Abstract
This was a nested sub-study of a randomised placebo-controlled trial of the effect of 6 months of treatment with rosiglitazone added to existing therapy on myocardial triglyceride (mTG) content in patients with type 2 diabetes (T2D) and prevalent cardiovascular disease (CVD) or at least one additional risk factor. The primary endpoint, mTG content, was measured with cardiac 1H-magnetic resonance spectroscopy. Of the 99 randomised participants selected for the imaging sub-study, 49 (48%) had complete and interpretable spectroscopy data (age = 58 years, duration of T2D = 9.5 years; 57% women and 69% non-white). There was no significant change in mTG in either group (−0.1 ± 0.6% and −0.05 ± 0.8% respectively) and the changes in mTG were not associated with changes in left ventricular structure or function. Compared with placebo, treatment with rosiglitazone for 6 months had no discernible effect on mTG or left ventricular function in this population with long-standing diabetes and CVD.
Introduction
The excessive cardiovascular disease (CVD) burden associated with type 2 diabetes mellitus (T2D) is conventionally attributed to a clustering of traditional risk factors including hyperglycaemia, hypertension, dyslipidaemia and obesity. More recently, studies by our group and others have demonstrated that individuals with impaired glucose tolerance and T2D have increased triglyceride content within the myocardium.1–3 This ectopic lipid deposition is purported to contribute to changes in cardiac structure and function frequently seen in individuals with glucose intolerance, referred to as ‘diabetic cardiomyopathy’.4,5 In animal models of obesity and T2D, thiazolidinedione (TZD) therapy attenuates myocardial triglyceride (mTG) deposition and normalises left ventricular (LV) systolic dysfunction.6–8 As substantial controversy exists surrounding the cardiac effects of these commonly used glucose-lowering medications,9,10 we conducted a nested imaging sub-study within a 6-month, randomised controlled clinical trial of rosiglitazone on cardiovascular performance. The sub-study assessed the effects of rosiglitazone on mTG content and its association with measures of cardiac structure and function in patients with T2D and increased CVD risk. Our a-priori hypothesis was that 6 months of rosiglitazone therapy would significantly reduce mTG content in patients with T2D, which would be associated with improved LV performance.
Research design and methods
This was a sub-study nested within a single-centre, prospective, randomised, double-blind, placebo-controlled, parallel-group trial evaluating the cardiovascular effects of rosiglitazone in patients with T2D and elevated CVD risk, with the study design and primary study results previously published (NCT00424762).11,12 In the present sub-study, we assessed the effects of rosiglitazone on 1 H-magnetic resonance spectroscopy (MRS)-derived mTG content and MRI-derived measures of LV structure and function. The study protocol was approved by the UT Southwestern Medical Center at Dallas Institutional Review Board, and all participants provided written informed consent in accordance with the Declaration of Helsinki.
Eligible patients had T2D as per American Diabetes Association criteria 13 and either a prior diagnosis of atherosclerotic CVD or >1 CVD risk factor in addition to T2D. 12 Exclusion criteria included TZD treatment in the 6 months prior to the study or documented TZD intolerance; previous heart failure; or abnormal liver function with transaminases ≥3 × upper limit of normal. Recruitment was designed to include equal representation from African American, Hispanic and Caucasian participants.
Study treatments
Participants were randomised 1:1 to receive rosiglitazone or matching placebo added to existing therapy for 6 months. A computer-based random number generator was used to create sequenced treatment assignments concealed in numbered, sealed opaque envelopes, with assignment concealed from all study personnel and patients throughout the trial. A senior research coordinator consented and enrolled all subjects and assigned participants to study group, with enrolment occurring between February 2005 and October 2006.
Study drug was initiated at a dose of 4 mg daily for the first month, followed by 8 mg daily thereafter, versus matching placebo. Throughout the study, glucose control was monitored and open-label non-TZD glucose lowering medications were used to achieve target haemoglobin A1C <7.0% in both study groups, commensurate with contemporary management guidelines for patients with T2D. 13 All assessments were conducted at UT Southwestern Medical Center.
