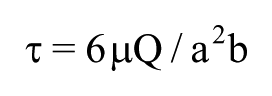Abstract
The interaction between platelets and endothelium in vivo is a complex phenomenon. Our aim was to develop an in vitro system that mimics the in vivo environment and investigate platelet function in a common pathological condition. Human umbilical vein endothelial cells were used and platelets from 28 type 2 diabetes patients were studied under shear stress conditions. Mean coefficient of variation of platelet aggregation was 10% in dynamic conditions in the presence of endothelium. Endothelial cells increased the concentration of inductor needed to achieve 50% platelet aggregation to adenosine diphosphate from 2.6 ± 1.3 in static conditions to 3.7 ± 1.3 µM in dynamic conditions. A similar pattern was observed when collagen was used for platelet activation. Incubation of endothelium with a nitric oxide inhibitor abolished this effect, indicating platelet inhibitory effect of endothelial cells is nitric oxide mediated. Platelet reactivity of healthy controls was less influenced by the presence of endothelial cells and displayed reduced basal platelet reactivity compared with platelets from diabetes patients. We show that platelet aggregation in diabetes as commonly reported in vitro may not fully reflect the in vivo pathophysiological process. Future studies are warranted to investigate other pathological conditions and analyse the effects of antiplatelet agents using this system.
Introduction
The endothelium is essential for the proper functioning of arterial circulation. The common risk factors for atherosclerosis also frequently affect endothelial function, inducing the release of pro-thrombotic, vascular growth factors and vasoactive substances.1,2 The interplay between endothelial cells and circulating blood has long been investigated. In fact, endothelial cells are constantly exposed to multiple physical forces generated by the movement of blood; in response to these physical phenomena, the endothelium releases nearby substances that have a critical role in vascular homeostasis. 3
Among the hemodynamic forces affecting endothelial function, shear stress, that is the frictional force exerted by the flowing blood on the surface of the vessel, has a central role. Several studies have demonstrated that endothelial cells are able to sense shear stress, generating changes in shape, function and gene expression. 4 This, in turn, affects cellular elements close to the endothelium: circulating cells such as leukocytes and platelets, whose adhesiveness and activation may change, and smooth muscle vascular cells, which regulate vessel diameter. 5 In physiological conditions, arterial endothelium is subjected to a wide range of pulsatile shear stress, depending mainly on vessel size and shape, with average values during the cardiac cycle ranging between approximately 10 and 20 dynes/cm2.6–8 In normal conditions, platelets are quiescent and do not adhere to the vessel wall, thanks to the anti-adhesive properties of endothelial cells, probably mainly due to the release of nitric oxide (NO) and prostacyclin (Prostaglandin I2, PGI2) by the endothelium, stimulated by shear stress.9,10
Adherence to the endothelial wall occurs when a vascular injury or the rupture of an atherosclerotic plaque exposes constituents that activate platelets through specific membrane receptors and biochemical pathways. 11 The modulation of platelet aggregation by endothelial cells stimulated by shear stress is not yet fully understood, however. In fact, apart from adhesiveness, the variation in platelet reactivity in physiological flow conditions (i.e. under controlled physiological shear stress) during contact with the endothelium has not been investigated in vitro.
Aim
The aim of this study was to assess the feasibility of and provide initial results for a platelet aggregation study in which platelets were allowed to interact dynamically with endothelial cells in vitro. Platelets were isolated from patients with type 2 diabetes, because this disease is known to cause platelet hyper-reactivity in terms of enhanced aggregation response, 12 thus addressing a relevant clinical context. The interaction between platelets and endothelial cells occurred in a flow chamber. This allowed the shear stress to be modulated so as to influence endothelial production of NO or PGI2. 13 The shear stress applied was identical to that in a large artery in patients with diabetes. 14 In a control arm, platelets from healthy donors were then analysed in the same experimental setting.
Methods
Experimental design
The experimental design included the following steps: (1) growth of human umbilical vein endothelial cells (HUVECs) in culture until confluence; (2) HUVECs on a culture dish were placed in a temperature-controlled flow chamber and subjected to shear stress identical to the mean shear stress in the common carotid arteries of patients with diabetes;
14
the medium used in the recirculation system contained platelets from patients with diabetes or healthy subjects; (3) a simplified Born test was conducted for the assessment of platelet function in four different phases: static conditions, dynamic conditions without HUVEC on the culture dish, dynamic conditions with HUVEC and dynamic conditions with HUVEC incubated with N(G)-monomethyl-
The sequence of these phases was randomly chosen on each day of the experiment in order to avoid any possible influence upon platelet ageing due on account of the increasing time from the point of blood withdrawal. For a series up to four observations in a day of experiments (covering a whole experimental duration up to 4.5 h), the platelets used were from a single patient/subject. Adenosine diphosphate (ADP) or collagen was randomly used as aggregating agents on different days of the experiment. Figure 1 depicts the experimental environment.
