Abstract
Incretin is a gut derived peptide hormone secreted in the intestine after food ingestion, and is degraded rapidly after secretion by dipeptidyl peptidase (DPP)-4. Incretin-based therapy, such as glucagon-like peptide (GLP)-1 and the DPP-4 inhibitor, has been proposed as a new therapeutic approach for the treatment of type 2 diabetic patients. In the past few years, growing evidence also demonstrated the cardioprotective effects of incretin-based therapy, especially during ischaemia-reperfusion (I/R) injury in both the animal models and in clinical studies. However, inconsistent reports exist regarding the use of these pharmacological interventions. In this article, a comprehensive review regarding both basic and clinical studies reporting the effects of GLP-1 and DPP-4 inhibitors on I/R hearts is presented and discussed. The consistent findings as well as controversial results are summarised, focusing on the effects of incretin on the infarct size, left ventricular function and haemodynamic improvement during an I/R injury.
Introduction
Diabetes mellitus (DM) has become a significant health problem in most nations with the number of patients dramatically soaring and expected to reach 366 million by the year 2030. 1 It has been shown that patients with type 2 diabetes mellitus (T2DM) have a two- to four-fold higher risk of coronary disease and stroke mortality.2–6 Although several new anti-diabetic drugs have been discovered in the past decades, the therapies have been limited by their adverse effects such as weight gain, hypoglycaemia, fluid retention and an unexpected cardiovascular risk.7–10 Therefore, new anti-diabetic drugs that could control hyperglycaemia and reduce the risk of cardiovascular events are of potential benefit to T2DM patients.
Incretin is a gut derived peptide hormone which enhances endogenous insulin secretion and reduces glucagon secretion, resulting in reduced blood glucose after food consumption.11–14 Its secretion is greatly influenced by ingestion. 15 The incretin hormone has been classified into glucose-dependent insulinotropic peptide (GIP) and glucagon-like peptide (GLP)-1. GIP is secreted by the enteroendocrine K cell of the proximal intestine, while GLP-1 is released from the enteroendocrine L cell of the distal intestine. GLP-1 is responsible for the majority of the incretin effect on pancreatic β-cell function. The secretion of GLP-1 is lower in patients with T2DM than normal, suggesting that this hormone contributes to the pathogenesis of the disease. 16
The circulating GLP-1 has two isotypes: GLP-1 (7-36) and GLP-1 (7-37), in which GLP-1 (7-36) is responsible for 80% of active GLP-1. 17 GLP-1 has insulinotropic, insulinomimetic and glucagonostatic effects. 18 GLP-1 binds to GLP-1 receptors (GLP-1Rs) leading to regulatory actions which are an enhancement of β-cell function and proliferation, enhancement of glucose-dependent insulin secretion from β-cell, activation of insulin biosynthesis, suppression of elevated glucagon secretion, suppression of food intake and slowing of gastric emptying.11,19 However, the therapeutic drawback is that the circulating GLP-1 level decreases rapidly, i.e. less than two minutes, after secretion due to being degraded by dipeptidyl peptidase (DPP)-4 enzymes and renal clearance.20–23 DPP-4 enzyme degrades GLP-1 (7-36) by removing an N-terminal dipeptide, resulting in its metabolite, GLP-1 (9-36), which has 1000-fold lower affinity to GLP-1Rs. 21 It has been shown that the inhibition of DPP-4 could enhance the level of intact GLP-1 and prolong its action time. 24 Thus, two classes of drugs have been recently used for incretin enhancement in T2DM, including GLP-1 analogues and DPP-4 inhibitors. While GLP-1 analogues (i.e. exenatide, liraglutide and albiglutide) increase the GLP-1 level to a supraphysiological level, DPP-4 inhibitors (i.e. vildagliptin, sitagliptin and saxagliptin) conserve and prolong intact GLP-1 availability within a physiological level.
Although the primary physiological function of GLP-1 is related to the control of plasma glucose, GLP-1Rs have been ubiquitously found in a variety of extra-pancreatic tissues including the central and peripheral nervous system, kidney, lung, gastrointestinal tract, blood vessel and heart both in rodents and humans.13,25 Since GLP-1Rs in the heart are similar to in the pancreas, 25 it has been suggested that drugs targeting incretin enhancement may potentially affect the heart.
DM has been shown to increase cardiovascular risk including increased incidence of myocardial infarction. 2 With the undesirable effects of several anti-diabetic drugs such as those in the thiazolidinedione group, i.e. rosiglitazone, that increased the cardiovascular morbidity and mortality,8,9 it is essential that any new anti-diabetic drug be investigated for both beneficial and harmful effects on the cardiovascular system. In this review, reports from basic and clinical studies regarding the effects of GLP-1 and DPP-4 inhibitor on cardiac function and infarct size in an ischaemic-reperfused heart are comprehensively summarised. Key results are critically discussed with emphasis placed on consistent findings. Inconsistent results are also highlighted to more fully explore what is known and what remains to be discovered.
Effects of GLP-1 on the infarct size
GLP-1 has been shown to have an infarct limiting effect in both in vitro and in vivo models of ischaemia-reperfusion (I/R) injury (Table 1). GLP-1 co-administered with DPP-4 inhibitor, valine pyrrolide (VP), could reduce the infarct size ranging from 39% to 58% in isolated Langendorff rat hearts whether given prior to ischaemia or during the reperfusion period.26–29 However, the administration of GLP-1 28 or DPP-4 inhibitor26–28 alone failed to reduce the infarct size. These studies suggested the synergistic effect of DPP-4 inhibition and exogenous GLP-1 on the infarct limiting effect in which DDP-4 inhibitor enhanced endogenous GLP-1 level and reduced exogenous GLP-1 degradation.
Summary of the beneficial effects of GLP-1 on the infarct size.