Study assessments
Assessments at baseline, 1, 2, 3 and 6 months included a clinical history and physical exam by a cardiologist with lab assessment including basic lab chemistry and haematology, as well as a measure of A1C with open-label adjustment of non-TZD hypoglycaemic medications as needed. Compliance with study drug prescription was determined at each study visit with pill counts. The primary outcome measure for the trial was integrated cardiovascular performance assessed by measuring oxygen consumption scaled to fat-free mass using a MedGraphics™ Cardiopulmonary Exercise System (CPX/D; Medical Graphics Corporation, St Paul, MN) for breath-by-breath measurement.12,14,15 Data for this outcome have been previously published. 11
Magnetic resonance studies
The pre-specified primary outcome measure for this sub-study was mTG. Cardiac MRI and spectroscopy studies to quantify mTG were performed using 1.5 Tesla Gyroscan Intera whole body system from Philips (Philips Medical Systems, Best, The Netherlands) equipped with spectroscopy and cardiac packages as previously described.1,12,16 All images and spectra were collected and analysed by a single investigator (JM) blinded to study treatment assignment using a commercially available workstation (MASS, Philips Medical Systems). Intraindividual variability for spectroscopy and imaging analyses from our group has been published previously.17–19
Cardiac 1H-magnetic resonance spectroscopy
At baseline and after 6 months of study treatment, mTG content was measured using cardiac 1 H-MRS with methods developed in our lab.1,12,16 In short, after anatomic imaging of the left ventricle in all three planes, a spectroscopic signal was acquired from a volume of interest (10 × 20 × 20 mm3) positioned within the interventricular septum with simultaneous cardiac triggering at end systole and respiratory gating at end expiration. Myocardial triglyceride was measured using the water signal for internal reference. Results were corrected for T2 relaxation of fat and water signals assuming monoexponential signal decay and expressed as a proportion of fat to water (%).1,16
Cardiac MRI
Measurements of LV structure and function were performed using standard cardiac imaging sequences of left ventricle.1,12,16 The imaging protocol included measures of LV volumes, mass, ejection fraction and early diastolic peak filling rate (PFR) as previously described. 1
Statistical methods
Baseline characteristics and imaging results were compared between groups using Student’s t-test or Wilcoxon rank-sum test for continuous variables and χ2 or Fisher’s exact test for categorical variables, as appropriate. A mixed effects linear regression model with repeated measures was used to compare changes in mTG content between the groups with adjustment for baseline differences in body weight and mTG content. Spearman Rho correlations and multiple linear regression modelling were used to test for associations between mTG content and LV morphology and function at baseline, with similar methods used to assess for associations between changes in mTG content and changes in the same parameters. All statistical testing was performed with SAS software (SAS Version 9.1.3 (Cary, NC, USA)) using 2-tailed tests, at a significance level of 0.05, with no correction for multiple comparisons.
The power calculations and sample size determination for the mTG sub-study have been published previously, designed to analyse differences in mTG between the treatment groups within each of three race-based strata. 12 Assuming a 33% rate of technically inadequate studies, we estimated that 17 subjects per race-by-treatment assignment and 102 subjects overall would provide ≥80% power to detect a difference of ≥30% change in mTG within each race-based stratum, and ≥90% power to detect a difference ≥32% in the overall cohort.
Results
Baseline characteristics and associations
Ninety-nine subjects, including 49 in the rosiglitazone group and 50 in the placebo group, participated in the imaging sub-study. Interpretable mTG measurements were obtained in 76 patients at baseline (33 in the placebo group and 43 in the intervention group). Of these 76, 49 patients, 23 in the placebo group and 26 in the intervention group, completed all phases of the study and had valid baseline and end-of-study mTG data.
There were no significant differences in baseline characteristics between those with and without complete paired mTG assessments (Table 1). Although baseline body weight was statistically different between the randomised treatment and placebo control groups, no other baseline differences were observed in medications, anthropometric or lab variables, or in cardiac morphology, function or triglyceride content (Tables 2–4). Among 76 participants with valid baseline data, mTG content was not associated with any cardiovascular risk factors. Of note, mTG was also not associated with body fat (ρ = −0.05, NS), age (ρ = −0.05, NS), duration of diabetes (ρ = 0.07, NS) or any measure of LV structure, mass or function.
