Abstract
Biopolymers have become prevalent in industries to prevent acid corrosion because of their robust structure and biodegradability. The current research investigates the effectiveness of the biopolymer (+)-arabinogalactan (AG) in inhibiting corrosion on AISI 5140 steel in H2SO4 media, employing a novel approach to experimental design called the custom design of experiment (DoE). Temperature and medium concentration have the greatest effects on the response, according to p-value analysis. The highest inhibitory efficiency (IE) at 50 °C, 0.1 M H2SO4 concentration, and 0.1 g/L of AG is predicted to be 75.57%. Density functional theory-based computational investigations demonstrate the potential of AG as a corrosion inhibitor, with chemisorption being the primary mode of adsorption. AG's adsorption on the metal surface is further confirmed by scanning electron microscope and energy-dispersive X-ray studies. Thus, this study pioneers the application of custom DoE in investigating AG's corrosion inhibition properties on AISI 5140 steel in H2SO4 medium.
Introduction
The susceptibility of metals to corrosion is determined by their chemical composition, microstructure, and the surrounding environment. Small areas function as micro galvanic cells due to the intrinsic heterogeneities in metals, leading to potential differences. The cause of this variation is due to the presence of various chemicals, crystal lattice defects, or phase volume percentages. Inhibitors can be used to mitigate these differences and improve corrosion resistance. Adsorption of an inhibitor molecule onto the metal surface leads to the creation of a barrier, thus leading to the prevention of corrosion. 1 Despite the availability of synthetic inhibitors with high efficiency, concerns regarding their toxicity have prompted a shift towards environmentally friendly alternatives, such as biopolymers. 2
Biopolymers are composed of polysaccharides and nucleic acid produced by living organisms and offer biodegradability attributed to their oxygen and nitrogen-rich atoms.3–5 These hetero atoms act as adsorptive centres to link the biopolymer with the metal surface.6,7 Few researchers have investigated the corrosion inhibition property of biopolymers.8–11 Jmiai et al. have investigated the corrosion mechanism of alginate as a green inhibitor for copper in 1 M HCl. Eighty-three percent maximum inhibitory effect was achieved when 0.1 g/L of alginate was used. 12 In a saline medium, I.B. Boto examined the corrosion-prevention ability of sodium alginate on API X60 steel and discovered a notable reduction in the metal's pitting corrosion. 13 Furthermore, the adsorption characteristics of two biopolymers Iota-carrageenan and Inulin on a mild steel/sulphuric acid interface were analyzed and identified as a mixed-type inhibition. 14 The inhibition mechanism of biopolymers involves the formation of a coordinate bond by the heteroatom and the metal surface. 15
Moreover, as the number of parameters increases, it becomes difficult to perform experiments since the inhibition efficiency depends on multiple aspects. If you take a factor-at-a-time strategy, you run into the risk of ignoring how these components interact. 16 To address this challenge, design of experiments (DoE) methodology has been created to expedite testing and enhance cost-effectiveness in industrial processes. By maximising response output and lowering trial numbers, this method maximises the effectiveness of industrial tests.
Response surface methodology (RSM) with DoE framework has been effectively implemented by researchers to optimise conditions for investigations on corrosion inhibition. 17 Naseri et al. analyzed the influence of various factors on the inhibitive effect of Clopidogrel using RSM and inhibition efficiency of 80–90% was achieved. 18 Similarly, Luo et al. used RSM for the optimisation of the extraction conditions of tannins from acron for corrosion inhibition studies, obtaining optimal conditions including the ultrasonic time (2.51 h), ultrasonic power (97.92 W), cellulose concentration (3.44 g/L), and extraction temperature (38 °C). 17 Likewise, Anadebe et al. maximised the inhibition efficiency of pigeon pea leaf extract as a corrosion inhibitor for mild steel; the highest inhibition efficiency was obtained at 0.9 g/L of the inhibitor. 19
In the present work, to reduce the number of trials involved, a novel approach, a custom design of DoE is employed in optimising the parameters involved in the corrosion inhibition study of (+)-arabinogalactan (AG). AG is a carbohydrate polymer consisting of arabinose and galactose monosaccharides. Due to its biodegradability and water solubility, it finds application as a stabiliser and thickener in the food industry. The large complex structure of arabinogalactan, consisting of a large number of oxygen atoms, suggests its use as a corrosion inhibitor. Despite of its biodegradability factor, the major challenge in the use of AG as a corrosion inhibitor is its hygroscopic nature and vulnerability towards degradation. 20 Therefore, care must be exercised in handling the inhibitor during the application stage while custom designs can be advantageous over classical designs since they can contain more experimental runs. The custom DoE design is based on filling the design space rather than just taking sample points predominantly around the boundaries of the design space. 21
Various studies have utilised custom DoE. For instance, Stein et al. have used a custom design DoE approach in process optimisation and prediction of the process of iron leaching from non-refractory grade bauxite. 22 Edrees et al. used a custom design DoE approach for the analysis of the chromatographic method for the simultaneous analysis of dimenhydrinate, cinnarizine, and their toxic impurities. 23 However, to the best of our knowledge, no attempt has been made to apply a custom design approach of DoE for the corrosion inhibition study of metals. Herein, the inhibitory effect of AG on the corrosion of AISI 5140 in H2SO4 medium is analyzed after process optimisation through custom DoE.
Methodology
Material preparation
The metals used for the corrosion study is annealed AISI 5140 steel (composition: Fe: 96.7%, C: 0.3%, Si: 0.275%, Mn: 0.78%, and Cr: 0.84%). The sample is cylindrical with dimensions of 12 mm diameter. Before each electrochemical analysis, the specimen was cleaned with emery sheets (80–800) in increasing sequence, disc polished with diamond paste (3 microns to 0.25 microns), and then cleaned with acetone and allowed to dry.
Medium preparation
The corrosive medium was prepared using Merck's analytical grade H2SO4 solution. To get the desired volume and concentration, it is diluted. Diluting the stock solution appropriately enabled the preparation of different concentration solutions (0.5 M, 0.25 M, and 0.1 M). From Tokyo Chemical Industry, biopolymer AG derived from larch wood was purchased. The necessary concentration of AG in the mentioned media was taken to prepare the inhibitor solution.
Corrosion studies using EIS and PDP method
For corrosion studies, molded AISI 5140 steel (composition: Fe: 96.7%, C: 0.3%, Si: 0.275%, Mn: 0.78%, and Cr: 0.84%) with an exposed area of 0.8 cm2 is utilised as the working electrode. Measurements were performed in an aerated, unstimulated condition at varying temperatures (30 °C to 50 °C) using a three-electrode assembly consisting of the working electrode, platinum counter electrode, and calomel reference electrode connected to a potentiostat (CH600E). The following settings were chosen for the EIS measurements: frequency: 100 Hz to 10 mHz; AC signal amplitude: 10 mV. After obtaining the impedance data, the graph is plotted. At a scan rate of 1 mVs−1, the polarisation curve was obtained at OCP between −250 mV and +250 mV. Tafel curves were plotted after the data was collected.
Surface morphology and elemental analysis
After immersing the annealed AISI 5140 steel (for 3 h) samples with and without the addition of (+)-arabinogalactan, the surface morphology studies were carried out. The samples were viewed at a 1000× magnification using a JEOL JSM-6380L scanning electron microscope (SEM). Energy-dispersive X-ray (EDX) analysis was used to carry out the elemental analysis.
Theoretical study using density functional theory
Gaussian 09 W software and GaussView 5.0.8 were used for the quantum chemical calculations. Numerous theoretical parameters were determined through the use of the B3LYP/6-31G (d, p) basis set in the calculations. These quantum chemical parameters include the energy of the highest occupied molecular orbital (EHOMO), the energy of the lowest unoccupied molecular orbital (ELUMO), ΔE (energy gap), electronegativity (χ), hardness (η) and softness (σ), global electrophilicity (ω), nucleophilicity (ε), and fraction of electron transferred (ΔN).
Optimisation of parameter using DoE
A custom design approach involving RSM was employed using Jmp software package JMP Pro 16.2.0 (SAS Institute InC.) to design, test, and assess the experiment. The major objective was to obtain a formula for predicting the influence of various factors on the inhibition efficiency along with identifying the optimal response. The concentration of the acid medium, concentration of AG and temperature were identified as the parameters under consideration that have a reliable impact on the output (corrosion inhibition efficiency). The concentration of medium was considered for three levels (0.1 M, 0.25 M, 0.5 M), concentration of AG (0.02 g/L, 0.04 g/L, 0.06 g/L, 0.08 g/L, 0.1 g/L) and temperature for three levels (30, 35, 40, 45, 50 °C), determined from initial preliminary test were chosen as categorical factors. A design predictive model is constructed in a randomised manner. For the set of experimental factors, the inhibition efficiency was calculated experimentally and fed into the model. The model calculates coefficients for each main effect, interaction effect, and quadratic effect. The optimised response is obtained.
Result and discussion
Quantum chemical calculations of AG
Quantum chemical calculations by density functional theory (DFT) method were utilised for understanding the inhibitory action of AG on AISI 5140 steel. The optimised structure, HOMO, and LUMO of AG is demonstrated in Figure 1. Localisation of electron density is observed in both HOMO and LUMO, confirming the presence of electron donating and electron accepting centres in the AG molecule.