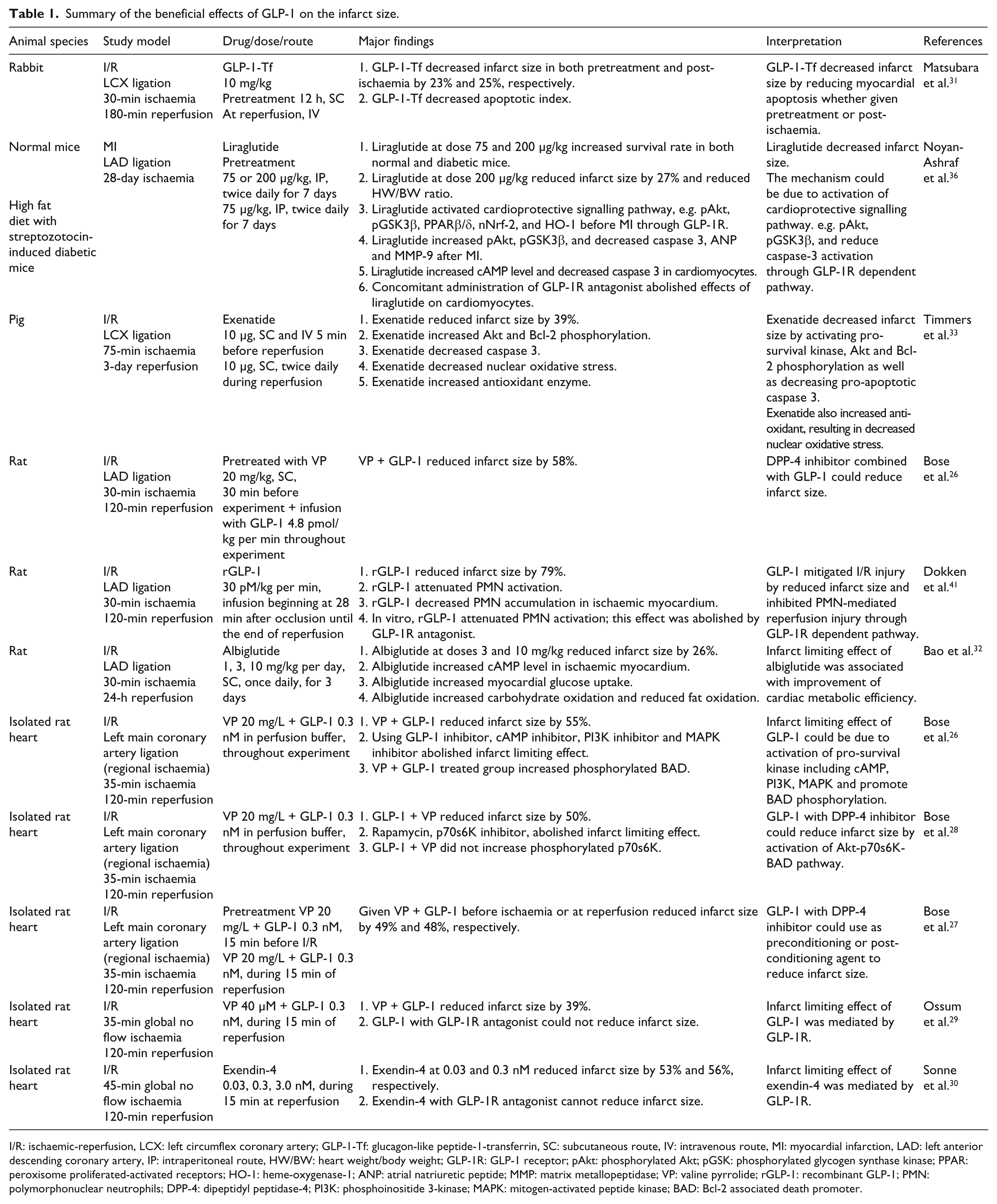
I/R: ischaemic-reperfusion, LCX: left circumflex coronary artery; GLP-1-Tf: glucagon-like peptide-1-transferrin, SC: subcutaneous route, IV: intravenous route, MI: myocardial infarction, LAD: left anterior descending coronary artery, IP: intraperitoneal route, HW/BW: heart weight/body weight; GLP-1R: GLP-1 receptor; pAkt: phosphorylated Akt; pGSK: phosphorylated glycogen synthase kinase; PPAR: peroxisome proliferated-activated receptors; HO-1: heme-oxygenase-1; ANP: atrial natriuretic peptide; MMP: matrix metallopeptidase; VP: valine pyrrolide; rGLP-1: recombinant GLP-1; PMN: polymorphonuclear neutrophils; DPP-4: dipeptidyl peptidase-4; PI3K: phosphoinositide 3-kinase; MAPK: mitogen-activated peptide kinase; BAD: Bcl-2 associated death promoter.
Another line of drug that has been used to enhance an incretin effect is a DPP-4 resistant GLP-1R agonist. Exendin-4, a hormone found in the saliva of the Gila monster, at 0.03 and 0.3 nM has been shown to reduce the infarct size, while 3.0 nM exendin-4 could not show this benefit, suggesting a biphasic infarct limiting effect. 30 Human transferrin (Tf) was also demonstrated to prolong the GLP-1 action. Administration of GLP-1-Tf could effectively reduce the infarct size in rabbits, given either before or after coronary occlusion. 31 Recently, albiglutide, another GLP-1 analogue, has been shown to increase cAMP in an ischaemic myocardium and improve cardiac metabolic efficiency by increasing glucose metabolism and reducing fat oxidation, which results in 26% reduction of infarct size. 32
In a pig model, exenatide has been shown to reduce the infarct size, 33 whereas the other two studies with a shorter period of I/R using recombinant GLP-1 (rGLP-1) 34 or liraglutide 35 could not demonstrate any improvement in infarct size (Table 2). This discrepancy could be due to a different duration and site of occlusion, therapeutic drugs and drug concentrations. This hypothesis is supported by a report by Noyan-Ashraf and colleagues, demonstrating that the optimal time and dose of liraglutide, GLP-1 analogue, played an important role in infarct size reduction, improved survival rate and contractile function in both normoglycaemic and diabetic mice. 36
Summary of the neutral effects of GLP-1 on the infarct size.