Comparison of baseline characteristics between patients with and those without complete paired myocardial triglyceride scanning data
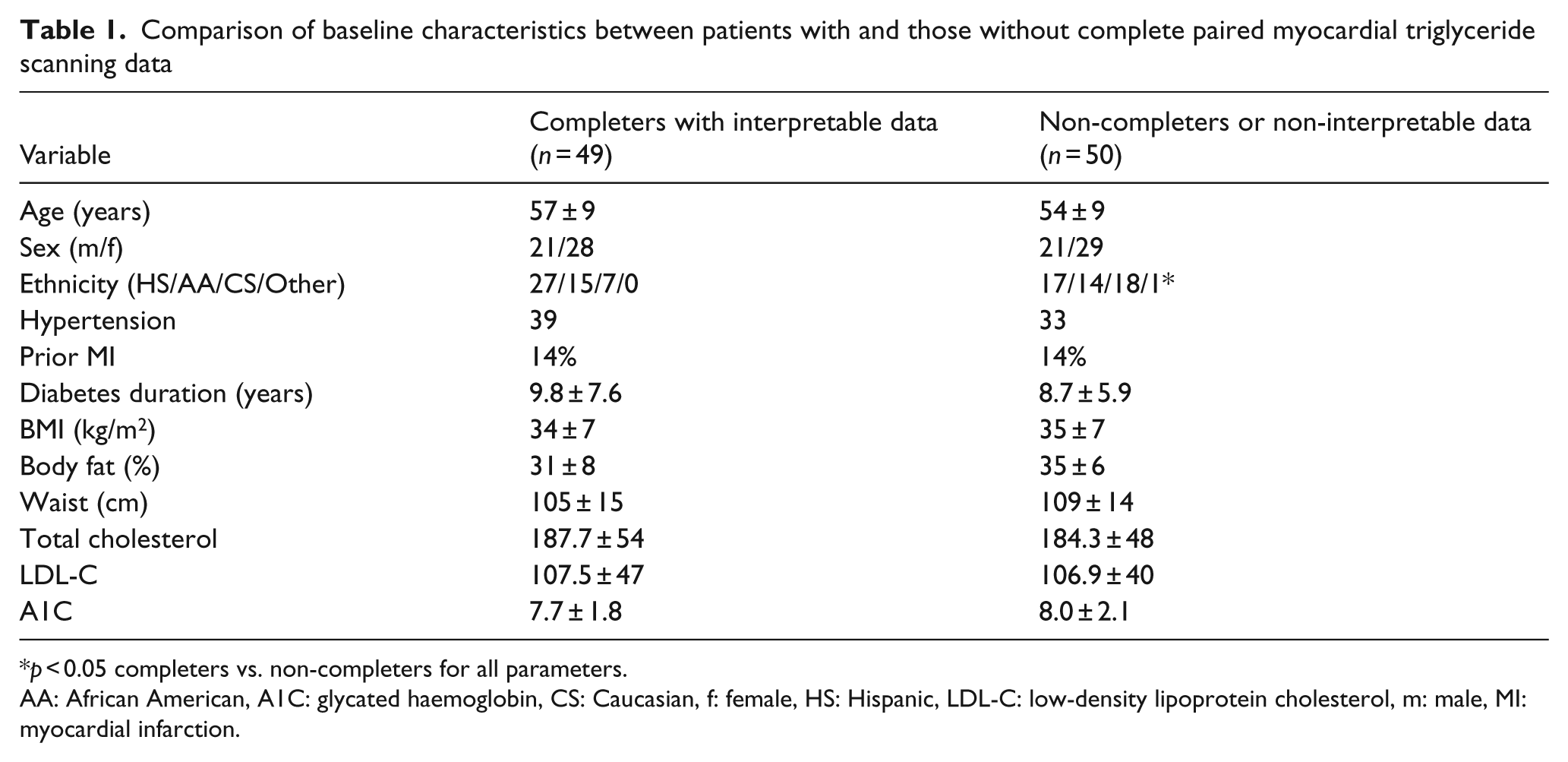
p < 0.05 completers vs. non-completers for all parameters.
AA: African American, A1C: glycated haemoglobin, CS: Caucasian, f: female, HS: Hispanic, LDL-C: low-density lipoprotein cholesterol, m: male, MI: myocardial infarction.
