Abstract
Diabetes augments the risk of hypertension. Although several factors have been implicated in the development of such hypertensive state, we designed this study to investigate blood pressure development, the activity of angiotensin-converting enzyme (ACE) in blood as well as sympathetic neurotransmission in the vas deferens of diabetic rats. We used streptozotocin (STZ)-induced diabetic rats (60 mg/kg) in order to evaluate the systolic blood pressure (SBP), ACE activity and peripheral sympathetic neurotransmission. We observed the following changes of parameters: increase of SBP, decrease of heart rate, augmentation of plasma ACE activity, enhancement of phasic and tonic vas deferens contractions elicited by electrical stimulation at 5 Hz, increase of maximal response to noradrenaline (NA) and decrease of adenosine triphosphate (ATP)-elicited contraction of vasa deferentia. The results reveal that in the development of hypertension in diabetic rats, augmentation of circulating ACE activity precedes the sympathetic dysfunction. Additionally, it seems that the purinergic and noradrenergic neurotransmission is compromised.
Introduction
Diabetes is a metabolic disease characterized by hyperglycaemia. Diabetes augments the risk of cardiovascular diseases, including arterial hypertension. 1 Hypertension and diabetes are becoming increasingly common; together, they greatly augment cardiovascular and renal damage, placing all diabetic hypertensive patients at high risk. 2 The common denominator of hypertensive/diabetic target organ-disease is the vascular tree. 3
There are several theories that describe mechanisms related to the development of hypertension in diabetics.1,4,5 For example, diabetic rats have a decrease in the levels of nitric oxide (NO) produced by acetylcholine in vascular endothelium and a higher incidence of oxidative stress in coronary arteries. 4
Common factors to diabetes and hypertension are oxidative stress and inflammation. Angiotensin-converting enzyme (ACE) is present in several tissues including vascular, brain, kidney and plasma.6–8 ACE catalyses the conversion of angiotensin I to angiotensin II; angiotensin II may trigger inflammatory effects and oxidative stress 9 that can elicit an increase of smooth muscle contraction.10,11
However, in diabetic patients, augmented sympathetic activity can increase heart rate (HR), leading to elevation of renin–angiotensin–aldosterone system (RAAS) activity; these factors directly influence the genesis of hypertension.9,12 The electrically stimulated isolated vas deferens is an excellent model to study sympathetic neurotransmission and hence its alterations in disease. For instance, studies indicate that diabetes induces an increase in the purinergic component of the response to sympathetic nerve stimulation of the rat vas deferens. 13
The mechanisms underlying the development of hypertension in diabetics are not fully understood. We therefore planned this study to investigate blood pressure development, circulating ACE activity as well as sympathetic neurotransmission in vas deferens from streptozotocin (STZ)-induced diabetic rats.
Methods
All experimental procedures were approved by the Ethical Committee of Federal University of São Paulo (protocol number: 1169/2011). Diabetes was induced by intraperitoneal administration of a single dose of STZ (60 mg/kg), and control rats were treated with drug-free vehicle. All animals were used for experimentation when 4-month-old. To obtain 30-day diabetic rats (D30), STZ was injected when rats were 3-month-old, and to obtain 60-day diabetic rats (D60), they were injected when they were 2-month-old.
The STZ (a glucosamine-nitrosourea) administered is taken up by the pancreatic cell glucose transporter GLUT-2, promoting an alkalinization of DNA and subsequent activation of poly[adenosine diphosphate (ADP)]-ribose, which breaks the DNA molecule and depletes nicotinamide adenine dinucleotide, leading to destruction of β-cells in the pancreatic islets of Langerhans. 14 In our study, the rats were considered diabetic when values of glycaemia were higher than 180 mg/dL after 48 h of diabetes induction; to measure glycaemia, we used a glucometer Accu-Chek Active® system of Roche (Brazil).
Non-invasive measurement of blood pressure from conscious rats
Systolic blood pressure (SBP) in non-anesthetized rats was measured by non-invasive tail-cuff plethysmography. 15 Before STZ administration, animals were monitored once a week, during 4 weeks, in order to let them adapt to this procedure. After this period, the animals were made diabetic as described above; the pressure was monitored once a week along the whole study.
