Abstract
A cross-sectional analysis of ethnic differences in dietary intake, insulin sensitivity and beta-cell function, using the intravenous glucose tolerance test (IVGTT), was conducted on 497 healthy adult participants of the ‘Reading, Imperial, Surrey, Cambridge, and Kings’ (RISCK) study. Insulin sensitivity (Si) was significantly lower in African-Caribbean (AC) and South Asian (SA) participants [IVGTT-Si; AC: 2.13 vs SA: 2.25 vs white-European (WE): 2.84 (×10−4 mL µU min)2, p < 0.001]. AC participants had a higher prevalence of anti-hypertensive therapy (AC: 19.7% vs SA: 7.5%), the most cardioprotective lipid profile [total:high-density lipoprotein (HDL); AC: 3.52 vs SA: 4.08 vs WE: 3.83, p = 0.03] and more pronounced hyperinsulinaemia [IVGTT–acute insulin response (AIR)] [AC: 575 vs SA: 428 vs WE: 344 mL/µU/min)2, p = 0.002], specifically in female participants. Intake of saturated fat and carbohydrate was lower and higher in AC (10.9% and 50.4%) and SA (11.1% and 52.3%), respectively, compared to WE (13.6% and 43.8%, p < 0.001). Insulin resistance in ACs is characterised by ‘normal’ lipid profiles but high rates of hypertension and pronounced hyperinsulinaemia.
Keywords
Introduction
Insulin resistance, beta-cell dysfunction and increased hepatic glucose production may all contribute to the development of impaired glucose tolerance (IGT) and type 2 diabetes mellitus (T2DM). 1 In the United Kingdom, people of South Asian and African-Caribbean origin are at exceptionally high risk of developing insulin resistance and T2DM. 2 Obesity, specifically central obesity, has been recognised as an important predictor of insulin resistance 3 –5 and is associated with the clustering of metabolic abnormalities, including the development of an atherogenic lipid profile, 6 that contribute to the high prevalence of coronary heart disease (CHD) in metabolic syndrome (MetS) and T2DM. 7,8 While the expression of insulin resistance within the latter two conditions is well recognised in South Asians, insulin resistance in the absence of obesity and dyslipidaemia but in the presence of hypertension in black African-Caribbeans 9 –11 suggests that MetS and T2DM may have a different metabolic aetiology in this population. Although cardiovascular disease rates are high, at least in the diaspora, 12 the clinical outcomes associated with T2DM are different in each group with respect to lower rates of myocardial infarction in African-Caribbeans, but high rates of stroke and renal failure, 13 –15 related to hypertension. 16 The underlying causes of this apparent disparity in the features of MetS and T2DM between black African-Caribbean and other ethnic groups are not well understood. Studies in black African-Americans, using the intravenous glucose tolerance test (IVGTT), have revealed differences in beta-cell function, resulting in greater acute insulin response (AIR) and relative hyperinsulinaemia for any degree of insulin resistance compared to other ethnic groups. 11,17 If greater AIR is a marker of increased insulin secretion, such prolonged hypersecretion may lead to relatively early ‘beta-cell exhaustion’ and a reduction in beta-cell function. Thus, this may suggest that T2DM is more strongly driven by beta-cell dysfunction, 18 –20 but this has not been reported in black communities residing in the United Kingdom.
The aim of this study was to determine ethnic variation in the expression of insulin resistance and associated metabolic risk factors and dietary intake in a large population of UK-dwelling black African-Caribbean, South Asian and white-European adults at high risk of the MetS. We hypothesised that the black African-Caribbeans and South Asians would be markedly more insulin resistant than the white Europeans; South Asians would be more frequently characterised by an atherogenic lipid profile, while black African-Caribbeans would express a cardioprotective lipid profile and impaired beta-cell function.