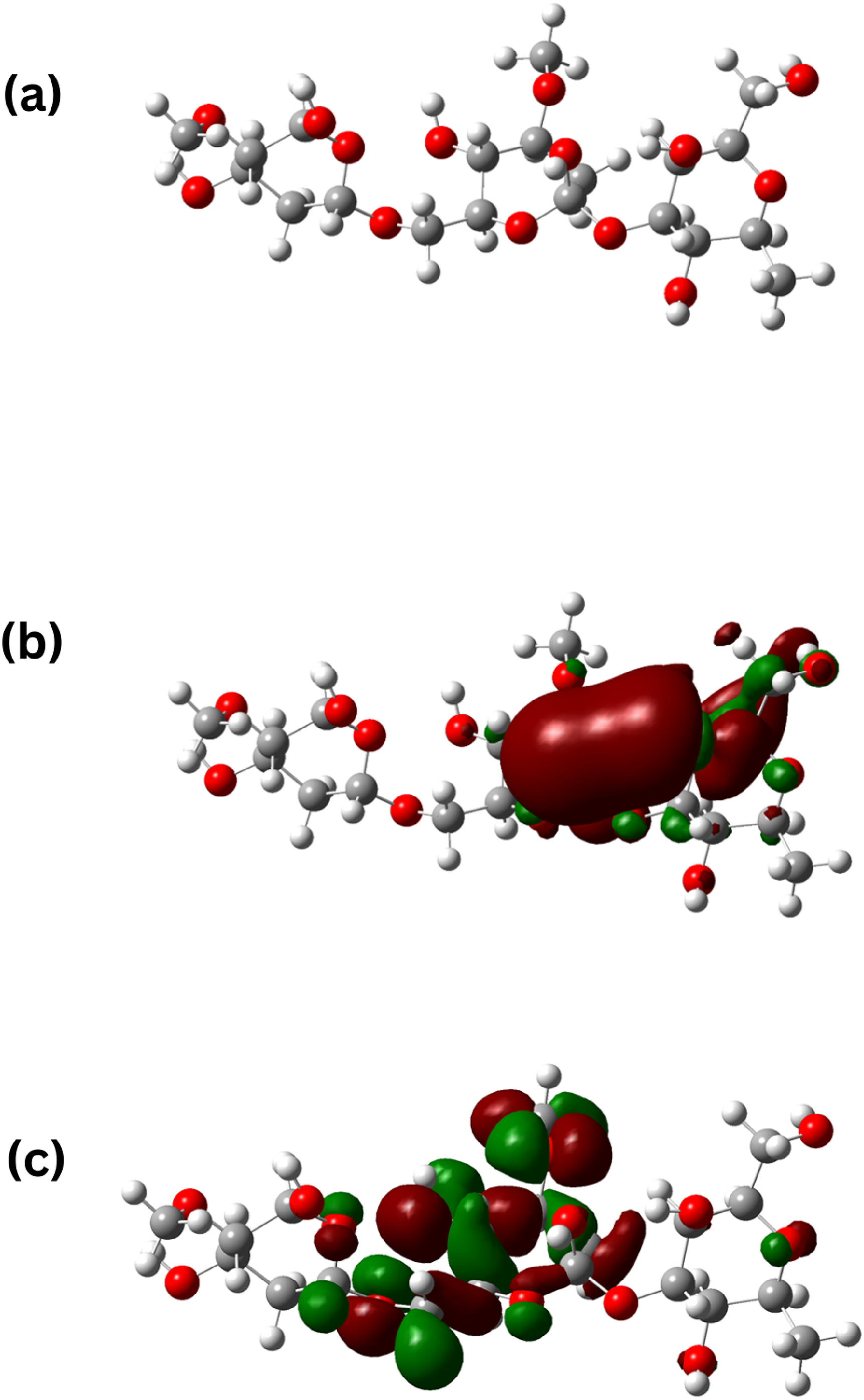
(a) Optimised structure (b) HOMO c) LUMO of AG.
Multiple parameters are calculated to comprehend the interaction between AG and the metal, including the energy of HOMO, LUMO, dipole moment (µ), energy gap (ΔE). The EHOMO value of AG indicates its electron-donating capacity. Thus higher the value of EHOMO, the higher will be the tendency to donate electrons to the lowest unoccupied d orbitals. The lower the value of ELUMO, greater the tendency to accept the electron.24,25 Therefore, the stability of the molecule is indicated by the value of ΔE. A stable complex will form when ΔE is lowered. From Figure 1(b), it can be observed that the HOMO is concentrated on the sites where there is a presence of oxygen atom. This can be attributed to the presence of lone pair on the oxygen atom, thus acting as an active site for adsorption. Table 1 lists the different quantum parameters that were obtained.
Quantum chemical parameters of (+)-arabinogalactan from DFT calculation.
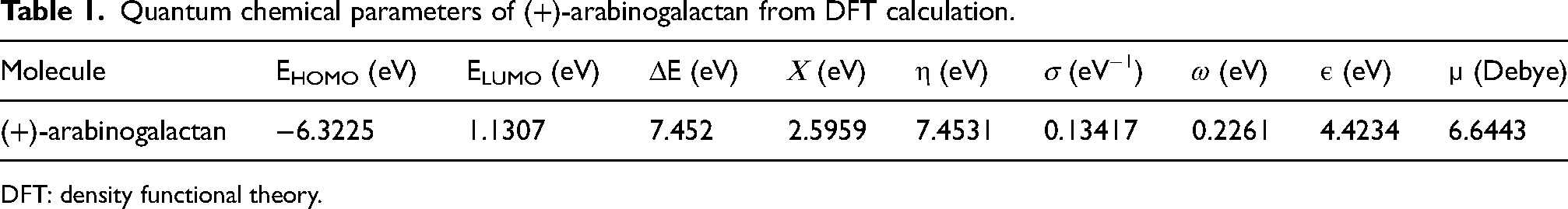
DFT: density functional theory.
A large dipole moment (µ) value indicates higher inhibitory activity.
26
For (+)-arabinogalactan the µ value was found to be 6.6443. This value exceeds the dipole moment of water, suggesting that dipole–dipole interaction may cause water molecules to be dispersed and adsorbed on the surface.
27
Koopman's theory can be applied to compute the values of ionisation potential (I) and electron affinity (A) which is given by the following equations
28
:

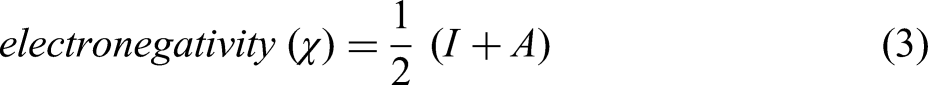
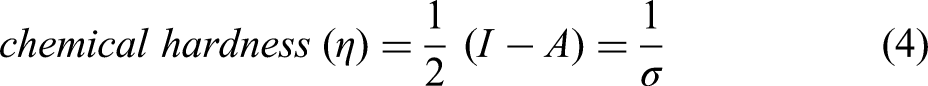
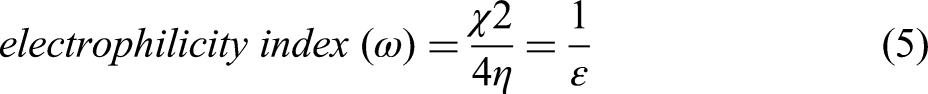
Global softness and hardness provide insight into molecular stability and reactivity. It is favourable to employ a molecule with a high global hardness and a low global softness as a corrosion inhibitor.
From the values of χ and η, the fraction of electron transferred (ΔN) can be obtained using the formula
29
:
To understand the electron adsorption centres in a molecule, Mulliken atomic charge distribution is examined. Figure 2 gives the Mulliken charge distribution for (+)-arabinogalactan. The negative charge is found to be accumulated on the heteroatom (oxygen atom), this negatively charged centre is the site for coordination for the molecule.
