Abstract
The importance of white matter injury induced by diabetes in stroke severity and prognosis is largely unknown. We aimed to investigate the relationship between diabetes-related white matter injury beyond stroke lesions with acute neurological deficits and clinical outcome after stroke. In total, 36 stroke patients within 3–7 days after onset were enrolled. Neurological deficits on admission were assessed by National Institute of Health Stroke Score, and poor outcome at 3 months was defined as modified Rankin score >2. White matter tracts were compared between patients with diabetic and non-diabetic stroke using fractional anisotropy from diffusion tensor imaging. Regional white matter abnormality with decreased fractional anisotropy was observed in diabetic patients (n = 18) when compared to non-diabetic patients (n = 18). Decreased fractional anisotropy in ipsilesional distal corticospinal tract was independently associated with higher National Institute of Health Stroke Score motor component score (β = −0.444, p = 0.005), and decreased fractional anisotropy in contralesional superior longitudinal fasciculus I was independently related to poor outcome (odds ratio, 0.900; p = 0.033). Our findings suggested that only white matter injury induced by diabetes in specific tracts like corticospinal tract and superior longitudinal fasciculus beyond stroke lesions has clinically relevant, providing insight into the mechanism of stroke recovery under the diabetic condition.
Introduction
White matter (WM) occupies nearly half of the human brain and is packed with predominantly myelinated axons interconnecting different parts of the brain. 1 WM injury is as important as grey matter damage in relation to functional recovery after stroke. The commonly affected WM by stroke lesions is corticospinal tract (CST) and the extent of CST damage has been found to be related to neurological deficits,2–4 as well as clinical outcome after stroke.3,5
In diabetic patients with acute stroke, in addition to CST tracts affected by ischaemic lesions, other WM tracts beyond stroke lesions are susceptible to the damaging effects of hyperglycaemia. 6 Early studies have demonstrated an association between diabetes and the severity of white matter hyperintensities (WMHs),7,8 indirectly suggesting diabetes may be a risk factor for WM injury. In recent years, several studies using diffusion tensor imaging (DTI) have reported significantly decreased integrity of WM microstructure in diabetic patients, including CST, corpus callosum (CC), uncinate fasciculus (UF) and external capsule (EC).9–11 Therefore, abnormal WM outside stroke lesions should exist in diabetic patients following stroke. However, the effects of WM injury induced by diabetes on stroke severity and prognosis are largely unknown. Elucidation of these issues may lead to further understanding the mechanism of diabetes-related stroke recovery.
In this study, we used fractional anisotropy (FA) derived from DTI to investigate the microstructure of WM difference between patients with diabetic stroke and non-diabetic stroke and to address the following questions: (1) whether patients with diabetic stroke had regionally decreased integrity of WM beyond stroke lesions, compared to non-diabetic stroke and (2) whether injured WM in specific regions was related to both acute neurological deficits and clinical outcome.
Materials and methods
Patients
The study was approved by the Human Ethics Committee of the Second Affiliated Hospital, Zhejiang University School of Medicine, and written informed consent was obtained from all subjects. Subjects from the ongoing prospective study which was designed to analyse the structural and functional changes during stroke recovery in diabetic and non-diabetic patients were enrolled for the current analysis.
Patients with acute ischaemic stroke from the Department of Neurology in our institution between June 2013 and July 2015 were recruited in this study. Inclusion criteria were (1) first-ever ischaemic stroke, (2) single infarct in the distribution of the middle cerebral artery, (3) no evidence of haemorrhagic infarction, (4) age >18 years, (5) admission between 3 and 7 days after stroke onset, (6) National Institute of Health Stroke Score (NIHSS) of 4–21 on admission and (7) no thrombolytic or recanalization therapies. Exclusion criteria were as follows: (1) atrial fibrillation, (2) history of neurological or psychiatric disorders and (3) pre-stroke modified Rankin scale (mRS) >1. In order to avoid WMHs effects, we further excluded stroke patients with periventricular or deep WMHs with a Fazekas et al. 12 score >1 on T2 fluid-attenuated inversion recovery (T2 FLAIR) images.
Demographic information and vascular risk factors including age, gender, diabetes, hypertension, hyperlipidaemia, smoking and drinking histories were recorded. Diabetic patients were diagnosed according to American Diabetes Association criteria. 13 The duration of diabetes and fasting plasma glucose (FPG) on admission was recorded. Patients with duration of diabetes less than 1 year were excluded in our study.
Neurological deficits were assessed by NIHSS total score (t-NIHSS) half hour before magnetic resonance imaging (MRI) scan. The degree of motor deficit was evaluated by the NIHSS motor component score (m-NIHSS). Clinical outcome at 3 months was evaluated by mRS (good outcome = mRS < 3). An experienced stroke neurologist who was blinded to the imaging findings assessed NIHSS and mRS.