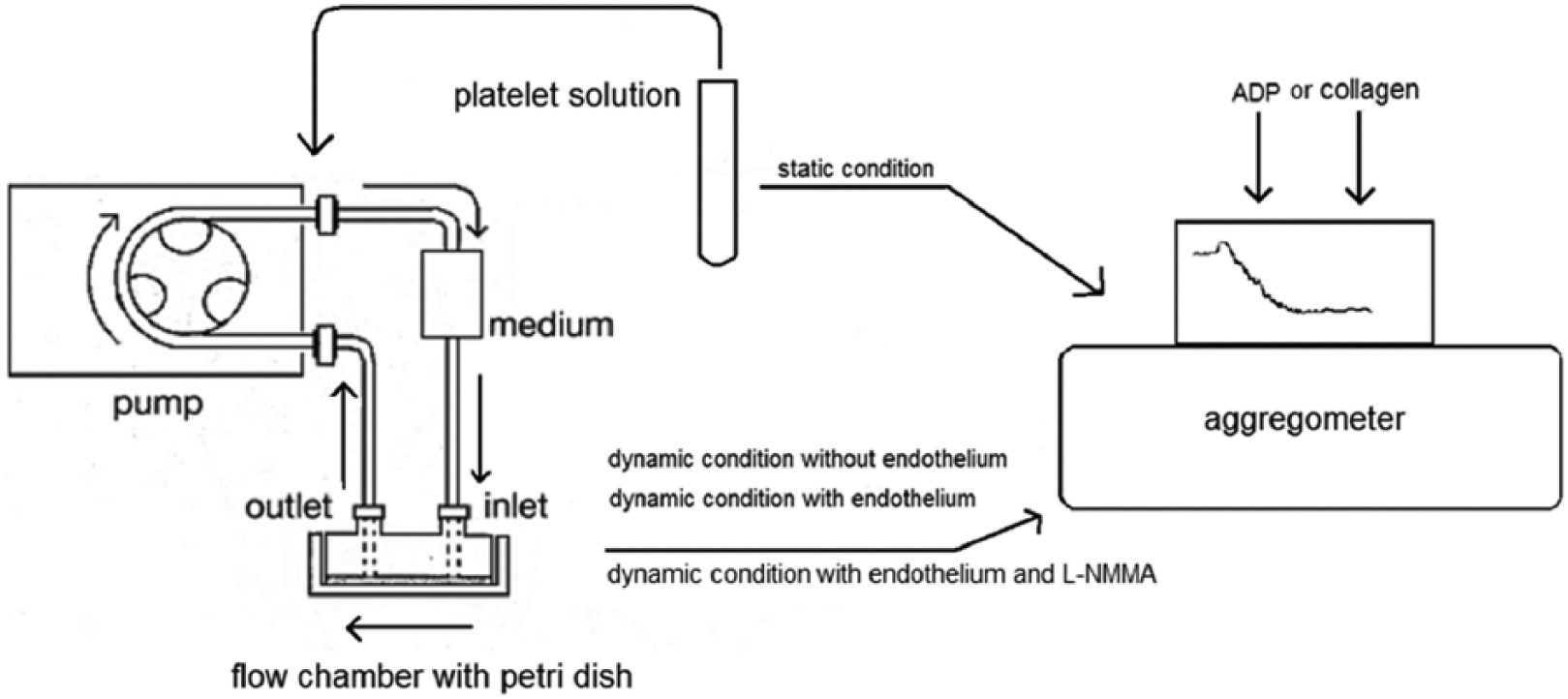
Aggregation stimulated by two inductors of a platelet solution kept in commonly used static conditions is compared to the aggregation after the passage of the platelets into a recirculating environment on a dish without adherent cells, with endothelial cells or with endothelial cells pre-treated with
Study participants
Twenty-nine type 2 outpatients with diabetes (62% male, 54.8 ± 3.5 years) were recruited from March 2011 to January 2012 following the acquisition of written informed consent; the procedures followed were in accord with the ethical standards of the responsible institutional committee. Exclusion criteria were as follows: poor clinical conditions, insulin use, antiplatelet therapy in the last 2 weeks, haemostasis disorders and inflammatory diseases.
Among them, 13 were randomly recalled in 2013 in order to repeat part of the experiments using NO inhibitors. Eleven age- and gender-matched healthy non-smokers volunteers (60% male, 50.3 ± 5.9 years) were then recruited as a control arm between June 2013 and March 2014 from the hospital staff.
Clinical and biochemical parameters
Systolic blood pressure (SBP) and diastolic blood pressure (DBP) were measured with a standardized sphygmomanometer on the right arm after the participant had rested for at least 5 min. Hypertension was defined as SBP/DBP ≥ 140/90 mmHg and/or use of antihypertensive agents. Height and weight were measured by routine methods. Body mass index (BMI) was computed as weight (kg) divided by height squared (m2); patients exceeding a BMI of 29.9 kg/m2 were considered obese. Blood glucose and lipids were measured by routine methods. Diabetes was defined as fasting blood glucose ≥ 126 mg/dL and/or use of antidiabetic agents. Hyperlipidaemia was defined as total cholesterol exceeding 200 mg/dL and/or triglycerides exceeding 150 mg/dL and/or use of lipid lowering drugs.
HUVEC preparation
HUVECs (Lonza, Walkersville, MD, USA) were expanded in a supplemented culture medium (ECM ScienCell, Carlsbad, CA, USA). Cells between passages 3 and 9 were seeded on 60-mm Petri dishes and used for the experiments when at confluence. In some experiments, cells were pre-treated with
Preparation of platelet suspension
A 25-mL sample of peripheral blood, anticoagulated with sodium citrate, was withdrawn without stasis from an antecubital vein. After centrifugation at 150g for 15 min at room temperature, the supernatant as platelet-rich plasma (PRP) was collected. The underlying cellular residue of the sample was again centrifuged at 1000g for 10 min; then, the supernatant was collected to obtain platelet-poor plasma (PPP).
The platelet suspension used was obtained by diluting PRP with PPP until reaching a concentration of 250,000 platelets/µL. 15 The number of platelets and the possible presence of platelet microaggregates were evaluated again after each experiment.