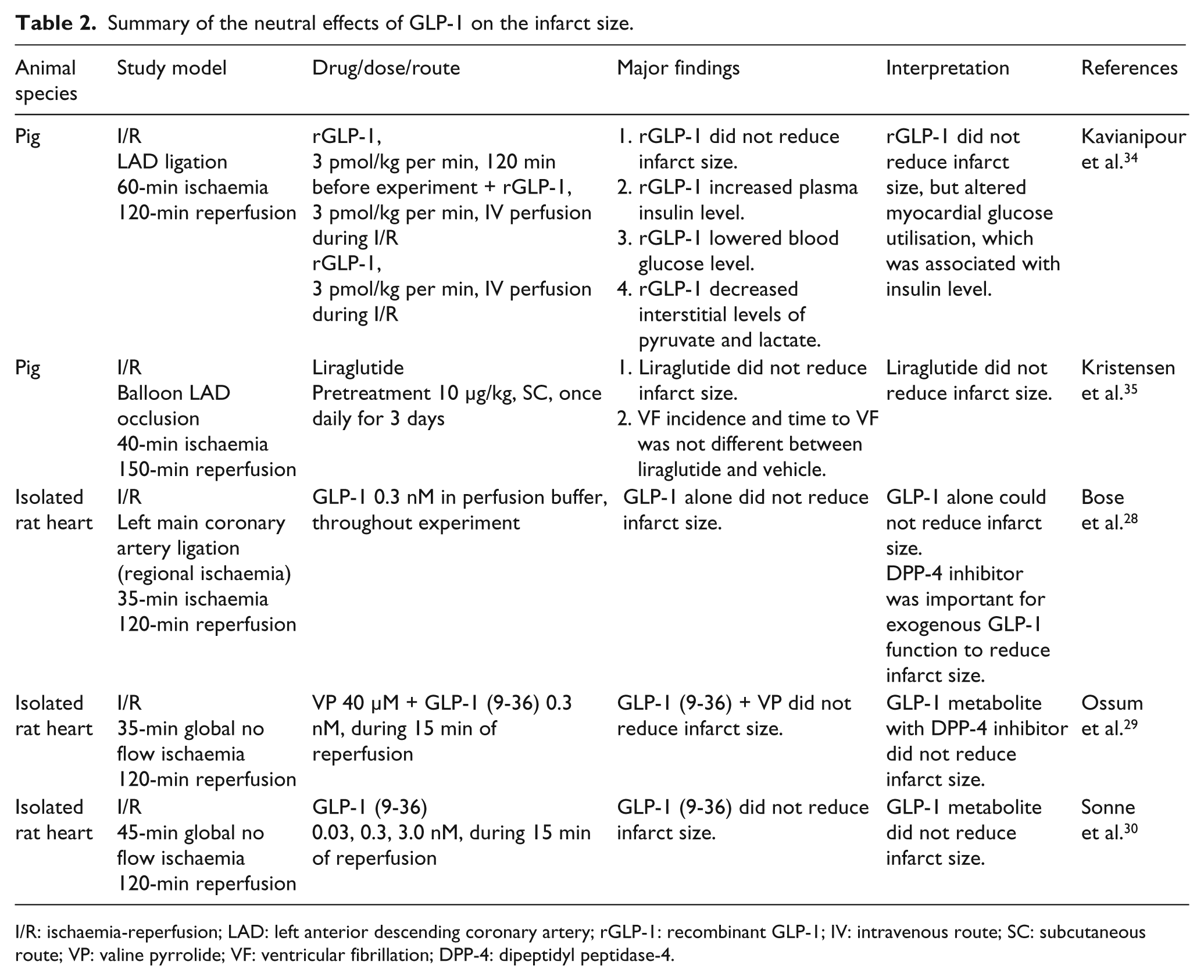
I/R: ischaemia-reperfusion; LAD: left anterior descending coronary artery; rGLP-1: recombinant GLP-1; IV: intravenous route; SC: subcutaneous route; VP: valine pyrrolide; VF: ventricular fibrillation; DPP-4: dipeptidyl peptidase-4.
Several mechanisms underlying the infarct limiting effect of GLP-1 have been proposed to be independent of weight loss. 36 First, GLP-1 has been shown to activate the cAMP-PKA pathway,26,32,36 the pro-survival kinase associated with reperfusion injury signalling kinase (RISK) pathway, 37 including the following: PI3K, Akt, MAPK, PPARβ/δ, Nrf-2, HO-1 and Akt-p70s6K-BAD pathways.18,26,27,33,36,38 Second, GLP-1 has been shown to reduce oxidative stress and increase antioxidants, leading to decreased apoptosis.26,31,33,36 Third, since proinflammatory cells such as neutrophils (PMNs) play an important role during the time of blood return to the heart,39,40 GLP-1 has been shown to attenuate the PMN activation and accumulation in the myocardium, thus reducing injuries caused by reperfusion. 41 Recent studies demonstrated that the infarct limiting effects were diminished with the administration of either GLP-1 metabolite, i.e. GLP-1 (9-36),29,30 or when giving GLP-1 with the GLP-1R antagonist, namely GLP-1 (9-39).26,29,30,36 Moreover, the administration of liraglutide failed to activate cardioprotective kinase in the GLP-1R depleted mice. Taken together, the effect of GLP-1 on the infarct size reduction is proposed to be a GLP-1R dependent mechanism.
Effects of GLP-1 on left ventricular function
Besides its infarct limiting effect, GLP-1 has been shown to improve left ventricular (LV) recovery from ischaemic myocardial stunning after reperfusion (Table 3). The administration of GLP-1 as a post-conditioning agent, which demonstrated a higher plasma GLP-1 level, could improve LV performance by decreasing the wall motion abnormality, and increasing the ejection fraction and LV developed pressure,29,31 compared with giving GLP-1 as a pre-conditioning agent, which has a lower plasma GLP-1 level. 31 Nevertheless, it is possible that the time for which GLP-1 was given as a preconditioning treatment in those studies was too short, thus allowing insufficient time for its action. This is supported by a report by Noyan-Ashraf and colleagues which demonstrated that the GLP-1 analogue required the optimal pre-treatment time and dose for the activation of the cardiac gene and protein to promote LV enhancement. 36 Other possible factors including routes and times of drug administration during I/R could also play an important role in this finding. Interestingly, like the infarct limiting effect, exendin-4 also improved the mechanical function in a biphasic manner. 30 In a clinically relevant swine model, exenatide improved regional and global systolic function. 33 In coronary artery disease patients with good LV function, a continuous infusion of rGLP-1 for 30 minutes before dobutamine stress echocardiography demonstrated an improved ejection fraction and LV regional wall function as well as mitigated post-ischaemic stunning, predominantly in the ischaemic wall segment. 42 The beneficial effect of a 72-hour GLP-1 infusion on the post- infarction recovery, including an increased ejection fraction and improvement of both global and regional wall motion was also observed in acute myocardial infarction patients with LV ejection fraction of less than 40% after successful primary percutaneous coronary intervention. 43 Long-term GLP-1 infusion also improved myocardial stunning, which was characterised by enhanced LV wall motion and relaxation in I/R dogs. 44
Summary of the effects of GLP-1 on left ventricular function.