MRI protocol
Brain MR was performed on a 3.0T scanner (MR750, GE Healthcare, USA) using an eight-channel brain phased array coil. DTI was acquired using an echo-planar imaging (EPI) sequence with following parameters: repetition time/echo time (TR/TE) = 5000/95 ms, slice thickness = 4 mm without slice gap, matrix = 256 × 256, field of view (FOV) = 24 cm, b value = 1000 s/mm2 with 30 directions. Diffusion weighted imaging (DWI) was acquired using an EPI sequence (TR/TE = 4000/79 ms, flip angle = 90°, slice thickness = 4 mm without slice gap, matrix = 256 × 256, FOV = 24 cm and b value = 1000 s/mm2 along three orthogonal directions). T2 FLAIR was acquired using a fast spin-echo sequence [TR/TE = 7500/152 ms, inversion time (TI) = 2100 ms, flip angle = 90°, slice thickness = 4 mm without slice gap, matrix size = 320 × 256 and FOV = 24 cm].
Image processing and analysis
The ischaemic lesion on DWI images was segmented using a semi-automated threshold algorithm to identify areas with an intensity greater than the mean plus 2 standard deviations of the value in the homologous contralateral region. 14 In order to exclude the effects of stroke lesions on FA map, all lesions were transformed so that they could be mapped to one hemisphere and further masked by lesion map. First, all right-sided lesions were flipped to the left side. Second, all lesions were normalized to a standard Montreal Neurological Institute (MNI) space, re-sliced with a voxel size of 1 × 1 × 1 mm3. Finally, the lesion overlap map was created (Figure 1) and further made as a binary lesion map.
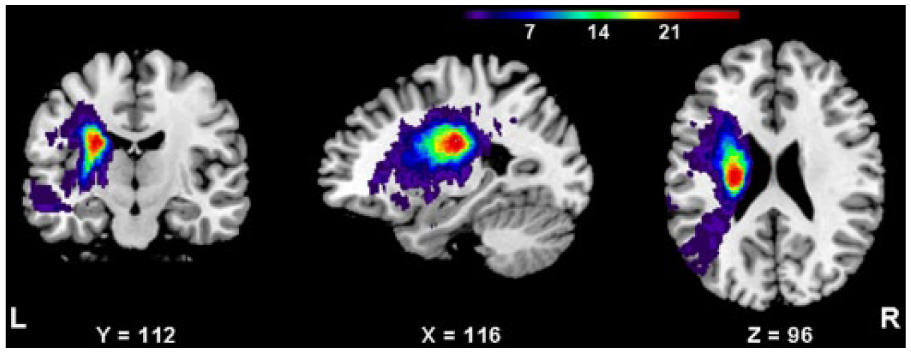
Lesion overlap map showing the distribution of patients’ lesions (n = 36). The colour bar indicates the number of patients with a lesion in that voxel.
DTI images were processed using FMRIB’s Software Library (FSL). The DTI data were corrected for head movement and eddy current distortions with the b0 image as a reference image. Diffusion tensor models were fitted independently for each voxel and FA maps were generated. For FA maps with right stroke lesions, they were right-left flipped. Then, all the FA maps were normalized to MNI space, re-sliced with a voxel size of 1 × 1 × 1 mm3. To exclude the influence of stroke lesions, FA maps were masked by the processed binary lesion map. Then, all the FA maps were smoothed using a Gaussian filter with full width at half maximum of 4 mm.
The WM differences between diabetic and non-diabetic stroke patients were performed by voxel-based analysis using Statistical Parametric Mapping 8 (SPM8). A threshold of FA >0.2 was used to restrict the significant clusters in WM regions. 15 Type I error was controlled with multiple comparison correction under the method of threshold-free cluster enhancement (TFCE) implemented in TFCE-toolbox in SPM8 by Christian Gaser (http://dbm.neuro.uni-jena.de/tfce/). Clusters of over 100 voxels with a corrected p value of <0.05 were considered significant and reported in our result. 16 Average FA values from significant clusters were extracted to analyse their relationships with FPG on admission, duration of diabetes and clinical scores in diabetic patients. Taking into account the possible influence of hemispheric lateralization on NIHSS, 17 we compared t-NIHSS and m-NIHSS between patients with left- and right-sided stroke lesions.
For each subject, the extent of CST damage was calculated by the volume of normalized stroke lesion overlap with CST which was extracted from Johns Hopkins University (JHU) WM template.