Subjects and methods
Study design
The study consisted of a cross-sectional comparison of insulin resistance and associated metabolic characteristics, including beta-cell function and habitual dietary intakes in South Asian, black African-Caribbean and white-European subjects as subgroups from the ‘Reading, Imperial, Surrey, Cambridge, and Kings’ (RISCK) study cohort that consisted of adults exhibiting characteristics of the MetS but otherwise healthy and free from chronic disease. 21
Study sample
A cross-sectional comparison of the MetS was performed on data from 794 men and women, aged 30–70 years, of South Asian, black African-Caribbean and white-European origin who were eligible for participation in the RISCK study; eligibility criteria excluded diabetes, cardiovascular disease and use of lipid-lowering medication (e.g. statins). Full details of the RISCK study methodology 22 and primary outcomes have been reported elsewhere. 21 In brief, participants were recruited from the general population local to each of five study centres using advertisements in local newspapers and databases of the previous study participation. Participants were scored for characteristics of the MetS (0–10 points), those scoring ≥4 points were eligible for participation. Dietary intakes (n = 503) were measured, and IVGTT (n = 497) performed on a subset of participants who completed the study.
All subjects gave informed written consent, and the study was approved by the National Research Ethics Service (Ref: ISRCTN29111298).
Methods
Anthropometry
Weight (kg), height (m), body mass index (BMI) (kg/m2) and waist circumference (cm) were measured for each subject. Body fat percentage was measured using a bioelectrical impedance method (Tanita BC418 MA). Blood pressure was measured using an automated sphygmomanometer, subjects were rested in a seated position, and the mean of three repeated measurements was recorded. All anthropometric measurements were performed in the morning after an overnight fast.
Biochemical measures
Blood samples were taken after a 12 h overnight fast. Samples for blood glucose, insulin and lipid analysis were collected in fluoride, lithium heparin and serum monovettes, respectively (Sarstedt, UK). All blood samples were processed within 2 h of collection. 21
The homeostatic model assessment (HOMA2) of insulin resistance (HOMA2-IR) was calculated from fasting glucose and insulin using the HOMA2 calculator software (Version 2.2, Diabetes Trials Unit, Oxford, UK). 23
The ratios of total to high-density lipoprotein cholesterol (HDL-C) and triglyceride to HDL-C concentrations were calculated as metrics of atherogenic dyslipidaemia. The prevalence of atherogenic dyslipidaemia, defined as a dichotomous state, was based on the combined occurrence of fasting triglyceride concentration of >1.70 mmol/L and HDL-C concentration of <1.04 mmol/L in males or 1.30 mmol/L in females.
Dietary intakes
Food diaries were used to record the habitual dietary intake of all participants (4-day food diaries, including 3 weekdays and 1 weekend day). Portion sizes were estimated using a photographic atlas 24 and household measures. Dietary records were checked and validated by interview with a nutritionist. 25 Nutrient intakes were estimated using the food-composition database software DINO (Medical Research Council Human Nutrition Research Unit, Elsie Widdowson Laboratory, Cambridge, UK), which includes the UK food-composition tables and is supplemented with additional data obtained from manufacturers and from in-house analyses and calculations. The database contained glycaemic index (GI) values for carbohydrate-containing foods. 26
IIVGTT
Insulin sensitivity (Si) was assessed by an IVGTT 27 at baseline and post intervention on all participants taking part in the dietary interventions. Data from the baseline IVGTT have been used in this analysis.
Si was calculated by mathematical modelling methods using the MINMOD Millennium programme (Version 6.02) [Boston, 2003, Diabetes Technol Ther 5(6): 1003–1015]. From the model insulin sensitivity (Si), glucose effectiveness (Sg), AIR and disposition index (DI) were calculated. 28
Statistical analysis
Data analyses were performed using SPSS 17.0 for Windows.
All variables were tested for normality, linearity, homogeneity of variances, homogeneity of regression slopes and for the reliability of covariates. Ranked values were used when variables were not normally distributed. Tabulated results are expressed as means and standard deviations (SDs), unless stated otherwise (geometric means, adjusted for age, gender and BMI, and 95% confidence intervals (CIs) are shown for Figure 1). Asymmetric data were log transformed and analysed using parametric methods.