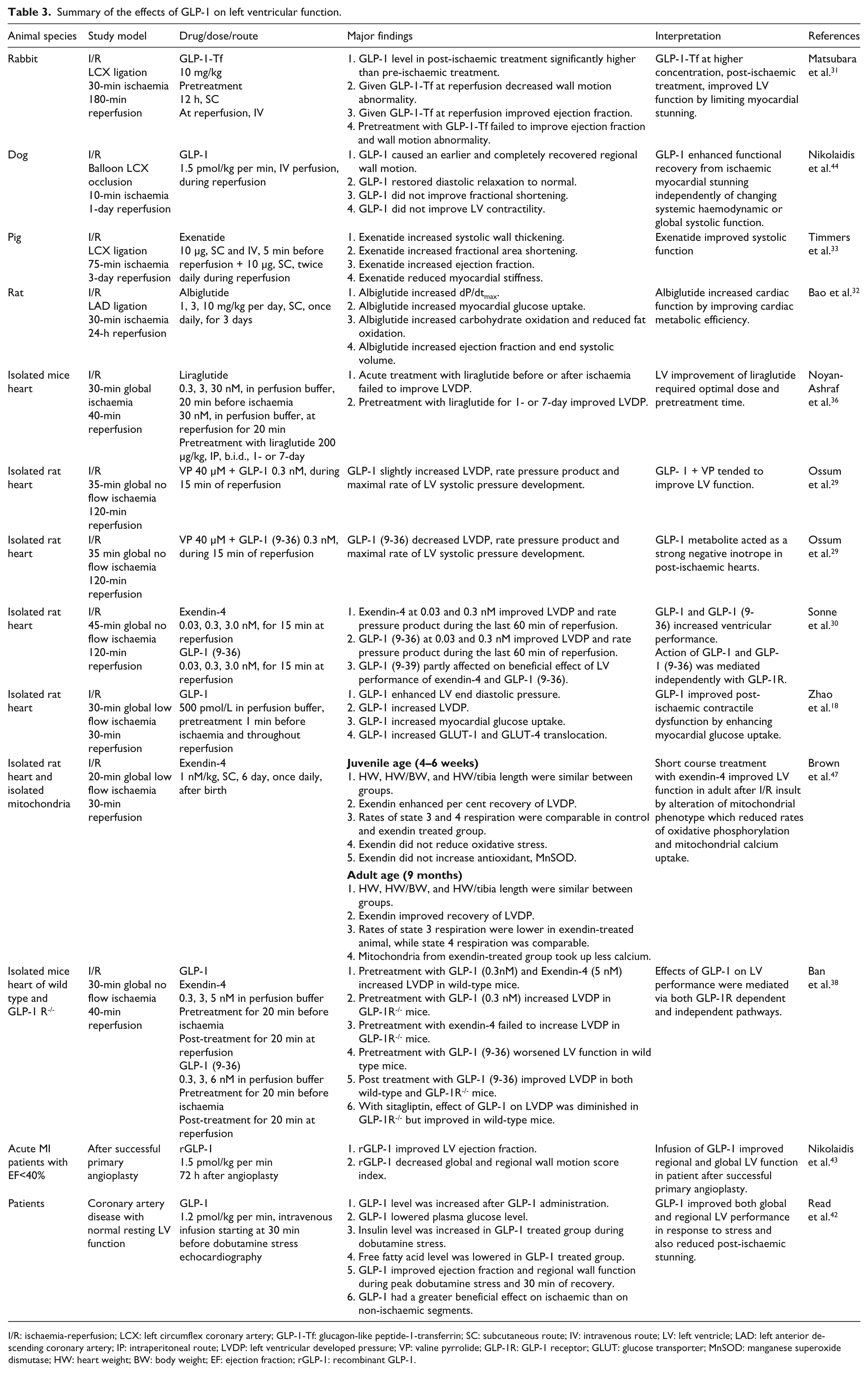
I/R: ischaemia-reperfusion; LCX: left circumflex coronary artery; GLP-1-Tf: glucagon-like peptide-1-transferrin; SC: subcutaneous route; IV: intravenous route; LV: left ventricle; LAD: left anterior descending coronary artery; IP: intraperitoneal route; LVDP: left ventricular developed pressure; VP: valine pyrrolide; GLP-1R: GLP-1 receptor; GLUT: glucose transporter; MnSOD: manganese superoxide dismutase; HW: heart weight; BW: body weight; EF: ejection fraction; rGLP-1: recombinant GLP-1.
Surprisingly, the benefit of exenatide and GLP-1 on LV function was also observed in the presence of GLP-1R antagonist and in GLP-1R deleted mice, suggesting that the improved LV function of GLP-1 may be mediated independent of GLP-1R.30,38 However, a recent study demonstrated that GLP-1 co-administered with sitagliptin, DPP-4 inhibitor, could not improve contractile functional recovery in GLP-1R deleted mice, whereas this treatment provided the benefit in wild-type mice. 38 Taken together, unlike the infarct limiting effect, the LV function amelioration of GLP-1 was mediated by both GLP-1R dependent and independent pathways. Although the administration of GLP-1 (9-36) could not reduce the infarct size,29,30 it exerts a functional recovery benefit which might mediate through the GLP-1 (9-36)-cGMP-NO pathway, independent of GLP-1R activation.30,38 However, the inotropic effect of GLP-1 (9-36) is still questionable. 29
Enhanced myocardial glucose uptake by up-regulation of glucose transporter (GLUT)-1 and GLUT-4 may be one of the underlying mechanisms to explain the beneficial effect of GLP-1 on LV functional improvement.18,32 In a normal physiological condition, the heart mainly uses fatty acid as a fuel in maintaining its function. Reduced availability of oxygen during low-flow ischaemia reduces the heart’s ability to produce energy from fatty acid oxidation and carbohydrates. The enhancement of glycolysis through diverse mechanisms or pharmacologic interventions can delay and prevent ischaemic damage and improve the recovery of the contractile function.45,46 GLP-1 has been shown to increase myocardial glucose uptake, which ameliorates the post-ischaemia myocardial dysfunction.18,32 This action of GLP-1 might be used as a therapeutic target for high risk cardiovascular disease patients, especially in insulin resistant T2DM. The long-term effect of GLP-1 analogue has been demonstrated in neonatal rats receiving exenatide once daily for six days. 47 This persistent beneficial effect is mediated by an altered mitochondrial phenotype which decreased the cardiac mitochondria calcium uptake and reduced oxidative phosphorylation, resulting in improved functional recovery after ischaemia reperfusion injury. Surprisingly, the cardioprotective effect of GLP-1 and GLP-1 analogue was not associated with any improvement in haemodynamic parameters (Table 4).26,31–35,44,47
Summary of the effects of GLP-1 on haemodynamic parameters.