Statistics
The clinical and imaging variables between two groups were compared using two sample independent t-test or Mann–Whitney U test for continuous variables and Pearson’s χ2 test or Fisher exact test for categorical variables. The associations between FA in significant clusters with FPG and duration of diabetes were analysed by partial correlation, controlling for age and gender. The relationships between FA in significant clusters with t-NIHSS and m-NIHSS were analysed using univariate and multivariate linear regression (enter method) and the standardized coefficient (β) was estimated. Variables that were significant at the p < 0.1 from the result of comparisons between patients with good and poor outcomes were entered into the multivariate logistic regression model (forward method) to analyse the independent factors associated with poor outcome. Odds ratio (OR) with its 95% confidence intervals (CI) was estimated. A two-tailed value of p < 0.05 was considered significant. Statistical analysis was performed using SPSS 20.0 for Windows software (IBM, USA).
Results
Patient characteristics
A total of 36 stroke patients were recruited, mean age was 59.1 ± 13.2 years (range, 35–81 years) and 17/36 (47.2%) were males. The time from symptom onset to MR examination was 5.5 ± 1.2 days (range, 3–7 days). t-NIHSS and m-NIHSS were 8.2 ± 2.8 (range, 4–14) and 4.6 ± 2.3 (range, 0–8), respectively. A total of 12/36 (33.3%) had poor outcome at 3 months. The median volume of ischaemic lesion was 6.0 cm3 (range, 0.2–38.8 cm3). In total, 18/36 (50.0%) had diabetes and all were receiving oral hypoglycaemic agents. The mean FPG was 10.0 ± 3.7 mmol/L (range, 4.9–21.0 mmol/L) and the mean duration of diabetes was 7.2 ± 4.4 years (range, 2–20 years).
There were no significant differences in clinical and imaging characteristics between patients with diabetic (n = 18) and non-diabetic stroke (n = 18), except FPG (10.0 ± 3.7 mmol/L vs 5.1 ± 0.7 mmol/L, p = 0.001, as shown in Table 1).
Clinical and imaging characteristics between diabetic and non-diabetic stroke.
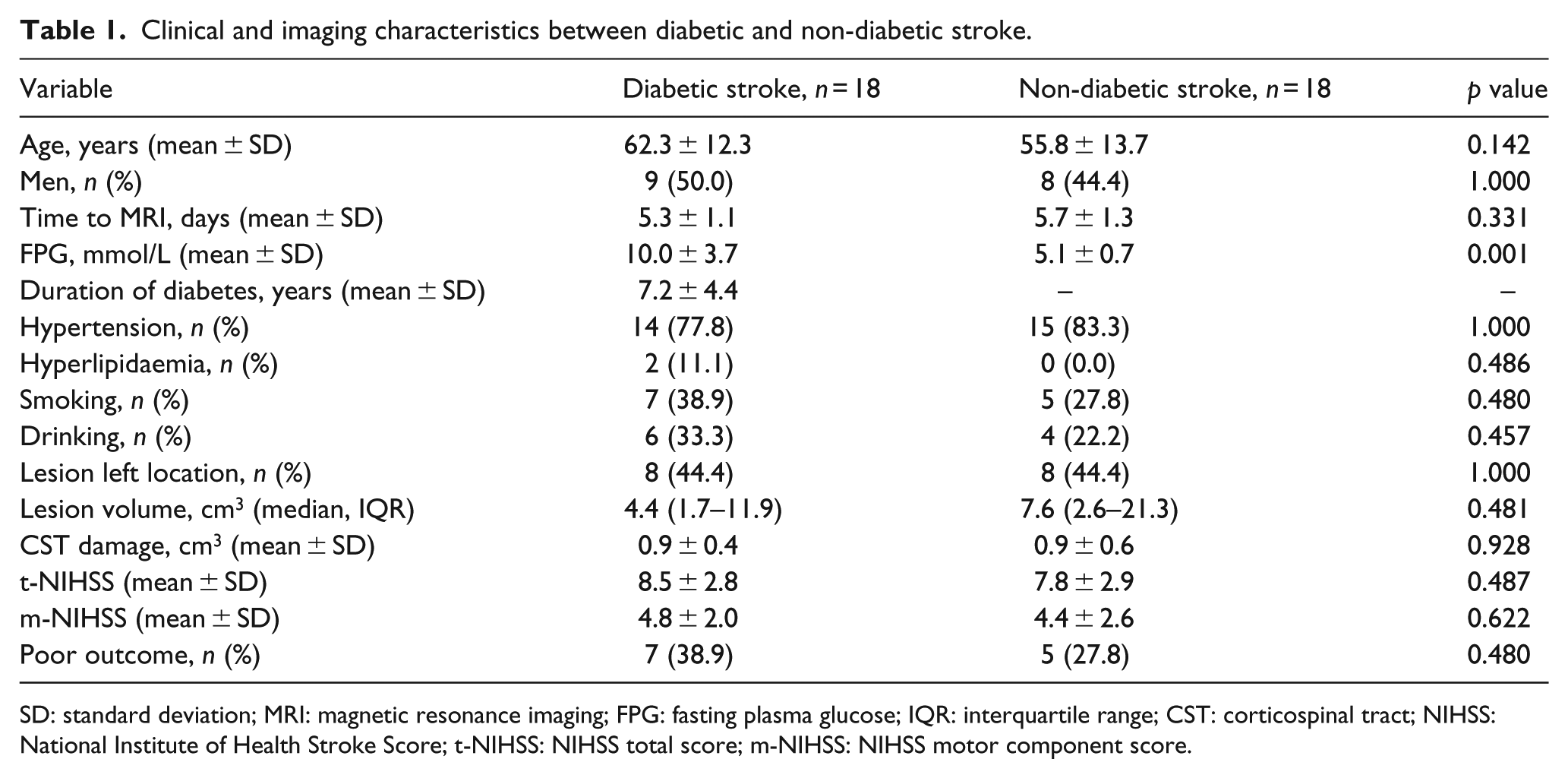
SD: standard deviation; MRI: magnetic resonance imaging; FPG: fasting plasma glucose; IQR: interquartile range; CST: corticospinal tract; NIHSS: National Institute of Health Stroke Score; t-NIHSS: NIHSS total score; m-NIHSS: NIHSS motor component score.
