Abstract
Purpose:
To investigate whether mesenchymal stem cells (MSCs) could inhibit transforming growth factor beta (TGF-β) signalling pathway by paracrine action.
Methods:
Bone marrow–derived MSCs were transplanted to streptozotocin-induced diabetic rats via tail vein. MSC-conditioned media were used with a model of mesangial cell fibrosis induced by high glucose in vitro.
Results:
At 8 weeks after MSC treatment, the renal function and the glomerulosclerosis as revealed by periodic acid Schiff stain was dramatically attenuated. The expression of collagen I, collagen IV and α-smooth muscle actin (SMA) in diabetic kidney was decreased, and E-cadherin increased after MSC treatment. The TGF-β signalling pathway was suppressed both in vivo and in vitro. MSCs secreted a significant amount of bone morphogenetic protein 7 (BMP7), in vitro, MSC-conditioned media inhibited TGF-β signalling stimulated by high glucose, and BMP7 neutralizing antibody blocked the inhibitory effect of MSC-conditioned media.
Conclusion:
MSCs ameliorated glomerular fibrosis in vivo and in vitro by inhibiting TGF-β/Smad signalling pathway via secretion of BMP7.
Keywords
Introduction
Diabetic nephropathy (DN) is the leading cause of end-stage renal disease worldwide, 1 with a significant increase in morbidity and mortality in patients with diabetes. 2 Considering that currently available treatments involving strict glycaemic and/or blood pressure control can delay but not eliminate the occurrence and development of DN, it is necessary to identify new strategies that could specifically target on DN.
Structural abnormalities of DN include hypertrophy of the kidney, an increase in the thickness of glomerular basement membrane, accumulation of extracellular matrix (ECM) components in the glomerulus (nodular and diffuse glomerulosclerosis (GS)), tubular atrophy and interstitial fibrosis, typically characterized by ECM accumulation. The transforming growth factor beta (TGF-β) is a multifunctional cytokine that regulates the proliferation and metabolism of ECM proteins 3 and considered as one of the main cytokines that aggravates DN.4,5 TGF-β binds to its receptor, TGF-β receptor IIβ, which activates the TGF-β receptor I (TβRI) kinase. Activated TβRI then phosphorylates receptor-regulated Smads (R-Smads), Smad2 and Smad3. Phosphorylated Smad2 and Smad3 form an oligomeric complex with Smad4. The complex translocates into the nucleus and regulates the target gene transcription. 6 Besides, TGF-β is also considered to induce epithelial-to-mesenchymal transition (EMT) of the injured tubule epithelial cells. 7 Up-regulation of TGF-β and its downstream Smad cascade is prevalent in many types of kidney diseases. More important, several experimental studies have shown that different therapeutic strategies which can inhibit TGF-β signalling pathway can prevent or even reverse the early features of DN.8–10 Thus, these results provide clear evidence that TGF-β activity contributes importantly to the development of diabetic GS.
Stem cell therapy becomes an attractive therapeutic strategy in acute and chronic kidney disease in animals in recent years.11–14 Because infusion of autologous mesenchymal stem cells (MSCs) is considered to be safe and has few adverse effects, it also seems to be promising for clinical use. In preliminary experiments, 15 we found that MSCs injected via the tail vein into rats with DN reduced blood glucose, microalbuminuria and ameliorated glomerular injury; however, the gross presence of stem cells was not found in the kidney, which was also demonstrated by other studies.16–18 So, we speculated that MSCs protected the kidney via paracrine action. It has been shown that MSCs can promote tissue repair through the paracrine action of various cytokines, to reduce adverse reactions of the kidney, so as to improve and promote the endogenous repair of kidney tissue in different experimental models, including DN.19–22 But it is unknown whether MSCs secrete cytokines which can inhibit TGF-β signalling pathway – the key mediator in the pathogenesis of DN in diabetic kidney.
Based on these findings, we tested our strategy by transplanting bone marrow–derived MSCs to streptozotocin (STZ)-induced diabetic rats and using MSC-conditioned medium (MSC-CM) with a model of mesangial cells fibrosis induced by high glucose to investigate whether MSCs could inhibit TGF-β signalling pathway by paracrine action and to provide novel insights into the treatment of DN.
Materials and methods
Diabetes models establishment
Female Wistar rats weighing 200–250 g were purchased from Animal Experimental Center of Shandong University, China. They were given free access to food and water throughout the study. After 1 week of adaptation, a single injection of STZ (60 mg/kg, Sigma Aldrich, St. Louis, USA) was given via intraperitoneal injection to induce diabetes following one-night fasting. The blood glucose level was measured after 72 h. The status of diabetes was confirmed as random glucose level higher than 16.7 mmol/L at three continuous days. The blood glucose levels were detected throughout the experiments with a glucometer ACCU-CHEK Advantage Meter (Andon Medical Electronics Co., Tianjin, China). All procedures were in accordance with the guidelines of the Animal Ethics Committee of the Shandong University, which are consistent with the National Institutes of Health Guide for the Care and Use of Laboratory Animals.
MSC preparation and transplantation
Bone marrow was collected from rats by flushing the femora and tibiae, and collected as previously described. 15 Cells were analysed by promoting their differentiation into osteocytes and adipocytes with specific differentiation media and flow cytometry analysis to confirm stem cell characteristics as previously described. 15 These cells were positive for CD29, CD90 and CD44, but negative for CD34, CD45 and CD11b, which was consistent with bone marrow–derived MSC characteristics. Third-passage cells were used for next experiments.
At 8 weeks after successful establishment of the diabetes model, the female Wistar rats were randomly assigned to three groups as follows.