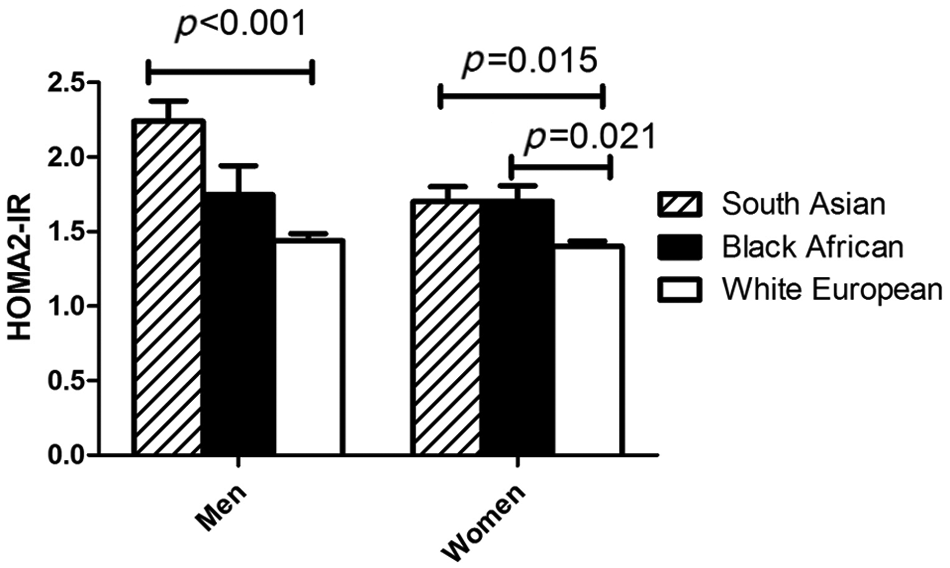
HOMA2-IR by gender and ethnic group.
A one-way between-group analysis of variance (ANOVA) was used with a Bonferroni post hoc test, to examine the differences in anthropometric and fasting metabolic characteristics, including insulin resistance (HOMA2-IR), between ethnic groups within each gender. The data shown are unadjusted mean values and SD.
A one-way between-group multivariate analysis of variance (MANOVA) was performed to investigate the differences in dietary intakes between ethnic groups, within each gender, adjusted for age and BMI with the Bonferroni multiple comparison test (probability adjusted for three comparisons). The data shown are unadjusted mean values and SD.
One-way between-group analysis of covariance (ANCOVA) was performed to examine the differences in Si, Sg, AIR and DI, from the IVGTT and HOMA2-IR, between ethnic groups. In the analyses of the effects of ethnicity within gender groups, age and BMI were covariates. In the analysis of the effects of ethnicity in the whole group, age and BMI were covariates and gender was a fixed factor. Within each ethnic group, differences between males and females have been tested using an independent samples t-test.
Results
Anthropometric, blood pressure and fasting biochemical measures of lipids and insulin resistance (HOMA2-IR) were available for 794 subjects, as shown in Table 1. The black African-Caribbeans were significantly younger, while the black African-Caribbean women had a higher BMI and proportion of body fat than all other groups. There were no significant differences in waist circumference between the groups, although a trend towards lower waist circumference was seen in the black African-Caribbean males. Total serum and low-density lipoprotein (LDL) cholesterol, and triglycerides were lower in the black African-Caribbeans compared with the other groups. HDL-C was higher in females than males for all ethnic groups, and lower in both the South Asian and black African-Caribbean females compared to the white participants. The ratio of total plasma cholesterol to HDL-C, an established metric of cardiovascular risk, was lower in black African-Caribbean females (mean = 3.5; SD = 0.83) compared with South Asian (mean = 4.1; SD = 0.97) and white-European females (mean = 3.8; SD = 1.07) (Table 1). The prevalence of atherogenic dyslipidaemia, assessed by the combined occurrence of raised triglycerides (≥1.70 mmol/L) and low HDL-C (≤1.04 mmol/L in males and ≤1.30 mmol/L in females), was greatest in South Asian females (26%) and lowest in African-Caribbean males (0%) and females (2%). Figure 1 shows estimates of insulin resistance (HOMA2-IR) in participants adjusted for age and BMI. HOMA2-IR was 43% higher in the South Asian compared to the white-European men [95% CI (21,64); p < 0.001], and 24% higher in South Asians [95% CI (8,40); p = 0.001] and 20% greater in black African-Caribbean compared with white-European women [95% CI (3,37); p = 0.014]. There were no differences in blood pressure between groups when subjects taking anti-hypertensive medication were excluded and means adjusted for age. The proportion of participants taking anti-hypertensive medication was highest in the black African-Caribbeans (19.7%) and lowest in the South Asians (7.5%).
Insulin sensitivity, serum lipids and anthropometric features by gender and ethnic group.