Flow chamber set-up and use
A flow chamber (31-001 GlycoTech, Rockville, MD, USA) was used to impose a laminar shear stress to the endothelial cells. This device is formed by two parts that adhere to each other and to the cell culture dish by the connection of a vacuum pump. The first part is essentially a plastic support with holes predisposed for the entry and exit of the circulating medium and for vacuum pump connection. A silicone gasket is placed below this support, giving the geometrical characteristics of the chamber chosen (length: 2 cm, height: 0.0254 cm, width: 0.5 cm, for a total volume of 25 mm3 and an endothelial surface of 100 mm2). The endothelial cell culture dish was placed under the gasket. The complete flow chamber was kept in a thermostatically controlled environment at 37 °C.
A peristaltic pump (Model 66-Small Peristaltic Harvard Apparatus, Boston, MA, USA) was connected to the flow chamber through a piece of 0.8-mm-diameter tubing (Tygon tubing, Bio-Rad Laboratories, Hercules, CA, USA). The output of the flow chamber was connected to a small collection tank to ensure recirculation. The circulating medium was the platelet suspension prepared as previously reported. The flow rate was 2 mL/min, corresponding to a wall shear stress of 9.9 dynes/cm2 on the endothelium in the flow chamber under laminar flow (Protocol number 7; GlycoTech). This value was chosen because it is nearly identical to the common carotid mean wall shear stress of patients with diabetes in vivo (9.7 dynes/cm2). 14 The wall shear stress value was calculated with the following equation, according to the flow chamber manufacturer specifications
where τ is the wall shear stress (dynes/cm2); µ is the medium viscosity (P); Q is the volumetric flow rate (mL/s); a and b are the flow chamber channel height and width, respectively (cm).
Medium viscosity (i.e. the viscosity of the platelet suspension) was measured twice in three patients at 37 °C with a cone-plate viscometer (Wells-Brookfield DV-III, Stoughton, MA, USA), at a shear rate of 600 s−1, corresponding to the wall shear rate of the flow chamber. Its mean value was 0.016 P and was used in the equation cited above.
Before starting, attention was paid to avoid air bubbles in the flow path. The circulating volume of the medium, which coincides with the full circuit volume, was 2 mL for each experiment. With a flow rate of 2 mL/min, and with the duration of each experiment being 20 min, platelets were directly in contact in dynamic conditions with the endothelium 20 times in each experiment. Furthermore, considering the chamber volume, the transit time of each platelet into the chamber (i.e. the duration of the dynamic contact between platelet and HUVEC) was approximately 1 s for each of the 20 passages. After the first passage, platelets probably came into contact with endothelium-derived flow-stimulated substances.
At the end of this step, the platelet solution was immediately collected and the extent of aggregation evaluated. A reading of platelet numbers before and after each recirculation was made in order to exclude any possible micro-aggregation during the dynamic steps. Careful microscopic observation of possible endothelial cell detachments from the culture dish under flow was made after every experiment, together with control of the tubing and flow chamber in order to detect any platelet clump.
Evaluation of platelet aggregation
Platelet function was evaluated within 5 h of blood withdrawal using a turbidimetric platelet aggregometer (Cronolog, Havertown, PA, USA) with the following aggregating agents (Mascia Brunelli, Milan, Italy): ADP, at concentrations ranging from 0.5 and 10 µM; collagen, at concentrations ranging from 0.5 and 5 µg/mL; concentrations used were upon individual platelet reactivity. After the first experimental series on 29 patients with diabetes showing a similar effect of both aggregating agents throughout the different experimental conditions, either ADP or collagen has been indifferently used afterwards. The aggregations were conducted at 37 °C under stirring at 1200 r/min for 3 min, and the resulting curves were analysed using the Aggrolink program (Cronolog). A dose–response curve was obtained for each inductor in each patient. 15 Then, the maximum amplitude, half maximal effective concentration (EC50), together with the EC50 slope for the dose-response curves were calculated. These parameters are inversely proportional to platelet reactivity. Figure 2 shows a sample of the series of traces needed to obtain platelet aggregation parameters for each patient/experimental condition/aggregating agent. Maximum amplitude is the maximal aggregation obtained in each patient with the maximal concentration of each inductor; EC50 represents the effective concentration of an inductor giving 50% of the maximum amplitude of aggregation; while the EC50 slope is an indicator of the velocity of the 50% platelet aggregating response.