Normal control group (normal, n = 12): animals without STZ injection received an injection of 0.9% saline instead of MSCs.
DN group (n = 12): animals with STZ injection received an injection of 0.9% saline instead of MSCs.
DN-MSC group (n = 12): animals with STZ injection received a tail vein injection of MSCs.
MSCs were transplanted via tail vein at a concentration of 2 × 106 in 0.5 mL serum-free media once a week for two continuous weeks as previously described. 23
Sample collection and renal function measurement
At 8 weeks after treatment, animals were housed in metabolic cages to collect 24-h urine. After measurement of body weight, rats in each group were given euthanasia by intraperitoneal injection of pentobarbital (50 mg/kg), blood samples were drawn from left ventricle, and kidneys were quickly removed, decapsulated, weighed and dissected into two parts, one of which was immediately frozen in liquid nitrogen and stored at −80°C for molecular biological studies and the other was dropped in 4% paraformaldehyde for histopathological analysis.
Blood and urine samples were centrifuged at 2000 g for 10 min to harvest the supernatant for the biochemical parameters measurement including serum glucose level, serum creatinine level, 24 h urine albumin excretion and urine creatinine level. The renal function was assessed by creatinine clearance (Ccr) calculated as urinary creatinine × urine volume/serum creatinine and expressed as millilitres per minute as previously described. 18 The renal mass index as revealed by the ratio of kidney weight to body weight was also calculated.
Renal histopathological measurement
To assess the glomerular sclerotic injury, renal tissues were embedded in paraffin, and 4-µm-thick sections were prepared for periodic acid Schiff (PAS) staining. GS was defined as the presence of dense, abundant deposition of PAS-positive material at the glomerular tufts, with occlusion of capillary loops and segmental hyalinization. GS was evaluated by determining the percentage of glomeruli exhibiting sclerotic lesions (%GS) as described previously; 13 200 consecutive glomeruli were examined for each rat.
Glomerular mesangial cell culture
HBZY-1 cells, a rat glomerular mesangial cell (GMC) line, purchased from the China Center for Type Culture Collection, were cultured in Dulbecco’s modified Eagle’s medium (DMEM) supplemented with 10% foetal bovine serum (FBS) and 100 U/mL penicillin and streptomycin. For collecting MSC-conditioned media (MSC-CM) for next analysis, the media from MSCs in 100 mm dishes were changed for 10 mL fresh complete media, then collected after 48 h of culture, at which time cell number was about 2 × 106, pooled and filtered through a 0.22 µm bottle-top filter system (Millipore Corp., Billerica, USA). To investigate the effect of MSCs on GMCs stimulated by high glucose through paracrine effects, cells were divided into three groups as follows: GMCs; co-culture of GMCs and MSCs with the ratio of 10:1 24 and GMCs cultured in MSC-CM. The cells in the three groups were stimulated with 30 mmol/L glucose for 72 h. The dose of glucose and the stimulating time were determined on the basis of our preliminary studies. GMCs cultured in normal media were taken as control. After 72 h, cells were collected to extract total protein to detect TGF-β, Smad2/3, phosphorylated Smad2 and phosphorylated Smad3 by Western blot method as described below. To assess the effect of bone morphogenetic protein (BMP7) on high glucose-induced TGF β signalling in GMCs, MSC-CM and fresh media were preincubated with or without bone morphogenetic protein 7 neutralizing antibody (BMP7-Ab) (R&D Systems, Lille, France, 10 µg/mL) for 1 h before its addition to GMCs cultures. Then, the high glucose stimulation was performed as described above. After 72 h, cells were collected to extract total protein to detect TGF-β, Smad2/3, phosphorylated Smad2 and phosphorylated Smad3 by Western blot method as described below.
Gene expression
Total RNA from kidney homogenates was extracted by the TRIzol method (Invitrogen, Carlsbad, USA). An amount of 1 µg total RNA was used for reverse transcription (RT) in a final volume of 20 µL using PrimeScript RT reagent Kit according to the manufacturer’s protocol (TaKaRa Biotechnology, Dalian, China). An amount of 2 µL complementary DNA (cDNA) was used for real-time polymerase chain reaction (PCR) with the SYBR Premix Ex Taq (TaKaRa Biotechnology) for TGF-β, collagen I, collagen IV, α-smooth muscle actin (SMA), E-cadherin and BMP7. The primers were as follows: TGF-β forward was 5′-CAT TGC TGT CCC GTG CAG A-3′ and the reverse 5′-AGG TAA CGC CAG GAA TTG TTG CTA-3′; collagen I forward was 5′-TCT AAG ACA TCC CTG GTC AC-3′ and the reverse 5′-GTC CTT CCA GAA GAA ACC TT-3′; collagen IV forward was 5′-TAG GTG TCA GCA ATT AGG-3′ and the reverse 5′-TCA CTT CAA GCA TAG TGG TCC G-3′; α-SMA forward was 5′-GAT CAC CAT CGG GAA TGA AGG C-3′ and the reverse 5′-CTT AGA AGC ATT TGC GGT GGA-3′; E-cadherin forward was 5′-CTG ATT CTG ATC CTC CTG CTC CT-3′ and the reverse 5′-ATC ATT TCG AAT CAC TTC CGG T-3′; and BMP7 forward was 5′-ACT GGA TCA TGG CAC CTG AA-3′ and the reverse 5′-CTG GAC GAT AGC ATG GTT GG-3′. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as an internal control with the forward and reverse primers being 5′-ACA AGA TGG TGA AGG TCG GTG-3′and 5′-AGA AGG CAG CCC TGG TAA CC-3′. All PCR reactions were performed in triplicate. The fold change in the expression of the target gene was calculated by the ΔΔ threshold cycle (CT) method and normalized to controls. All primers were designed and generated by TaKaRa Biotechnology.