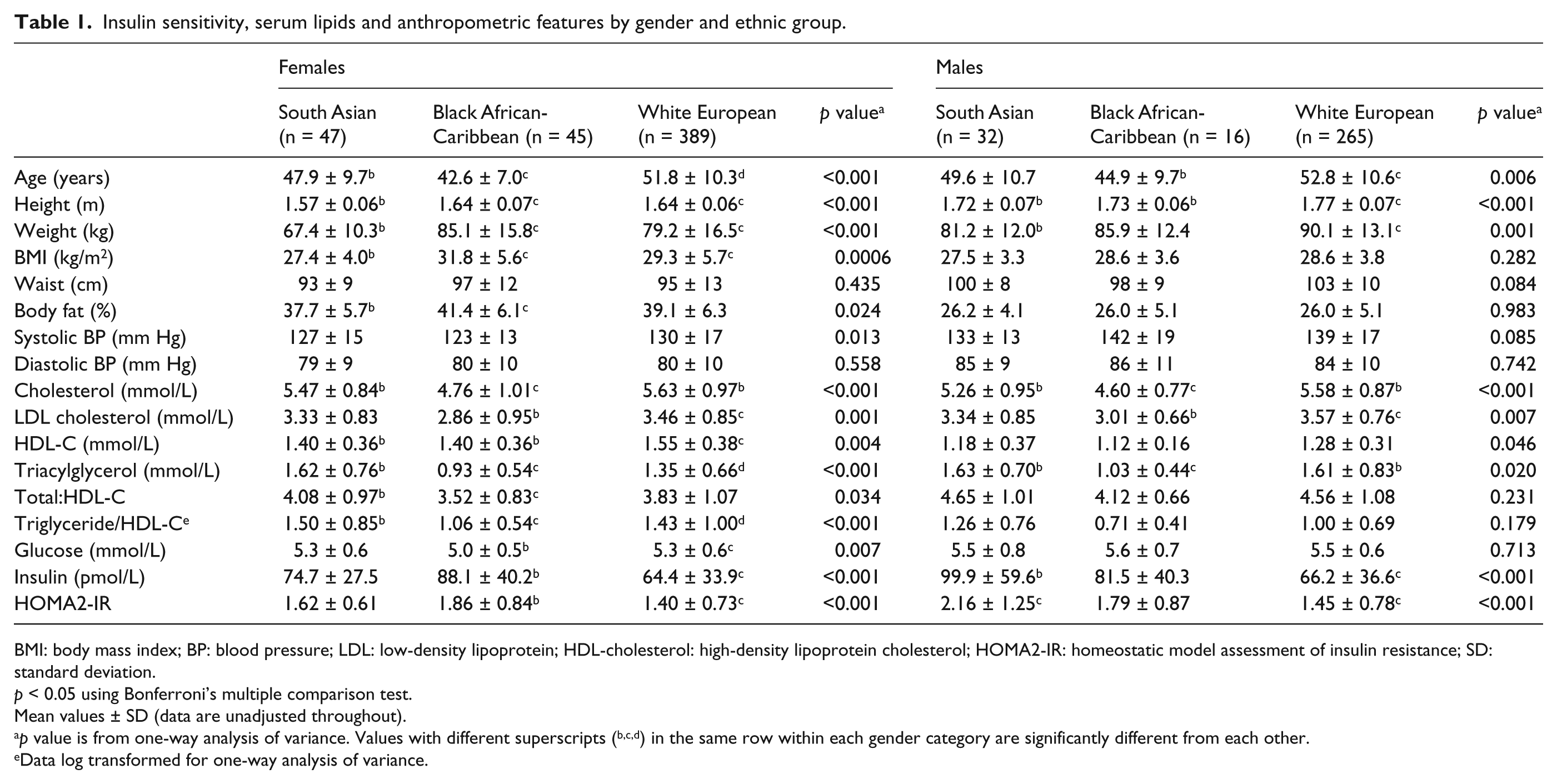
BMI: body mass index; BP: blood pressure; LDL: low-density lipoprotein; HDL-cholesterol: high-density lipoprotein cholesterol; HOMA2-IR: homeostatic model assessment of insulin resistance; SD: standard deviation.
p < 0.05 using Bonferroni’s multiple comparison test.
Mean values ± SD (data are unadjusted throughout).
p value is from one-way analysis of variance. Values with different superscripts (b,c,d) in the same row within each gender category are significantly different from each other.
