Abstract
Aim:
The aim of this study was to estimate the impact of diabetes and obesity on cerebral autoregulation in pregnancy.
Methods:
Cerebral autoregulation was evaluated in women with gestational diabetes, type 2 diabetes mellitus and/or overweight (body mass index ⩾ 25 kg m−2) and compared to a cohort of euglycaemic pregnant women. The autoregulation index was calculated using simultaneously recorded cerebral blood flow velocity in the middle cerebral artery and blood pressure. Autoregulation index values of 0 and 9 indicate absent and perfect autoregulation, respectively.
Results:
Autoregulation index in women with either diabetes (
Conclusion:
Cerebral autoregulation is not impaired in pregnant women who have non-vasculopathic diabetes or overweight. This suggests that the increased risk of pre-eclampsia in diabetic and overweight women is not associated with early impaired cerebral autoregulation.
Introduction
Pre-eclampsia (PE) is a multisystem disease, which complicates 2%–8% of pregnancies. 1 The exact pathogenesis is still unknown, but maternal obesity and insulin resistance are believed to be important contributing factors.2,3 While the risk of PE is increased with pre-gestational diabetes, the association between PE and gestational diabetes (GDM) is less pronounced, and studies are confounded by co-existing obesity, which in itself is an independent risk factor for PE.
Patients with diabetes have increased cardiovascular complications, including stroke. These complications could arise due to endothelial dysfunction, which is common in early and otherwise uncomplicated type 2 diabetes mellitus (DM2). 4
Cerebral autoregulation is a physiological process that maintains blood flow at an appropriate level, despite changes in blood pressure (BP). Studies on cerebral autoregulation in non-pregnant patients with DM2 have shown conflicting results, showing either impaired autoregulation5,6 or no difference 7 compared with control subjects. Differences in disease duration, severity and complications may underlie these inconsistencies. Preliminary evidence indicates impaired cerebral autoregulation in PE when compared to normotensive controls. 8
The effect of diabetes in pregnancy on the autoregulatory capacity is not known. Based on the increased risks of PE and cerebrovascular complications in patients with pre-gestational diabetes, but a less pronounced relationship with GDM, we hypothesize that the cerebral autoregulation is impaired in DM2, but not in GDM. Consequently, the primary aim of this study was to estimate the impact of diabetes and obesity on cerebral autoregulation in pregnancy.
Materials and methods
We conducted a prospective cohort study in non-labouring pregnant women without a history of cerebrovascular disease and with an estimated gestational age (EGA) >20 weeks. Cases included women with GDM, DM2 or overweight [pre-pregnancy body mass index (ppBMI) ⩾ 25 kg m−2] who were otherwise healthy. The referent population included healthy normotensive pregnant women with a ppBMI < 25 kg m−2. All women were screened for glucose intolerance using the 1-h 50 g glucose challenge. Study participants were recruited from the outpatient clinics at Texas Children’s Pavilion for Women in Houston and North Austin Maternal-Fetal Medicine in Austin, Texas, during a routine prenatal care visit. One experienced examiner (T.R.V.V.) performed all measurements. The study was approved by the local Institutional Review Boards at Baylor College of Medicine in Houston, Texas, and North Austin Medical Center in Austin, Texas, and informed written consent was obtained from each participant prior to data collection.
Inclusion criteria comprised pregnancy and maternal age greater than 18 years. Women were excluded if they had chronic medical illnesses (other than diabetes), used antihypertensive medications, smoked, used illicit drugs or if they developed PE during their pregnancy. Furthermore, in the control and study groups, we excluded anyone who developed GDM after enrolment. Women with diabetes were excluded if they had underlying proliferative vasculopathy (White’s classification D or higher). GDM was diagnosed according to the guidelines of the American College of Obstetricians and Gynecologists (ACOG). 9 GDM patients were categorized into A1 (diet-controlled) and A2 (medication-controlled). Women diagnosed with diabetes prior to 16 weeks of gestation were classified as ‘pre-gestational’.
Cerebral autoregulation was assessed using a combination of transcranial Doppler (TCD) and continuous non-invasive BP measurement 10 and is expressed as the autoregulation index (ARI), with 0 being absent and 9 perfect cerebral autoregulation. 10
Simultaneous TCD evaluation of both middle cerebral arteries (MCAs) was carried out using two 2 MHz pulsed, range-gated probes (Spencer Technologies, Seattle, WA), held in place using a head frame. If only one MCA could be found, that one side was used in the analysis.