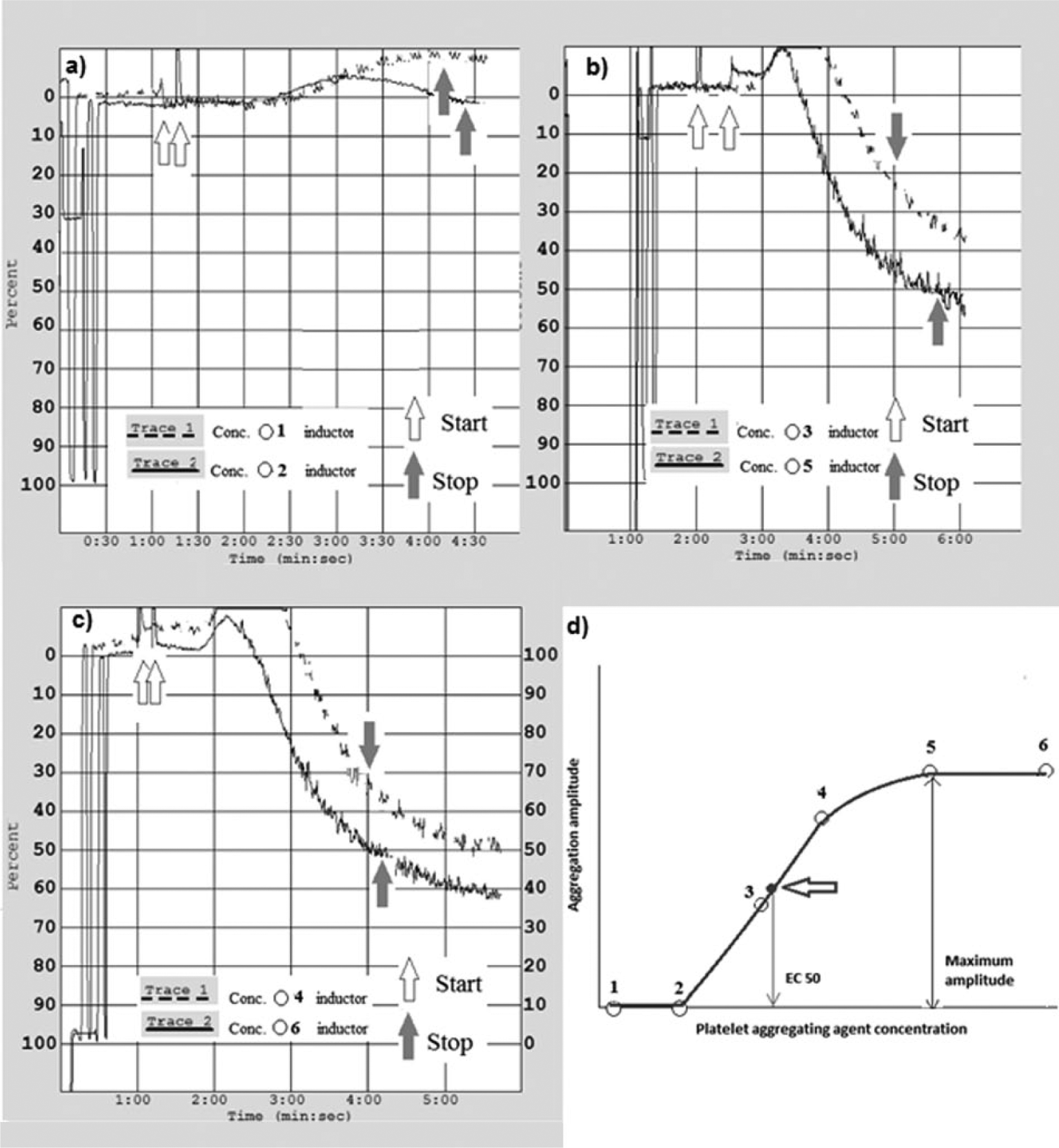
An exemplificative sequence of the complete traces (a–c) needed to calculate (d) platelet aggregation parameters (EC50) and its slope, maximal aggregation) for a single patient tested with a single aggregating agent in a single experimental condition. Start: aggregating agent injection; Stop: aggregating response after 3 min.
Statistical analysis
Maximum amplitude, EC50, and the EC50 slope for platelet aggregation each showed a normal distribution. Coefficients of variation (CVs) were calculated to test the reproducibility of these platelet aggregation measurements, as follows: platelet aggregation measurements with various inductors at various concentrations in all three experimental conditions were repeated twice in 14 patients with diabetes; then, the arithmetic mean between the two examinations was calculated, and the ratio between an individual examination value and the average value was determined as the mean CV, taken as an absolute value. CVs were grouped according to the experimental conditions and not according to the aggregating agents used.
A general linear model (GLM) was used for repeated measures and the Huynh–Feldt test was used to verify the within-subject effect of circulation and of HUVEC presence on several points of analysis of platelet aggregation (i.e. static, dynamic, dynamic with HUVEC, dynamic with HUVEC plus
Results
Reproducibility
Cell confluence, which was verified after each experiment, was lost in only one patient, randomized in the collagen group; this was due to detachment of the endothelial cells, for unknown reasons. The results of this experiment were excluded from the following analyses. No platelet aggregates were found in the circulating medium or adherent to the flow chamber and the circuit after each experiment. In addition, platelet numbers were unchanged after each experiment (mean ± standard deviation of platelet count: 252 ± 8 vs 241 ± 9 × 103 platelets/µL, before vs after perfusion, respectively). The reproducibility data of the platelet aggregation measurements were as follows: CV = 3.9% in static conditions (n = 12 duplicate measurements); CV = 6.1% in dynamic conditions (n = 9 duplicate measurements) and CV = 10.5% in dynamic conditions + HUVEC (n = 19 duplicate measurements).
Platelets from patients with diabetes
The clinical data for all the patients with diabetes in the study (n = 28) are summarized in Table 1. The prevalence of arterial hypertension was 42%, the prevalence of obesity was 35% and hyperlipidaemia was 39%. None of the patients had experienced major cardiovascular diseases such as myocardial infarction or stroke or peripheral arterial disease. All patients were on metformin, plus glinides or sulphonylureas.
Clinical and biochemical characteristics of patients with diabetes (n = 28).