Baseline characteristics by treatment group among patients with complete baseline and end-of-study myocardial triglyceride data
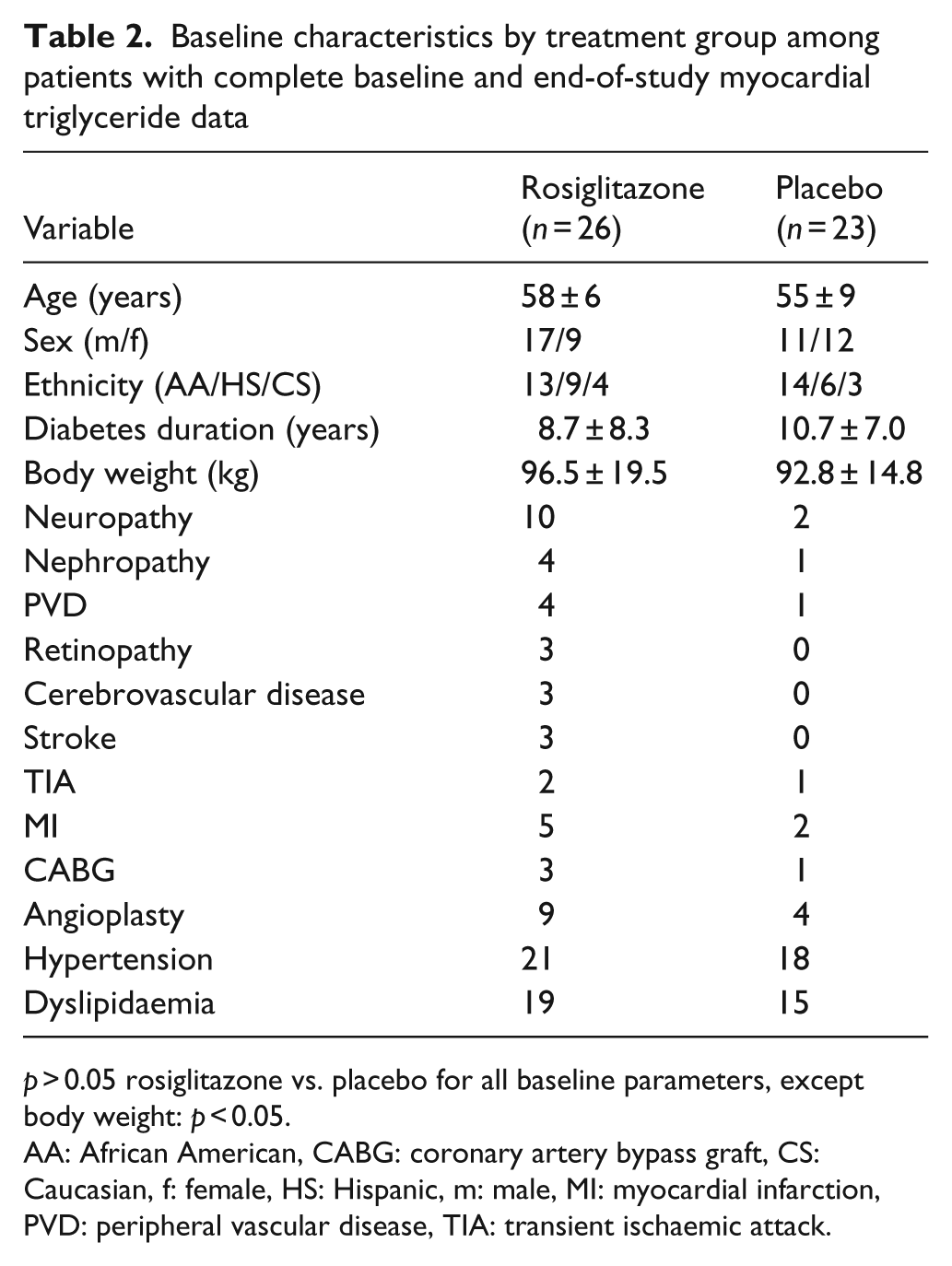
p > 0.05 rosiglitazone vs. placebo for all baseline parameters, except body weight: p < 0.05.
AA: African American, CABG: coronary artery bypass graft, CS: Caucasian, f: female, HS: Hispanic, m: male, MI: myocardial infarction, PVD: peripheral vascular disease, TIA: transient ischaemic attack.
Baseline and end-of-study anthropometric and laboratory measures by study treatment group
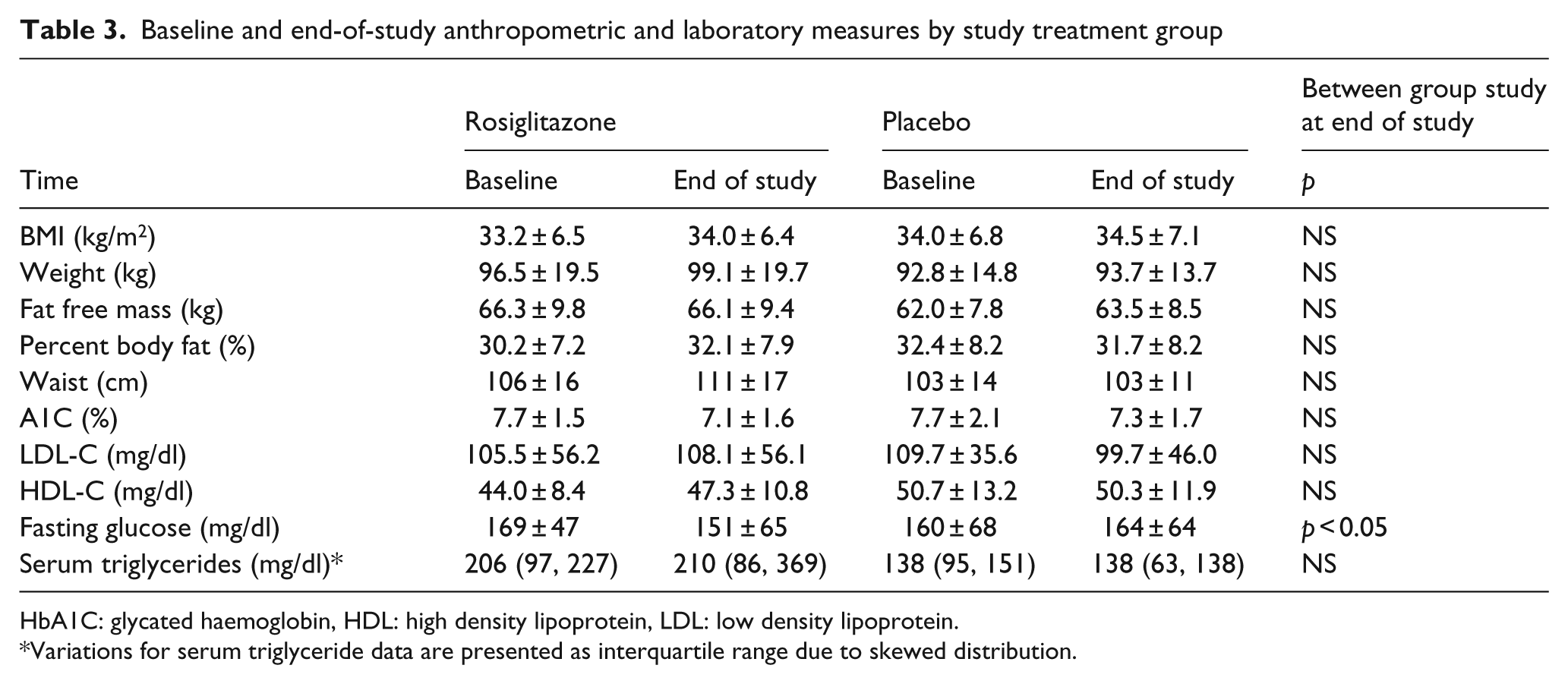