Protein expression
BMP7 concentration in the MSC-CM was measured using enzyme-linked immunosorbent assay (ELISA) kit (Bio-Swamp Life Science, Shanghai, China) according to the manufacturer’s instruction. Meanwhile, the standard media were performed as control. The protein expression levels of BMP7 in renal cortical homogenates were also quantified by ELISA. All measurements were tested in triplicate.
The total protein of renal tissues and mesangial cells was extracted by total protein extraction kit (DBI Bioscience, Ludwigshafen, Germany) according to the manufacturer’s instruction, and the protein concentration was quantified using the Bradford method (Biyotime, Shanghai, China). Each protein lysates (50 µg) were run on 10% sodium dodecyl sulphate (SDS)-polyacrylamide gels and transferred to nitrocellulose membranes. The membranes were blocked with 5% skim milk in tris-buffered saline (pH 7.4) containing 0.1% Tween 20 (TBST) for 1 h at room temperature and then incubated for 16 h at 4°C with primary antibodies. After being washed extensively, the membranes were incubated with horseradish peroxidase–conjugated secondary antibody for 2 h at room temperature in 5% skim milk/TBST. Proteins were detected by enhanced chemiluminescence (Millipore Corp., Billerica, USA). Protein levels were normalized to β-actin levels. Primary antibodies used in this study were as follows: rabbit polyclonal antibody to TGF-β (1:500; Bioworld, St. Louis, USA); rabbit polyclonal antibody to collagen I (1:5000; Abcam, Cambridge, USA); rabbit polyclonal antibody to collagen IV (1:1000; Abcam, Cambridge, USA); rabbit polyclonal antibody to α-SMA (1:500; Abcam, Cambridge, USA); rabbit monoclonal antibody to E-cadherin (1:1000; Abcam, Cambridge, USA); rabbit polyclonal antibody to phosphorylated Smad2 (1:1000; Abcam, Cambridge, USA); rabbit polyclonal antibody to phosphorylated Smad3 (1:500; Abcam, Cambridge, USA) and rabbit polyclonal antibody to Smad2/3 (1:200; Abcam, Cambridge, USA).
Statistical analysis
All results were expressed as means ± standard deviation (SD). Statistical analysis was performed by SPSS software (version 11.0; SPSS Inc., Chicago, IL, USA). One-way analysis of variance was applied to analyse the difference among different animal groups. Statistical significance was set at p < 0.05.
Results
MSCs ameliorated renal function and glomerular fibrosis in diabetic rats
Diabetic rats showed increased blood glucose, 24 h urine albumin excretion, Ccr and renal mass index at 16 weeks after STZ injection compared to normal control rats. The transplantation of MSCs after the induction of hyperglycaemia ameliorated these indexes significantly (Figure 1).
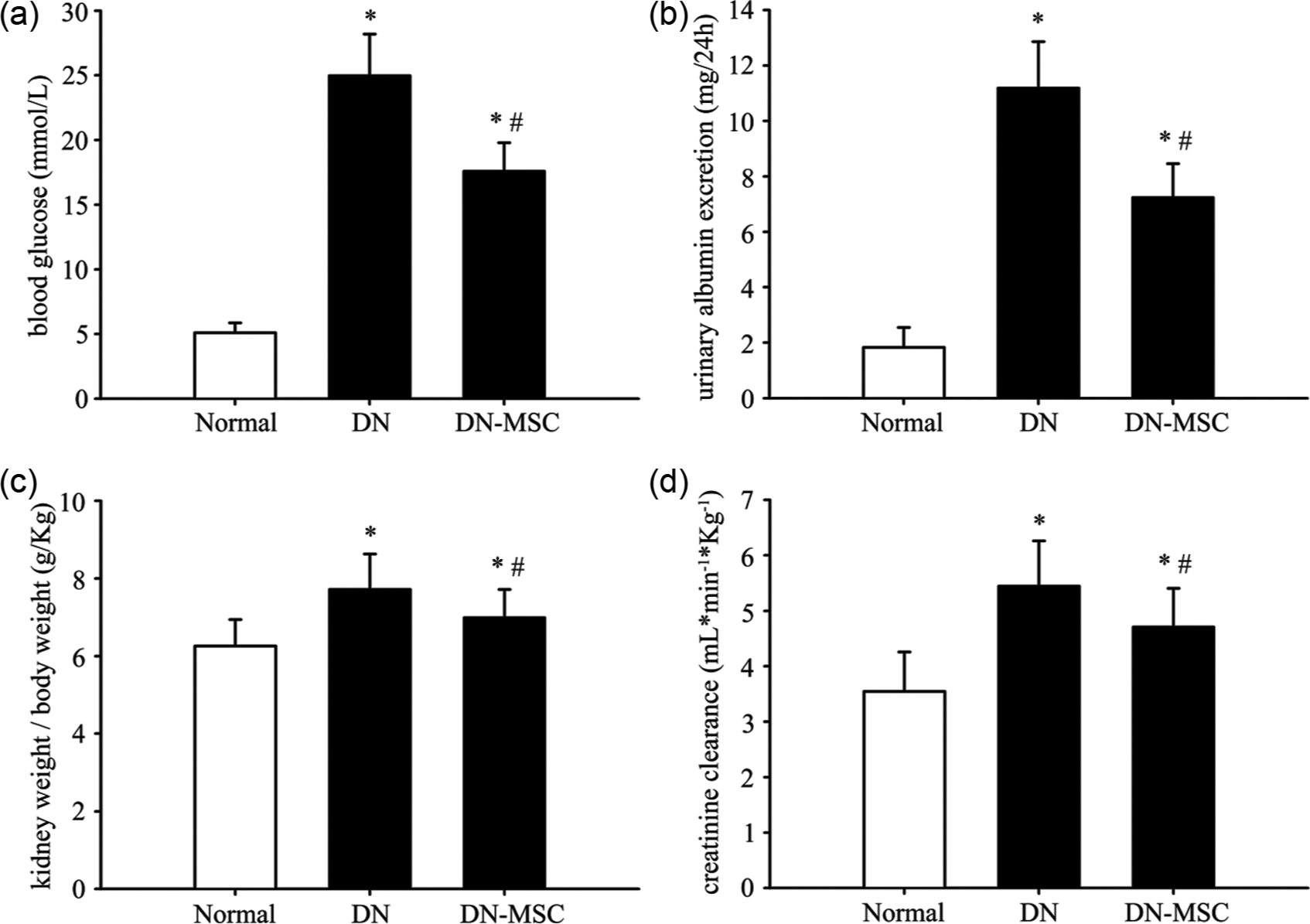
Effect of MSCs on physical and biochemical parameters: (a) blood glucose, (b) urinary albumin excretion, (c) kidney-to-body weight and (d) creatinine clearance in three groups of rats.
