Abstract
Aims:
Osteoprotegerin (OPG) is a glycoprotein from tumour necrosis factor receptor superfamily, responsible for osteoclastogenesis inhibition and associated with arterial calcification and stiffness. We describe the association between metabolic syndrome (MS) and OPG in type 2 diabetes mellitus patients.
Methodology:
We consecutively enrolled 1220 patients from our institution’s Diabetes Centre from August 2011. Anthropometric data such as fasting blood/urine were obtained, and OPG was measured by enzyme-linked immunosorbent assay (ELISA).
Results:
Mean (standard deviation (SD)) of age and diabetes duration was 57.4 (10.9) years and 11.2 (8.9) years, respectively. Prevalence of MS was 64.3% (95% confidence interval (CI): 61.3%–67.2%) and associated with significantly higher OPG (5.44 vs 4.47 pmol/L) and microvascular complications. The presence of microvascular complications was associated with higher OPG: nephropathy (5.54 (2.20) vs 4.65 (1.70) pmol/L,
Conclusion:
Higher OPG levels were associated with risk of MS and microvascular complications. Studies are needed to test whether OPG could be a useful biomarker identifying patients at risk of vascular complications and whether further exploration of this pathway may lead novel therapeutic options.
Introduction
Osteoprotegerin (OPG) is a soluble glycoprotein from the tissue necrosis factor (TNF) receptor superfamily. It is a decoy receptor binding to receptor activator of nuclear factor κB ligand (RANKL), preventing osteoclastogenesis and thus inhibiting bone resorption. 1 OPG is also expressed in vascular tissues including endothelial cells and vascular smooth muscle cells, 1 with studies in OPG knockout mice showing early onset of osteoporosis and severe vascular medial calcification. 2 While this may suggest a possible protective effect of OPG on vascular endothelium, clinical studies suggest OPG as a risk factor of progressive atherosclerotic cardiovascular disease, being increased in coronary artery disease, arterial calcification and cardiovascular risk factors, including diabetes mellitus (DM). 3
Metabolic syndrome (MS) comprises a constellation of vascular risks including obesity, dyslipidemia, hyperglycaemia, hypertension, insulin resistance and a pro-inflammatory state. As OPG may modify inflammatory response, we postulate that OPG may play a role in MS and its associated risks. While in postmenopausal women in the general population, OPG was shown not to have a significant association with MS, 4 data looking at this association in patients with type 2 DM (T2DM) are scarce. This study aims to address this gap, exploring the associations of OPG with the presence of MS and long-term complications in T2DM.
Methods
We consecutively enrolled adults, 21–90 years old, with T2DM seen in our institution from August 2011 as part of the ongoing SMART2D program. 5 SMART2D is a cross-sectional study aimed at understanding risk factors of long-term complications of diabetes. All patients were observed after a 10-h overnight fast, with venous blood and urine collected. Anthropometric measurements and screening for complications (including retinal assessment by a trained diabetologist or ophthalmologist) were performed.
Biochemical analyses were performed in our institution’s Referral Laboratory. Serum total cholesterol (TC), triglyceride (TG), high-density lipoprotein cholesterol (HDL-C) and low-density lipoprotein cholesterol (LDL-C) levels were measured using an automatized autoanalyzer (Roche COBAS INTEGRA 700). Plasma glucose was obtained by enzymatic methods. Urinary spot albumin and creatinine were measured using commercial assays (Immulite, Surrey, UK and Roche Diagnostics, Mannheim, Germany). Plasma OPG was measured using a highly sensitive quantitative sandwich enzyme-linked immunosorbent assay (ELISA) (BioVendor, Modrice, Czech Republic) (intra- and inter-assay coefficients of variation were 2.36% and 5.97%, respectively).
Institution’s Ethical Review Committee approved the study, with all participants providing written informed consent. To analyse the association of OPG with MS, from the 1220 patients enrolled as of August 2012, 600 were randomly selected using SPSS version 19.0.