Abnormal WM in diabetic patients
Compared to non-diabetic patients, diabetic patients had a significantly decreased FA in the contralesional superior longitudinal fasciculus I (SLF-I), posterior cingulum (PC), EC, UF, posterior corpus callosum (PCC), bilateral CST at the level of cerebral peduncle (CST-CP), contralesional CST at the level of pons (CST-pons) and middle cerebellar peduncle (MCP). In addition, diabetic patients had a significantly increased FA in the contralesional MCP (superior to the level of MCP with decreased FA). Pearson’s correlation revealed that increased FA in MCP was negatively correlated with decreased FA in MCP (r = −0.329, p = 0.050). The location of all abnormal WM is shown in Figure 2 (detailed information is listed in Table 2).
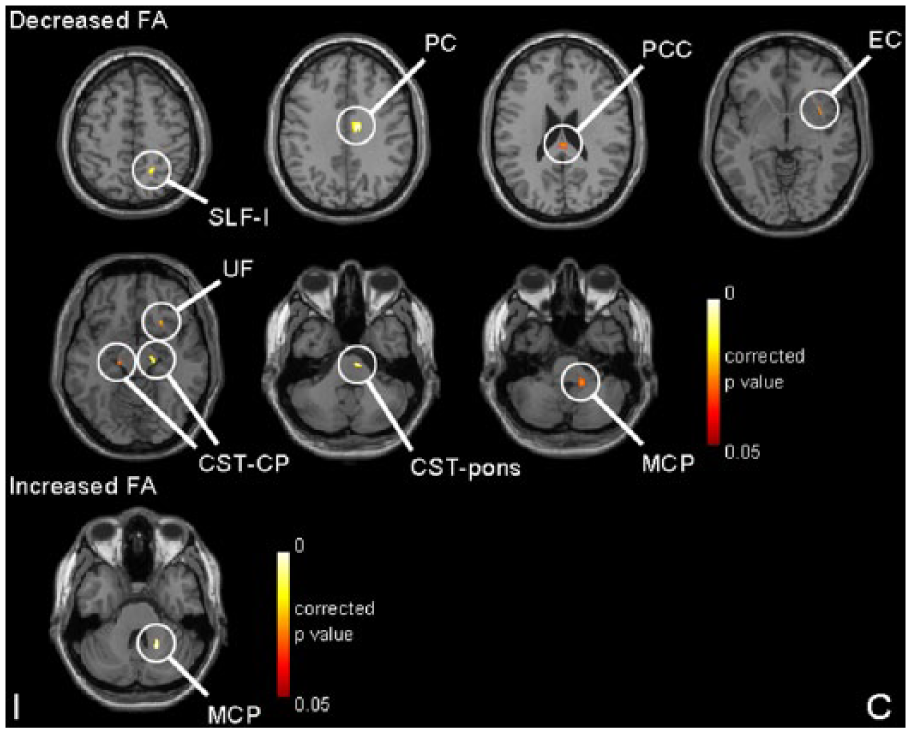
Abnormal white matter tracts with decreased and increased fractional anisotropy (FA) after voxel-based analysis in patients with diabetic stroke.
Voxel-based FA comparison between patients with diabetic stroke and non-diabetic stroke.