PAS staining showed that kidneys of DN group rats exhibited profound ECM deposition in the mesangium at 16 weeks after STZ injection. But MSC treatment ameliorated these pathological abnormalities remarkably as revealed by the GS increased by approximately tenfold in the DN group, but dramatically decreased in MSCs group (Figure 2).
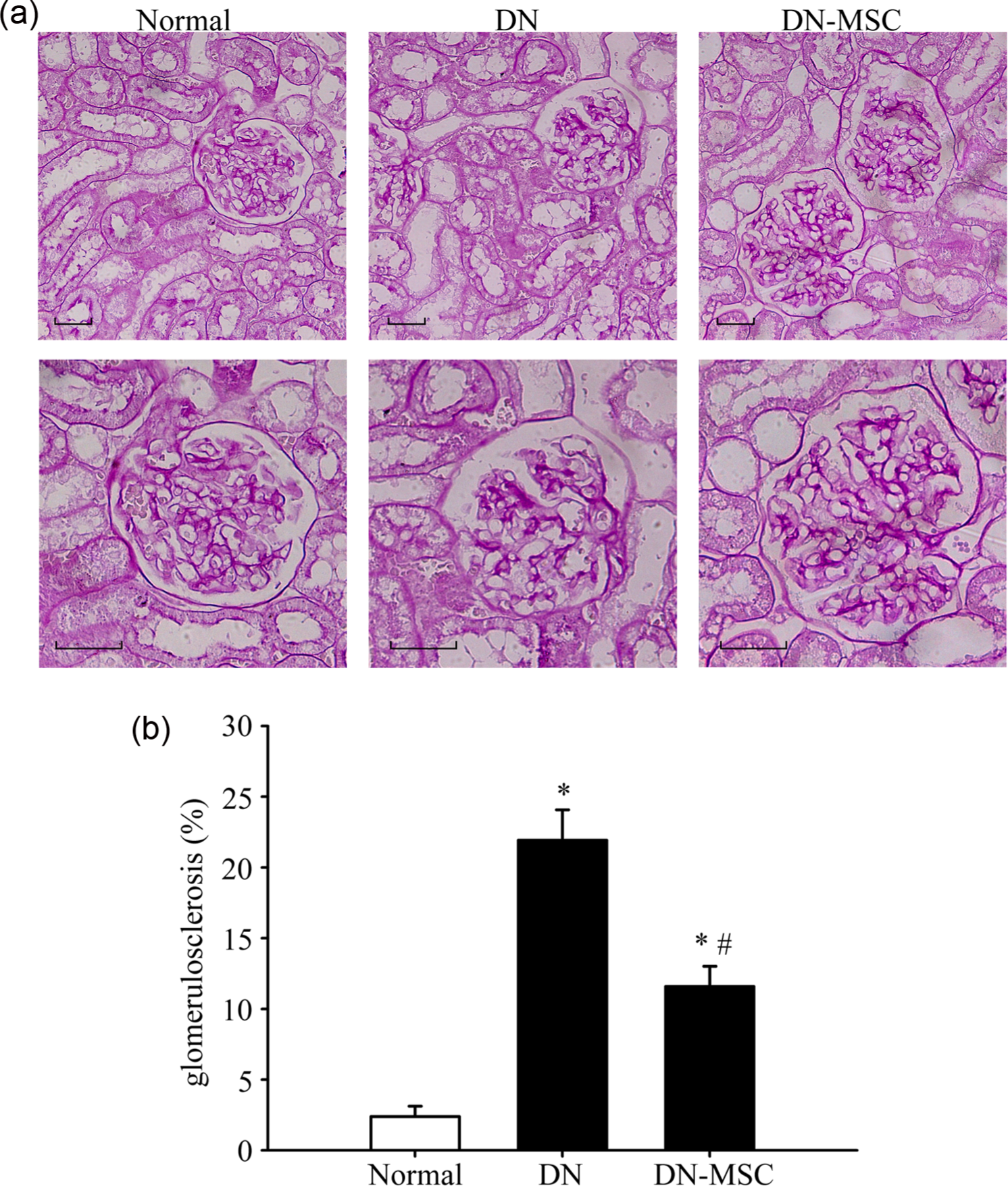
Effect of MSCs on renal histopathological changes: (a and b) representative images of periodic acid Schiff (PAS) staining in three groups and (c) quantitative analysis of glomerulosclerosis (GS) evaluated by the percentage of glomeruli exhibiting sclerotic lesions (%GS).