Data collection
Blood pressure was taken from participants in a sitting position, after 5 min resting using an automated blood pressure monitor (Dinamap Pro 100V2, Freiburg, Germany). Ankle pressures were measured using a standardized Doppler ultrasonic device (8 MHz; Smartdop™ 20EX, Kawasaki, Japan), after 5-min rest in the supine position. Ankle brachial index (ABI) was calculated as described previously. 6 Neuropathy assessment was made by a neurothesiometer (Horwell Scientific, Yorkshire, UK) for vibration and a 5.07 (10-g) monofilament on non-calloused plantar sites for light touch.
Definitions
MS was defined according to the National Cholesterol Education Program (NCEP) Adult Treatment Panel (ATP) III criteria, with cut-off for waist circumference modified for Asians. 7 MS was present when at least three of the following criteria were satisfied: (1) waist circumference > 90 cm (men) and > 80 cm (women), (2) TG level ≥ 1.7 mmol/L, (3) HDL-cholesterol <1.0 mmol/L (men) and <1.3 mmol/L (women), (4) blood pressure ≥130/85 mmHg or known treatment for hypertension and (5) fasting glucose level of ≥ 6.1 mmol/L or known treatment for T2DM.
Estimated glomerular filtration rate (eGFR) was calculated using the Modification of Diet in Renal Disease (MDRD) equation. Nephropathy was present if urine albumin to creatinine ratio (ACR) > 30 µg/mg or eGFR < 60 mL/min/1.73 m2. Neuropathy was present if an abnormal finding in monofilament (failure to detect at ≥ 3 of 10 points) or neurothesiometer testing (≥ 25 V) was detected. Peripheral artery disease (PAD) was defined to be present if ABI ≤ 0.9. 6
Statistical analysis
All analyses were performed using SPSS version 21 (SPSS Inc, Chicago, IL, USA). Results for continuous variables were presented in mean (standard deviation (SD)), for binary variables in percent, and analysed using Student’s T-test and Chi-square respectively, while non-parametric tests were used when not normally distributed. OPG levels were divided into tertiles, and logistic regression models were used to estimate the odds ratio (OR) for each risk factor of MS, adjusting for age and gender. All statistical tests were two-sided with a level of significance being <0.05.
Results
The characteristics of the entire cohort with T2DM (
MS was present in 64.3% (95% CI: 61.3%–67.2%) (Supplementary Table 2). Obesity and elevated blood pressure were the next two most common components of MS after abnormal glucose in our patients with T2DM (68.3% with obesity, 58.2% with abnormal blood pressure, 37.7% with low HDL-cholesterol and 36% with raised TGs). Compared to those without MS, those with MS were more likely to be female, less likely to be Chinese and had higher risk of nephropathy (OR = 2.806 (95% CI: 2.122–3.710,
Patients with MS had higher OPG (5.44 (2.21) vs 4.47 (1.92) pmol/L,
Multiple logistic regression for risk factors for metabolic syndrome.