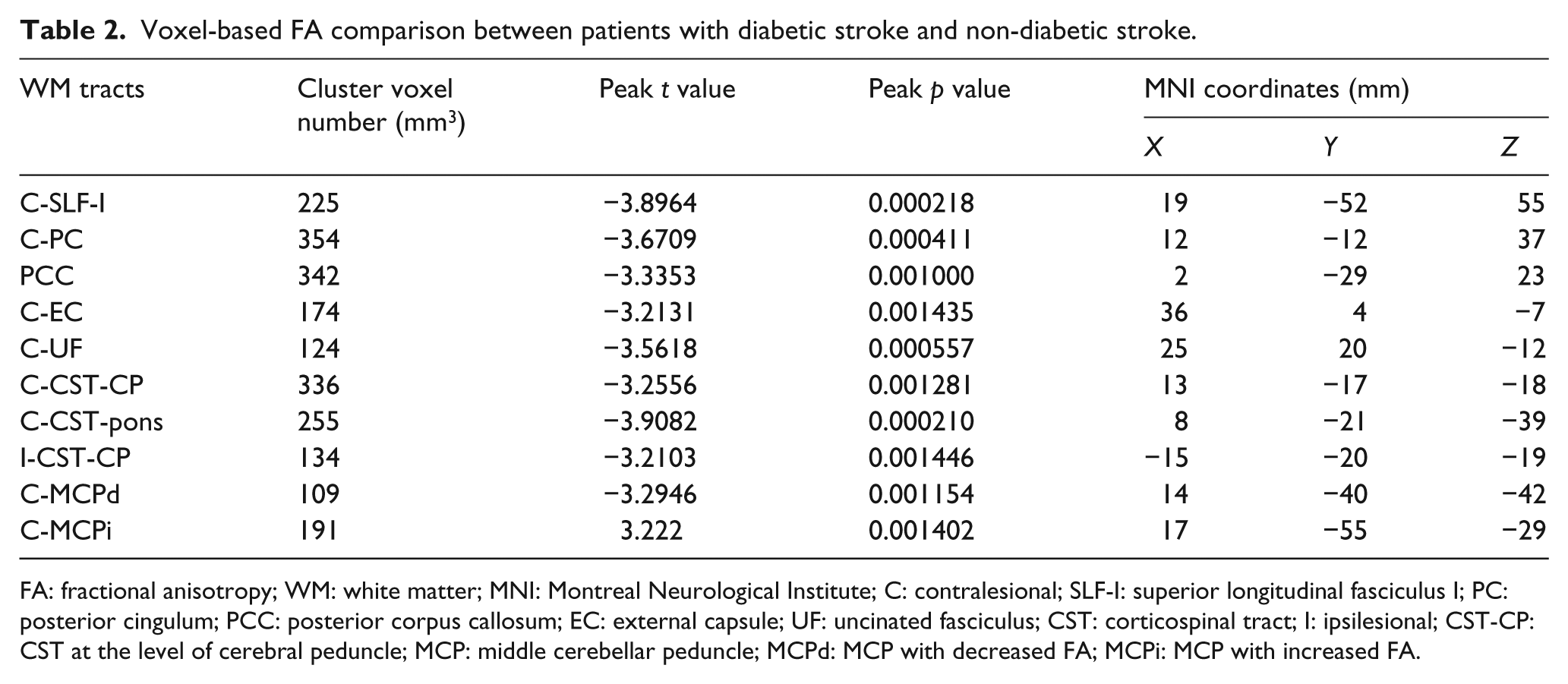
FA: fractional anisotropy; WM: white matter; MNI: Montreal Neurological Institute; C: contralesional; SLF-I: superior longitudinal fasciculus I; PC: posterior cingulum; PCC: posterior corpus callosum; EC: external capsule; UF: uncinated fasciculus; CST: corticospinal tract; I: ipsilesional; CST-CP: CST at the level of cerebral peduncle; MCP: middle cerebellar peduncle; MCPd: MCP with decreased FA; MCPi: MCP with increased FA.
There were significantly negative correlations between FA in the contralesional SLF-I (r = −0.335, p = 0.053), UF (r = −0.438, p = 0.010), CST-pons (r = −0.357, p = 0.038) and MCP (FA decreased) (r = −0.349, p = 0.043) with FPG, and a significant positive correlation between FA in the contralesional MCP (FA increased) and FPG (r = 0.480, p = 0.004). No significant correlations were found between FA in significant WM clusters with duration of diabetes.
WM injury related to neurological deficits
There were no significant differences in t-NIHSS (8.6 ± 2.8 vs 7.9 ± 2.9, p = 0.460) and m-NIHSS (4.1 ± 2.1 vs 5.1 ± 2.5, p = 0.240) between patients with left-sided strokes (n = 16) and right-sided strokes (n = 20). Among the abnormal WM tracts, only decreased FA in the ipsilesional CST-CP was negatively related to m-NIHSS (β = −0.335, p = 0.046, as shown in Figure 3), but not related to t-NIHSS (β = −0.155, p = 0.368). After controlling for age, gender, time to MRI examination, lesion location (left or right side), lesion volume and degree of CST damage, multivariate linear regression analysis revealed that in addition to lesion volume (β = 0.351, p = 0.009) and degree of CST damage (β = 0.543, p < 0.001), decreased FA in the ipsilesional CST-CP remained independently related to m-NIHSS (β = −0.444, p = 0.005).