Collagen I and collagen IV are the main components of ECM in mesangial area. The messenger RNA (mRNA) and protein expression of collagen I and collagen IV in kidney as detected by real-time RT-PCR and Western blot method were remarkably increased in the DN group compared with the normal group. But the transplantation of MSCs attenuated collagen accumulation in diabetic kidney remarkably (Figure 3(a), (b), (e) and (f)).
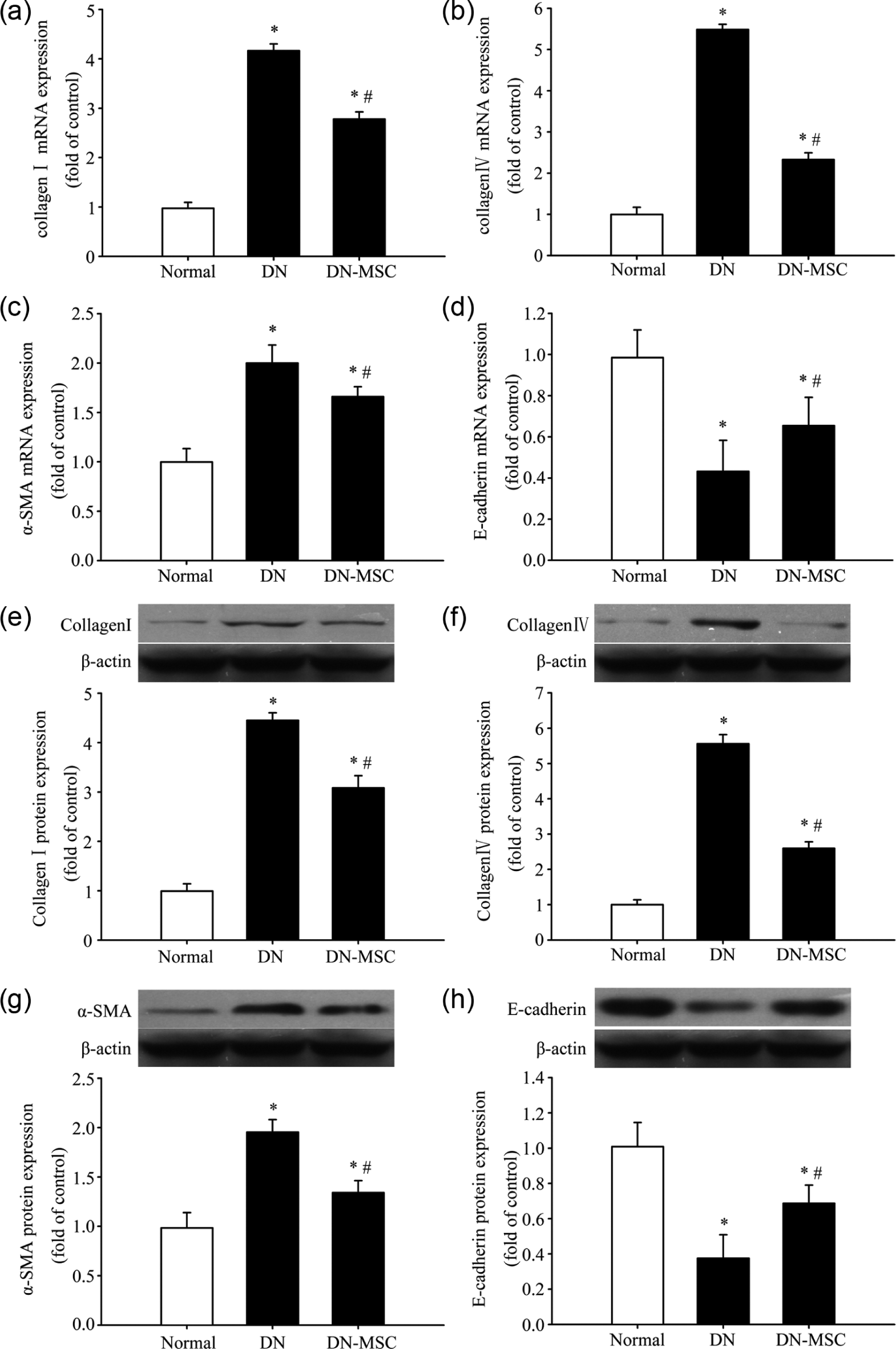
Effect of MSCs on glomerular fibrosis in diabetic kidney. At 8 weeks after treatment, the mRNA and protein expression of collagen I (a and e), collagen IV (b and f), α-SMA (c and g) and E-cadherin (d and h) in kidney tissue were measured using real-time RT-PCR and Western blot, respectively.
We next measured pro- and anti-fibrotic cytokines, which are well-known markers of DN. MSC transplantation effectively prevented α-SMA protein upregulation and E-cadherin protein downregulation in diabetic kidney (Figure 3(g) and (h)). The mRNA expression of α-SMA and E-cadherin showed a similar tendency as protein expression (Figure 3(c) and (d)).
MSCs suppressed TGF-β/Smad signalling in diabetic kidney
The mRNA and protein expressions of TGF-β in diabetic kidney were dramatically increased compared with the normal control group, and the phosphorylations of Smad2 and Smad3 were also increased, whereas the mRNA and protein expressions of TGF-β and phosphorylations of both Smad2 and Smad3 were significantly decreased in the MSCs group compared with the DN group (Figure 4), suggesting that MSCs suppressed TGF-β/Smad signalling in diabetic kidney.