HbA1C: glycated haemoglobin, HDL: high density lipoprotein, LDL: low density lipoprotein.
Variations for serum triglyceride data are presented as interquartile range due to skewed distribution.
Baseline and end-of-study magnetic resonance imaging measures by study treatment group
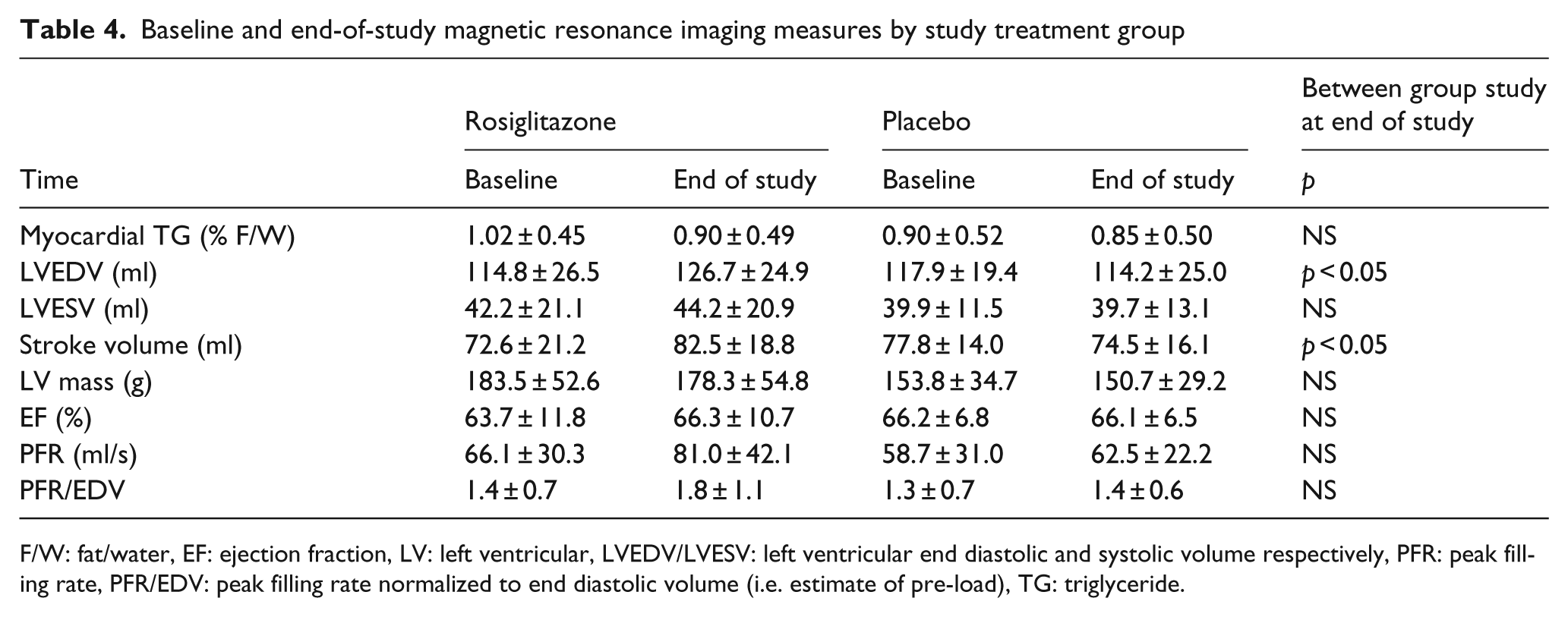
F/W: fat/water, EF: ejection fraction, LV: left ventricular, LVEDV/LVESV: left ventricular end diastolic and systolic volume respectively, PFR: peak filling rate, PFR/EDV: peak filling rate normalized to end diastolic volume (i.e. estimate of pre-load), TG: triglyceride.