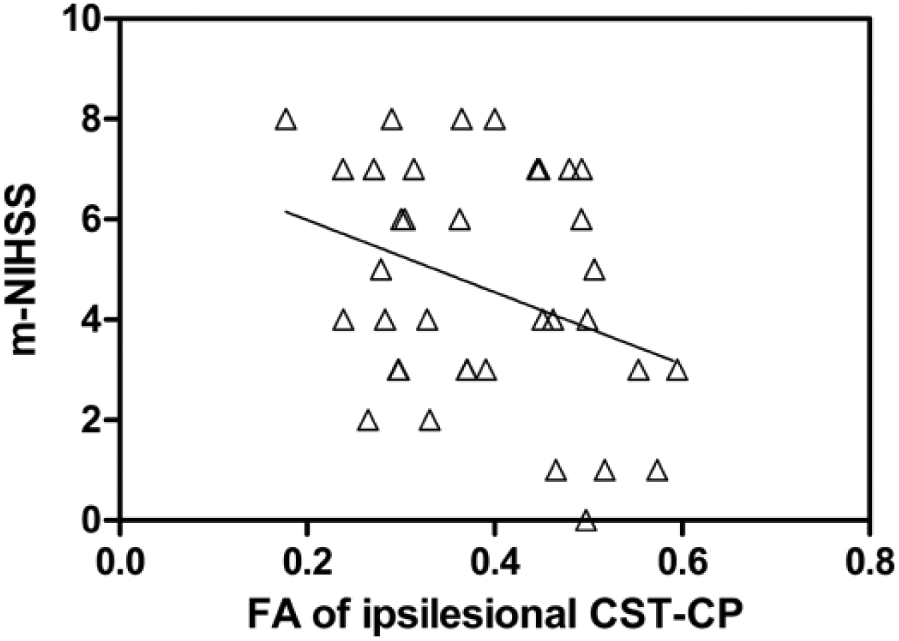
Scatter plot of motor component of National Institute of Health Stroke Score (m-NIHSS) in relation to fractional anisotropy (FA) in the ipsilesional corticospinal tract at the level of cerebral peduncle (CST-CP).
WM injury related to poor outcome
FA in the ipsilesional CST-CP (0.34 ± 0.10 vs 0.42 ± 0.10, p = 0.045), contralesional CST-CP (0.53 ± 0.09 vs 0.46 ± 0.11, p = 0.081) and SLF-I (0.24 ± 0.09 vs 0.31 ± 0.07, p = 0.022) were found to be lower in patients with poor outcome than those with good outcome. Besides, patients with poor outcome had older age (p = 0.051), shorter time to MRI examination (p = 0.077), higher t-NIHSS (p = 0.030) and m-NIHSS (p = 0.066), and lesion volume (p = 0.016, as shown in Table 3). After entering the factors including age, time to MRI examination, t-NIHSS, lesion volume and FA in the bilateral CST-CP and contralesional SLF-I in the multivariate regression model, only decreased FA in the contralesional SLF-I was independently related to poor outcome in diabetic stroke (OR, 0.900; 95% CI, 0.817−0.992; p = 0.033).
Clinical and other imaging characteristics between good and poor outcomes in stroke patients.
