodies were photographed and analysed using MATLAB software. (a) Regions of TJP localization were converted to white areas while (b) non-TJP areas appeared black. The corresponding punctate TJP spots on some blood vessels are encircled in (a) original and (b) MATLAB images. The percentage of total area occupied by all the white spots against a black background of all images per brain region was calculated and used as the quantitative representation of TJP localization. Using IHC and MATLAB, localization of claudin-5 (c) and occludin (d) in the anterior (A), middle (M) and posterior (P) brain regions was quantified.
The pattern of occludin expression in the cortical microvasculature differed from claudin-5 for all three brain regions (Figure 7(d) to (f)). In the anterior cerebral cortex, the DMHC-Darapladib group exhibited the highest level of occludin expression, followed by DMHC and controls (Figure 7(d)). Differences among the treatment groups attained statistical significance. In the middle region of cortex, all treatment groups showed comparable levels of occludin expression (Figure 7(e)), whereas in the posterior region of cortex, the occludin expression pattern was opposite to that of the anterior cortical regions, with the lowest expression measured in the DMHC-Darapladib group and the highest in controls (Figure 7(f)). The difference in occludin expression reached statistical significance in the posterior brain region. In terms of significance to barrier function, these results are in accordance with the current thinking that claudin-5 may be more important for the functional integrity of the BBB than occludin.
Chronic DMHC alters claudin-5 and occludin gene expression in the cerebral cortex
Changes in claudin-5 and occludin gene expression in response to DMHC and Darapladib treatment were assessed in the middle brain region only of the pig cerebral cortex by RT-PCR using beta-actin as a reference (Figure 8). Unlike results for protein localization and expression, patterns of claudin-5 and occludin gene expression were similar, with the highest expression levels in the Darapladib-treated DMHC group, followed by DMHC and controls (Figure 8(a) and (b)). Differences in both claudin-5 and occludin gene expression between the Darapladib-treated DMHC and control groups approached statistical significance.
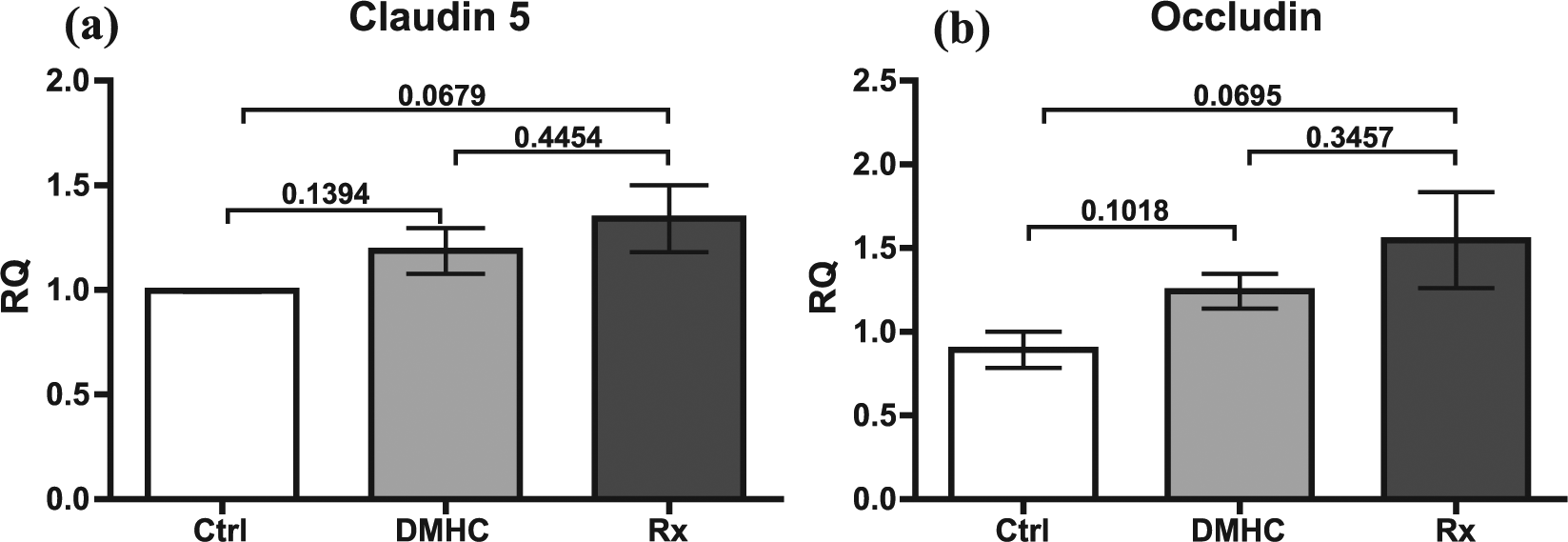
DMHC induction and Darapladib treatment alter TJP gene expression. Gene expression studies of (a) claudin-5 and (b) occludin carried out for the middle brain cortex of age-matched control (Ctrl), DMHC and Darapladib-treated (Rx) pigs. Relative quantification of claudin-5 and occludin gene expression was carried out against β-actin, a housekeeping gene. Scale bar: 50 µm.