Determination of ACE activity
Once a month, about 0.5 mL of blood was taken from the tail artery with a heparinized syringe; plasma was separated by centrifugation (10 min, 2500 r/min, 4°C) and stored at −20°C. Plasma ACE activity was determined using the indirect fluorometric method described by Santos et al.
6
The method is based on the hydrolysis of hypuril-
Functional study of sympathetic system and pharmacological reactivity of vas deferens
Vasa deferentia from male diabetic and control rats were isolated and mounted under 1.0 g tension in 10-mL organ baths containing a nutrient solution with the following composition (mM): 138 NaCl, 5.7 KCl, 1.8 CaCl2·2H2O, 15 NaHCO3, 0.36 NaH2PO4·H2O and 5.5 glucose, prepared in glass-distilled deionized water, bubbled with air and maintained at 32°C, pH 7.3.16 –18 One end of the vas deferens was fixed to the organ chamber, and the other end was attached by means of a silk surgical suture to a force–displacement transducer (model FT 302; CB FT-302, iWORX, NH, USA) connected through a bridge amplifier to a PowerLab recorder (ADInstruments, Castle Hill, NSW, Australia) coupled to a computer. Contractions were recorded and the data stored in Chart 4.2.1 software (ADInstruments, Warwick, USA).
Pharmacological reactivity
Cumulative concentration–response curves were made for the adrenergic agonist noradrenaline (NA) (10−8−3.10−4 M), 16 to quantify for possible alterations of receptors, indicated by the respective pharmacological parameters, 19 such as Eam (maximum contraction induced by an agonist A) 16 and pD2, as the apparent affinity of the agonists, measured as the negative log of ED50. 19 Contractions induced by single concentration of the indirect agonist tyramine (10−4 M) were also measured, to check for changes on the release of endogenous NA. We also performed time-effect curves for KCl (80 mM) or adenosine triphosphate (ATP) (1 mM) to verify the time-course of smooth muscle contraction induced by depolarization.
Nerve-mediated contraction
For the study of contractions, elicited by sympathetically released NA, the vas deferens was mounted as described above and placed between two parallel platinum electrodes for electrical field stimulation, as previously described.17,18 After an equilibration period of 30 min, the organ was subjected to electrical field stimulation of 60 V, 1 ms duration and frequencies of 2 and 5 Hz, by means of a Grass S88 stimulator (Grass USA). In neurogenic contractions induced by the electrical field stimulation, two steps can be distinguished, namely, phasic (purinergic, ATP dependent) and tonic (noradrenergic). Both components were measured for comparisons between groups. 20
Statistics
The circulating levels of ACE and the concentration–response curves were analysed by a GraphPad Prism computer program. The parameters obtained were expressed as mean ± standard error of mean (SEM). Statistical analysis was made by using the one-way ANOVA and Tukey’s test; significance was accepted at p < 0.05.
Results
Diabetes induction by STZ was evaluated using blood glucose level monitoring. In control animals, glycaemia was 133 ± 8 mg/dL, while in diabetic animals, it was 472 ± 23 mg/dL, showing a 3.54-fold increase.
The rats treated with STZ exhibited clinical signs of diabetes such as polydipsia or polyuria. Concerning the evolution of body weight, at the third week of STZ treatment, diabetic rats had a significant weight loss, time-dependent changes of body weight in control, D30 and D60 groups (Figure 1(a)).