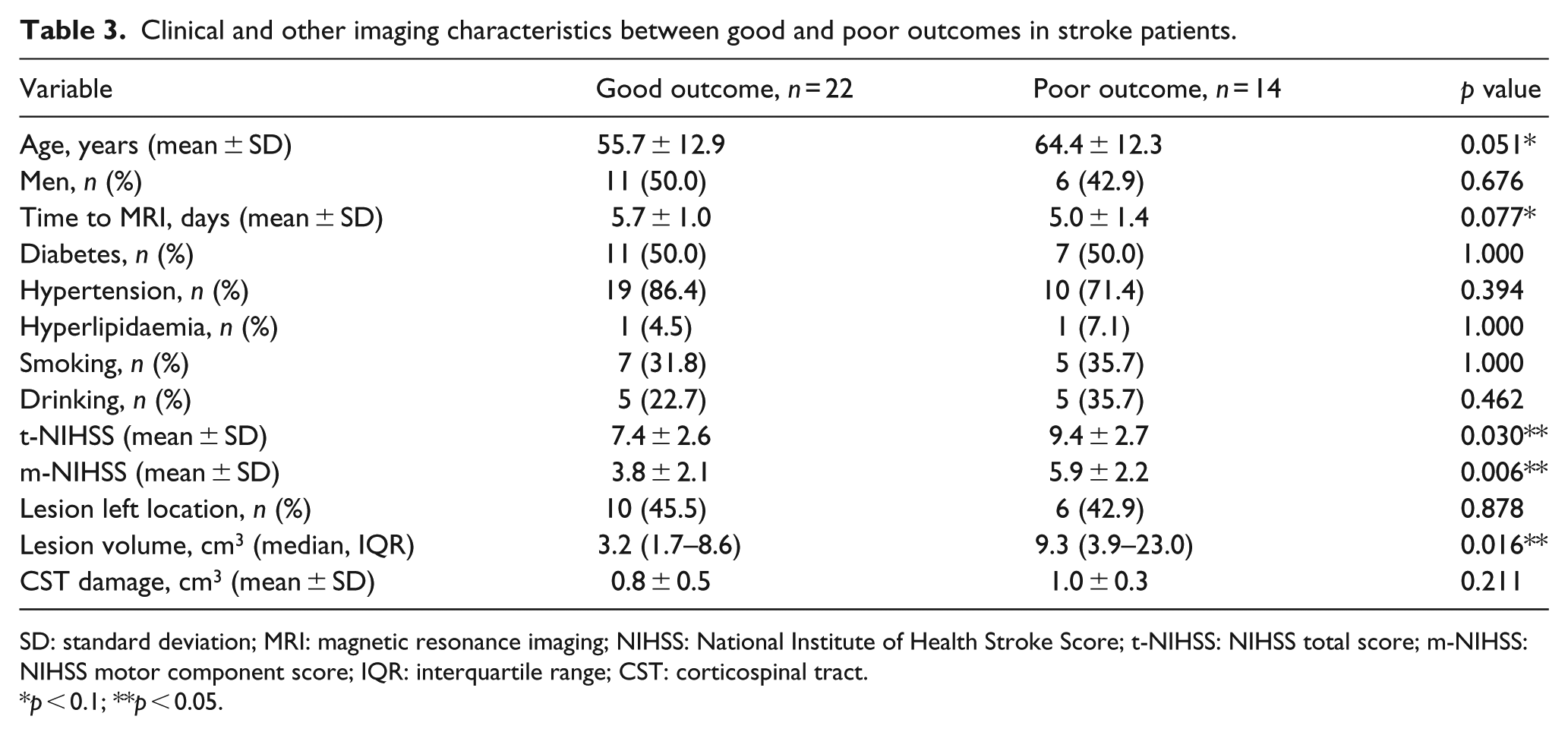
SD: standard deviation; MRI: magnetic resonance imaging; NIHSS: National Institute of Health Stroke Score; t-NIHSS: NIHSS total score; m-NIHSS: NIHSS motor component score; IQR: interquartile range; CST: corticospinal tract.
p < 0.1; **p < 0.05.
Discussion
WM fills nearly half of the brain, but receives disproportionately less scientific attention when compared to grey matter. WM forms various groups of association, commissural and long projection fibres, and thus represents a complex network of ‘communication highways’ that avoid local traffic and serve for rapid undisturbed delivery of signals in the brain. In the last decade, the importance of WM tracts like CST and CC in functional disability after stroke has become clear.4,18 In addition to stroke lesion itself, vascular risk factor such as diabetes could contribute to WM injury as well. 19 Thus, WM could be compromised before the occurrence of stroke in diabetic patients. Improvement in understanding of WM neuropathology in the clinical settings of acute stroke with diabetes has clinical implications for exploring new pharmacologic treatment strategies.
In this exploratory study, we observed that there were regionally decreased FA in WM beyond ischaemic lesions in diabetic patients. Decreased FA in the ipsilesional distal CST was an independent factor related to acute motor function deficits after stroke, in addition to lesion volume and CST damage caused by stroke lesion. And, decreased FA in the contralesional SLF-I was independently associated with poor stroke outcome at 3 months. These findings suggest that WM injury at specific regions induced by diabetes is clinically relevant, involving in both stroke severity and prognosis.