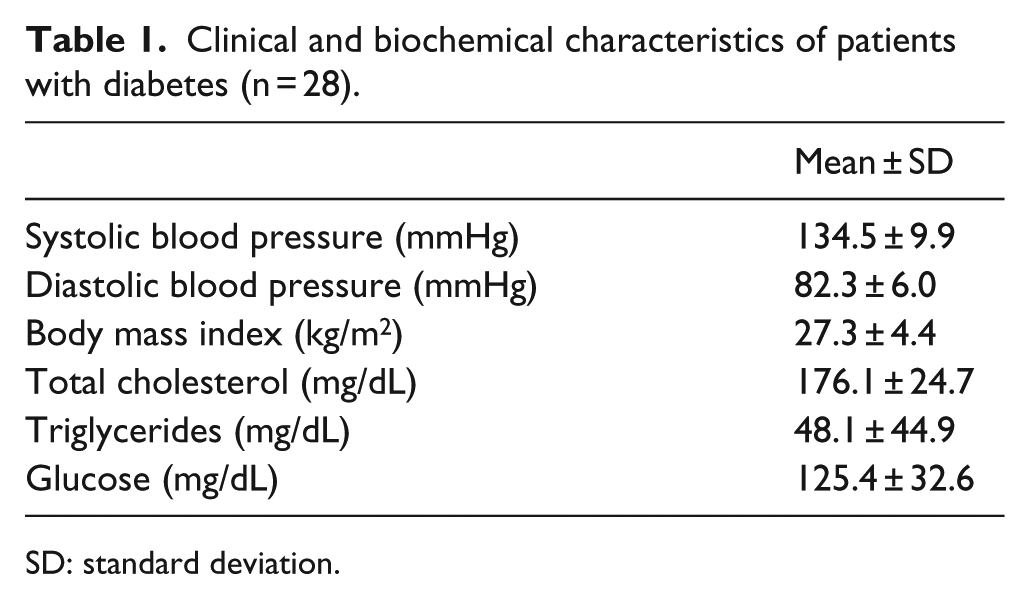
SD: standard deviation.
First, the 28 recruited patients with diabetes were randomly divided into two groups, 14 analysed using ADP and 14 using collagen. Compared to static conditions, the EC50 of ADP-stimulated platelet aggregation (i.e. the ADP concentration needed to obtain 50% of maximal aggregation) increased when platelets were being circulated in the tubing-flow chamber system and increased even more markedly with endothelial cells present (Table 2). The concentration of inductor needed to obtain 50% of the maximal velocity of aggregation to ADP, measured via the EC50 slope, was also increased by contact with HUVEC (Table 2). No differences were noted between the three experimental conditions, however, in terms of the maximal platelet aggregating response to ADP (data not shown). Collagen stimulus, meanwhile, gave similar results with respect to platelet aggregation (Table 3).
ADP-stimulated platelet aggregation (n = 14 patients with diabetes).
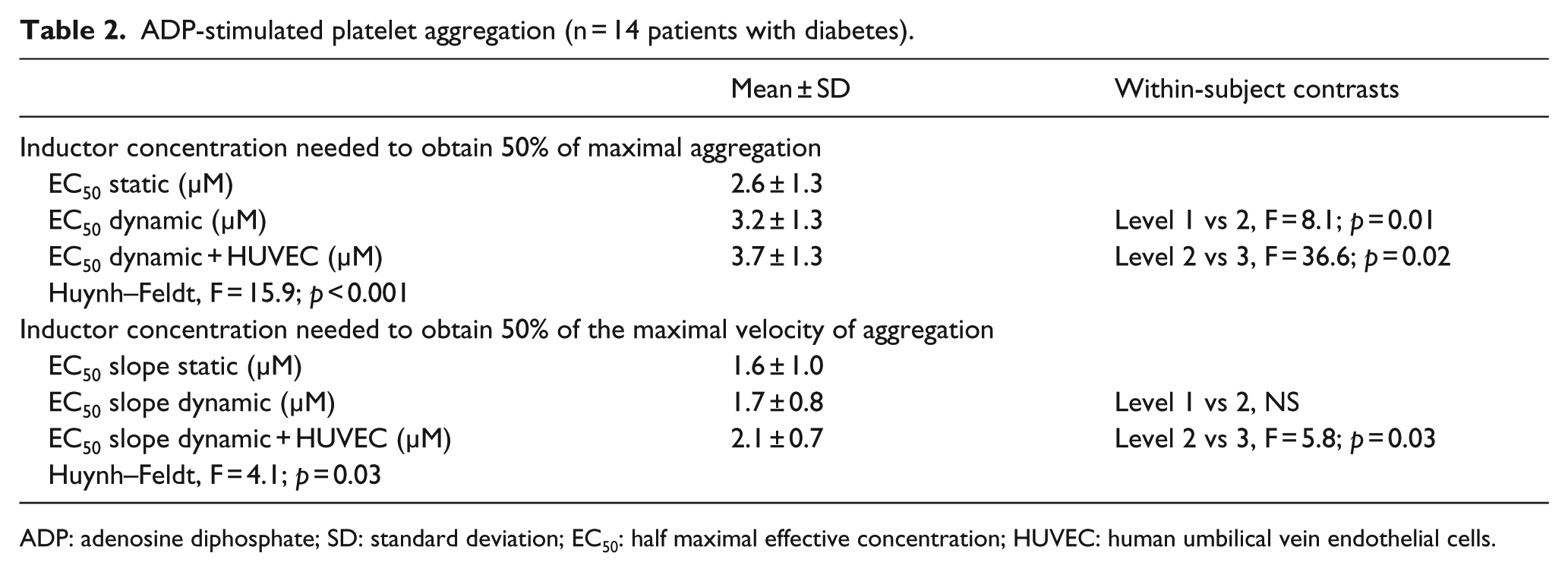
ADP: adenosine diphosphate; SD: standard deviation; EC50: half maximal effective concentration; HUVEC: human umbilical vein endothelial cells.
Collagen-stimulated platelet aggregation (n = 14 patients with diabetes).