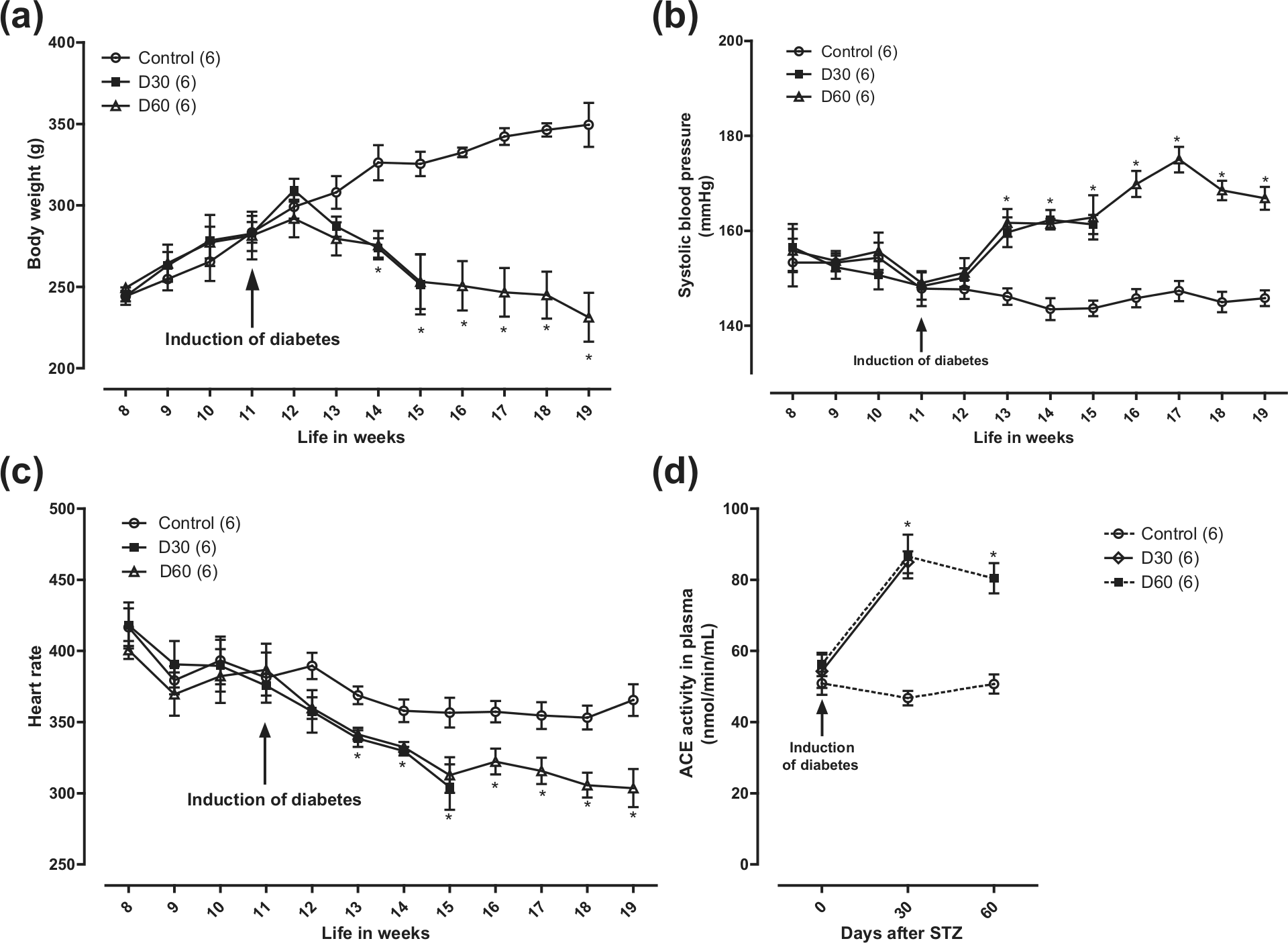
Time-dependent changes of (a) body weight, (b) systolic blood pressure, (c) heart rate and (d) plasma ACE activity in control animals and after 30 (D30) or 60 (D60) days of STZ injection.
The SBP of animals was monitored by a non-invasive indirect method in the rat tail for 4 weeks before diabetes induction and continued to be monitored weekly after induction. In the second week, the SBP was 146 ± 1.7 mmHg in the control group and 160 ± 3.13 mmHg in diabetic animals (Figure 1(b)); this elevation of blood pressure was maintained throughout the study period.
The elevation of blood pressure in diabetic animals was accompanied by a decrease in HR that developed gradually during the first 4 weeks after STZ injection. Control animals had an initial HR of 416 ± 13.2 bpm, while after 30 days post STZ injection (D30), the HR decreased to 304 ± 15 bpm. From 4 to 8 weeks post STZ (D60), the HR stabilized at around 303 ± 11 bpm (Figure 1(c)). HR also decreased with time in control animals, from 416 ± 13.2 to 365 ± 11 bpm by week 8.
Of interest was the finding that diabetes elicited an augmentation of circulating ACE activity. So, in control rats, the initial ACE activity was 50.9 ± 3.2 nmol/min/mL, a figure that was maintained fairly similar after 30 or 60 days of the experiment duration. However, in STZ-treated rats, ACE activity augmented to 84.9 ± 3.1 nmol/min/mL in D30 and to 80.4 ± 4.2 nmol/min/mL in D60 rats, representing about 1.6- and 1.57-fold above control, respectively (Figure 1(d)). We also assessed the contractions of vasa deferentia elicited by tyramine that is known to release NA from sympathetic nerve terminals; using this protocol, no difference was found between control and diabetic rats.