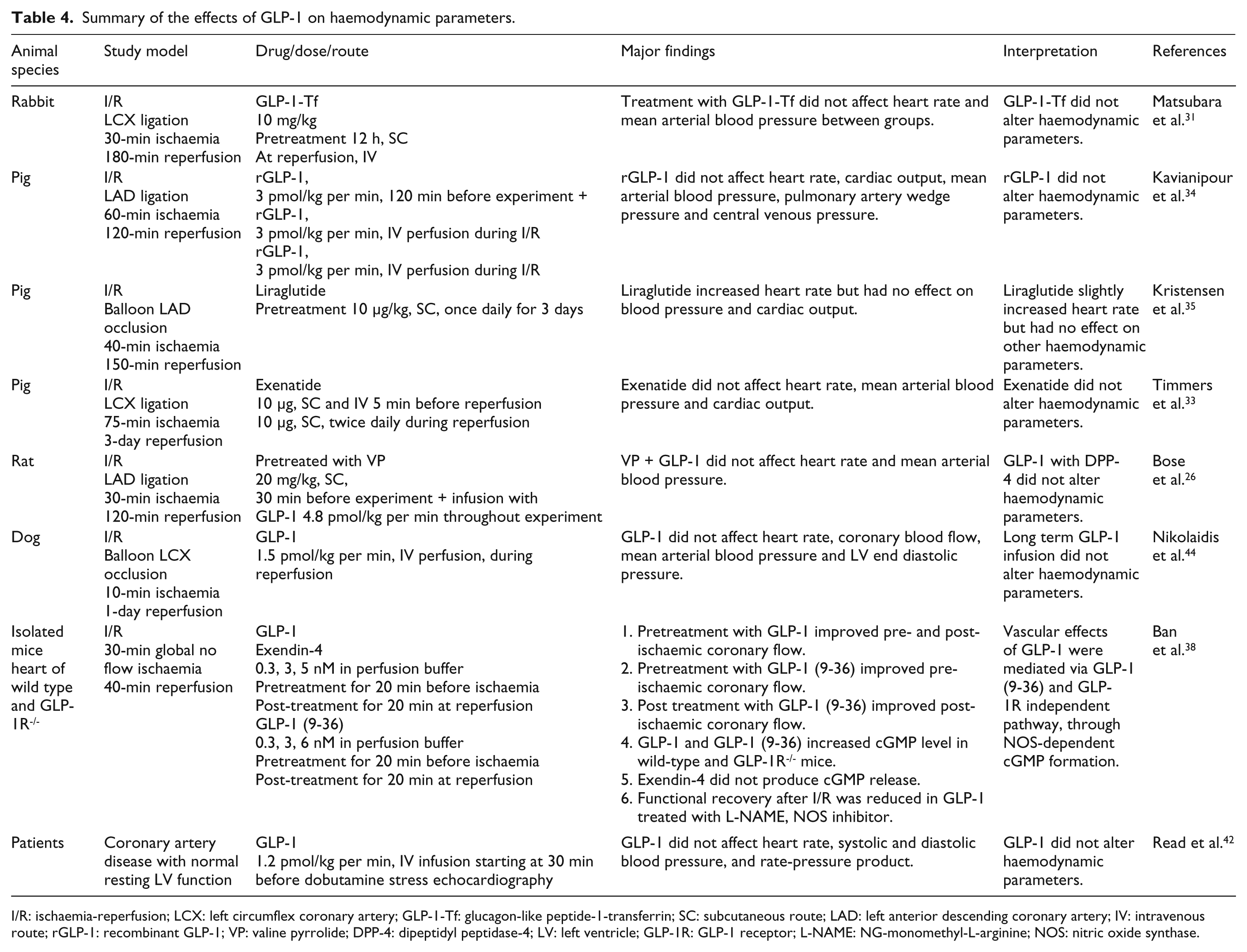
I/R: ischaemia-reperfusion; LCX: left circumflex coronary artery; GLP-1-Tf: glucagon-like peptide-1-transferrin; SC: subcutaneous route; LAD: left anterior descending coronary artery; IV: intravenous route; rGLP-1: recombinant GLP-1; VP: valine pyrrolide; DPP-4: dipeptidyl peptidase-4; LV: left ventricle; GLP-1R: GLP-1 receptor; L-NAME: NG-monomethyl-L-arginine; NOS: nitric oxide synthase.
Unlike GLP-1, information regarding the effects of DPP-4 inhibitor on the I/R heart is scarce and controversial.48–50 DPP-4 inhibitors inhibit the enzyme activity of DPP-4, resulting in decreasing degradation rate, thereby maintaining higher GLP-1 levels.24,51,52 Although growing evidence demonstrated the infarct limiting effect of DPP-4 inhibitors (Table 5), many studies suggested that the physiological level of GLP-1 may not be sufficient to reduce the infarct size (Table 6). Besides the infarct limiting effect, the beneficial effect of DPP-4 inhibitors on LV function after I/R injury has also been documented (Table 7).
Summary of the beneficial effects of DPP-4 inhibitor on infarct size.