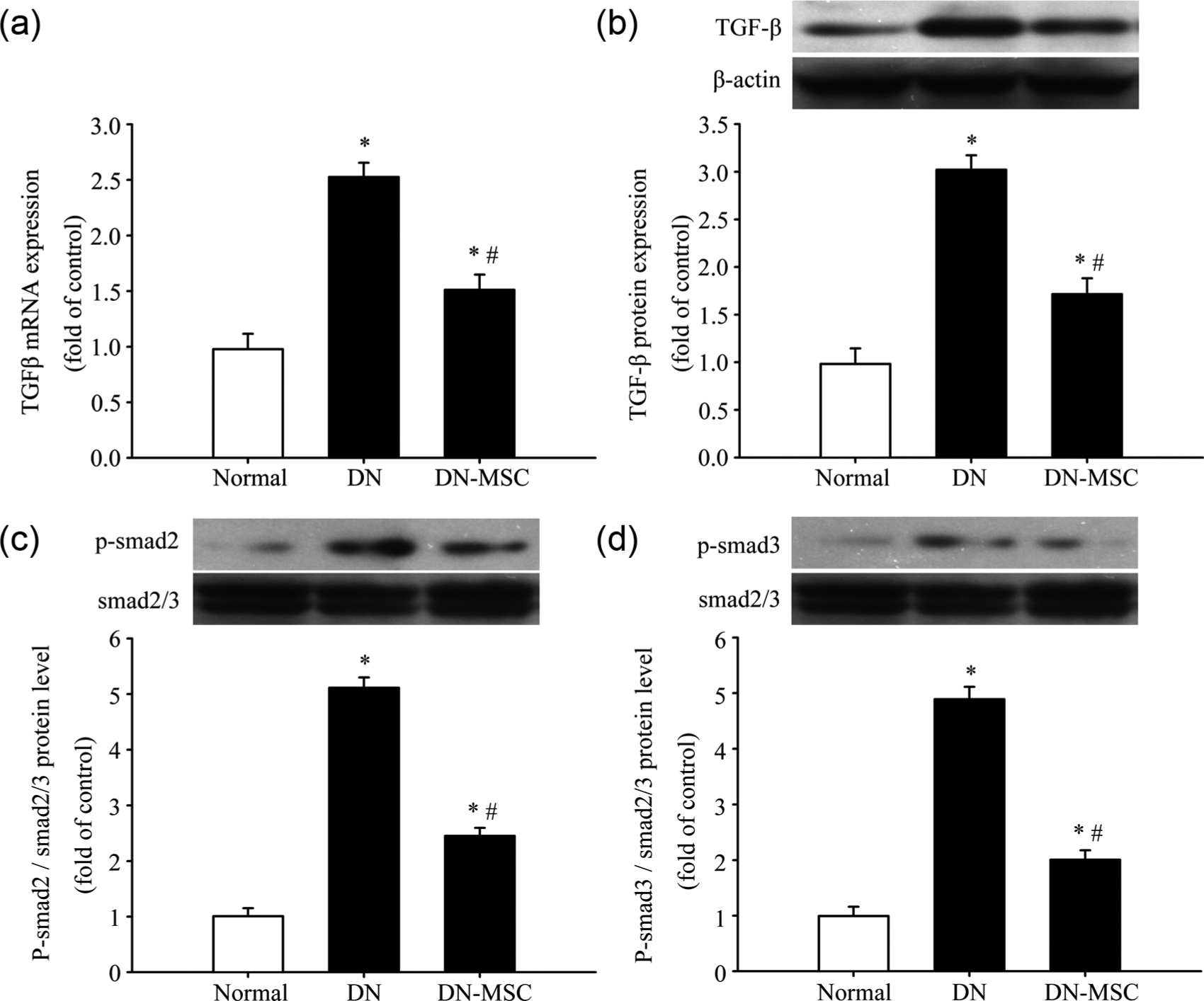
Effect of MSCs on TGF-β/Smad signalling in diabetic kidney. At 8 weeks after treatment, (a) the mRNA expression of TGF-β in kidney tissue was measured by real-time RT-PCR, and the protein expression of (b) TGF-β, (c) phosphorylated Smad2 and (d) phosphorylated Smad3 was measured by Western blot.
MSC-CM suppressed glucose-induced TGF-β/Smad signalling in GMCs
Glucose stimulation significantly induced the expression of TGF-β and phosphorylations of Smad2 and Smad3 compared with that in control cells, whereas co-culture of GMCs with MSCs resulted in a significant reduction of TGF-β protein expression and Smad2 and Smad3 phosphorylations compared with levels in vehicle-treated cells. Importantly, the MSC-CM has the similar effect as the MSCs (Figure 5(d)), suggesting that MSC-CM suppressed glucose-induced TGF-β/Smad signalling in GMCs; in other words, MSCs acted to suppress the glucose-induced TGF-β/Smad signalling in a paracrine manner in GMCs.