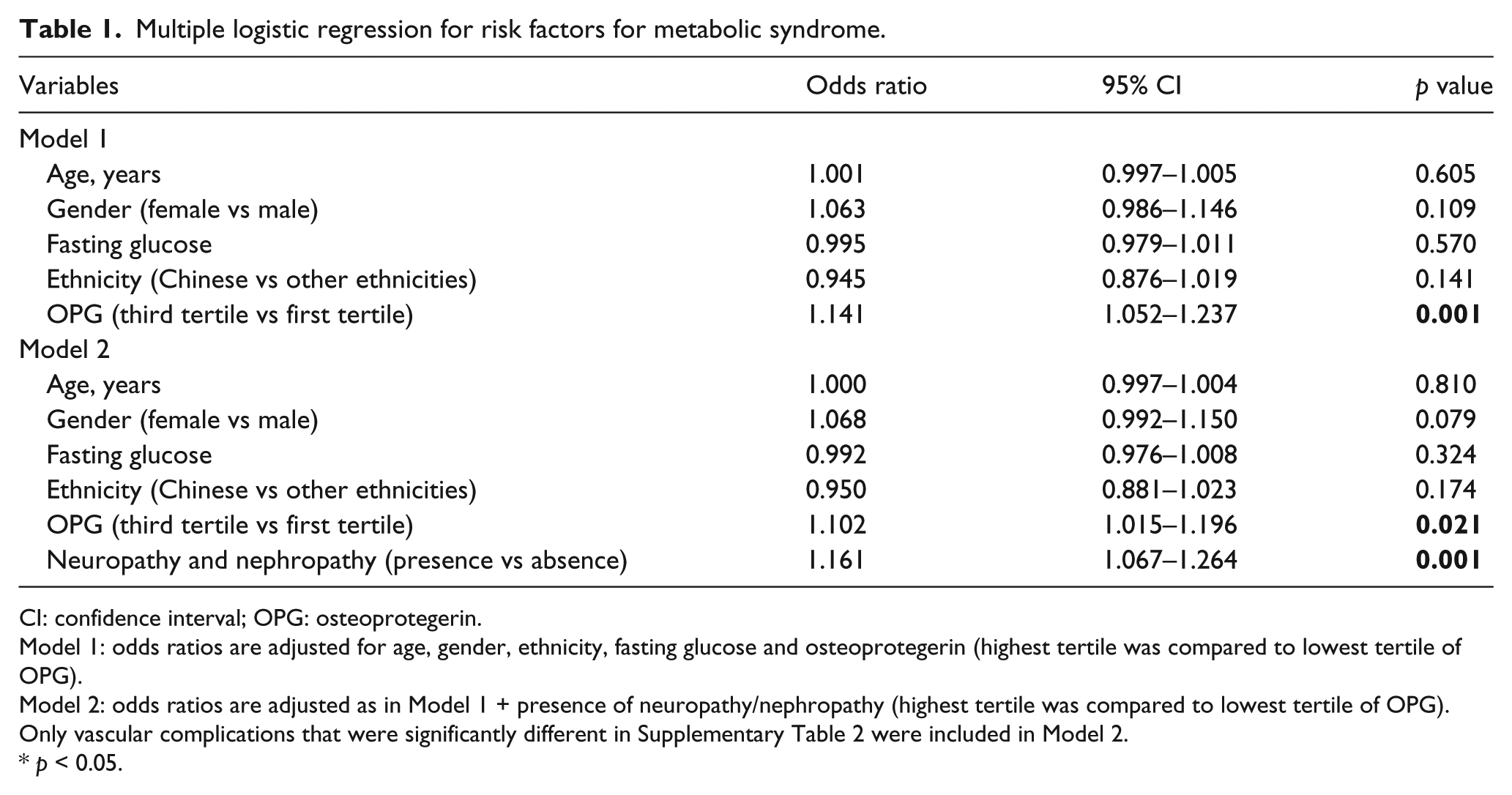
CI: confidence interval; OPG: osteoprotegerin.
Model 1: odds ratios are adjusted for age, gender, ethnicity, fasting glucose and osteoprotegerin (highest tertile was compared to lowest tertile of OPG).
Model 2: odds ratios are adjusted as in Model 1 + presence of neuropathy/nephropathy (highest tertile was compared to lowest tertile of OPG). Only vascular complications that were significantly different in Supplementary Table 2 were included in Model 2.
Nephropathy (5.54 (2.20) vs 4.65 (1.70) pmol/L,
Discussion
In patients with T2DM, we found OPG to be a significant predictor of MS. Independent of the presence of MS, elevated OPG was also associated with increased risk of microvascular complications of nephropathy and neuropathy.
Previous studies looking at the association between OPG and MS revealed contrasting findings. While no correlation between OPG and MS was seen in a population-based study in postmenopausal women, 4 higher OPG levels were associated with MS in younger women with previous gestational diabetes. 8
OPG expression is inhibited by insulin administration. 9 As insulin resistance is strongly associated with the development of MS, it is likely insulin resistance may contribute to elevated OPG.