Discussion
DM and HC are considered major risk factors for a number of diseases, including diabetic retinopathy and Alzheimer’s disease.1–3,10–13 In humans, DM and HC often occur concurrently, and this close interrelationship has been linked to well-known deleterious effects on the vasculature, particularly in the elderly, some of which may have their origins in vascular inflammation.11,12,14–16 An earlier study investigating the role of inflammation in atherosclerosis in a DMHC swine model identified lysophosphatidyl choline (LPC) and oxidized non-esterified fatty acids (oxNEFAs) as proinflammatory and proapoptotic lipid mediators associated with recruitment and activation of macrophages, apoptosis and impaired removal of dead cells. 17 LpPLA2, an enzyme secreted by T-lymphocytes, monocyte-derived macrophages and mast cells cleave oxidized phosphatidyl choline molecules, generating LPC and oxNEFAs. 17 Darapladib, a potent inhibitor of LpPLA2, was shown to block the progression of arterial atherosclerotic plaques to a high-risk phenotype by reducing inflammatory macrophage infiltration in a DMHC swine model. 8 Subsequently, we reported that DMHC increased BBB permeability, selective binding of IgG to cortical pyramidal neurons and increased intraneuronal deposition of Aβ42 in the cerebral cortex in these same DMHC pigs and that these effects were partially ameliorated by Darapladib. 6 In addition, a recent placebo-controlled phase IIa study on the efficacy of Darapladib treatment on individuals suffering from diabetic macular oedema demonstrated modest improvements in their vision and in the extent of macular oedema within 3 months of treatment. 18
The above-mentioned effects of DMHC on the cortical vasculature and retina prompted us to further investigate its influence on the retina in the pig model. In this study, we observed an increase in the permeability of the BRB of DMHC pigs. Pigs have many similarities in their metabolism and physiology to that of humans. Importantly, their organs are large enough to be supported by vessels of comparable hierarchy. For this reason, pigs are an ideal animal model to study abnormalities associated with blood vasculature and their relationship with downstream pathologies in both the eye and brain. 7 Also, unlike rodent models, pigs are naturally vulnerable to atherosclerosis and DM and can develop cardiovascular anomalies that mimic those in humans. In this study, we found that chronic DMHC did not cause changes in the overall thickness of the pig retina but was associated with significant decreases in the thicknesses of specific layers within the retina, namely, the GCL, ONL and IS/OS layer, compared to controls. However, the IPL showed increased thickness compared to other treatment groups. In addition, thinning of the GCL and INL was associated with a reduction in number of cells populating these layers. In DMHC pigs treated with Darapladib, the thicknesses of the GCL, ONL and IS/OS layer remained comparable to controls, possibly due to inhibition of cell loss within these layers. Darapladib treatment also maintained IPL thickness comparable to controls.