However, it is well established that the contractions elicited by transmural electrical stimulation of the vas deferens are due to the simultaneous release of ATP and NA from sympathetic nerve terminals, as a result of action potential generation. 21 With electrical stimulation at 2 Hz, no difference was found between groups. However, at 5 Hz stimulation, we found an increase in the phasic response (PR) (ATP dependent) of 51% at D30 and 56% at D60 (Figure 2). The tonic response (TR) (NA dependent) was not significantly changed in D30; however, it was increased at D60 (23.4%).
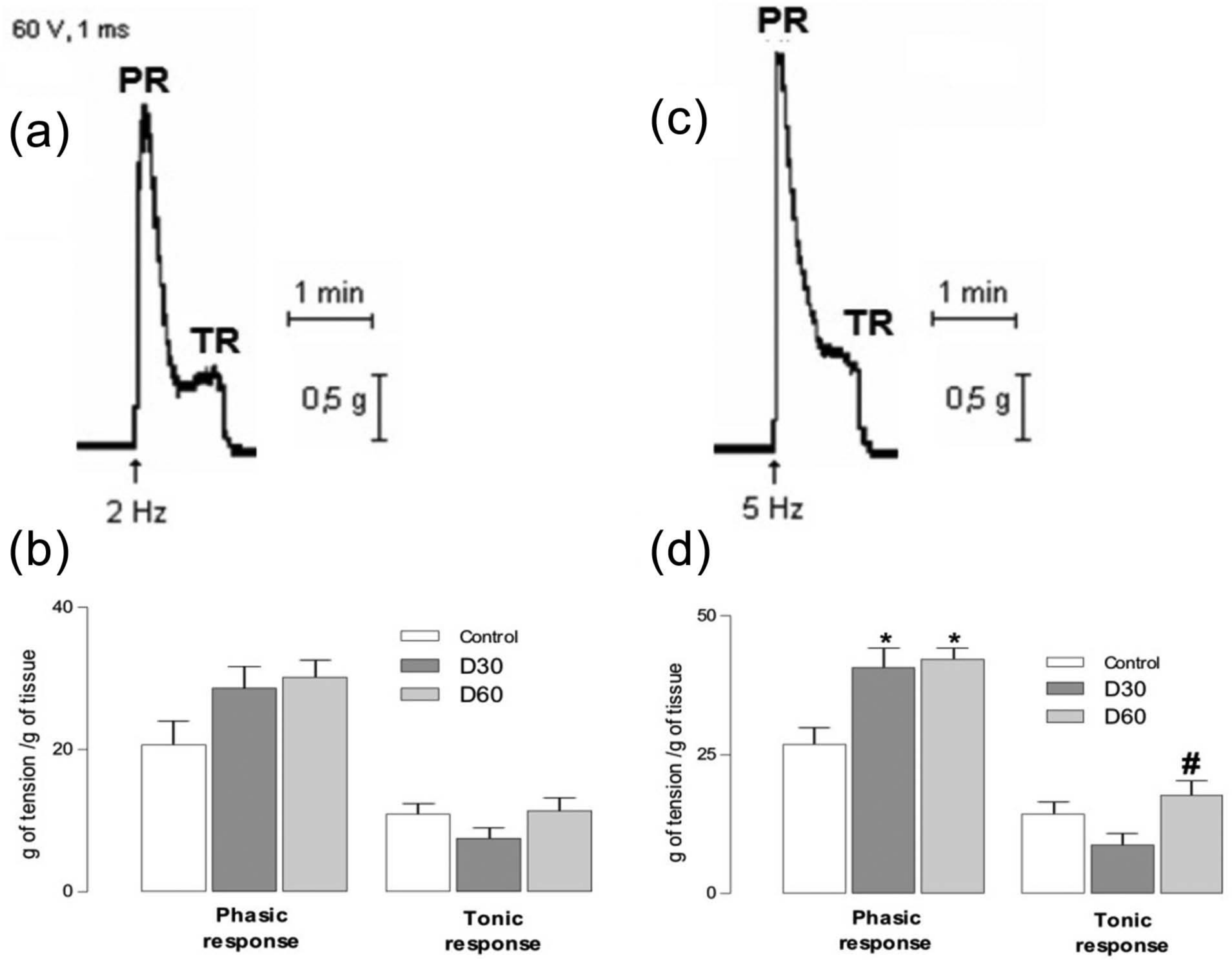
(a and c) Recording of electrical stimulation at 2 and 5 Hz with PR and TR. (b and d) Field electrical stimulation of vas deferens from control, D30 and D60 rats.