While others have reported OPG to be higher in patients with albuminuria, 10 we found OPG also to be significantly associated with nephropathy and neuropathy. The mechanism for these associations remains unclear, but this remains after adjustment for eGFR, suggesting that factors other than reduced renal clearance of OPG in nephropathy may play a contributory role. OPG produced in endothelium and vascular smooth muscle cells exerts anti-apoptotic effects by binding to the TNF-related apoptosis inducing ligand (TRAIL). 1 As OPG interferes with TRAIL-induced NFκB, activation and TRAIL-induced apoptosis, OPG may act as a response mechanism to counter increase the increased expression of TRAIL in proximal tubular cells. It is thus possible that increased OPG expression is a response to injury, with a resultant unintended cascade of events leading to worsening of microvascular complications, and this will need to be explored in future studies.
The discovery of modifiable risk factors for microvascular disease is essential, with components of MS representing one established option. Our data suggest that OPG may be an independent predictor of MS and microvascular complications. In murine models, a humanized monoclonal antibody (denosumab), with a high affinity and specificity for RANKL, reduced prednisolone-induced vascular calcification in the aorta. 11 Vessel wall–derived OPG was able to inhibit calcium deposition in ApoE−/− OPG−/− knockout animal models, indicating the ability of endogenous OPG to inhibit vascular calcification, further suggesting that systemic elevation of OPG may be an adaptive response factor. 12 Taken together, it remains unclear whether targeting RANK-RANKL may have a role in modifying vascular disease.
The strength of this study includes the ability to obtain detailed assessment for each patients, with all assessments, performed by trained and accredited staff, ensuring data quality. Limitations include the cross-sectional nature of our study, which precludes conclusions regarding the temporal nature of the observations.
In conclusion, in this study on patients with T2DM, higher OPG levels were associated with risk of MS and microvascular complications. With OPG being expressed in osteoblasts, endothelial cells and vascular smooth muscle cells, the crosstalk of these cells and their role in T2DM, MS and vascular complications need further exploration, to enhance understanding of the pathogenesis of diabetes and its complications. Further research is needed to study whether OPG may be a useful biomarker to help identify patients at high risk of vascular complications and whether further exploration of this pathway may lead to discovery of new therapeutic targets.
Footnotes
Acknowledgements
The authors would like to acknowledge the Research Nurses from the Clinical Research Unit from our institution who helped with data collection and data entry and thank all the patients who volunteered for this study. Singapore Clinical Research Institute contributed to the study protocol, database design and implementation as well as the study conduct. Study data were collected and managed using REDCap electronic data capture tools hosted at Singapore Clinical Research Institute. REDCap (Research Electronic Data Capture) is a secure, web-based application designed to support data capture for research studies, providing (1) an intuitive interface for validated data entry, (2) audit trails for tracking data manipulation and export procedures, (3) automated export procedures for seamless data downloads to common statistical packages and (4) procedures for importing data from external sources. The guarantor, S.T., accepts full responsibility for the work and/or the conduct of the study, had access to the data and controlled the decision to publish. All authors contributed to the intellectual development of this article. S.T. conceived and coordinated the whole work and prepared the manuscript; P.S.L.T., L.S.C., S.C.F., L.J.J., N.X.W. and Y.L.Y. participated in the planning of the study and key discussions and edited the manuscript. All authors are part of the SMART2D study (Singapore study of MAcro-angiopathy and microvascular Reactivity in Type 2 Diabetes).
Declaration of conflicting interests
All authors declare that there are no conflicts of interest related to this manuscript.
Ethical approval
Ethics approval was obtained from National Healthcare Group Institutional Review Board prior to study commencement. Informed consent was obtained from all participants before conduct of study.
Funding
This study was supported by a grant from the National Medical Research Council of Singapore and NMRC/PPG/AH(KTPH)/2011.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