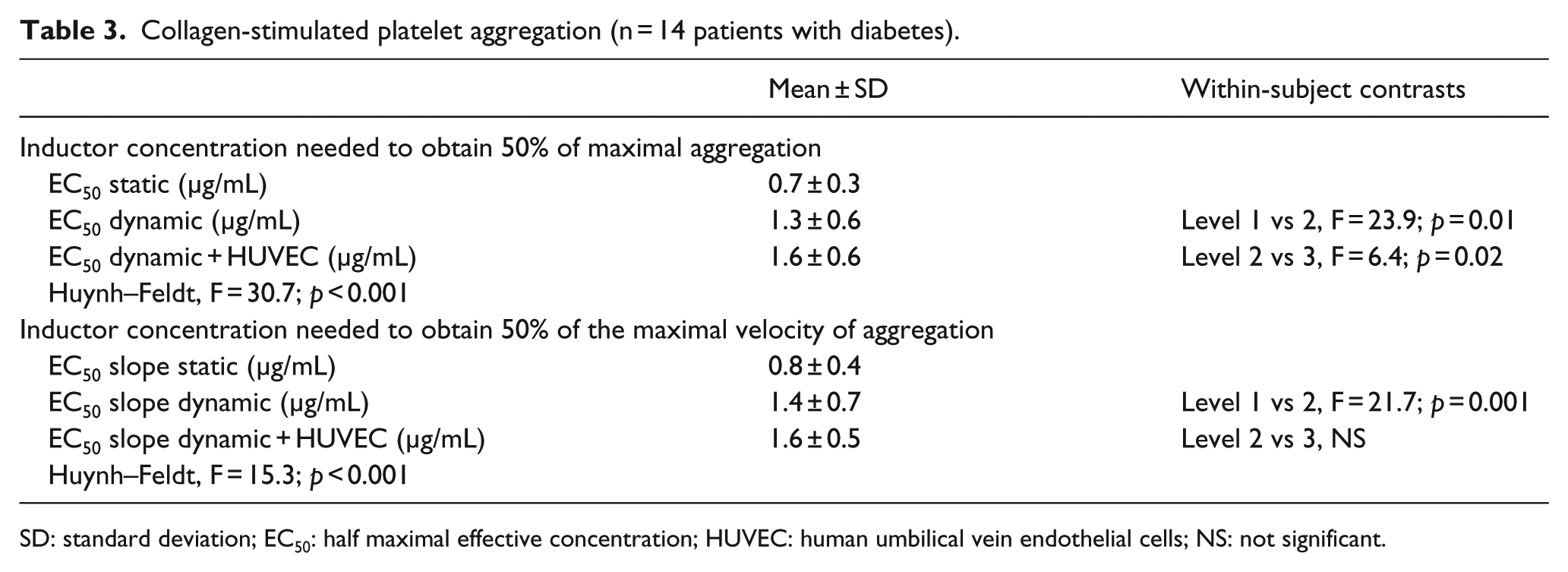
SD: standard deviation; EC50: half maximal effective concentration; HUVEC: human umbilical vein endothelial cells; NS: not significant.
Therefore, platelet reactivity to both aggregating agents, in terms of both quantity of aggregation and aggregating velocity, was blunted by the simulated circulation and even more by the extent of contact between the platelets and endothelial cells, whereas the maximal platelet aggregating capacity was not influenced. In order to verify if shear stress-stimulated endothelium decreased platelet aggregation at least in part by NO, 13 of them were randomly recalled in order to test HUVEC pre-treated with
ADP (µM)- or collagen (µg/mL)-stimulated platelet aggregation (n = 13 patients with diabetes).
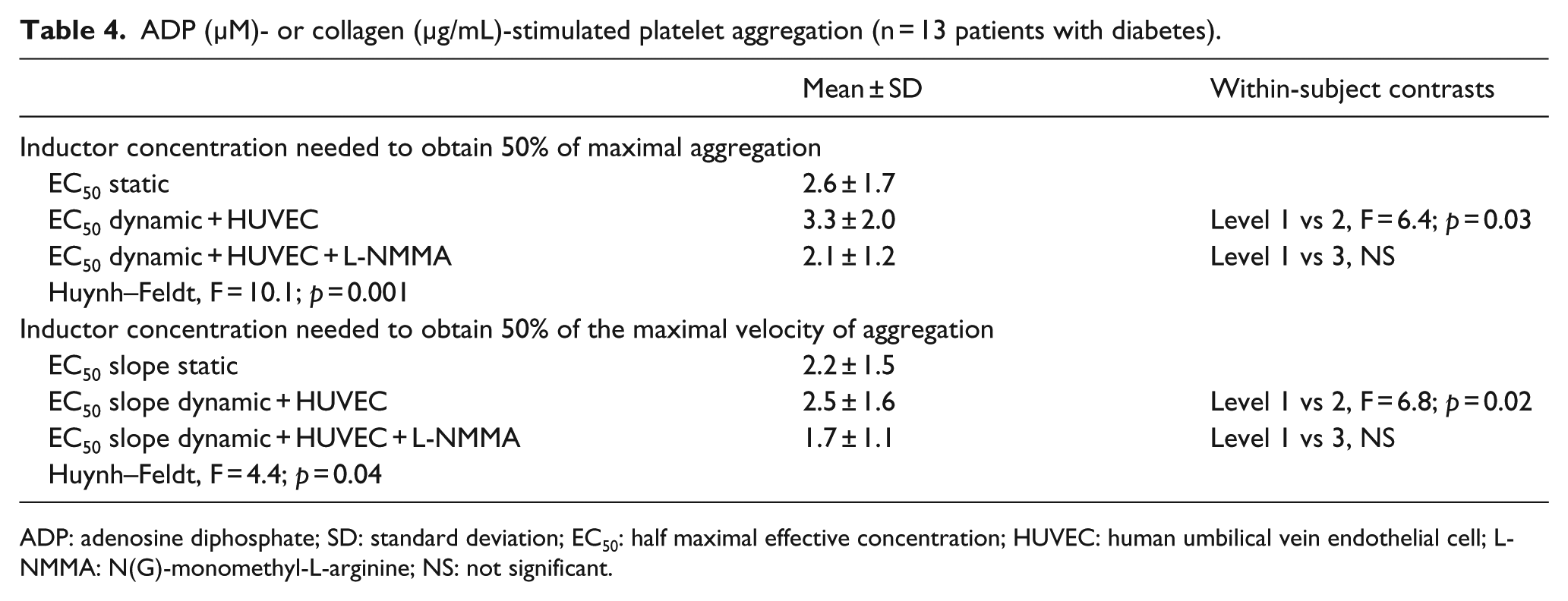
ADP: adenosine diphosphate; SD: standard deviation; EC50: half maximal effective concentration; HUVEC: human umbilical vein endothelial cell; L-NMMA: N(G)-monomethyl-L-arginine; NS: not significant.