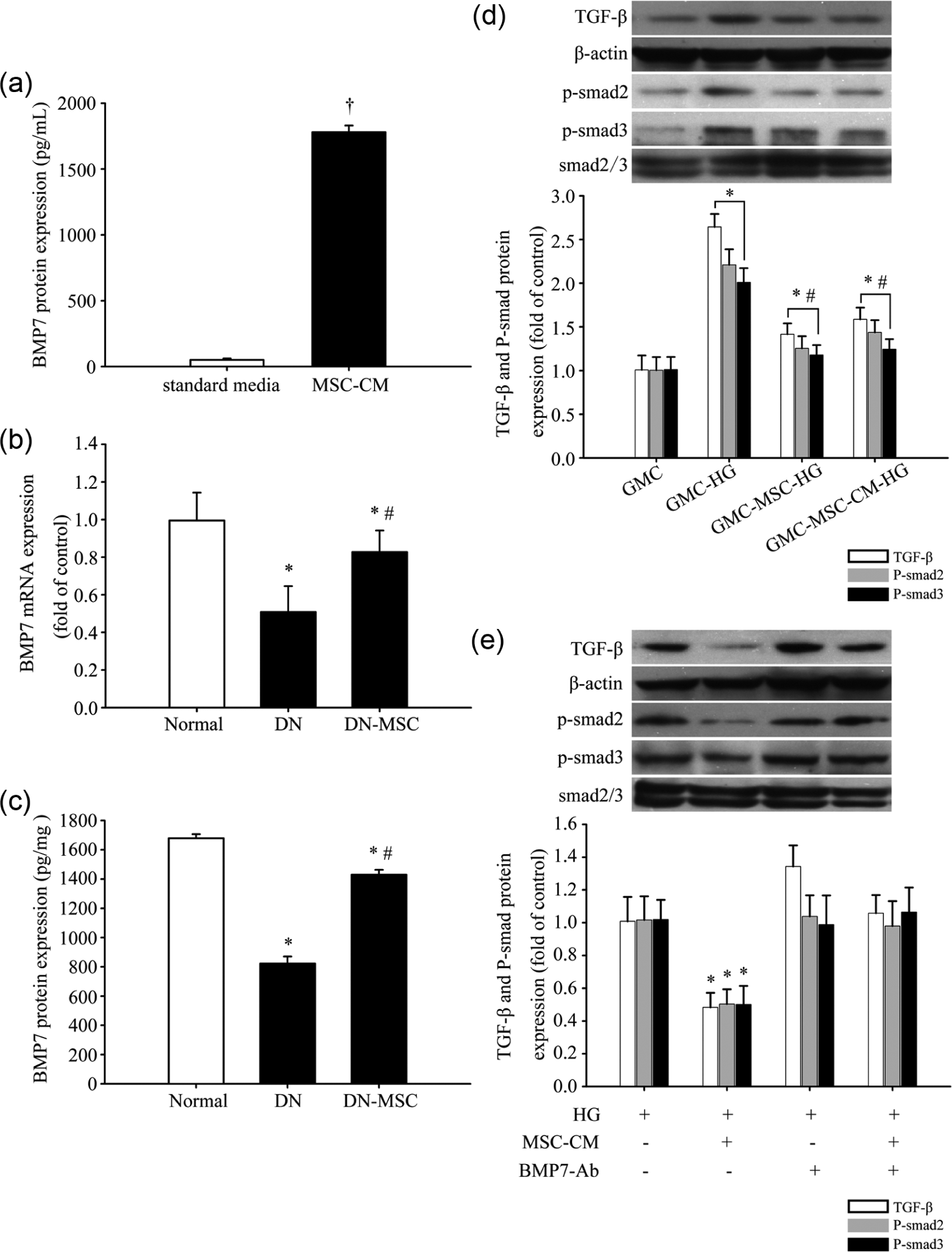
MSCs suppressed the glucose-induced TGF-β/Smad signalling via secretion of BMP7. (a) BMP7 in conditioned media obtained from MSCs was measured by ELISA (†p < 0.05 versus standard media). (b and c) BMP7 mRNA and protein expression in kidney tissues in three groups of rats measured by ELISA (*p < 0.05 versus normal group, #p < 0.05 versus DN group). (d) TGF-β, phosphorylated Smad2 and phosphorylated Smad3 protein expressions in four groups of GMCs as measured by the Western blot analysis and the quantitative analysis (GMC-HG indicates GMCs stimulated by high glucose; GMC-MSC-HG indicates co-culture of GMCs and MSCs stimulated by high glucose; GMC-MSC-CM-HG indicates GMCs cultured in MSC-CM stimulated by high glucose; *p < 0.05 versus GMC, #p < 0.05 versus GMC-HG). (e) Western blot analysis showed that MSC-CM inhibited upregulation of TGF-β expression, Smad2 and Smad3 phosphorylation after 72 h of high glucose stimulation and BMP7-Ab blocked the inhibitory effect of MSC-CM (*p < 0.05).
MSCs secreted BMP7 and inhibited TGF-β signalling in GMCs
A significant amount of BMP7 was detected in MSC-CM by ELISA. In other words, MSCs secreted the renoprotective cytokine BMP7 (Figure 5(a)). In diabetic kidney tissue, the mRNA and protein expressions of BMP7 were significantly decreased as compared with the normal group, but the level was much higher in MSCs group than in the DN group (Figure 5(b) and (c)).
To investigate the effect of BMP7 on TGF-β signalling, BMP7-Ab (10 µg/mL) was added or not into MSC-CM and fresh media for 1 h before its addition to GMCs. Western blot analysis showed that MSC-CM inhibited upregulation of TGF-β expression and phosphorylations of Smad2 and Smad3 after 72 h of high glucose stimulation, and BMP7-Ab blocked the inhibitory effect of MSC-CM (Figure 5(e)), suggesting that MSCs suppressed the glucose-induced TGF-β/Smad signalling via secretion of BMP7.
Discussion
The major findings of this study were that, in STZ-induced DN rat models, MSCs treatment effectively reduced blood glucose, urinary albumin excretion, Ccr, renal mass index and ameliorated glomerular fibrosis. The possible mechanisms underlying these effects involved inhibiting TGF-β/Smad signalling pathway via secretion of BMP7.