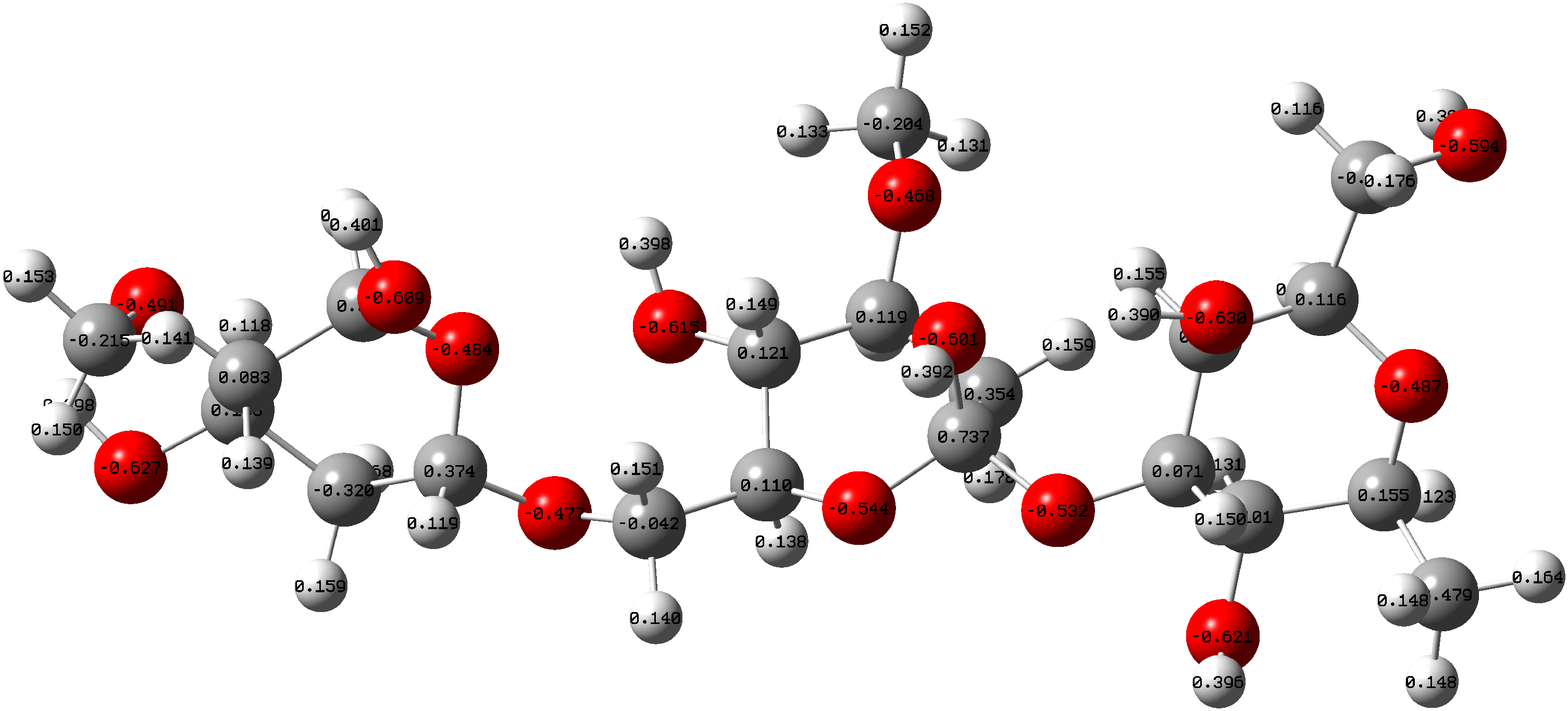
Mulikken charge distribution of (+)-arabinogalactan.
Design of experiment for the corrosion inhibition study of AG on AISI 5140 steel in different concentrations of H2SO4 media
The optimisation of the parameters for the inhibition study of AG is performed using an RSM based I-optimal custom design in JMP Pro 16 software.22,31 The main aim of the experiment is to maximise inhibition efficiency. The inhibitor concentration and temperature are selected as a five-step categorical factor and the concentration of the medium is selected as a two-step categorical factor.
For each factor, an experimental range is selected by preliminary analysis and is represented in Table 2. The JMP software creates an experimental design as given in Table 3 in a random order containing 13 runs including a centre point. The parameters that are not varied (that are not considered as varied factors) including the metal used, and time of immersion was kept constant.
Experimental levels of each factor.

Custom design based on response surface methodology.
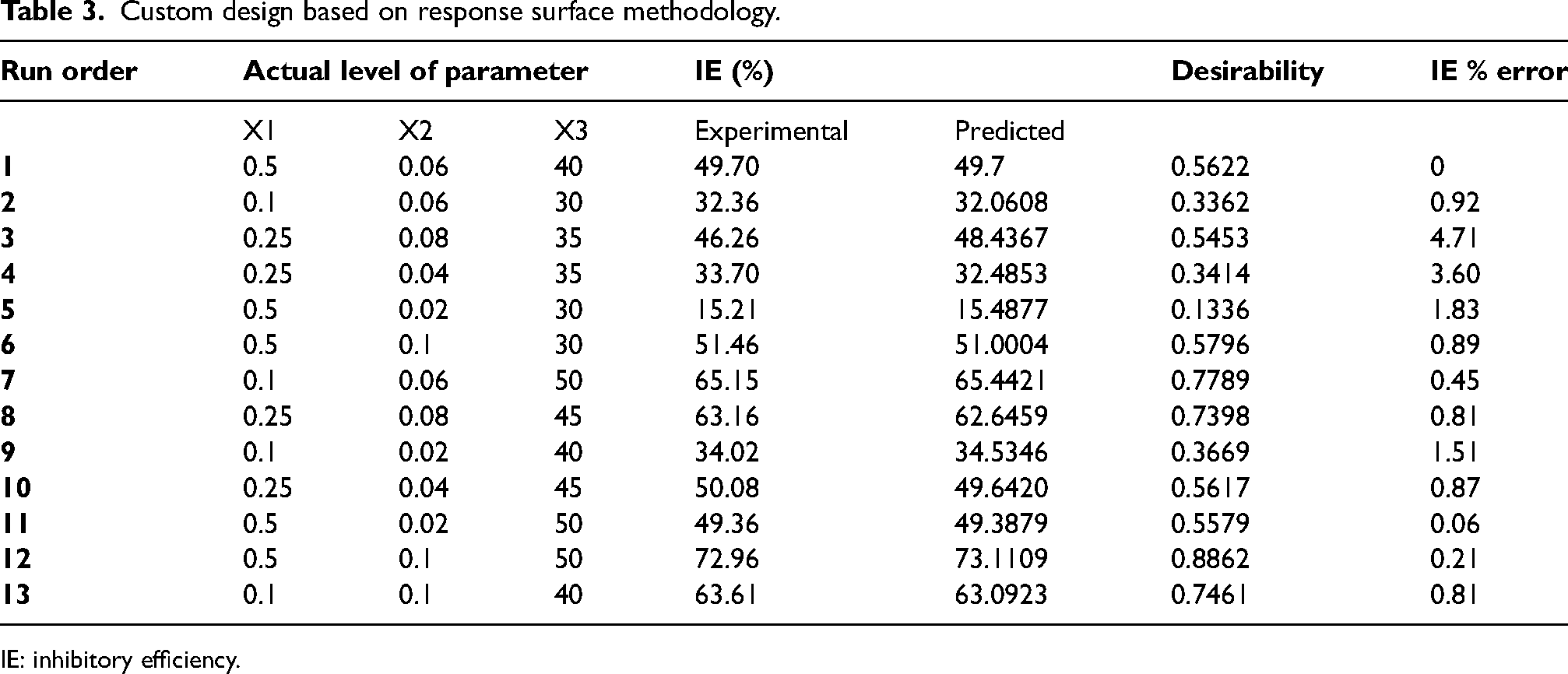
IE: inhibitory efficiency.
The experimental design is statistically evaluated to fit all the variables. The overall effects and interactions in the created experimental design were evaluated based on the p-value. Table 4 shows the significance of the overall effects in the 95% confidence level. From Table 4, a rise in the p-value was observed from top to bottom. The lesser the p-value, the greater will be the effect of the corresponding interaction on the response. Thus main effects, temperature, and concentration of the medium are found to show the greatest influence on the response.
Effect summary report of model fitting.

All the main effects, two-way interactions, and quadratic effects were included during the model fit. Some of the effects were found to be not significant but it was kept in the model since its removal resulted in a decrease in the model fit.
The analysis of the variance for the inhibition efficiency is performed and is given in Table 5. It is observed that the response (inhibitory efficiency (IE) %) is significant with an F-ratio of 135.44. The r-square value is found to be 0.99, which means that 99% of the variability in the response (IE %) is explained using the fitted regression model. The model-leverage plot (Figure 3) by taking the inhibition efficiency (actual) in the y-axis and the inhibition efficiency (predicted) in the x-axis shows that the values are close to the red line. This indicates that the values are within the 95% confidence level. A model is considered valid, when there is a small difference between the actual and predicted value. As observed, the experimental results obtained are randomly distributed along a diagonal line. This illustrates this model's high accuracy in predicting the inhibition efficiency of AG.