Data log transformed for one-way analysis of variance.
Data from the IVGTT were available in a subgroup of 497 participants, the characteristics of which did not differ from the total cohort (data not shown). Adjusted (for age and BMI) mean values showed that the black African-Caribbean women had the lowest Si and a significantly higher AIR than the other ethnic groups (Table 2); furthermore, AIR was significantly higher in African-Caribbean females than males. When data for both genders were combined and adjusted for age, BMI and gender, both the South Asians and black African-Caribbeans expressed lower Si than the white participants. The South Asian and black African-Caribbean males also had lower Sg than the white participants, but there were no differences among the female participants.
Indices of insulin sensitivity and insulin secretion in response to glucose from the IVGTT by gender and ethnic group.
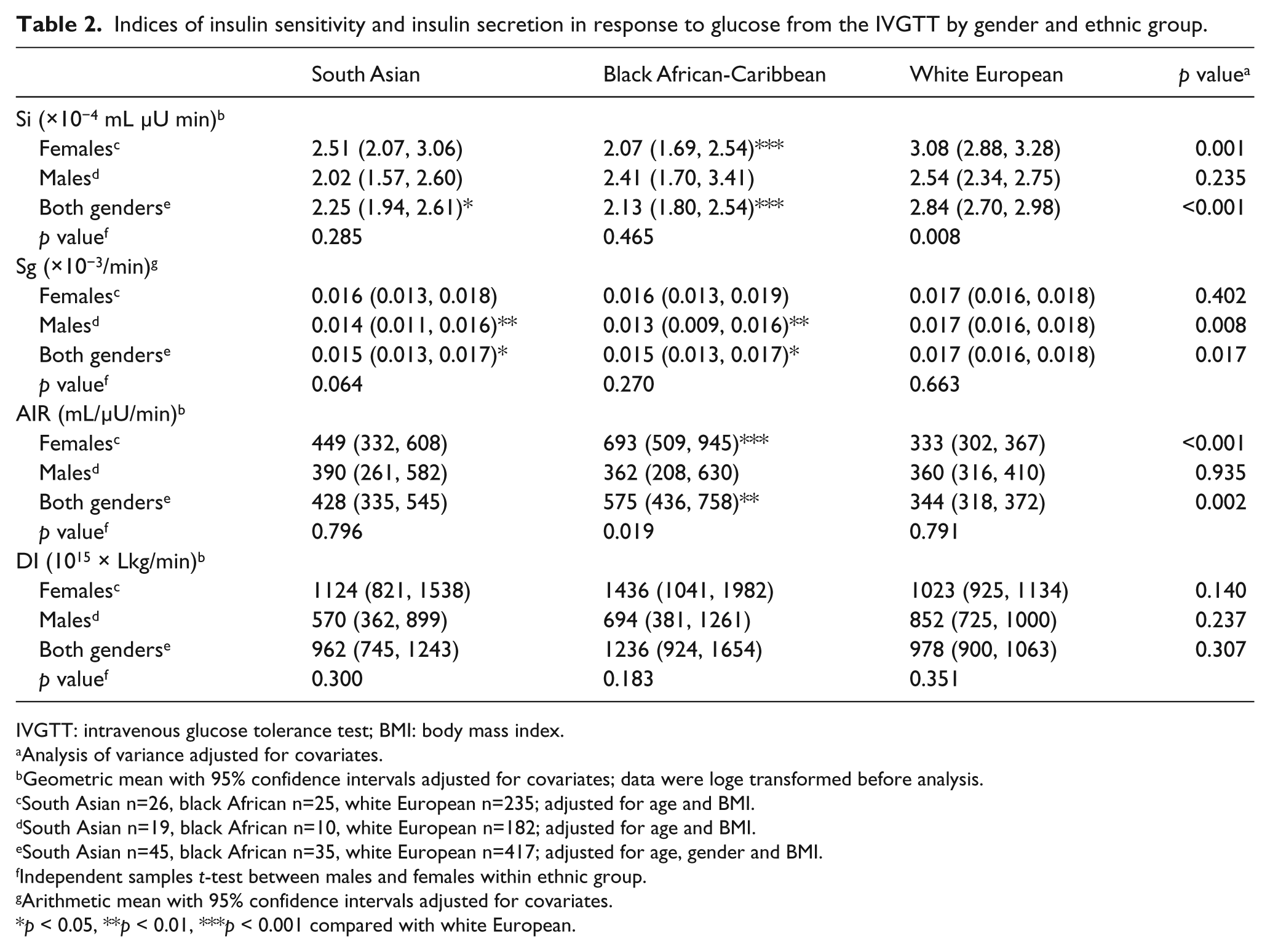
IVGTT: intravenous glucose tolerance test; BMI: body mass index.