BP was continuously measured non-invasively using finger arterial volume clamping (Finometer PRO; Finapres Medical Systems, Amsterdam, The Netherlands) with the servo-adjust switched off after an acclimatization period of at least 5 min, when a stable waveform was achieved with the servo-adjust on. This was subsequently calibrated with the brachial BP. The BP tracing also served to mark each cardiac cycle. End-tidal CO2 (EtCO2) was measured with a nasal cannula (Nellcor Oximax N-85; Covidien, Mansfield, MA).
All data were recorded and analysed as described previously,8,10 calculating mean BP, bilateral cerebral blood flow velocity (CBFV), EtCO2 and heart rate for each beat and using the first harmonic of BP and CBFV to obtain critical closing pressure (CrCP) and resistance area product (RAP). Cerebral autoregulation was determined from the CBFV responses to spontaneous fluctuations in mean arterial BP as described previously using fast Fourier transform (FFT) and compared to 10 template curves proposed by Tiecks et al. and used as described in previous studies8,10 Cerebral haemodynamic parameters are reported as the average over a 7-min baseline recording.
All data sets were checked for normalcy of distribution (Kolmogorov–Smirnov test). Data are reported as mean and standard deviation, or median (range) as appropriate. Analyses were performed using analysis of variance (ANOVA) with Bonferroni’s post hoc test or ANOVA on ranks with Dunn’s post hoc test (both comparisons vs the control group) (Sigmastat 2004; Systat Software, Richmond, CA).
Results
A total of 36 women with DM (GDMA1, GDMA2 and DM2, 12 in each group), 24 overweight women without DM and 24 control women were enrolled.
One woman with GDMA1, two with DM2, three with overweight/obesity and one woman in the control group later developed PE and were excluded from the analysis.
Maternal demographics were similar for both groups, except for body mass index (BMI) and gestational age at delivery (Table 1). Patients with DM2 used insulin (
Patient characteristics.
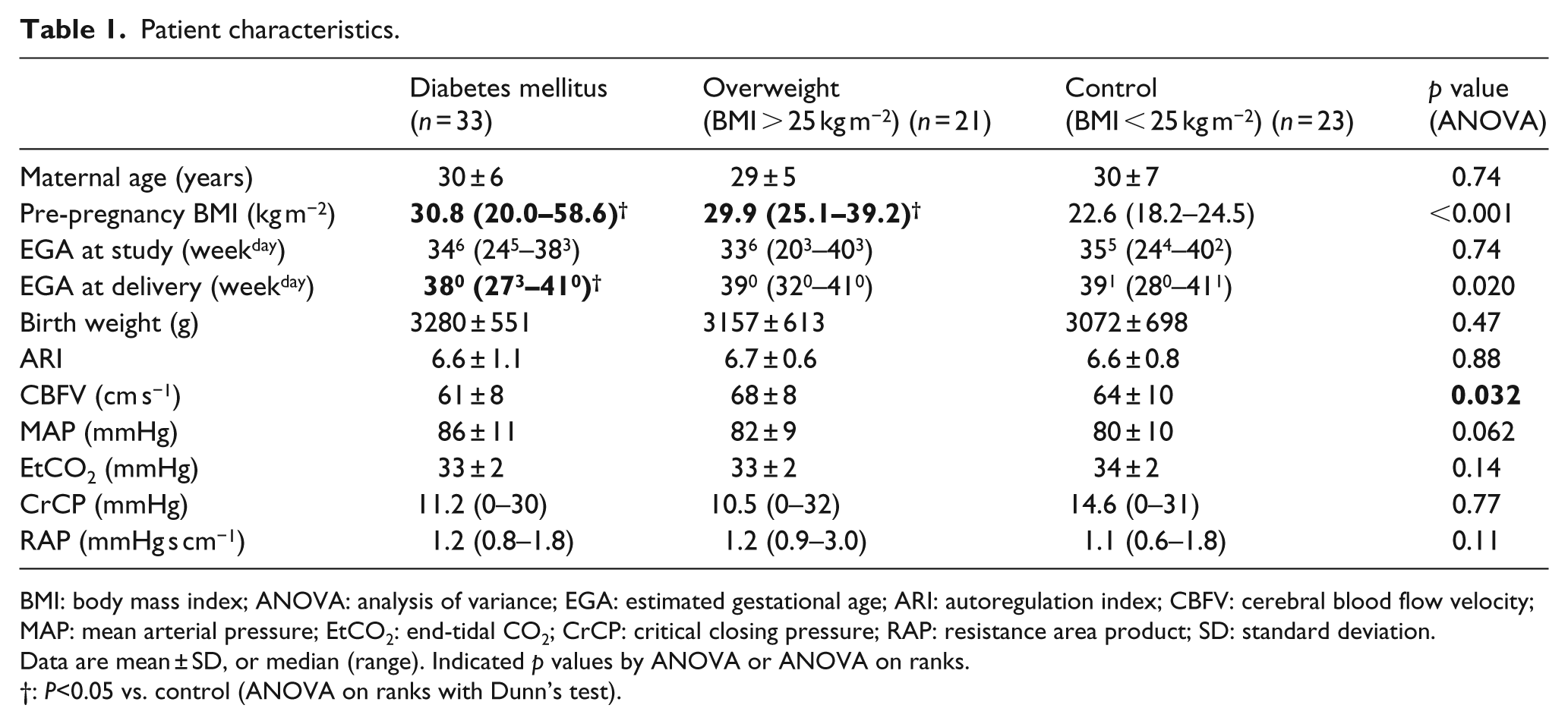
BMI: body mass index; ANOVA: analysis of variance; EGA: estimated gestational age; ARI: autoregulation index; CBFV: cerebral blood flow velocity; MAP: mean arterial pressure; EtCO2: end-tidal CO2; CrCP: critical closing pressure; RAP: resistance area product; SD: standard deviation.