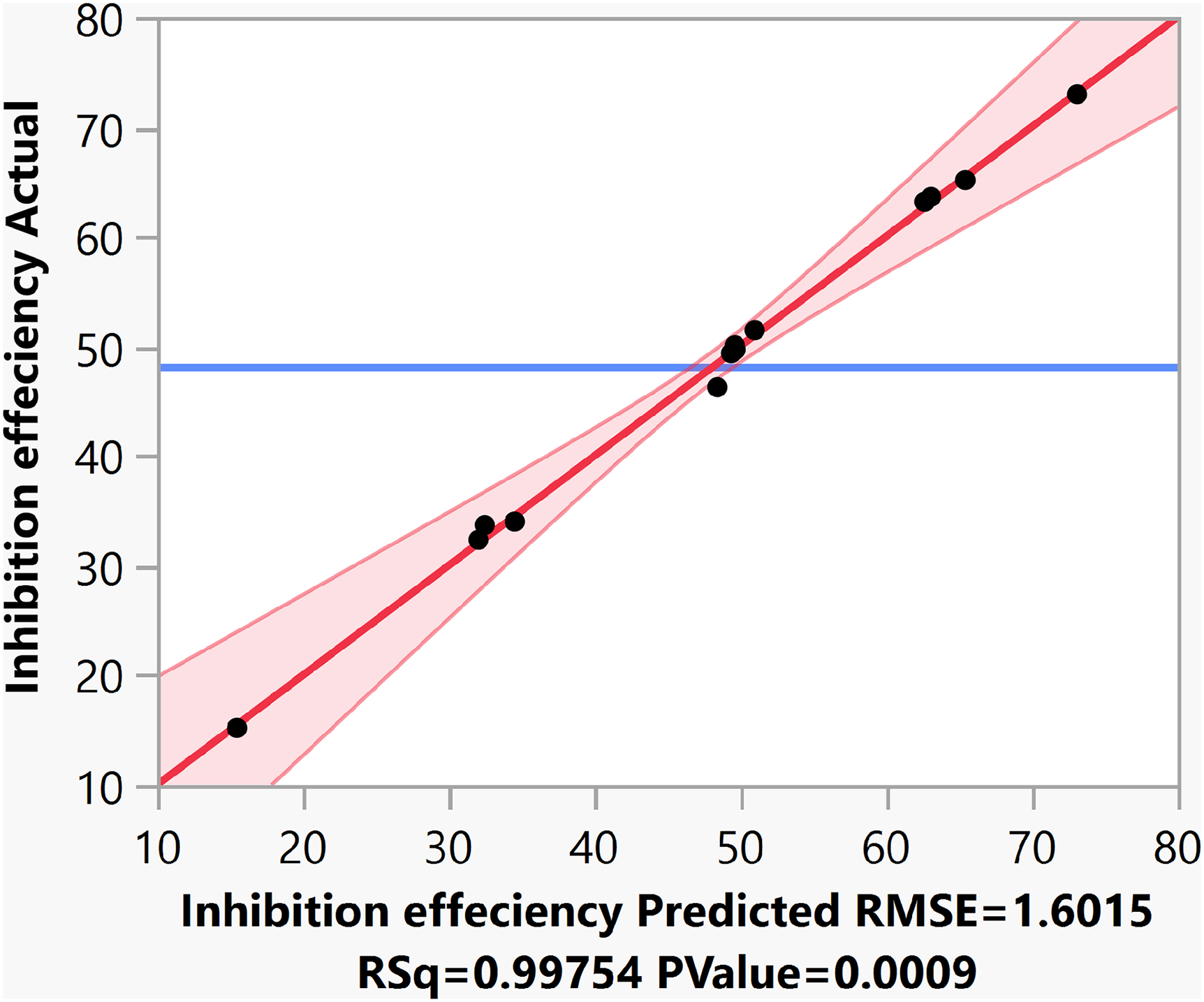
Model-leverage plot for inhibition efficiency.
Analysis of variance.

The interaction between factors occurs when the response change is different for two variables. 32 The interaction effect plot is demonstrated in Figure 4. It is clear that there is less significance for the interaction between various factors except for the quadratic effect of the concentration of acid. From the ANOVA analysis, it is evident that the only two-way interaction effect with a p-value <.05 is the interaction between the concentration of the medium and temperature. The p-value of other two-way interactions reiterates that there is no intercorrelation between the three factors.
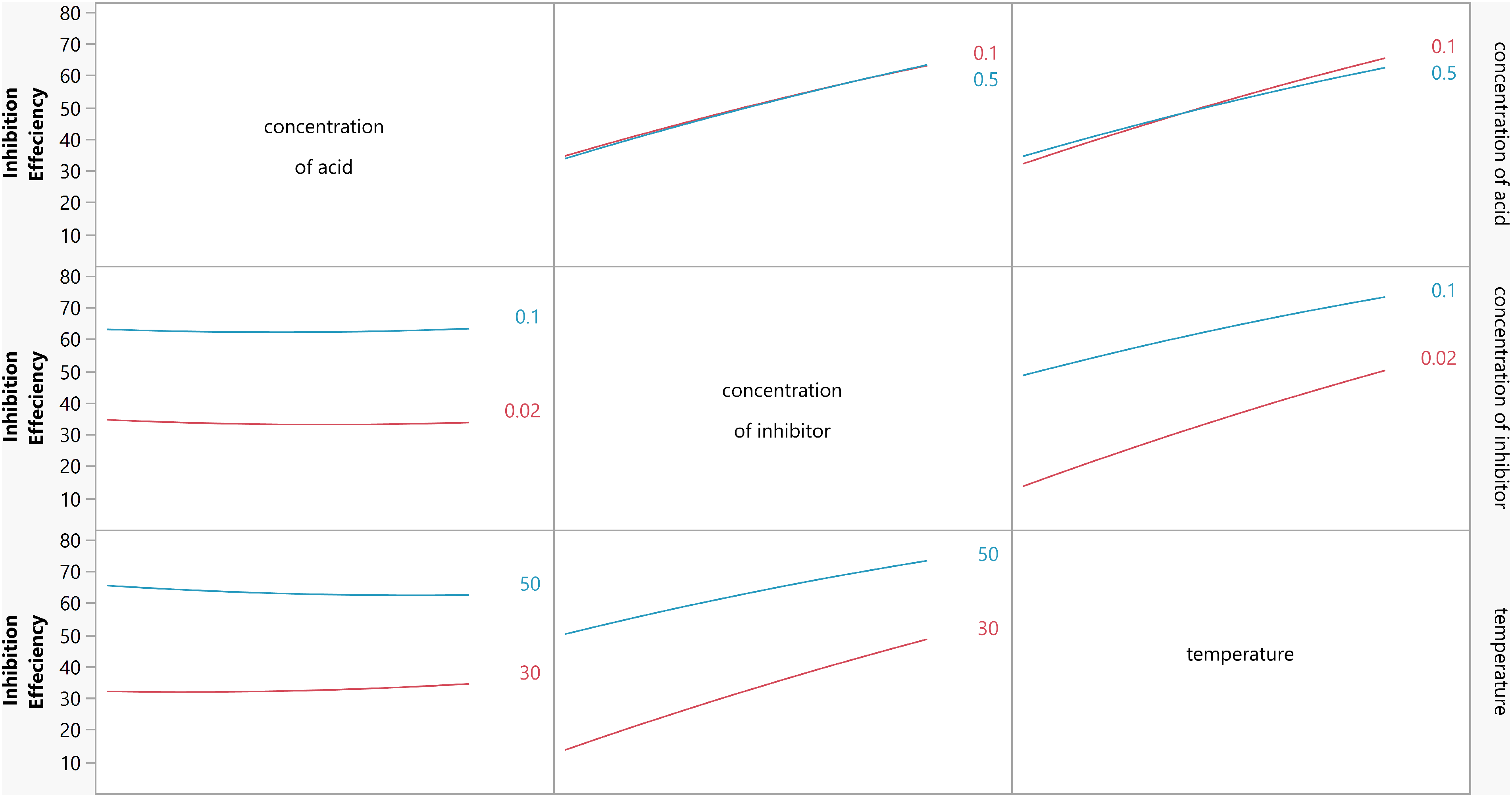
Interaction plot for inhibition efficiency.
The interaction effect of the various process variables on the inhibition efficiency of AG is represented using a surface plot in Figure 5. From Figure 5(a), it is observed that at a particular concentration of the medium, IE % elevates with the increase in the concentration of the inhibitor. The elevation in the IE % with the increase in the temperature and inhibitor concentration can be observed. This suggests that the mechanism of adsorption of AG on AISI 5140 steel is through chemisorption.
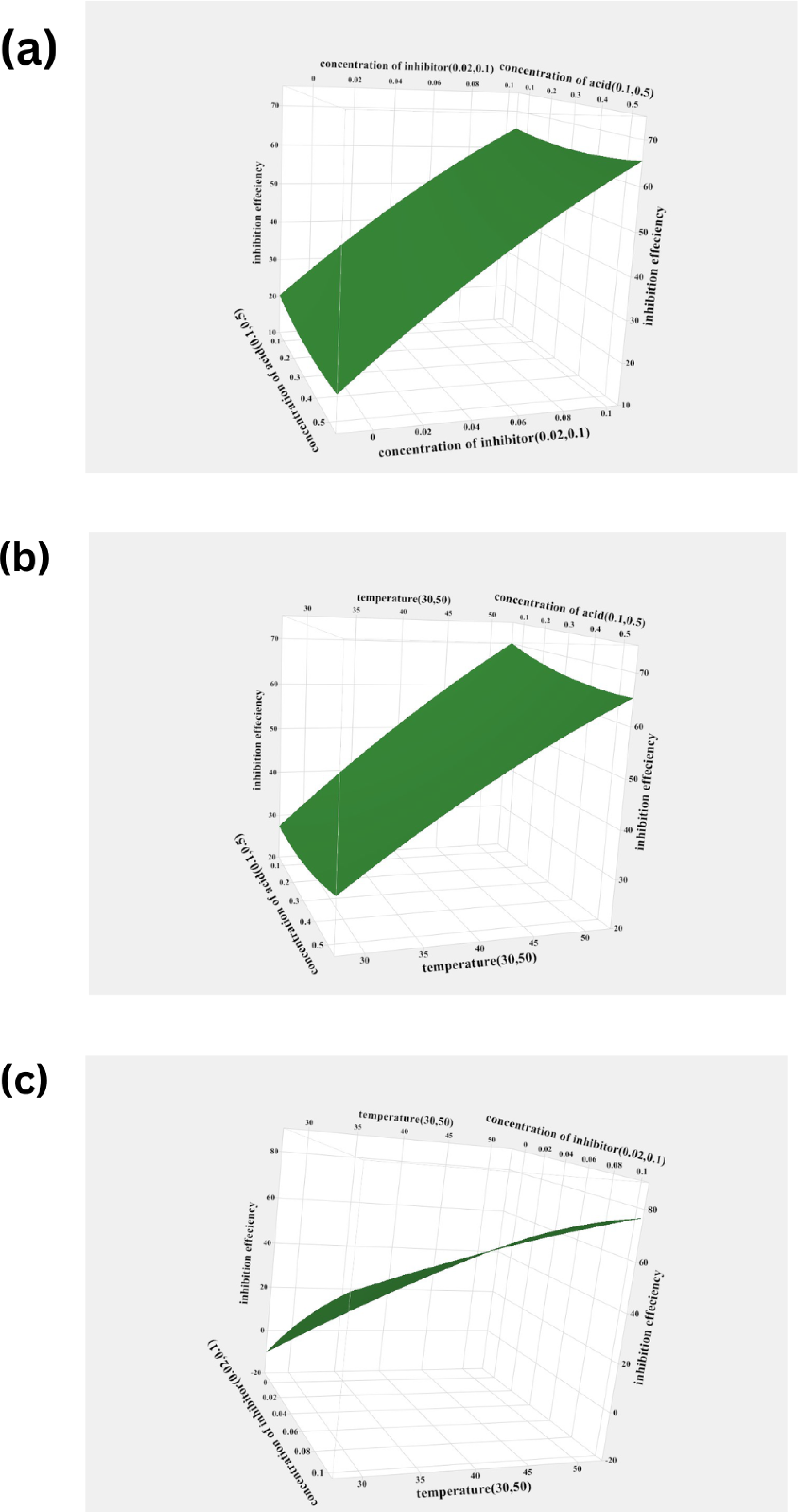
Surface plot for inhibition efficiency versus (a) X1, X2 (b) X1, X3 (c) X2, X3 at hold value (X1 = 0.3 M, X2 = 0.06 g/L, X3 = 40 °C).