Analysis of variance adjusted for covariates.
Geometric mean with 95% confidence intervals adjusted for covariates; data were loge transformed before analysis.
South Asian n=26, black African n=25, white European n=235; adjusted for age and BMI.
South Asian n=19, black African n=10, white European n=182; adjusted for age and BMI.
South Asian n=45, black African n=35, white European n=417; adjusted for age, gender and BMI.
Independent samples t-test between males and females within ethnic group.
Arithmetic mean with 95% confidence intervals adjusted for covariates.
p < 0.05, **p < 0.01, ***p < 0.001 compared with white European.
Dietary data were available for 503 participants (Table 3). The dietary analysis, adjusted for age and BMI, shows that South Asian and black African-Caribbean men and women had a lower proportion of energy from saturated fat [saturated fatty acid (SFA)] and higher proportion of energy from carbohydrate (CHO), specifically starch, and higher glycaemic loads compared to the white participants.
Habitual dietary intakes and sources of dietary energy in participants.
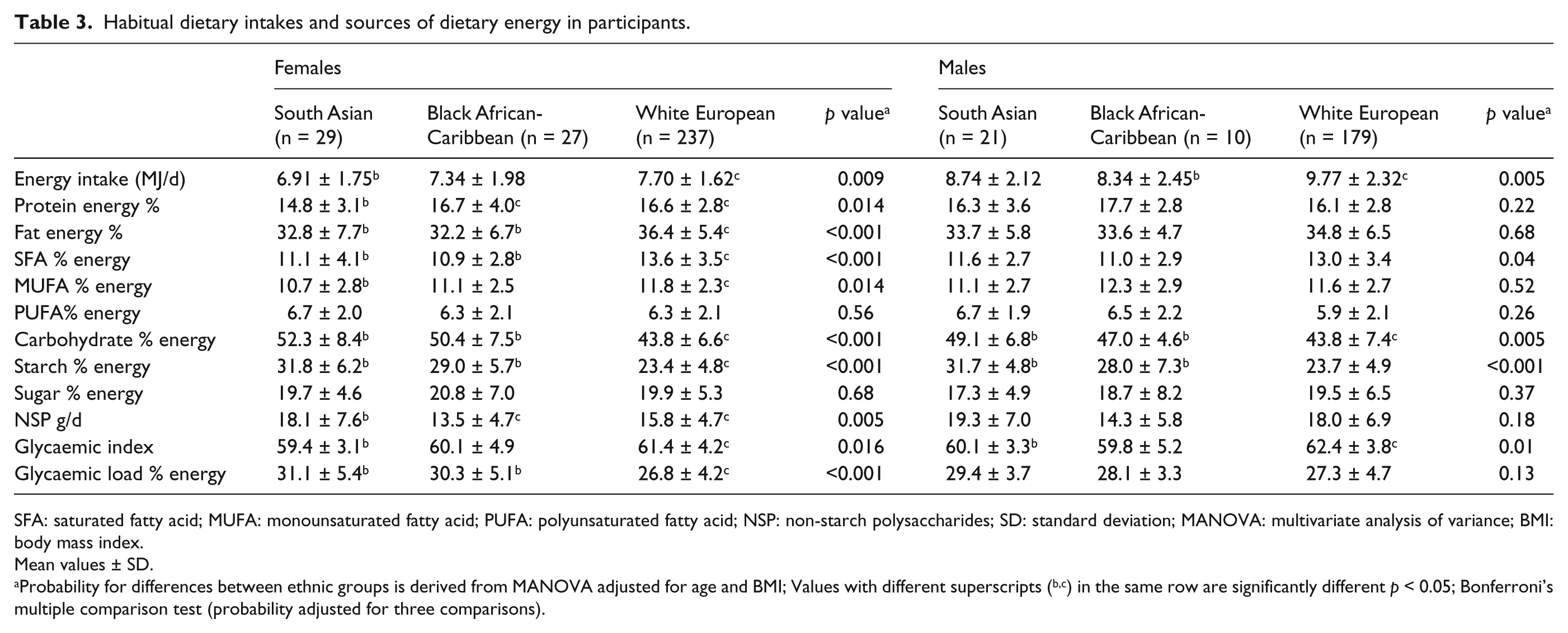
SFA: saturated fatty acid; MUFA: monounsaturated fatty acid; PUFA: polyunsaturated fatty acid; NSP: non-starch polysaccharides; SD: standard deviation; MANOVA: multivariate analysis of variance; BMI: body mass index.