Cumulative concentration–response curves were made for the adrenergic agonist NA (10−8−3.10−4 M). The maximal effect of concentration–response curves for NA was enhanced by 192% in D60 when compared with control (Figure 3); however, the pD2 was unchanged.
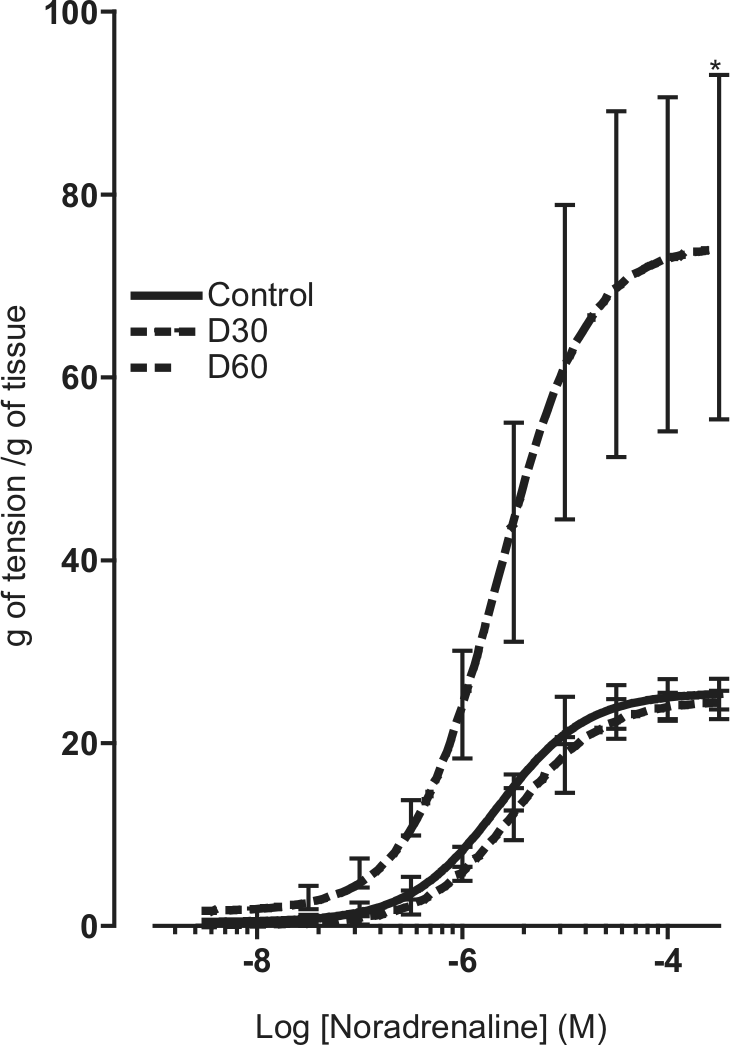
Concentration–response curves for noradrenaline in vasa deferentia from control animals and diabetic rats at 30 (D30) and 60 days (D60) after streptozotocin treatment.
The time curve of ATP-elicited vas deferens contraction considerably differed in control and diabetic animals. The maximum amplitude decreased by 42% in D30 animals and by 60% in D60 animals, compared with controls. However, the TR to KCl was potentiated in D30 (32.4%) (Figure 4(d)).