Response optimisation
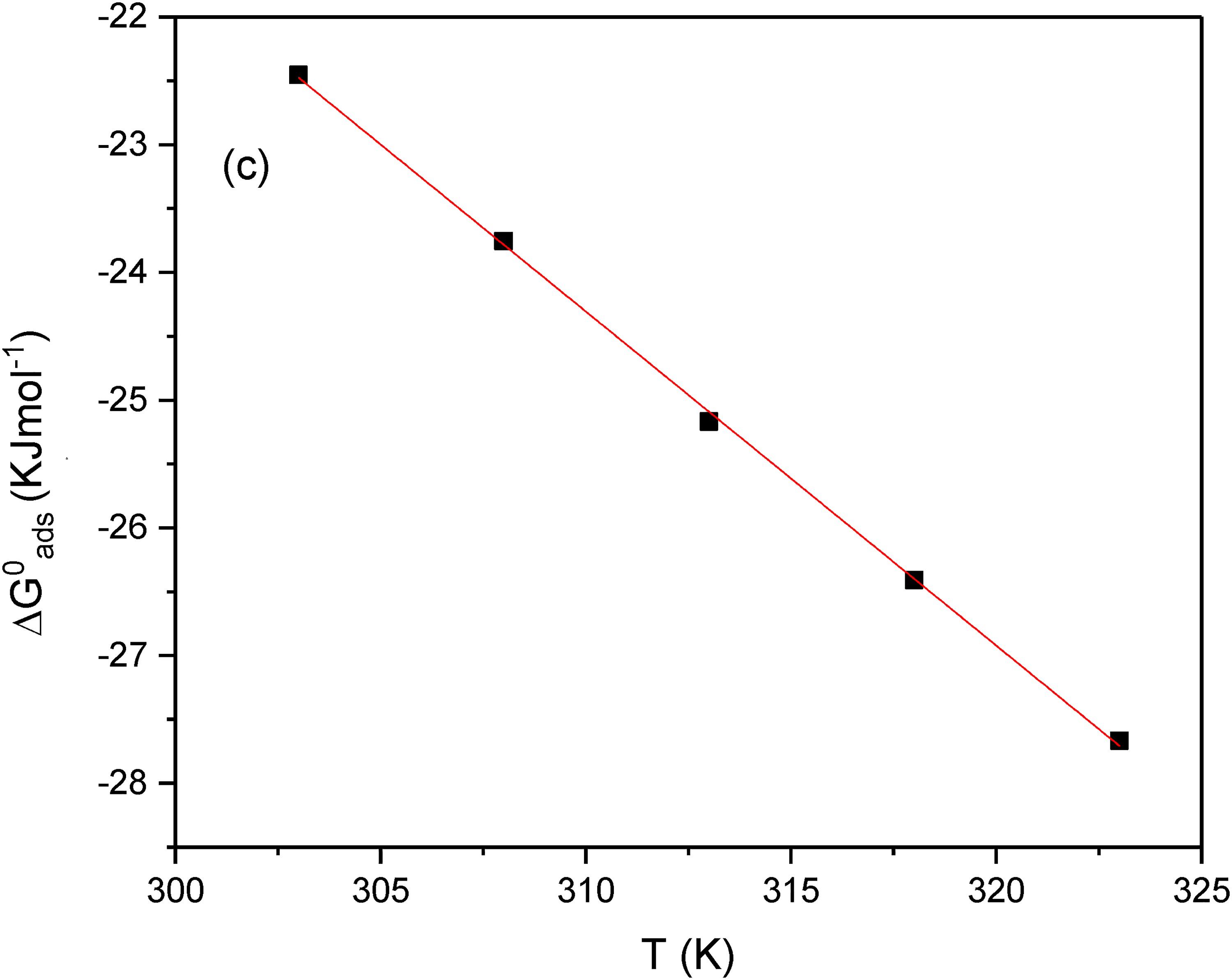
Optimisation of the response is performed to achieve maximum inhibition efficiency. The highest possible inhibition efficiency for AG was achieved by taking into account the desirability function approach. The optimisation is performed by setting the goal values for the factors and setting the inhibition efficiency to have maximum desirability. The predicted optimised parameters obtained, and the desirability is shown in Figure 6. Maximum IE % of 75.57% was predicted at 0.1 M H2SO4 concentration, 0.1 g/L of AG, and a temperature of 50 °C. The validation of the optimised result was performed by confirmation experiment in these factors. The Tafel parameters for the confirmation experiment is given in Table 6. Figure 7 shows the tafel plot for the confirmation experiment and the highest inhibition efficiency of 75.25% is achieved. Thus, validating the accuracy of the predictive mode of calculation.
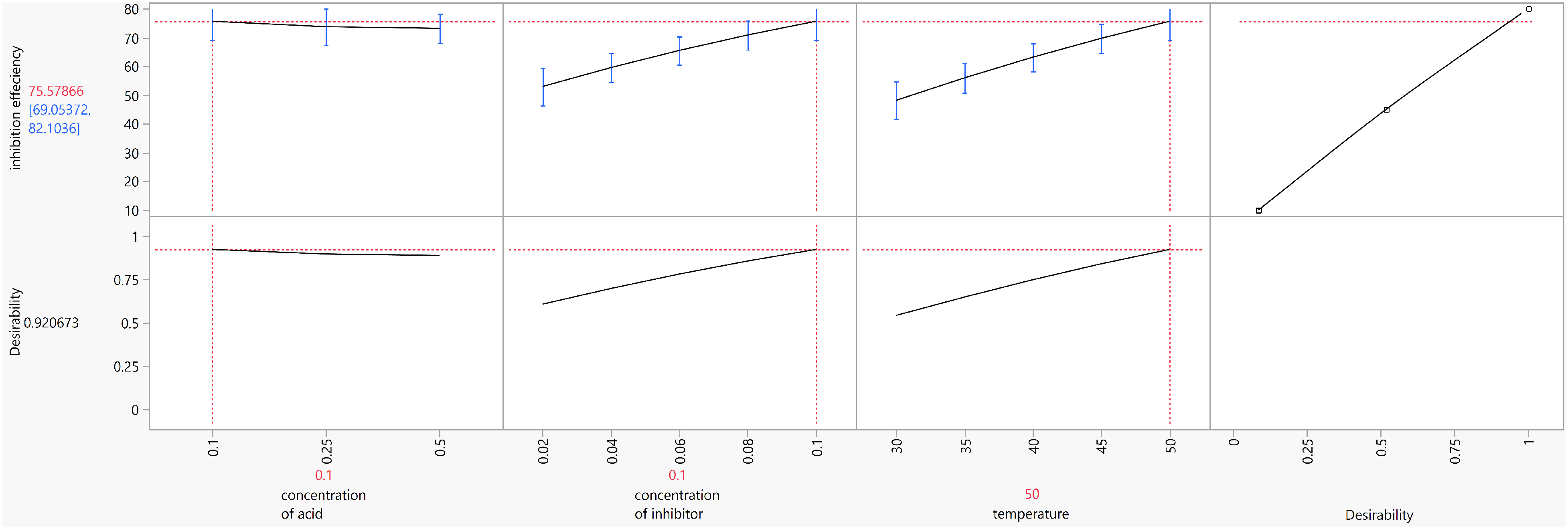
Response optimisation plot for inhibition efficiency.