Several studies carried out in patients and animal models with DM have reported an increased BRB permeability and altered tight junction protein expression in the retina.4,19–23 In this study, a DMHC-associated BRB compromise was revealed by detection of the selective binding of IgG to ganglion cells within the GCL. IgG is normally excluded from the retinal interstitium and thus, as in the brain, can be used as an effective reporter molecule to identify sites of increased BRB permeability. The effects of chronic binding of IgG on ganglion cell functions are unknown. However, in our previous study that investigated the effects of chronic DMHC on the cerebral cortex in these same pigs, we demonstrated a selective and increased binding of IgG to neurons and intraneuronal Aβ42 deposition in the regions exhibiting a compromised BBB. 6 Similarly, in an in vitro study, binding of IgG to cortical pyramidal neurons dramatically increased the accumulation of soluble, exogenous Aβ42 peptide in adult mouse brain slice cultures. 9 These results strongly suggest that the influx of IgG into the brain and retina enables IgG binding to cell surfaces and promotes chronic endocytosis which, in the case of cortical pyramidal neurons, has been linked to intracellular Aβ42 deposition, a feature presumably contributing to early AD pathogenesis and disease progression. 9 In a similar manner, binding of IgG to ganglion cells may interfere with the normal function of their cytoplasmic processes. Our data suggest a direct link between the influx of plasma components including IgG into the retina and the reduced thickness and increased cell loss within individual retinal layers. The fact that Darapladib treatment curtailed IgG influx via the BRB into retinal layers and stabilized the thickness of these retinal layers and the cell numbers populating them further supports a causal link between blood–tissue barrier–associated plasma influx, inflammation and degenerative changes.
DM has also been linked to activation of Müller cells resulting in enhanced levels of GFAP expression in the retina.24 –28 In this study, using the porcine model, we also looked for any early telltale signs of diabetic retinopathy such as reactive gliosis.24,26–28 Increased GFAP expression is widely used as a reliable biomarker of glial activation that usually occurs in response to injuries resulting from trauma, disease, inflammation or chemical insults. 29 Here, we show that chronic DMHC enhances gliosis in the retina as evidenced by increased expression of GFAP by Müller cells. Two types of glial cells populate the mammalian retina: Müller cells and astrocytes. 30 The cell bodies of Müller cells are positioned within the GCL, and their cytoplasmic processes extend to the inner- and outer-limiting membranes of the retina.28,30–32 Astrocytes, however, are less abundant in the retina and form a single layer in the GCL with close proximity to the inner-limiting membrane.25,31,32 Under normal, healthy conditions in the retina, GFAP is predominantly expressed by astrocytes, while Müller cells rarely express it.24,27 However, under conditions of DM or retinal injury as shown here, Müller cells become the dominant glial cell type expressing GFAP.24,26–28 This change hints at ongoing pathological changes resulting from inflammation, oxidation or degeneration in the retina, possibly as a result of chronic DMHC.21,33,34 The fact that retinas of Darapladib-treated DMHC pigs showed significantly lower expression levels of GFAP suggests that Darapladib can have beneficial effect by inhibiting or alleviating DMHC-associated pathological changes in the retina.
Although claudin-5 and occludin and their relationship to the BBB have been extensively studied, their individual contributions to the structural and functional integrity of the BBB remain unresolved.35,36 Previous studies have implicated a role for DM in BBB compromise through altering the expression of tight junction proteins.37,38 For example, Hawkins and coworkers 37 demonstrated lower levels of occludin expression in streptozotocin-induced diabetic rats. Similarly, Hoffman et al. 38 reported a reduction in claudin-5 and occludin expression in the blood vessels of the frontal cortex, hippocampus, occipital cortex, basal ganglia and cerebellum in two adolescent cases of diabetic ketoacidosis. Using hypercholesterolaemic rats, Kalayci et al. 39 reported increased BBB permeability with lowered expression of occludin in the cerebral cortex and significantly lowered expression of occludin in association with increased BBB permeability in the hippocampus and olfactory bulbs of New Zealand white rabbits fed with high-cholesterol diet.