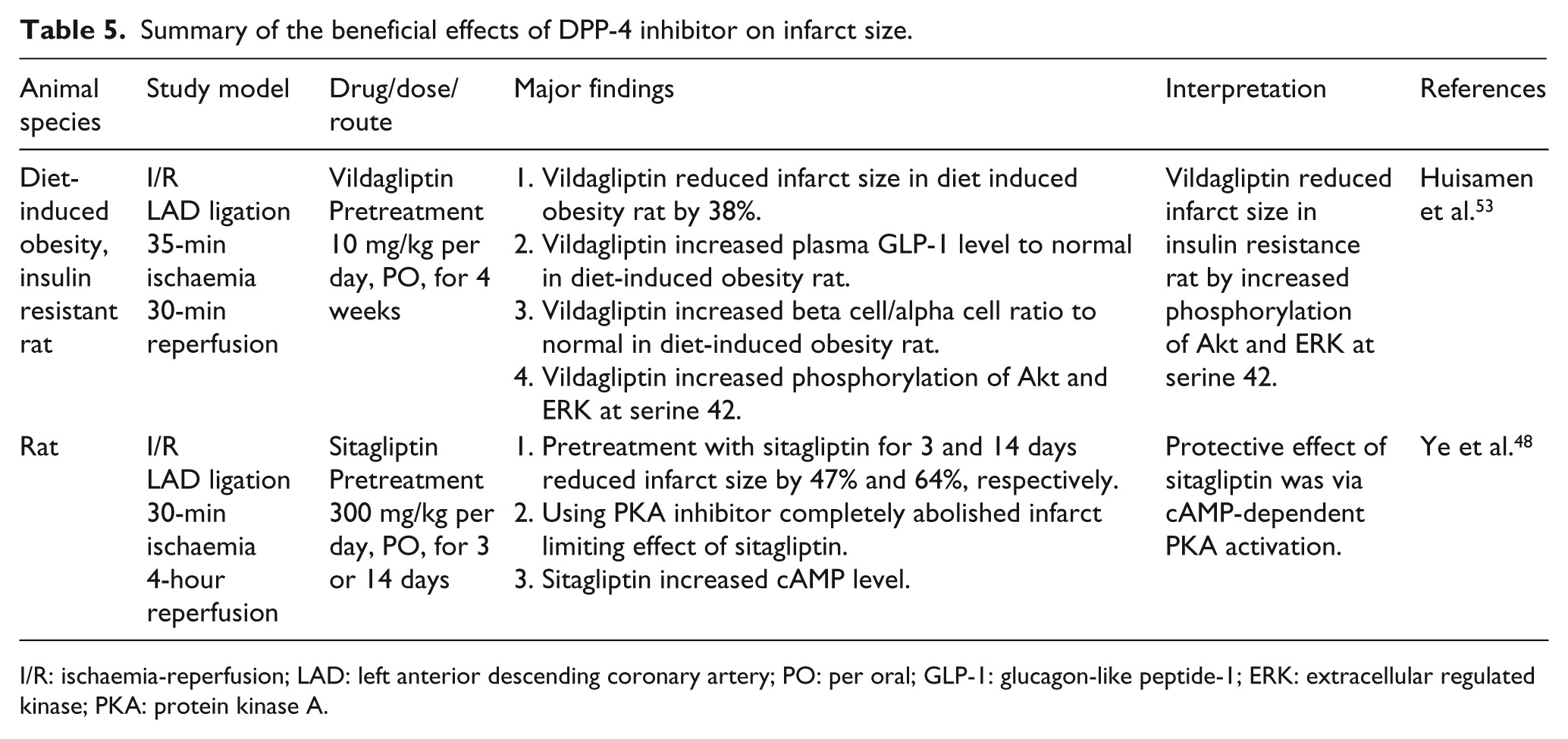
I/R: ischaemia-reperfusion; LAD: left anterior descending coronary artery; PO: per oral; GLP-1: glucagon-like peptide-1; ERK: extracellular regulated kinase; PKA: protein kinase A.
Summary of the neutral effects of DPP-4 inhibitor on infarct size.
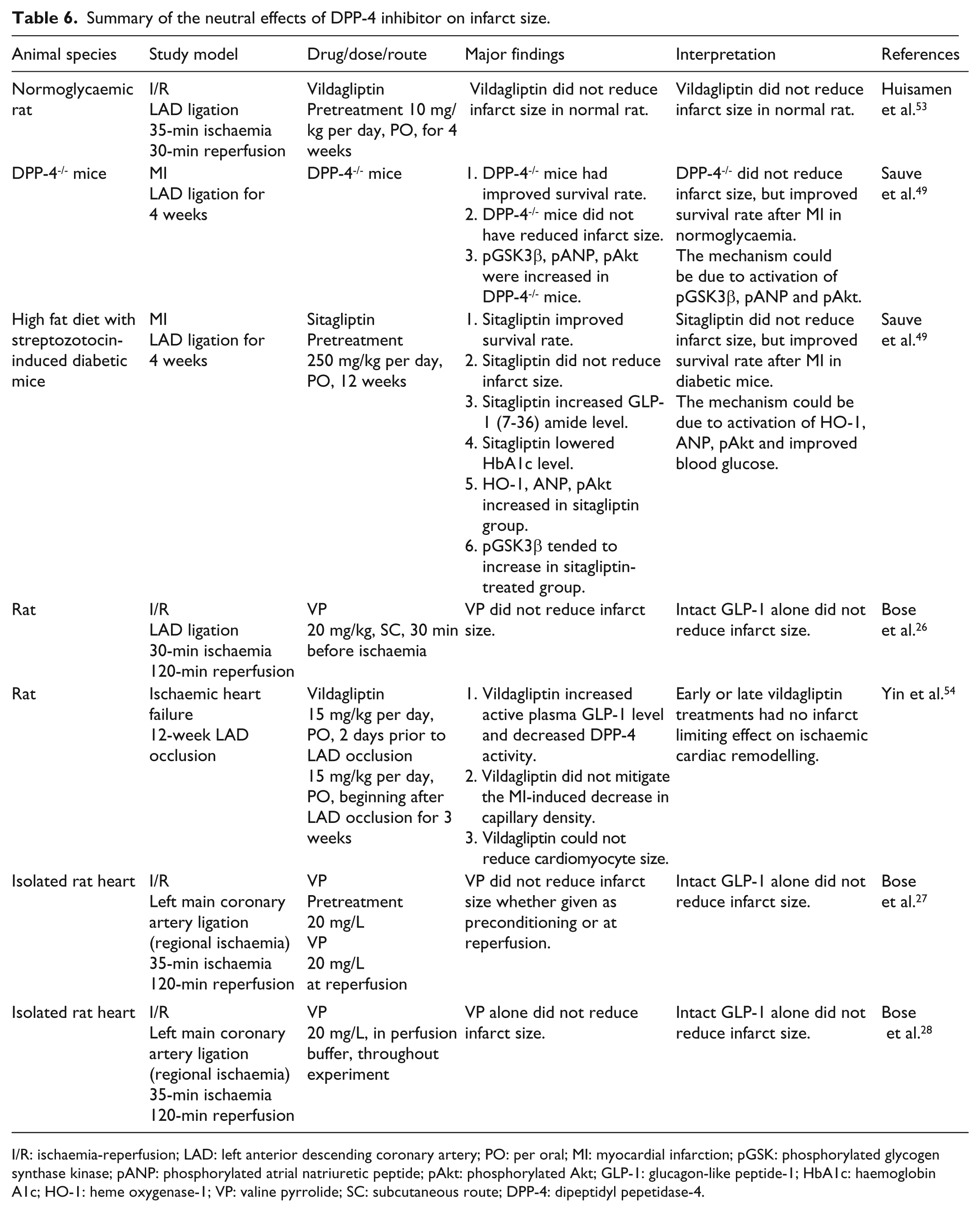
I/R: ischaemia-reperfusion; LAD: left anterior descending coronary artery; PO: per oral; MI: myocardial infarction; pGSK: phosphorylated glycogen synthase kinase; pANP: phosphorylated atrial natriuretic peptide; pAkt: phosphorylated Akt; GLP-1: glucagon-like peptide-1; HbA1c: haemoglobin A1c; HO-1: heme oxygenase-1; VP: valine pyrrolide; SC: subcutaneous route; DPP-4: dipeptidyl pepetidase-4.