Our study showed that patients with diabetes had focal WM injury in several fibre tracts, including SLF-I, PC, PCC, EC, UF, CST and MCP. The majority of damaged WM tracts were in line with previous DTI studies reported in diabetic subjects.9–11 It has been reported that diabetic patients with poor glycaemic control are more likely to be associated with WM damage. 20 We further found that the degree of injured WM in SLF-I, UF, CST-pons and MCP at the contralesional hemisphere was related to higher FPG. The mechanism for WM damage related to diabetes is still not clearly known. Chen et al. 6 reported that hyperglycaemia decreased neuronal and oligodendrocyte cell survival and neuronal dendrite outgrowth, inducing severe WM and axonal damage in diabetic mice. They also found that hyperglycaemia increased matrix metalloproteinases (MMP) expression and inhibition of MMP decreased neuronal and oligodendrocyte death. 6 Another hypothesis is that hyperglycaemia initiates disruption of the blood brain barrier (BBB) with subsequent damage to the WM tracts. 21 Notably, the release of MMP is also the key factor in the process of BBB damage through disrupting the tight junction proteins and opening the BBB. 21
Both lesion volume and degree of CST damage are well known to be related to neurological deficits and motor disability. 22 This study further showed that WM injury in the ipsilesional CST-CP was also independently related to motor function deficits. The distal CST degeneration induced by diabetes may explain the previous finding that diabetic patients have more pronounced neurological deficits with higher NIHSS after stroke.23,24 However, t-NIHSS and m-NIHSS were slightly higher in diabetic patients in our study, which did not reach significant difference, possibly because of small sample size. In previous studies, distal CST degeneration occurred at least 2 weeks after stroke, which was caused by ischaemic lesions through Wallerian degeneration.25,26 In our study, the abnormal CST was detected at 3–7 days after stroke onset when the process of Wallerian degeneration is not present because it usually begins at the chronic stage of stroke in the human brain. 27 Therefore, decreased FA in distal CST in our study should depend on the history of diabetes.
Although stroke patients with poor outcome had lower FA in distal CST at the bilateral hemispheres and in SLF-I at the contralesional hemisphere, only reduced FA in the contralesional SLF-I was associated with poor outcome, independently of age, time to MRI examination, stroke severity and lesion volume. There is less known about the role of SLF in stroke recovery. Anatomically, SLF is a multiple-component fibre bundle which connects between the parietal and frontal lobe and consists of three subcomponents: SLF I–III. 28 Jang and Hong 29 using DTI probabilistic tractography method found that SLF-I originated from the superior parietal lobule and medial parietal cortex and then terminated to the supplementary motor area and premotor area. An early study from Petrides and Pandya 30 using an autoradiographic technique found a similar pathway of SLF-I. Therefore, SLF-I may be a node of functional connectivity between motor network and other networks. During the course of stroke recovery, in addition to motor network, other networks such as default mode network and sensorimotor network are also associated with functional recovery. 31 Damage of SLF-I may hamper the functional integration within brain networks.
Focal WM injuries in the ipsilesional distal CST and contralesional SLF-I were clinically relevant in stroke patients, indicating that only WM damage in the critical inter-hub pathway may have an impact on behaviours after stroke. It is unexpected that WM injuries in posterior part of CC and distal CST at the contralesional hemisphere, which are also important fibres in regulating motor fuction, 32 were not related to neurological deficits after stroke. This finding reflects that the neurological deficits after stroke, especially motor impairment, mainly depend on the extent of damage in CST at the ipsilesional hemisphere. It is difficult to interpret the lack of correlation between CC and contralesional CST abnormalities with prognosis, since there is currently inadequate knowledge about the mechanism of stroke recovery.
In diabetic patients, in addition to regionally decreased FA in WM tracts, increased FA was also detected in the region of MCP. Moreover, the increased FA in MCP was related to higher level of FPG. MCP is part of the cortico-ponto-cerebellar pathway that connects the cerebral motor cortex with the contralateral cerebellum and preferentially targets the lateral regions of the cerebellum. 33 Previous animal studies have revealed that an elevated FA in WM regions is consistent with WM reorganization with increased density of axon and myelin.34,35 Interestingly, in our study, the location of MCP with increased FA was superior to the level of MCP with decreased FA and there was also a negative correlation between increased FA and decreased FA in MCP. Therefore, the increased FA in MCP may be in relation to WM reorganization to compensate the adjacent WM degeneration.
Our study clearly has some limitations: first, flipping right-sided strokes to left may not be the optimal method in understanding the association of FA with outcomes measured by NIHSS and mRS because it has been reported that patients with right hemispheric lesions have a lower NIHSS compared to left-sided strokes. 17 In our sample, NIHSS score was similar between patients with left- and right-sided strokes and the abnormal WM related to NIHSS located outside of stroke lesions. Second, patients with moderate or severe WMHs have been excluded in our study, largely reducing the confounding of WMHs, yet we cannot exclude the possibility that some mild degree of WMHs might have affected the FA values. However, it is difficult to include elderly patients with no evidence of WMHs, especially for patients who have vascular risk factors.
In conclusion, regional WM injury related to hyperglycaemia was detected in stroke patients with diabetes and only WM injury in specific tracts including CST and SLF beyond stroke lesions has, respectively, impact on motor impairment and prognosis. Our results provide insight into the mechanism of stroke recovery under diabetic condition.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This study was funded by the National Natural Science Foundation of China [No.81271530] and the Zhejiang Provincial Natural Science Foundation of China [No.LZ14H180001].