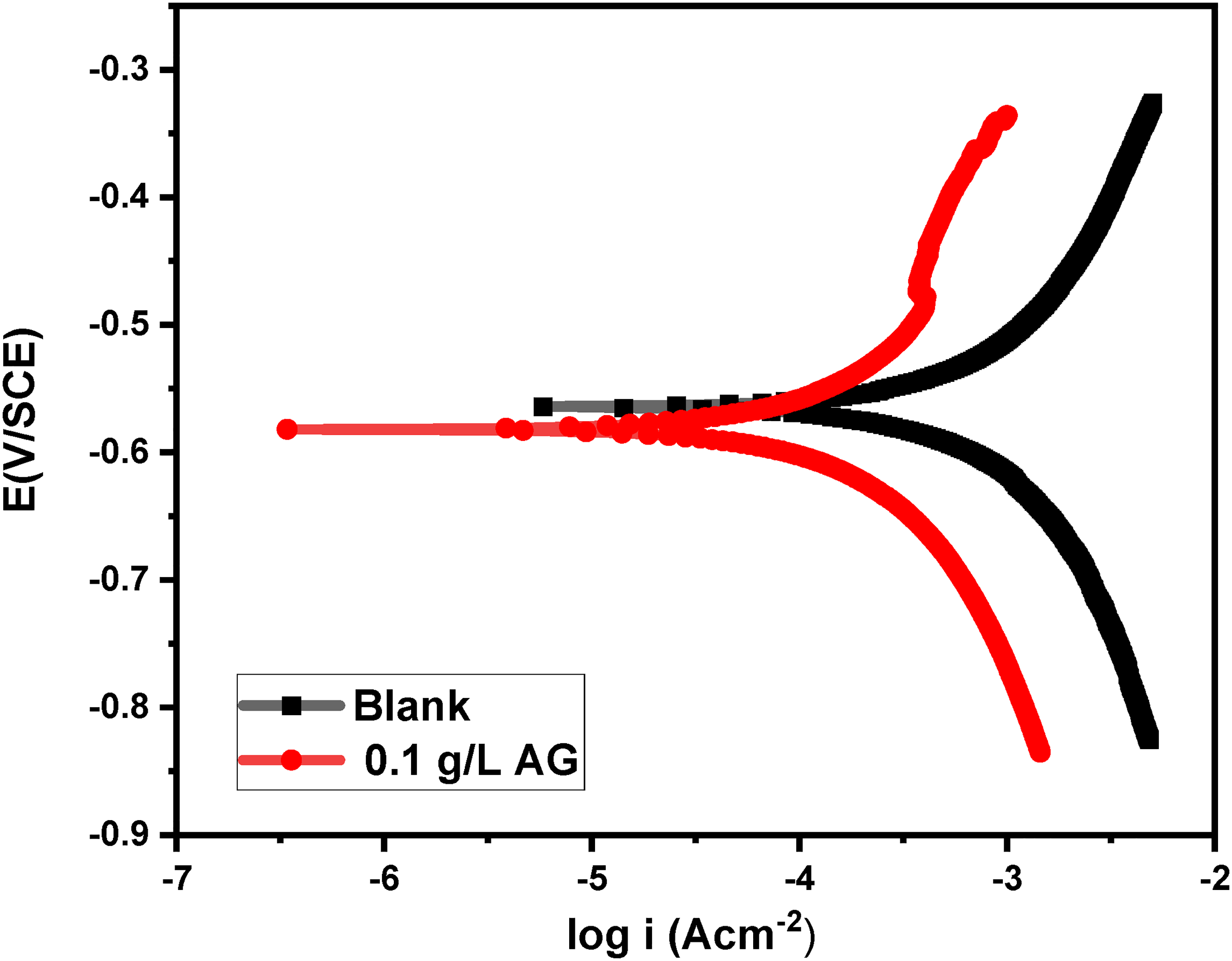
Tafel plot for corrosion inhibition of 0.1 g/L of AG in 0.1 M H2SO4 medium (IE% = 75.28).
Potentiodynamic polarisation parameters confirmation experiment of AG in 0.1 m H2SO4 medium at optimised parameters.
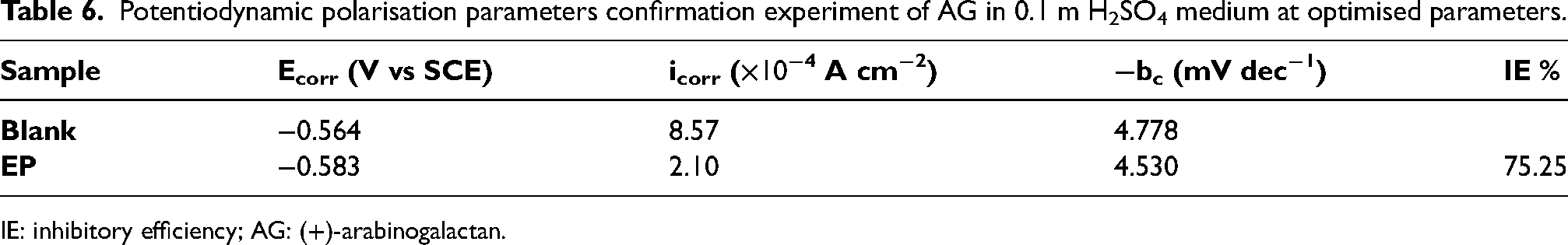
IE: inhibitory efficiency; AG: (+)-arabinogalactan.
Effect of kinetic and thermodynamic factors
The inhibition efficiency of AG was studied at different temperatures in H2SO4 (0.5 M) media. Irrespective of the media, the IE % elevates with the increase in temperature. As temperature rises, the active sites for corrosion also increase, and adsorption also increases. Furthermore, the activation parameters (activation energy (Ea), enthalpy of activation (ΔH#), and entropy of activations
Activation parameters for the corrosion inhibition study of (+)-arabinogalactan on annealed AISI 5140 steel in H2SO4 (0.5 M) medium.

AG: (+)-arabinogalactan.
Thermodynamic parameters.

Transition state theory is used to find the enthalpy of activation (ΔH#) and entropy of activation (ΔS#) given by
34
: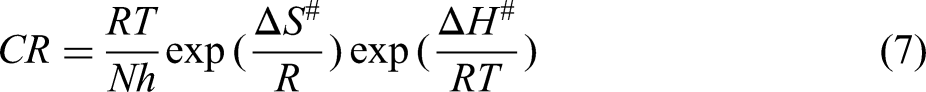
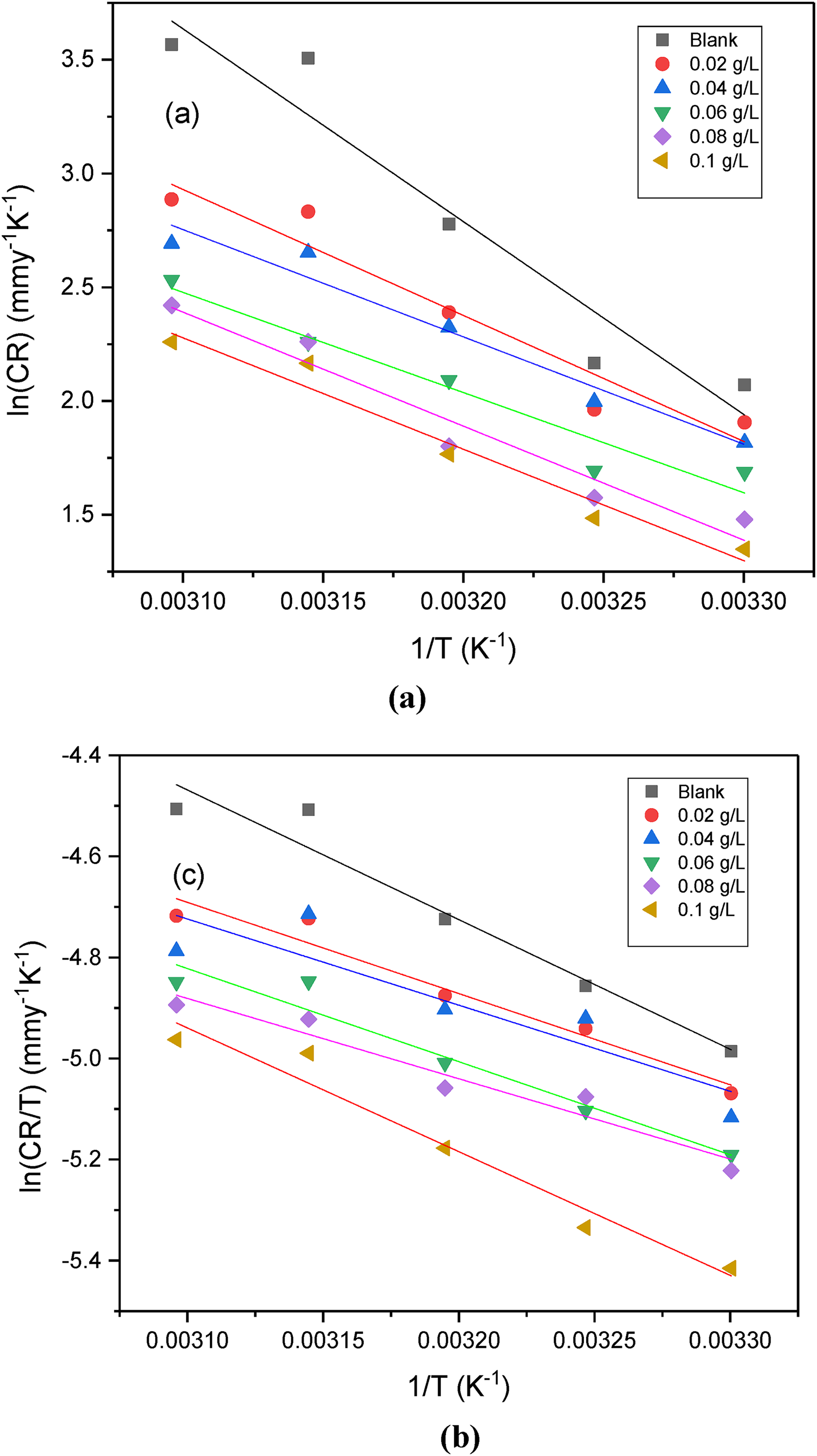
Plots of (a) ln (CR) versus 1/T (b) ln (CR/T) versus 1/T plot of annealed AISI 5140 steel in H2SO4 (0.5 M) solution.