As we have discussed in our previous article, 15 the diabetes model with a single intraperitoneal injection of STZ we chose in this study is well documented to produce hyperglycaemia and insulinitis similar to their human counterparts. 25 At 8 weeks after STZ injection, diabetes rats develop severe hyperglycaemia, albuminuria, enhanced Ccr and glomerular pathological changes, all of which are characteristics of early stage of DN. So, we chose this time-point to conduct MSCs treatment to explore the effect of MSCs on DN.
In this study, we found that MSCs transplantation intravenously reduced blood glucose level significantly, which has also been documented by other studies.16,23 But there are also studies which have shown that MSCs have no effect on hyperglycaemia.18,22 By comparing these studies, we thought that the different transplantation route, the diabetes aetiology and/or stage, the source of stem cells and technical differences in the handling of donor MSCs may be the reasons that lead to this discrepancy. There are two possible mechanisms underlying the effect of MSCs on reducing blood glucose, one of which is that MSCs can differentiate into insulin-producing pancreatic cells; the other is that MSCs can enhance the regeneration of pancreatic β cells. A recent study 26 reveals that transplantation of multipotent stromal cells from human bone marrow to diabetic non-obese diabetic (NOD)/severe combined immunodeficiency (SCID) mice increases the number of mouse islets and mouse insulin-producing cells, and new islets appear to bud off pancreatic ducts that are the source of islets during early development of the pancreas, thus deducing that MSC can enhance proliferation, migration and differentiation of the nearby endogenous pancreatic stem/progenitor cells. In our other article (not published now), we had another experimental group with the similar blood glucose level to MSC-treated group by insulin treatment and found that although insulin treatment had effect on amelioration of renal parameters and glomerular fibrosis, the effect was much inferior to that of MSCs treatment. So, the data supported the speculation that the effect of MSCs treatment was not all due to the improvement of hyperglycaemia in MSCs group. There should be other mechanisms underlying the effect of MSCs on treating DN.
In our previous study, 15 MSCs were transducted with lentiviral-transduced green fluorescent protein as a cell tracker to assess the intrarenal localization of MSCs. We found that at 24 h after transplantation, a small amount of MSCs was present around the glomeruli and near vessels in kidney of MSC-treated diabetic rats, but at 8 weeks after transplantation, there was only few MSCs found in glomeruli and not found elsewhere, at which time the protective effect of stem cells on diabetic kidney still existed. There are also similar findings in other studies.16–18 So, it was likely reasonable to speculate that MSCs protected the diabetic kidney primarily via paracrine action, not cell-to-cell effect. Recently, more studies suggest the essential role of paracrine mechanism in renoprotective effect of MSCs. Administration of human embryonic–derived MSC-CM rescues kidney function in rats with established chronic kidney disease. 20 Furthermore, Eliopoulos et al. 21 found that human marrow–derived MSC-CM reduce cisplatin-induced tubular epithelial cell death, which is effectively inhibited by anti-vascular endothelial growth factor (VEGF) antibody, thus suggesting that VEGF secreted by MSCs may play an important role in renoprotection. Li et al. 27 also found that human adipose–derived MSCs prevent podocytic apoptosis and injury induced by high glucose mainly through secreting epithelial growth factor.
BMP7 is a 35-kDa homodimeric secretory protein and a member of the TGF-β superfamily. BMP binds to serine/threonine kinase receptor Types I and II; after ligand binding, Type II receptor phosphorylates Type I receptor, which phosphorylates R-Smads (Smad1, Smad5 and Smad8). Subsequently, these R-Smads form an oligomeric complex with Smad4 and translocate to the nucleus, where they activate transcription of target genes. BMP7 promotes various aspects of repair processes in various types of kidney cells: it antagonizes TGF-β-dependent fibrosis 28 and reduces apoptosis of tubular epithelial cells and podocytes. 29 Furthermore, BMP7 attenuates the augmented inflammation caused by anti-TGF-β strategies and reduces the expression of inflammatory cytokine. 30 In this study, we found that bone marrow–derived MSCs secreted a large amount of BMP7, and the expression of BMP7 in diabetic kidney after MSCs treatment increased dramatically. The expression of TGF-β and phosphorylation of Smad2 and Smad3 were significantly decreased in diabetic kidney after MSCs treatment, suggesting that MSCs suppressed TGF-β/Smad signalling in diabetic kidney. In vitro, MSC-CM had a similar effect as MSCs on suppressing TGF-β/Smad signalling pathway, which is effectively inhibited by anti-BMP7 antibody, thus suggesting that BMP7 secreted by MSCs may play an important role in renoprotection. Although the transplanted MSCs could be found within the kidney, the amount was very small, and the protective effect of MSCs on DN still existed at which time there was only few MSCs found in kidney. Besides, one study has found that human embryonic–derived MSC-CM rescues kidney function in rats with chronic kidney disease. 20 So, we speculated that the therapeutic effect of MSCs on DN was more likely to be systemic. It needs to be further explored whether the bone marrow–derived MSC-CM has protective effect on DN.
Taken together, the present data showed that bone marrow–derived MSCs ameliorated glomerular fibrosis in diabetic rats by inhibiting TGF-β/Smad signalling pathway via secretion of BMP7.
Footnotes
Acknowledgements
We thank the Laboratory Animal Center of the Second Hospital of Shandong University for the excellent rat care. We also thank Central Laboratory of the Second Hospital of Shandong University for providing the excellent experimental equipment. Guangju Guan and Gang Liu contributed equally to this work.
Declaration of conflicting interests
The authors declare that there is no conflict of interest.
Funding
This work was supported by the Innovation Foundation of Shandong University (No. 2009JC022) and the Development of Science and Technology Plan Projects in Shandong Province (2013GSF12104).