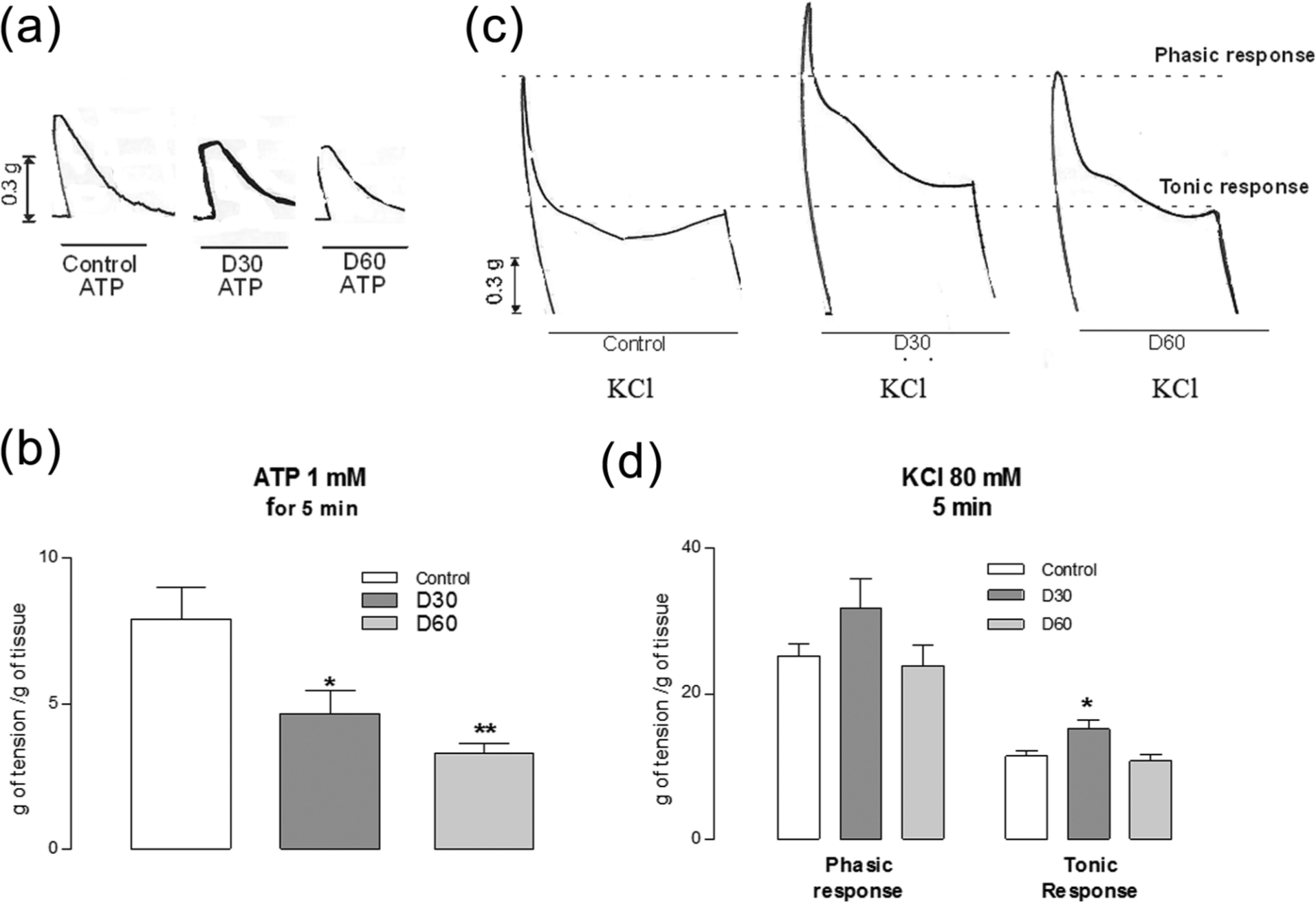
Changes of reactivity of the smooth muscle of vasa deferentia, stimulated with ATP or a solution containing 80 mM K+. (a) Example records of ATP-elicited responses in a vas deferens from control and streptozotocin-treated animals at 30 (D30) and 60 days (D60). (b) Peak amplitude of ATP-elicited contraction, measured in grams of tension per gram of tissue (ordinate) in control, D30 and D60 rats. (c) Example records of the K+-elicited contraction of vasa deferentia from control, D30 and D60 animals. (d) Averaged values of tonic and phasic amplitudes in K+-stimulated vasa deferentia from control, D30 and D60 animals.
Discussion
STZ-elicited diabetic rats of this study were compared well with models in the literature 22 in that the animals progressively lost weight and developed a prompt hyperglycaemic state. The diabetic rats also developed a sustained hypertensive state (Figure 1(b)) that was accompanied by a progressive decline of HR (Figure 1(c)). This bradycardia could be due to a secondary reflex compensation due to hypertension. A decrease of HR and coronary blood flow in STZ-induced diabetic rats has been previously observed. 23 The fact that diabetic rats develop hypertension is well documented.23,24 However, the mechanism underlying such blood pressure elevation is as yet unclear.