Adsorption isotherm
To validate the adsorption of AG, adsorption isotherms are used. The best-fit adsorption isotherm is selected by fitting the data with adsorption isotherms such as Langmuir, Freundlich, Temkin, Flory- Huggins, etc. The best fit is considered for which the average correlation factor (vR2) is close to unity. The corrosion inhibition data of AG on AISI 5140 steel shows the best fit for Langmuir adsorption isotherm. The data gives a straight line when a plot of C/θ versus C is drawn (Figure 9). From this plot, various parameters such as equilibrium adsorption constant (K), R2 are calculated and shown in Table 7.
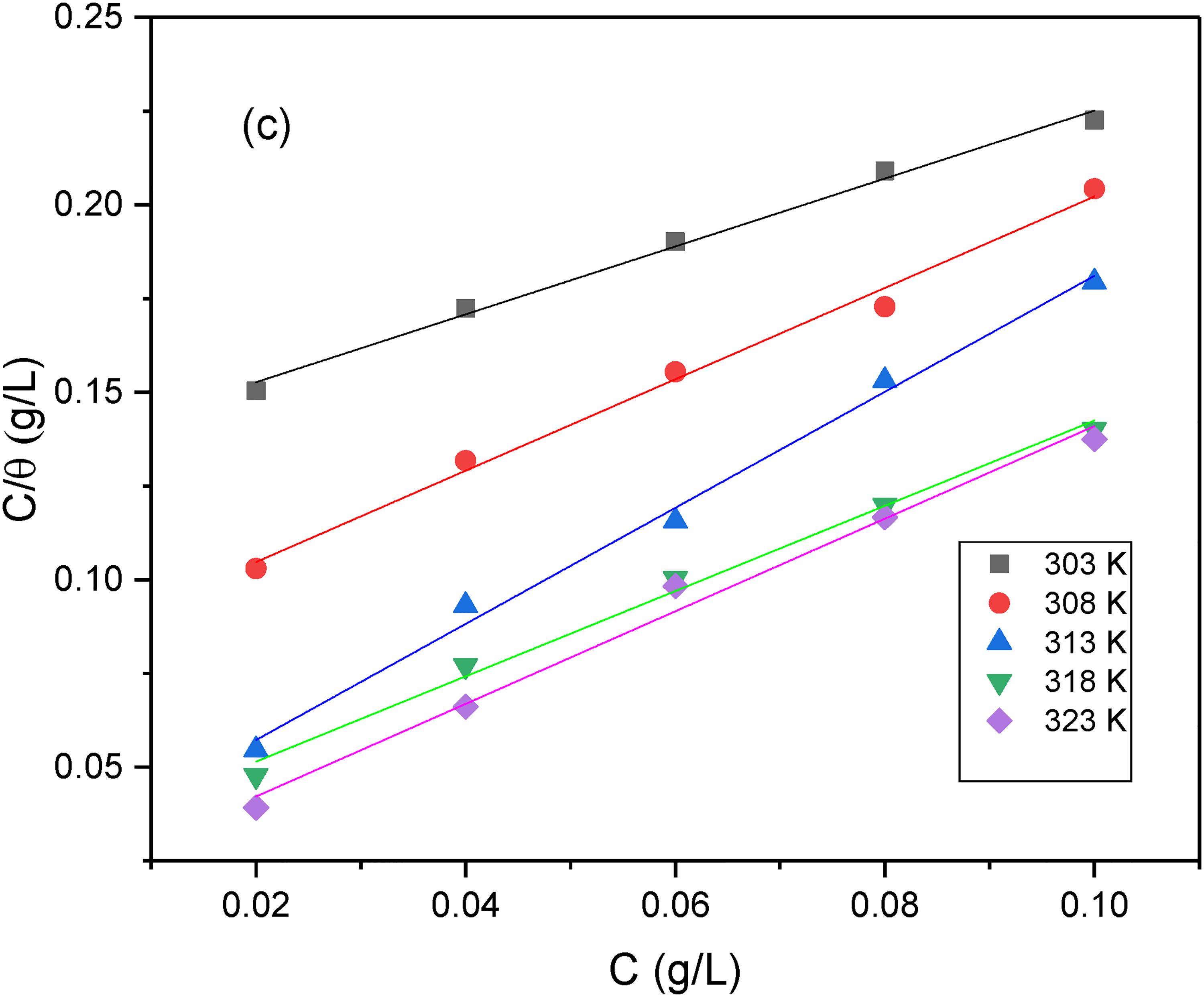
Langmuir adsorption isotherm for the adsorption of AG on 5140 steel in H2SO4 (0.5 M) medium.

Plot of
SEM and EDX analysis
SEM images of corrosion inhibition study of annealed AISI 5140 before and after the addition of AG in H2SO4 (0.5 M) is represented in Figure 11. The microscopic images reveal that the maximum smoothness of the surface is obtained when AG is added to the medium irrespective of the metal. The smoothness of the surface attained after the addition of AG is due to the adsorption of the molecule onto the metal surface. The adsorbed AG is found to be uniformly distributed, thus covering most of the cavities, thereby preventing corrosion.

SEM images of (a) freshly polished annealed AISI 5140 steel, (b) AISI 5140 steel + H2SO4 (0.5 M), (c) AISI 5140 steel + H2SO4 (0.5 M) (+)-AG.
The EDX images of the corrosion inhibition study of annealed AISI 5140 steel before and after the addition of AG in H2SO4 (0.5 M) is represented in Figure 12. The elemental composition of the analyzed specimen is given in Table 9. Since the backbone of the molecule AG is composed of C and O atoms, its adsorption leads to an increase in the C and O content when AG is added to the medium. From Table 9 it is clear that the addition of AG leads to an increase in the percentage composition C and O. This concludes that the adsorption of AG on the metal substrate and thereby prevents corrosion.
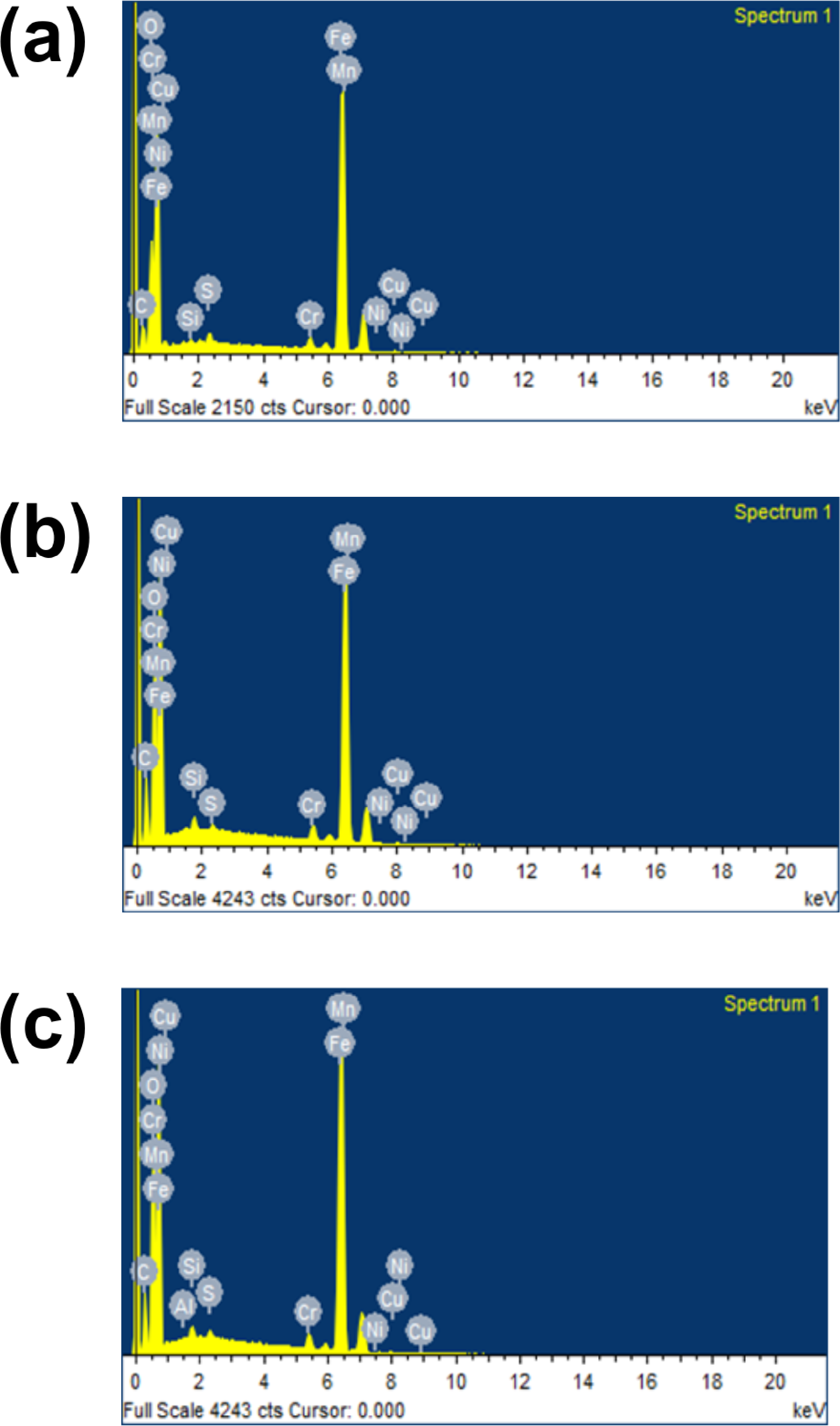
EDX analysis images of a) freshly polished annealed AISI 5140 steel b) AISI 5140 steel + H2SO4 (0.5 M) c) AISI 5140 steel + H2SO4 (0.5 M) +AG.