This study revealed quantitative changes in the gene and protein expression of claudin-5 and occludin in the DMHC pig cerebral cortex. Both proteins are expressed in BVECs that serve as a key structural correlate of the BRB and BBB. In the cerebral cortex in the anterior and middle regions of the brain, DMHC pigs showed the greatest expression of claudin-5, followed by Darapladib-treated DMHC and control pigs. The Darapladib-treated DMHC group exhibited less claudin-5 expression than the DMHC group, but the differences between these two groups did not reach statistical significance. Interestingly, treatment of DMHC animals with Darapladib resulted in a reduced expression of claudin-5 to levels similar to controls. Our data show that chronic DMHC and treatment of DMHC animals with Darapladib had a clear impact on claudin-5 expression. We suggest that the overall increased expression of tight junction proteins under condition of DMHC could be a part of a compensatory mechanism implemented by BVECs in response to DMHC-induced BBB breach. In contrast to claudin-5, expression of occludin varied from one brain region to another and failed to show any reproducible trend. This may be, in part, explained by occludin’s limited role in maintaining BBB integrity.40–43 Analysis of gene expression using RT-PCR failed to reveal a clear correlation between claudin-5 and occludin gene and protein expression. These variations could be due to technical limitations. However, it is well known that messenger RNA (mRNA) expression levels are often poorly correlated with protein expression.44–46 After mRNA transcription, the generation of functional proteins involves numerous processes associated with post-transcription, translation and post-translation. Under these conditions, it is still generally accepted that a more direct demonstration of the presence of a particular protein in the tissue provides the most accurate and reliable real-time assessment of the expressional status of any given protein in a cell or tissue, as was done in this study. 46
This is the first study reporting the combined effect of DM and HC on BRB permeability, regression of retinal cell layers and expression of claudin-5 and occludin in brain vasculature in a DMHC swine model. The intricate pathways regulating these changes are poorly understood and need further investigation. These observed changes in DMHC animals could be possibly due to the inflammatory changes targeting blood vessels resulting from chronic hyperglycaemia and HC, as is thought to be the case in DMHC-induced atherosclerosis in this pig model. 17 Vascular inflammation is further implicated since pathological changes were reversed by Darapladib, a potent inhibitor of LpPLA2. In addition, in a recent study carried out in streptozotocin-diabetic rats, BRB compromise due to diabetes was alleviated by Darapladib treatment. 47
This study has some limitations. The first is the small sample size in each treatment group, due primarily to the high cost of maintaining and monitoring the DMHC pig model. Despite this shortcoming, we believe that the data obtained from this pig model are more directly relevant to human pathology and treatment as pigs have gyrencephalic brain, while rodents have lissencephalic brains. This anatomic advantage in pig brains includes a vascular hierarchy comparable to that of humans. Also, like humans, pigs are prone to naturally acquired DM and obesity. Another potential limitation is the young age of the animals used in this study. By the time of sacrifice, the pigs were less than 2 years old, an age that is not equivalent to the age range of humans afflicted with DM and AD. The primary reason for the selection of younger pigs for this study was to avoid ageing-associated co-morbid changes in the vasculature. Also, it is noteworthy to point out that the induction of DM by injection of streptozotocin more resembles type 1 DM than type 2, although type 1 DM and type 2 DM share co-morbidities. Finally, because the BBB and BRB share similar barrier functions and tight junction proteins, for practical reasons, claudin-5 and occludin protein and gene expression studies were carried out on blood vessels in the cerebral cortex instead of the retina. Although both the BBB and BRB clearly show increased permeability under conditions of chronic DMHC, it is certainly possible that BVECs in the vasculature of the cerebral cortex and retina respond differently to DMHC and Darapladib in terms of tight junction protein expression and barrier function.
Clinical perspectives
These findings support the interesting possibility that detection of BRB compromise in the eye may be generally predictive of a similar compromise of the BBB in the brain, thus providing diagnostic utility. Fluorescein angiograms have been routinely used by ophthalmologists to assess functional integrity of the BRB because of the ease and greater predictability for early diagnosis of diabetic retinopathy. Using this approach, BRB breakdown can be detected years ahead of the clinical diagnosis of overt and symptomatic diabetic retinopathy. Since the BBB and BRB are functionally similar, affected by similar stressors, and are apparently compromised years prior to the onset of their respective diseases, monitoring BRB integrity may prove to be useful in gauging the overall health and functionality of the BBB in the brain. 48 Thus, detecting increased BRB permeability in the eye may help in pre-symptomatic diagnosis or at least a determination of risk for developing various neurodegenerative diseases in which breakdown of the BBB is thought to be a key component. Also, since Darapladib has been shown to have the beneficial effects of reducing BBB and BRB permeability under conditions of DMHC in the pig model, this drug and others with similar action may aid in the slowing or prevention of diseases like diabetic retinopathy and age-related macular degeneration in the eye and Alzheimer’s disease in the brain.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.G.N. and R.L.W. are recipients of research grants from GlaxoSmithKline; R.G.N. has filed a patent for use of Darapladib in the treatment of neurodegenerative diseases. C.H.M. is an employee of GlaxoSmithKline, the manufacturer of Darapladib.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by GlaxoSmithKline and Osteopathic Heritage Foundation.