Treatment effects
Among patients with complete paired data, pre-treatment mTG was not significantly different between study groups (1.02% vs. 0.90%; p = 0.50). Six months of treatment with rosiglitazone added to existing therapy resulted in modest but significant improvements in fasting glucose (166 ± 44 mg/dl to 150 ± 67 mg/dl; p < 0.05) and A1C (7.6 ± 1.4% to 7.1 ± 1.6%, p = 0.029). In contrast, in the placebo group, no significant changes were observed in fasting glucose (160 ± 68 mg/dl to 164 ± 64 mg/dl, p = 0.713) or A1C (7.7 ± 2.1% to 7.3 ± 1.7%, p = 0.087; Table 3).
Results of cardiac MRI and MRS measures are presented in Table 4. There were no significant changes within either group in mTG during the study, and no difference between the groups in mTG at study completion. Similarly to results previously reported on the overall study cohort, 11 in this subset of patients with mTG measurement, left ventricular end-diastolic volume (LVEDV) increased significantly with rosiglitazone treatment (114.8 ± 26.5 ml to 126.7 ± 24.9 ml), with no such changes evident in the placebo group (117.9 ± 19.4 ml to 114.2 ± 25.0 ml; p < 0.01 group × time effect). Similar differences were observed in resting stroke volume, which increased significantly with rosiglitazone treatment (72.6 ± 21.2 ml to 82.5 ± 18.8 ml, p = 0.004) but not in the placebo group (77.8 ± 14.0 ml to 74.5 ± 16.1 ml, p = 0.3). There were no other significant changes in measures of cardiac structure or function within groups or differences between groups after 6 months of study treatment, including any change in LV diastolic function.
In bi-variable associations, the change in mTG was not associated with changes in LV stroke volume (ρ = −0.18, p = 0.21) or LV peak filling rate (ρ = −0.20, p = 0.16); however, it was positively associated with LV ejection fraction (ρ = 0.40, p = 0.045) within the 26 patients in the treatment arm (Table 5).
Bi-variate correlations between myocardial triglyceride at baseline and the change over time with various parameters of cardiovascular and LV function and morphology
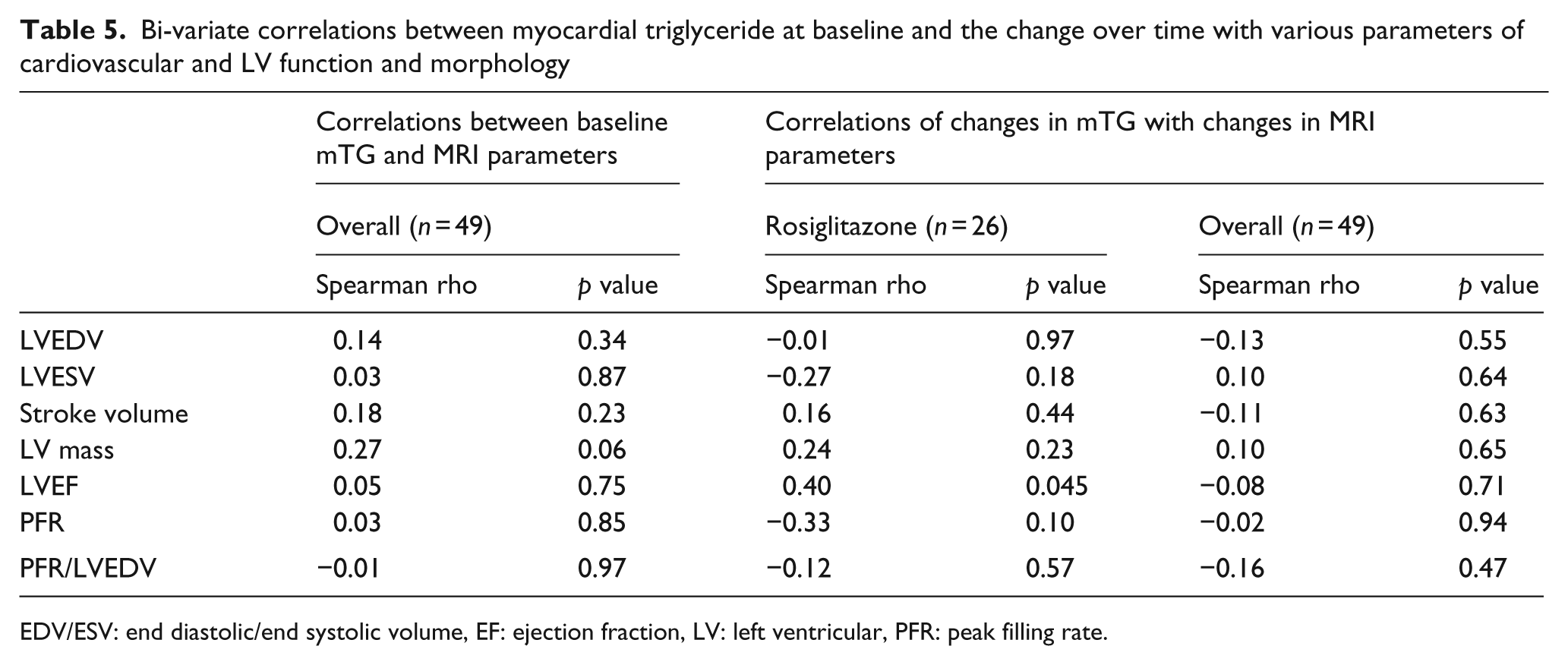
EDV/ESV: end diastolic/end systolic volume, EF: ejection fraction, LV: left ventricular, PFR: peak filling rate.
Conclusions
The primary observation from this randomised, controlled trial is that 6 months of treatment with rosiglitazone added to existing therapy had no discernible effects on mTG content, compared with placebo. Additionally, absolute measures and changes in mTG were not significantly associated with cardiac structure or function in patients with long-standing T2D and elevated CVD risk.