Results of elemental mapping of EDX measurement.

EDX: energy-dispersive X-ray; AG: (+)-arabinogalactan.
FT-IR of the adsorbed XL on the metal surface
The FT-IR spectrum of pure XL on ASI 5140 steel in the presence of XL is given in Figure 13. The bands at 3342 and 2900 cm−1 correspond to the stretching vibrations of –OH and –CH groups of XL. The H-O-H bending vibrations at 1635 cm−1 is also observed. The band at 1460 cm−1 arises due to bending vibration of –CH2 groups. A sharp peak at 1040 cm−1 corresponds to the C-O-C stretching of XL. A peak at 1375 cm−1 is due to the CH bending vibrations. 37 The characteristic band at 897 cm−1 indicates the stretching vibration of the β-(1,4) glyosidic linkage between the xylan molecules. 38 In the case of metal with XL, there is shift in these bands to lower frequency region and there is the presence of new bands are also observed. 39 This observation strongly suggests the presence of an interaction between xylan (XL) and the metal surface. The shift in the band at CO band at 1040 cm−1 indicates the coordination by Fe2+ of AISI 5140 steel with XL. The shift in –CO band also confirmed the same. The characteristic band at 897 cm−1 indicating the stretching vibration of the β-(1,4) glyosidic linkage between the xylan molecules are also prominently observed in case of XL on metal.
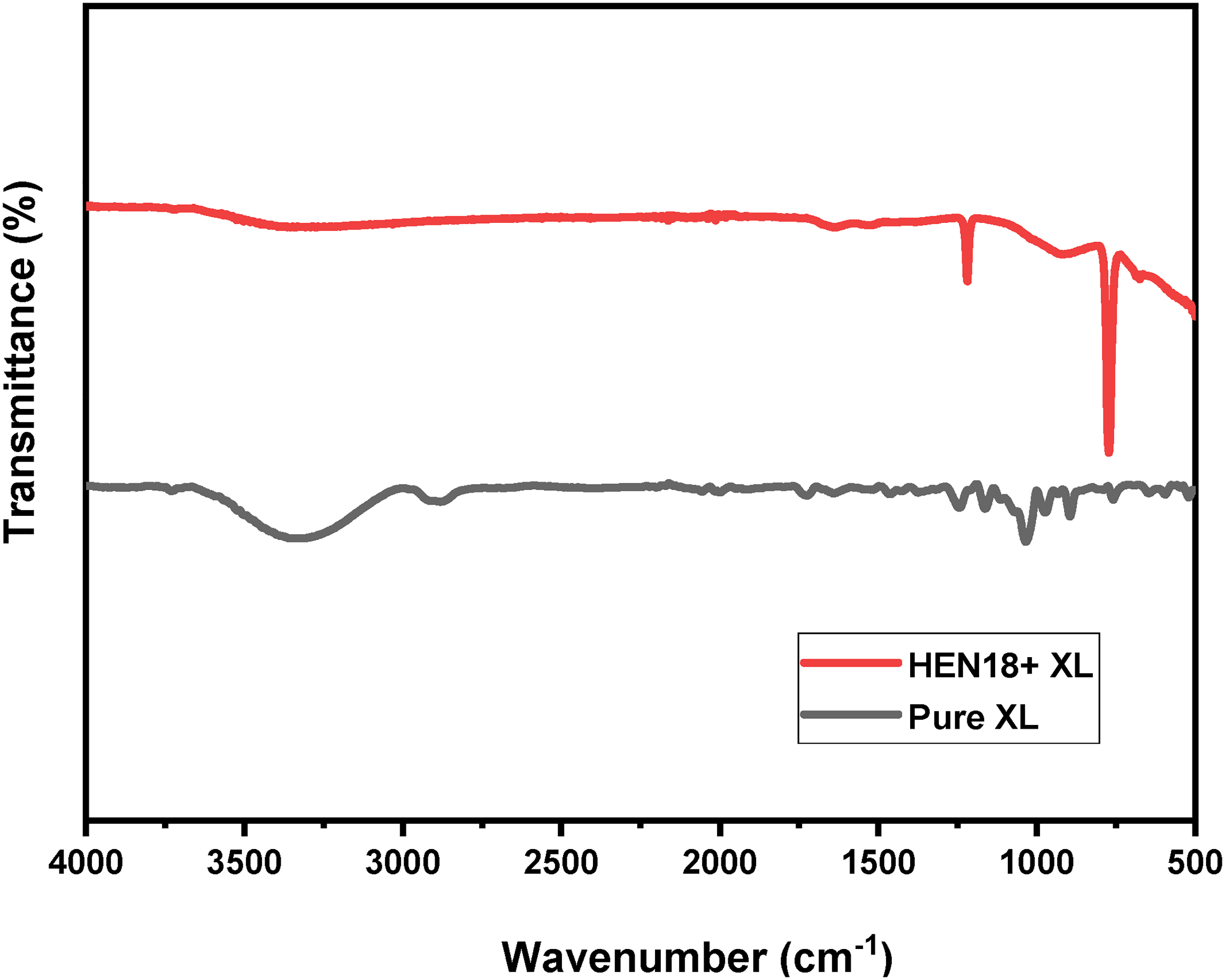
FT-IR spectrum of XL absorbed in annealed AISI 5140 steel in NaCl medium.
Mechanism of corrosion inhibition of AG
The corrosion inhibition mechanism can be classified as a surface phenomenon. The adsorption phenomenon is due to the replacement of the water on the metal surface with the inhibitor molecule present in the medium. 40 The corrosion inhibition property of AG can be attributed due to the presence of O atom in arabinose (the building blocks of AG; Figure 14). From the kinetic and thermodynamic studies, it is confirmed that the mechanism of AG adsorption is through chemisorption. The active cathodic sites are occupied by AG by competing with the H+ ions on the metal surface and form a metal–AG complex, thus diminishing the cathodic reaction. 41 The lone pair of electrons present in the O atoms forms a coordinate bond with the vacant orbital of the metal leading to the formation of a coordinate bond with the metal atom. 42
Mechanism of corrosion inhibition of AG on the surface of AISI 5140 steel.
Conclusion
This work illustrates the use of AG as a corrosion inhibitor for AISI 5140 steel and creates a model to optimise the parameters for this using DoE. The quantum chemical calculation of AG was performed and found that the negative charge is accumulated on the heteroatom (oxygen atom). This indicates the coordination of the molecule to the metal surface through these negatively charged centres. The DFT studies showed that the mechanism of inhibition can be due to the chemisorption mechanism of AG. This was further validated using DoE, where the linear increase in inhibition efficiency was observed with an elevation in temperature indicating a chemisorption mechanism. The kinetic and thermodynamic study proved that AG adsorption follows chemisorption. The Langmuir isotherm is found to be the best fit for the corrosion inhibition study of AG. A maximum inhibition efficiency of 75.57% was predicted at 0.1 M H2SO4 concentration, 0.5 g/L of AG, and a temperature of 50 °C. The SEM and EDX analysis confirmed that AG gets adsorbed onto the surface of the metals and prevents corrosion. This custom design approach of DoE was successfully utilised for predicting the corrosion inhibition study of AG. Thus, minimising the number of trials and optimising the results for this study.
Footnotes
Acknowledgements
The authors extend their acknowledgment to the Department of Chemistry and Central Instrumentation Facility, Manipal Institute of Technology (MIT), Manipal Academy of Higher Education (MAHE), Manipal, for carrying out the work and the characterisation facilities.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Manipal Academy of Higher Education (Grant No. MAHE/CDS/PHD/IMF//2021). The first author expresses gratitude to MAHE, Manipal, for intramural funding and the Dr TMA Pai scholarship.