Mean values ± SD.
Probability for differences between ethnic groups is derived from MANOVA adjusted for age and BMI; Values with different superscripts (b,c) in the same row are significantly different p < 0.05; Bonferroni’s multiple comparison test (probability adjusted for three comparisons).
Discussion
In accordance with the findings of previous studies, 9,29 this study presents evidence that South Asian and black African-Caribbean ethnicities at risk of MetS express lower Si than white Europeans. There was also a lack of expression of an atherogenic lipid profile (raised fasting serum triglycerides and low HDL-C), in black African-Caribbeans, in which the phenotype of insulin resistance was characterised by a marked alteration of beta-cell function, independently of age and BMI. The highest prevalence of atherogenic dyslipidaemia, defined by the combined occurrence of high triglycerides and low HDL-C, was in the South Asian females, which may be due to greater visceral adipose tissue accumulation, even though their BMIs were lower and their waist circumferences were not different to the other ethnic groups.
Previous studies in African-Americans have described a profound hyperinsulinaemic response (AIR) to an intravenous glucose challenge, 17,19 in both healthy and insulin-resistant populations, independent of adiposity. 30 The ability of pancreatic beta-cells to increase insulin secretion in response to insulin resistance can be assessed by the DI in the IVGTT. Hasson et al. 20 recently demonstrated reduced Si with a compensatory higher AIR and DI in African-American adolescents. Our data show similar responses to an intravenous glucose challenge in UK-based migrants who are likely to have had less exposure to a Westernised environment, due to more recent migration, than African-Americans. We also identified marked differences between our black African-Caribbean males and females, with female subjects showing a significantly higher AIR than the males, suggesting higher endogenous secretion of insulin. This may suggest that there is a different phenotype of insulin resistance between black African-Caribbean males and females; however, our study is small and insufficiently powered to conclude this and larger studies are needed. Our black African-Caribbean males had higher fasting glucose concentrations than the females, which, in combination with the AIR data, may provide evidence for beta-cell exhaustion and progression to IGT; however, they were more insulin sensitive than the females. These apparent gender differences are novel and warrant further investigation in a larger African-Caribbean population ideally with matching for glucose tolerance status.
Studies in African-Americans examining insulin resistance and the associated compensatory responses have recognised a reduction in the amount of insulin extracted by the liver in response to glucose stimulation. 17,19 It has been proposed that this provides an alternative compensatory response to insulin resistance that raises peripheral insulin levels without a need to increase insulin secretion and may conserve beta-cell function. 19
Our dietary data revealed a markedly lower intake of saturated fat and higher intake of CHO in South Asian and black African-Caribbean groups compared to white- European populations, an observation that is consistent with earlier findings. 31 –34 Both the South Asian and black African-Caribbeans within the present study consumed diets that were relatively high in CHO and starch, with higher glycaemic loads. Further study is needed to determine the impact of these dietary features on Si and beta-cell function. A lower intake of saturated fat would be expected to exert a cholesterol-lowering effect, 35 while a high intake of CHO would be expected to exacerbate the features of the MetS. 36 The higher dietary glycaemic load may drive an atherogenic metabolic profile as a result of postprandial portal hyperinsulinaemia, which may then promote the production of triglyceride-rich lipoproteins. However, in this situation, the similar intake of macronutrients, but dissimilar plasma lipid profiles between Asians and African-Caribbeans, supports significantly different hepatic insulin dynamics and impact of diet on metabolism in African-Caribbeans. Goree et al. 37 have recently investigated the impact of metabolic phenotype and ethnicity on the effects of diet on Si and beta-cell responsiveness and identified significant differences in the beta-cell response to diet between African-American and white participants, with a pronounced down-regulation of beta-cell responsiveness in the African-American participants. It is noteworthy that in the present study, black African-Caribbean females were characterised by distinct obesity and high proportions of body fat compared to males from that ethnic group and other ethnic groups that may have impacted on these comparisons. Further studies in larger populations will be required to establish whether high CHO diets exert greater adverse effects on Si and insulin secretion in the presence of obesity.