Platelets from healthy subjects
In 11 age- and sex-matched healthy volunteers taken as a control groups, platelets were stimulated with ADP (n = 4) or collagen (n = 7) in all the four experimental conditions. Basal platelet reactivity was less in this group (for collagen – the most used aggregating agents in healthy group – EC50 static: 0.7 ± 0.3 µg/mL in patients with diabetes versus 1.6 ± 1.8 µg/mL in healthy subjects, p = 0.05), whereas maximal aggregation was unchanged (data not shown).
In all the controls, ADP- or collagen-stimulated platelet aggregation showed the same trend of patients with diabetes, but this effect was not constant among individuals (see Figure 3 for a comparison among static and dynamic + HUVEC conditions), and therefore, the results were not significant (EC50 static: 1.8 ± 1.8 µg/mL; EC50 dynamic: 2.1 ± 1.8 µg/mL; EC50 dynamic + HUVEC: 2.3 ± 1.8 µg/mL; EC50 dynamic + HUVEC +
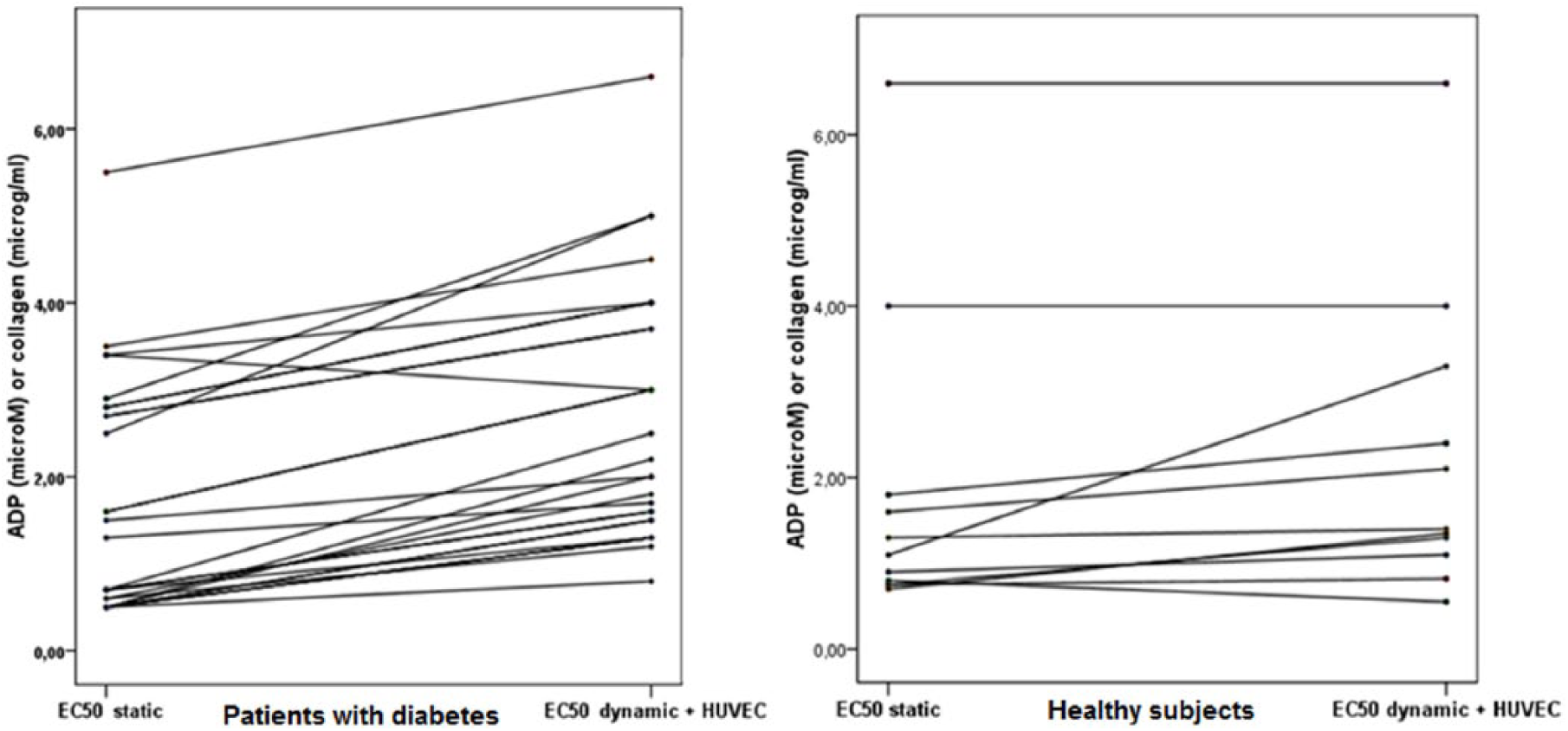
ADP- or collagen-stimulated platelet aggregation in 28 patients with diabetes (left) and in 11 healthy subjects (right).