Among several other mechanisms that could explain hypertension development associated with diabetes is a decrease of nitric oxide production with concomitant vasoconstriction 4 or an increase of oxidative stress. 25 More relevant to the present study are the old 26 and recent observations 27 that the plasma ACE activity is increased in diabetic patients. In this sense, our data showed an elevation of circulating ACE activity in diabetic rats D30 and D60 (Figure 1(d)). Enhanced ACE activity correlates with diabetic nephropathy in humans, 27 suggesting that augmented angiotensin II synthesis could also be a contributor factor in development of hypertension in diabetes. This is compatible with clinical data showing that treatment with blockers of angiotensin II receptors or with ACE inhibitors reduces cardiovascular events in diabetic patients. 26
An interesting finding was that ACE elevation somehow preceded the full development of systolic hypertension. Thus, at day 30 after STZ treatment, ACE elevation was already maximal and displayed a tendency to decay at day 60 (Figure 1(d)). In contrast, the SBP continued its increasing tendency from days 30 to 60 after STZ administration.
Augmentation of ACE activity and thus of angiotensin II synthesis has also been associated with enhanced oxidative stress that in turn may contribute to sympathetic dysfunction and to the development of diabetic hypertension. 25 In line with this hypothesis are the observations that in control rat vas deferens, angiotensin II enhances the purinergic component 28 as well as the noradrenergic component 29 of the electrically induced contractions.
In diabetic rats, we have found an increase of the electrically induced contractile response of vasa deferentia at 60 days, but not at 30 days after STZ injection (Figure 2). Because the elevation of ACE was similar in D30 and D60 rats (Figure 1(d)), it seems that the enhanced sympathetic response, likely due to excess synthesis of angiotensin II, is a delayed phenomenon with respect to ACE changes. This delayed presynaptic response also correlated with a delayed postsynaptic response, namely, the pronounced increase of exogenous NA-elicited responses in D60, which was not observed in D30 rats (Figure 3). A similar enhanced response to NA was previously reported. 29
It is well established that the contractile response elicited by electrical field stimulation of the rat vas deferens consists of an initial phasic component due to ATP release that is followed by a tonic component due to NA release. 21 Because we found an enhanced PR in D60, we expected that the response to exogenously added ATP should also be augmented; however, we found the opposite (Figure 4(b)). This decreased purinergic response correlates well with a decreased similar response recently found in heart atria of STZ-treated rats. 5 As for the TR, we also found an apparent discrepancy between the vas deferens tonic contractions elicited by endogenous NA, which only elicited a mild increase in D60 (Figure 2(d)), and the drastic increase elicited by exogenous NA (Figure 3). Our data are only in partial agreement with the earlier observation of an increase of the purinergic and noradrenergic components of electrically induced vas deferens contractions of diabetic rats.
The apparent discrepancy between the vas deferens responses to endogenously released and exogenously added ATP and NA in diabetic D60 rats deserves a last comment. ATP is co-stored with NA in large-dense cored vesicles at sympathetic nerve terminals. 30 The enhanced PRs were only seen at frequencies of 5 Hz stimulation (Figure 2(d)) but not at 2 Hz (Figure 2(b)). The selective release of ATP or NA at the lower and higher stimulation frequencies 20 could explain that the PRs (due to ATP release) could be potentiated to a greater extent compared with the TR (Figure 2(d)). It is also curious that while the response to exogenously added ATP was considerably decreased in D60 rats, those elicited by exogenous NA were drastically enhanced. These differences in the response of presynaptic-mediated and postsynaptic-mediated responses in D60 animals suggest that diabetes is causing distinct purinergic and noradrenergic dysfunctions.
In conclusion, the results of this study are compatible with the hypothesis that in STZ-elicited diabetic rats, the ACE elevation precedes the full development of hypertensive state. Furthermore, the ACE elevation also precedes the dysfunction of the sympathetic nervous system that contributes to the development of hypertension. Finally, diabetes seems to clearly affect in a different mode the purinergic and noradrenergic neurotransmission.
Footnotes
Declaration of conflicting interests
The authors declare that they have no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