A limitation of this study was that the RISCK study cohort was selected on the basis of being at risk of MetS, 38,39 and may therefore not be representative of the wider population. Furthermore, IVGTT analysis was only available for 497 participants who completed the 6-month dietary intervention phase, giving relatively small group sizes for the different ethnic populations. Nevertheless, there were no significant differences between the eligible and completer groups, which provide evidence for the validity of our findings. In the African-Caribbean group, the female participants were characterised by markedly higher BMI than the male participants that may have impacted on our findings, although the IVGTT outcomes have been adjusted for BMI. An additional limitation was the lack of information on C-peptide that prevented our ability to dissociate insulin secretion from hepatic insulin extraction – an important phenomenon in African-Caribbean populations.
In conclusion, this study presents novel data to show that UK-dwelling black African-Caribbeans under-express the classic insulin-resistant phenotype by having less central obesity and an absence of dyslipidaemia. We present novel findings that suggest that insulin resistance in this group is coupled with markedly altered beta-cell function, and that these effects may be exaggerated in females.
Footnotes
Acknowledgements
Fiona Lewis, Samantha Bowen, L Chen, Robert Gray, Nuala Booth, Gary Moore, Roy Sherwood, Anthony Leeds, A Shah, G Saran, J Niehuser-Saran and JA Cockburn (Nutritional Sciences Division, King’s College London, London, UK); Margaret Griffin and Nicola Harman (Faculty of Health & Medical Sciences, University of Surrey, Guildford, UK); Christine Williams, Rachel Gitau, Katie Newens, Sean Lovegrove, Ana Rodriguez-Mateos and John Wright (Department of Food and Nutritional Sciences, University of Reading, Reading, UK); Carmel Moore, Mark Chatfield, Celia Walker, Hannah Farrant, Claire Lawrence, Edel Magee, Kit Tsoi, Darren Cole, Steve Austin, Hanneke Mfuni, Kate Guberg, Anna Gent, Celia Greenberg, Caroline Stokes, Mario Siervo and Rosemary Hall (MRC Human Nutrition Research, Cambridge, UK); Claire Howard, Namrata Dhopatkar, Bushra Siddiqui and Anne Dornhurst (Nutrition and Dietetic Research Group, Department of Investigative Medicine, Imperial College London, London, UK). Statistical advice was provided by Dr Peter Milligan of King’s College London, School of Medicine.
Authors’ contributions
Conceived, designed and conducted the original study: S.A.J., J.A.L., B.A.G., T.A.S. and G.S.F. Modelled IVGTT data: L.B. Initiated and conceived the current study: G.S.F. and L.M.G. Conducted the statistical analyses, interpretation of data and wrote the article: L.M.G. and G.S.F. Critical revision of the article for its intellectual content: all the authors.
Conflict of interest
The authors and their research groups have a number of links with the food industry. In a personal capacity, G.S.F. is a consultant to Coca-Cola, Premier Foods, and Unilever; T.A.S. has acted as a consultant to Seven Seas, is a member of the Scientific Advisory Committee for the Global Dairy Platform, the external scientific review committee of the Malaysian Palm Oil Board and chairs Cadbury’s Global Nutrition Advisory Panel. T.A.S., B.A.G., J.A.L., S.A.J. and G.S.F. have received ad hoc honoraria for lectures or writing articles. In a non-personal capacity, B.A.G. was formerly a member of an expert group known as the Fat Panel, which was supported by Dairy Crest, Kerry Gold and Unilever; S.A.J. is a member of Scientific Advisory Boards for Coca-Cola, Heinz, PepsiCo, Nestlé and Kellogg’s; S.A.J. and J.A.L. sit on government advisory boards that also include food industry members. All research groups received products from a range of food companies gratis for research purposes, including Archer Daniel Mills, Croda, Matthews Foods, Nestle, PepsiCo, Jordan, GSK and Unilever. L.M.G. and L.B. reported no conflicts of interest.
Funding
The RISCK study was funded by the Food Standards Agency (FSA) (N02031).