Previous studies in animal models of obesity and T2D demonstrated that the TZDs troglitazone and rosiglitazone attenuate the development of myocardial steatosis.6–8 Human studies of pioglitazone, and now rosiglitazone, have failed to replicate these findings in patients with T2D for several reasons. First, early studies began treating animals prior to the onset of frank diabetes and the development of profound myocardial steatosis, 6 while human studies for the most part are treating older patients with varying degrees of glycaemic control and CVD risk. 8 Second, to replicate the finding that reducing mTG improves LV function requires a population of patients with T2D with significant cardiac steatosis free from conditions that would otherwise influence LV function, in particular coronary artery disease. To date, studies have enrolled patients with T2D with a wide range of mTG values and varying degrees of coronary artery disease. Future studies wishing to replicate animal studies would need to treat with a TZD agent young overweight individuals who display impaired glucose tolerance and defined myocardial steatosis in the absence of coronary artery disease.
Prior results from our lab demonstrated that 6 months of treatment with pioglitazone and insulin significantly reduced mTG content in new patients with T2D. 20 The present observations did not replicate this effect but are in accord with a recently published randomised controlled trial using a similar 6-month regime of pioglitazone. 21 Significant differences between study designs and patient populations likely explain the disparate results between experimental trials of the effect of TZD therapy on mTG in humans. For example, previous studies were restricted to Caucasian men, with a shorter duration of T2D and much lower overall CVD risk. 21 Additionally, some studies have included patients receiving insulin to improve glycaemic control 20 while others have not. 21 Collectively, this degree of discordance across studies is potentially due to (1) differential biology of rodents and humans, (2) differences in effects of various drugs within the TZD class, (3) differences in duration of therapy or (4) differences in ethnic make-up of patients studied across trials.
Our group and others have previously demonstrated that cardiac steatosis is a distinctive feature of impaired glucose tolerance and T2D in humans independently of CVD and heart failure.1,3 In both animal studies and studies of older adults, increased mTG is inversely associated with LV performance,6,22–25 leading some to believe that steatosis contributes in part to the idiopathic cardiomyopathy commonly observed in individuals with insulin resistance or glucose intolerance.24–26 Myocardial TG values observed in the present trial were similar to those reported in previous investigations of patients with T2D using 1H-MRS technology,20,21,24,25 despite the increased CVD risk and extended duration of T2D. While experimental studies have yielded negative findings, cross-sectional studies using echocardiographic measures of LV performance have demonstrated that patients with T2D with elevated mTG display significant impairment in LV diastolic function.24,25 As we were unable to demonstrate any measurable effects of rosiglitazone on mTG, we are unable to determine whether a causal association exists between mTG and LV function.