Summary of the effects of DPP-4 inhibitor on left ventricular function.
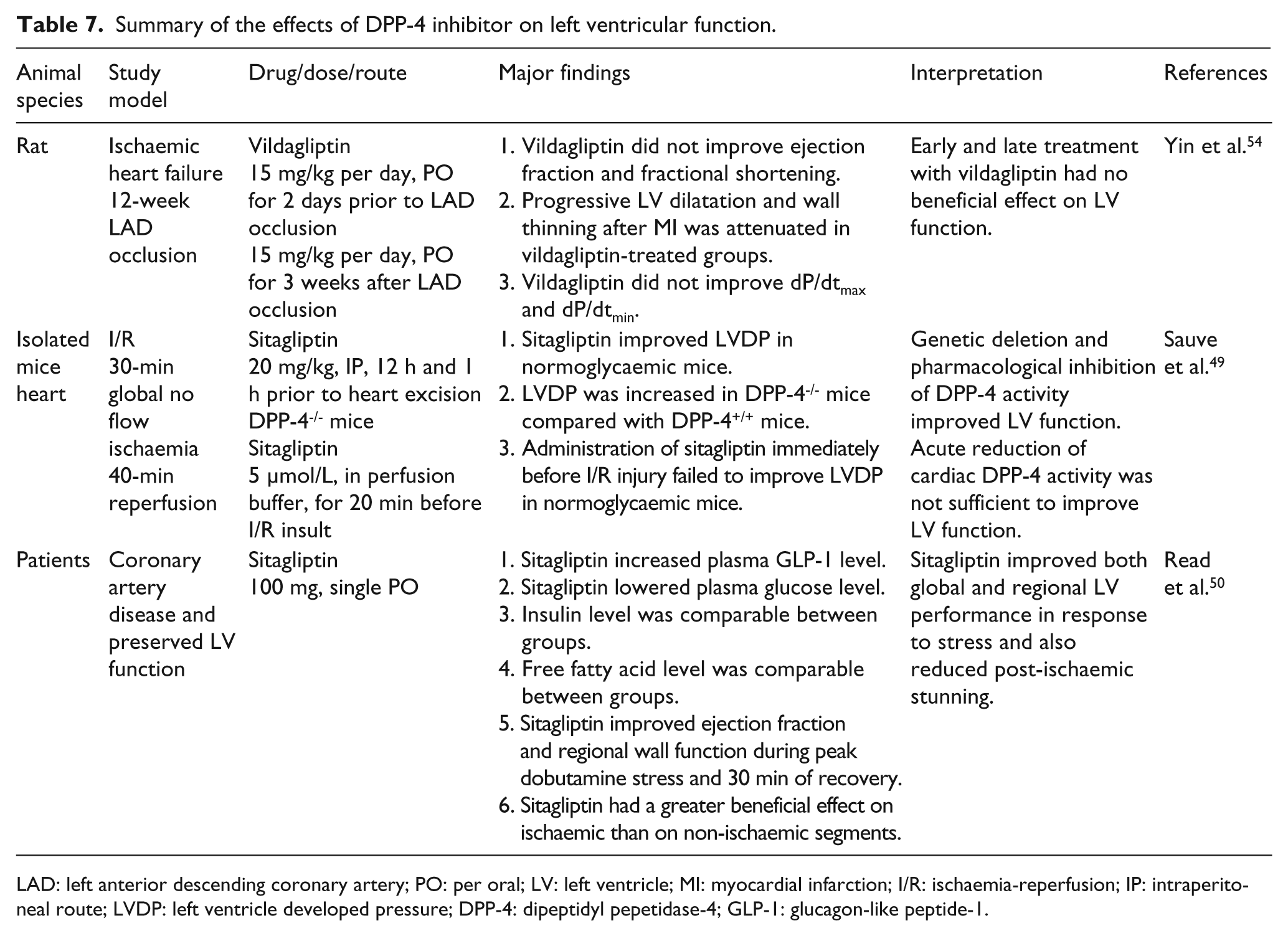
LAD: left anterior descending coronary artery; PO: per oral; LV: left ventricle; MI: myocardial infarction; I/R: ischaemia-reperfusion; IP: intraperitoneal route; LVDP: left ventricle developed pressure; DPP-4: dipeptidyl pepetidase-4; GLP-1: glucagon-like peptide-1.
Effects of DPP-4 inhibitor on the infarct size
As DM increases cardiovascular risk including myocardial infarction, pretreatment with vildagliptin, DPP-4 inhibitor, has been shown to attenuate the infarct size in insulin resistant rats. 53 This cardioprotective effect was also observed in normoglycaemic rats with pretreatment of sitagliptin. 48 A longer period of pre-treatment with sitagliptin resulted in smaller infarct size, suggesting that the plasma drug level might influence its cardioprotective effect. The infarct limiting mechanism of DPP-4 inhibitor in these studies was proposed to be due to the activation of Akt and cAMP-PKA pro-survival kinase in RISK pathway as observed in GLP-1 administration.48,53 However, some studies demonstrated that prolonged availability of intact GLP-1 caused by a DPP-4 inhibitor failed to demonstrate the infarct limiting effect.26–28,54 Moreover, inhibition or reduction of DPP-4 activity using DPP-4 deleted mice or pretreatment with sitagliptin before ischaemia in high fat diet-induced diabetic mice could not reduce the infarct size, but improved the survival rate by activation of cardioprotection kinase such as GSK3β, ANP and Akt. 49 These discrepant results suggest that the different animal models, the duration of ischaemia in the heart models and the minimal dose for a specific time may play a pivotal role in the reduction of the infarct size. Tables 5 and 6 summarise significant effects of DPP-4 inhibitors on the infarct size.