Data are mean ± SD, or median (range). Indicated
: P<0.05 vs. control (ANOVA on ranks with Dunn’s test).
When compared with the control group, women with GDM, DM2 and high BMI did not have any significant ARI differences. There was also no difference noted between GDMA1, GDMA2 and DM2 (ARI 6.5 ± 1.5, 6.4 ± 1.0 and 6.9 ± 0.6,
Discussion
In this study, dynamic autoregulation in pregnancies complicated by diabetes was examined and compared to normotensive and euglycaemic pregnant controls with BMI < 25 kg m−2. The findings indicate that cerebral autoregulation is not impaired in women with (uncomplicated non-vasculopathic) diabetes in pregnancy. Furthermore, the functionality of autoregulation is equally effective in diet- and medication-controlled GDM and in euglycaemic women with and without pre-pregnancy obesity.
GDM, 11 DM2 4 and obesity 4 all are associated with endothelial dysfunction and chronic inflammation. The effect of diabetes and subsequent endothelial dysfunction on cerebral autoregulation is less clear. Non-pregnant DM2 patients with good glycaemic control and no major complications showed normal autoregulation. 7 Others found affected dynamic autoregulation in DM2 with 6 and without microvascular disease. 5 We did not find evidence suggesting affected dynamic cerebral autoregulation in our group of pregnant women with DM2, but comparison with the aforementioned studies is difficult due to differences in age, disease duration and severity, and gender. None of our patients had microvascular complications or autonomic neuropathy, which is associated with the development of PE, 12 and all patients had excellent glycaemic control (as shown by daily glucose monitoring), relatively mild hyperglycaemia and short disease duration that allowed for preserved cerebral autoregulation.
Another explanation for the absence of impaired autoregulation in this study might be pregnancy in and of itself. The ARI found in all groups seems to be in the high normal range when compared to non-pregnant subjects. 13 This might be the result of the relative hypocapnia seen in pregnancy which improves the autoregulatory capacity and the hormonal changes of pregnancy which enhance endothelial function.
One of the strengths of this study is the inclusion of both women with diabetes and two groups of women without diabetes (ppBMI < 25 and >25 kg m−2). Interpretation of the effect of GDM in pregnancy is complicated because these women are often obese, a condition known to be associated with insulin resistance, endothelial dysfunction, a pro-inflammatory state and PE. 3
This study has some limitations, which merit discussion. We did not measure glycosylated haemoglobin (HbA1c) as a marker of glycaemic control. Patients with diabetic complications (vasculopathy) were excluded from our study and clearly may have demonstrated a different response given the nature of the disease. Furthermore, we only studied autoregulation within the context of spontaneous fluctuations in BP during rest, which is a mainly myogenic activity. Therefore, we cannot exclude the possibility of cerebral blood flow (CBF) changes induced by metabolic activity such as might be present in patients with impaired CO2 cerebrovascular reactivity. 7 Finally, the women were studied at a wide range of gestational age, but with a comparable median gestational age. The effect of advancing gestational age on ARI is not known; however, we did not find a correlation between gestational age and ARI (data not shown).
In conclusion, our findings suggest the presence of normal functioning dynamic cerebral autoregulation in normal weight and high-BMI pregnant women with pre-gestational and GDM. Whether this holds true for patients with advanced diabetic complications remains to be determined by future studies.
Footnotes
Declaration of conflicting interests
The authors report no conflict of interest.
Funding
Internal funding only.