Previous reports by our group from this trial have documented the changes in cardiac volumes in patients treated with rosiglitazone. 11 Based on the observed increase in brain natriuretic peptide and body weight, coupled with declines in haematocrit and albumin, in patients treated with rosiglitazone, we believe the changes in LV volume are related to an expanded plasma volume.11,27. We also recently documented that the duration of diabetes was a predictor of plasma volume expansion and peripheral oedema. 28
Validity of the null findings
Several factors may have contributed to the null findings, including type II statistical error and characteristics of the patient population studied. With 26 and 23 patients in treatment and placebo arms respectively and a standard deviation of 0.49 for baseline mTG, we had 80% power to detect a mean difference of 0.40% fat/water levels in the myocardium and 90% power to detect a difference of 0.46% fat/water between experimental and control arms. While the population mean of mTG at baseline was very close to predicted, the larger variance in the study outcome and a rate of attrition higher than anticipated precluded our ability to rigorously exclude treatment effects smaller than 0.4. However, effects this small would not be likely to yield substantial clinical effects. Furthermore, by studying patients with longer duration of T2D and with previous high CVD risk burden, the effects of TZD medications on mTG may be attenuated or confounded by the presence of underlying coronary artery disease and hypertension or the confounding effects of other CVD risk mitigating therapies, though these effects remain completely undefined. Finally, the low rate of successful acquisition of mTG data in patients reduced our sample size and may have introduced sampling bias. The reasons for the poor yield in this study could be explained by the population studied, with the technical challenges of image acquisition in an older cohort with significant comorbidity as well as the high overall rate of study attrition.
Limitations
Similarly to previous studies in which mTG has been measured in humans,1,22,29 the present study was restricted by the technological limitation of uninterpretable data in a significant portion (25%) of patients in each wave of testing and a relatively high rate of overall study drop out, as previously reported (28%), 11 yielding fully interpretable data over the course of the study for only 50% of enrolled patients. Although the proportion of participants with uninterpretable data is decreasing as experience in the technique accumulates, participant movement in the MRI magnet and the presence of arrhythmia and ectopy remain limitations inherent in the protocol. In addition, the present study was limited by the lack of sensitivity of cardiac MRI-derived measures of LV function adopted for this study. More sophisticated ultrasound techniques or cardiac tagging 30 would have provided a more sensitive measure of LV systolic and diastolic function. Finally, we are unable to determine whether longer treatment duration or use of another TZD medication would yield any different results. Larger randomised controlled trials of longer treatment duration in patients with established cardiac steatosis are needed to conclusively assess the effectiveness of this class of drug on cardiac lipid content.
Six months of rosiglitazone therapy had no effect on mTG content or LV function in patients with T2D and increased cardiovascular risk. Accordingly, the potential cardiac-specific benefits of modulating mTG content in patients with T2D with any drug therapy remain unproven.
Footnotes
Funding
This work was supported by research grants from GlaxoSmithKline (GSK); Biosite, Inc.; the Donald W. Reynolds Foundation; and the United States Public Health Services General Clinical Research Center (grant #M01-RR00633) from National Institutes of Health/National Center Research Resources-Clinical Research. JMM was supported by a Target Obesity postdoctoral fellowship from the Canadian Institutes of Health Research, Canadian Diabetes Association and the Heart and Stroke Foundation of Canada. The funding organisations did not participate in study design, data collection, analysis or interpretation, writing of the report, or the decision to submit the paper for publication. Representatives of GSK reviewed the manuscript prior to submission, but did not contribute to its content. The corresponding author had full access to all of the data in the study, takes responsibility for the integrity of the data and the accuracy of the data analysis, and had final responsibility for the decision to submit for publication.
Acknowledgements
Clinical Trial Registration: NCT00424762. JMM, DKM, SMA and LSS collected data. JMM and CRA researched data. JMM and DKM wrote the manuscript. LSS, SMA, RS, MOG, MHD and JAL reviewed/edited the manuscript.
Conflict of interest
DKM reports receiving honoraria from Pfizer and Takeda and consultancy fees from Novo Nordisk, Tethys Bioscience, and AstraZeneca. MHD reports receiving honoraria from GlaxoSmithKline. LSS reports receiving grant support from GlaxoSmithKline and Takeda. SMA, JMM, JAL, RS, MOG and CRA report no financial relationships.