Effects of DPP-4 inhibitor on left ventricular function
DPP-4 inhibitors also exert a cardioprotective effect by the improvement of post-ischaemia myocardial stunning. Table 7 summarises the effects of DPP-4 inhibitors on LV function. In DPP-4 deleted mice and mice pretreated with sitagliptin for 12 hours prior to aortic occlusion, improved LV developed pressure after I/R injury was observed. 49 However, acute treatment with sitagliptin for 20 minutes prior to I/R insult failed to improve ventricular performance. 49 The discrepancy in these findings could be due to the differences in the time of drug administration as well as study models (in vivo versus ex vivo), and might suggest that the benefit of DPP-4 inhibitor required an optimal time and an intact cardiovascular system process. However, a recent report in rats with ischaemic heart failure demonstrated that early or late treatment with vildagliptin had no beneficial effect on ventricular performance. 54 In a clinical study, single dose treatment with sitagliptin improved regional and global wall function, which improved post-ischaemic stunning in patients with coronary artery disease awaiting revascularisation. 50 These improvements are independent of insulin 50 or haemodynamic parameter alterations.49,50,53 Table 8 summarises the effect of DPP-4 inhibitors on haemodynamic parameters.
Summary of the effects of DPP-4 inhibitor on haemodynamic parameters.
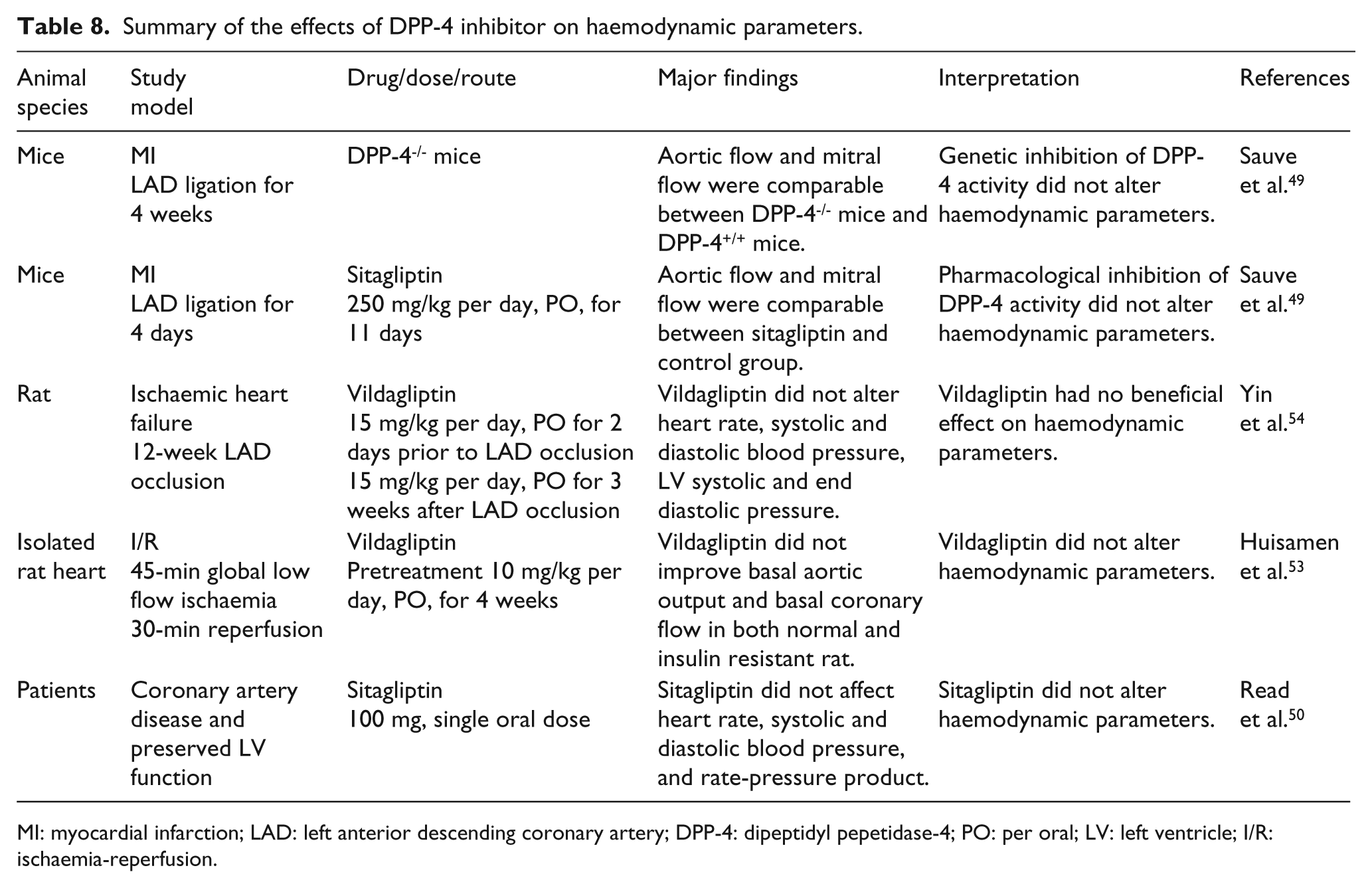
MI: myocardial infarction; LAD: left anterior descending coronary artery; DPP-4: dipeptidyl pepetidase-4; PO: per oral; LV: left ventricle; I/R: ischaemia-reperfusion.
Conclusion
Growing evidence suggests that incretin exerts a cardioprotective effect during I/R injury. The infarct limiting effect is mediated through GLP-1R, while LV function improvement is mediated through both GLP-1R dependent and independent (via GLP-1 (9-39)) pathways. The underlying mechanisms were due to the activation of prosurvival kinase, increased antioxidant and myocardium glucose utilisation, reduction of oxidative stress and pro-apoptotic kinase, and attenuation proinflammatory cell activation and accumulation in the myocardium. Although many mechanisms have been proposed, the exact mechanisms have never been fully elucidated. Therefore, further investigations are required to determine the cardioprotective mechanisms of incretin. Ultimately, large clinical trials are the essential step to validate those effects reported in animal studies and to warrant their clinical use in the I/R condition.
Footnotes
Funding
This work was supported by the Thailand Research Fund Royal Golden Jubilee PhD Project (KC and NC) and the Thailand Research Fund (grants nos RTA5280007 (NC) and BRG5480003 (SC)).
Conflicts of interest
No conflicts of interest have been declared.