Discussion
The present data demonstrate that it is feasible to evaluate platelet–endothelium interaction in vitro, in terms of its influence on platelet aggregation and in a model resembling in vivo dynamic conditions. Furthermore, this first application of this new method to a diabetes context shows that platelet reactivity is reduced in dynamic conditions compared with static conditions by endothelial NO synthase (NOS) activity, suggesting that the in vitro platelet aggregation as commonly reported might not fully reflect the in vivo pathophysiological condition, at least in type 2 patients with diabetes. In fact, healthy subjects showed the same trend, but the results were less constant into the group and the statistical significance not reached. This might be due to the absence of platelet hyper-reactivity in healthy subjects versus patients with diabetes, when investigated in static conditions. 12
Flow chambers have been frequently used for the observation of endothelial cells in vitro, 5 and although they have some limitations in terms of physical forces’ displacement in the chamber, 16 they have already been used to study the adhesion of platelets on endothelial cells. The oldest study demonstrates that when experiencing physiological shear stress, platelets do not adhere to confluent endothelium. 17 Afterwards, various stimuli were added to the system in order to induce and evaluate platelet adhesion to the endothelium. Tanahashi et al. 18 verified brain endothelial adhesion of platelets from PRP under microscopy and at low shear stresses, after pretreatment of the endothelium with thrombin or pretreatment of the platelets with thromboxane or ADP. Again, endothelial pretreatment with tissue plasminogen activator modulated platelet adhesion. 19 Activated platelets also modulated monocyte–endothelium interactions. 20
To our knowledge, no experiments have evaluated changes in platelet aggregation when in contact with endothelial cells in dynamic conditions resembling physiological shear stress conditions. In this study, endothelium subjected to shear stress serves to reduce platelet aggregation capacity to both ADP and collagen by an average of 20% in comparison to the absence of HUVECs in dynamic conditions. This effect occurred at a typical level of common carotid shear stress for type 2 diabetes and with platelets from patients affected by the same disease. This reduction occurred for non-maximal aggregation stimuli, whereas the maximum platelet aggregating capacity was preserved in every experimental condition.
This platelet deactivation seems to be due to endothelial production of antiplatelet agents stimulated by shear stress, such as NO, 21 and may be finalized in order to avoid improper platelet activation in vivo after mild stimuli, such as vortex formations or collisions at arterial bifurcations or with cardiac valves. Furthermore, the reduction in platelet reactivity disappears at the highest stimuli, suggesting that platelet function should be ensured for strong stimuli in vivo, such as those indicating acute vascular events.
The exposure of platelets to physical forces resembling arterial circulation, even if without endothelium, also reduced their agonist-induced aggregation; it was verified in patients with diabetes, and as a trend in healthy people. Looking at always changing platelet radial position into the tubing and the chamber, it can be assumed that the mean shear stress imposed on platelets in the present device is always less than 10 dynes/cm2, that is, the wall flow chamber shear stress, apart for some short areas of turbulent flow at the inlet and outlet of the flow chamber. This down-regulation of platelet aggregation will be the focus of future research because this study was not designed to analyse these findings. It is possible, however, that shear stress–enhanced platelet production of NO contributed to the present results.
Platelet NO production is less studied and somewhat controversial. It is known that platelets produce NO, mainly by an endogenous constitutive endothelial-type NOS. 22 Platelet-derived NO, both in resting 23 and stirred (i.e. under shear stress) conditions, reduces aggregation, probably in order to elevate the cytoplasm cyclic guanosine monophosphate (cGMP), both of the originating and nearby platelets. 24 An inducible NOS activity has been found in washed platelets, 25 while platelet RNAs of both NOS subtypes have also been found. 26 Megakaryocytes, and some granulocytes, have constitutive and inducible NOS subtypes. 27 Looking at in vivo studies, platelets from patients with acute coronary syndrome showed reduced NOS activity. 28 Otherwise, a mouse model showed that the impact of platelet NO production on platelet reactivity is limited to systemic circulation. 29
Another puzzling aspect is the possible biphasic effect of platelet derived NO, with a reduction in aggregation only at higher NO concentrations. 30 Similarly, high shear rates, but still within physiologic ranges, serve to activate platelet NOS as an antiaggregating stimulus, 31 whereas it has long been known that supraphysiological shear stress strongly activates platelet aggregation. 32
Limitations
The present first application of this new method was mainly addressed to patients with diabetes. Diabetes is known to induce platelet hyper-reactivity in terms of enhanced aggregation response, 33 with a worsening in patients with poor glucose control. 34 Studies in other disease groups are awaited.
Second, the integration of other blood components, mainly erythrocytes, in the experimental system, might be important. In fact, apart from the interaction between NO and haemoglobin, red blood cells should displace platelets from the centre of the flow chamber approaching them to the endothelial surface, as occurs in vivo. Third, analyses of this approach of shear stress variation, in terms of pulsatile or oscillating forces, might be useful. 35
Finally, even if, compared to other methods, the one described here more closely reproduces what happens physiologically in the bloodstream, there are still technical limitations that prevent the realization of a system which is completely superimposable to the in vivo situation, particularly in terms of the platelet transit times on the endothelium and endothelial/platelet proportions.
Conclusion
In conclusion, this study demonstrates the feasibility of in vitro dynamic evaluation of platelet–endothelium interaction in terms of influence on platelet aggregation. This platelet aggregation study model, in its first application in an environment partially resembling the in vivo conditions of type 2 patients with diabetes, showed a reduced platelet reactivity with respect to usual static conditions of analysis. In healthy people, the effect was less evident. In both groups, platelet desensitization is mediated by endothelial NOS activity. The present model has the potential to be applied to different diseases and more complex experimental conditions.
Footnotes
Acknowledgements
The authors are grateful to Dr Ivan Presta for his advices in cell cultures and Dr Marta Hribal for cell supply.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.