Abstract
Background:
Frailty correlates with poor clinical outcomes and is not routinely assessed in neurorehabilitation inpatient settings.
Methods:
We recruited adults from two neurorehabilitation units. We administered six validated tools for assessing frailty and collected data around length of stay, discharge, readmission and change in rehabilitation outcome measures.
Results:
Seventy-eight participants aged between 31 and 84 years were recruited with a range of neurological diagnoses. Frailty prevalence ranged between 23% and 46%, depending on the scale used, with little agreement between tools. Frailty status did not correlate with age, gender, length of stay, discharge destination and rehabilitation outcome measures. One-year readmission was higher in participants rated as frail by the Frail-Non-Disabled Questionnaire, the FRESH-screening questionnaire and the Clinical Frailty Scale.
Conclusion:
Frailty ascertainment was variable depending on the tool used. Three frailty indices predicted readmission rate at 1 year but no other outcome measures. Therefore, frailty tools may have limited utility in this clinical population.
Introduction
Frailty describes loss of physiological reserve and vulnerability to adverse events. It was initially described in older people because it increases in prevalence with age and has explanatory value in understanding how older people respond differentially to acute illness. 1 Frailty is associated with adverse outcomes regardless of age. 2 The term ‘frailty’ should not be used in populations with life-long impairments but may help understand other patient groups with acquired disabilities who deteriorate physiologically in response to acute insults.
Prognostication in neurorehabilitation is difficult because of multiple complex interacting exposures that determine outcomes. 3 Identifying (and addressing) frailty may improve outcomes or help to inform discussions around prognosis and treatment planning.
Two main paradigms describe and measure frailty. The first describes a cascade of homeostatic decompensation and is measured by the presence or absence of physiological parameters (phenotypic frailty). 4 The second counts deficits across a number of domains and expresses severity of frailty as a ratio of deficits present to potential deficits (accumulation of deficits). 5 Both models select different individuals as being frail but predict adverse outcomes equally well and consensus papers 6 have suggested that frailty indices should be selected based upon feasibility and utility in a given clinical context. Most frailty indices have been designed and are primarily used in older populations, and caution should be used when applying them to younger populations. 7
Against this background, we set out to evaluate frailty indices in inpatients undergoing neurorehabilitation with the following objectives:
To compare the prevalence of frailty as measured by different frailty measures
To assess differences in frailty between different diagnoses and ages
To assess whether frailty status impacted rehabilitation outcomes
To assess feasibility of assessing frailty using questionnaires during inpatient neurorehabilitation
To assess ease of use of commonly used questionnaires either with patient or next of kin
Methods
This was a two-site study in the East Midlands of England. The sites comprised a 19-bedded level 2b unit and a 25-bedded level 2a unit based within large NHS acute provider trusts.
The study was sponsored by the University of Nottingham and received ethical approval from Yorkshire and The Humber – Leeds East Research Ethics Committee (ref: 18/YH/0361).
Participants were recruited between February 2019 and December 2020, with a hiatus between March and July 2020 (Derby) and March and September 2020 (Nottingham) due to the COVID-19 pandemic.
Patients were eligible for inclusion if they were inpatients in participating units for purposes of neurorehabilitation and aged 18 years or older. They were excluded if they or a consultee were unable or unwilling to give consent.
Potential participants who had capacity to consent to participate in the study gave written informed consent. Participants who lacked capacity and could not give consent were recruited following a discussion with a relevant consultee, such as a family member or friend, in keeping with the provisions of the Mental Capacity Act 2005. 8
Baseline constituted the date of admission to the rehabilitation unit. Data collection at baseline comprised participant demographic and clinical details, including diagnosis, comorbidities, age, gender, date of admission to rehabilitation unit and frailty status.
We selected frailty assessment tools which:
Asked about the period of time before the hospital admission as being representative of a patient’s baseline function
Did not require invasive testing for example, blood tests/muscle biopsies
Did not rely on tests which could have been altered by recent inpatient stay or intercurrent illness (e.g. serum albumin)
Could be answered by next of kin/consultee if the participant was cognitively impaired and unable
Could be carried out quickly and simply on an inpatient unit, with minimal disruption to daily care
Had been used and validated in clinical settings
Required no special training to perform
On this basis we recorded:
The Clinical Frailty Scale (CFS) – uses a nine-item scale to categorise individuals on the spectrum of ‘very fit’ to ‘terminally ill’ 9
Fatigue/Resistance/Ambulation/Illness/Loss of weight scale (FRAIL) – a five-item questionnaire (covers fatigue/resistance/ambulation/illnesses/loss of weight); individuals are divided into ‘frail’, ‘pre-frail’ or ‘robust’ depending on their score 10
Frail non-disabled questionnaire (FIND) – a brief, five item questionnaire which asks about walking, climbing stairs, weight loss, fatigue and physical activity; it categorises respondents into ‘robust/disabled/frail/frail and disabled’ 11
FRESH-screening instrument – a four-question tool (around mobility/fatigue/falls/assistance); individuals are characterised as ‘normal’ or ‘frail’ 12
Groningen Frailty Index (GFI) – a 15-question questionnaire that covers the domains of mobility, vision, hearing, nutrition, comorbidity, cognition, psychosocial and physical fitness, with the total score dividing respondents into ‘frail’ or ‘non-frail’ 13
Modified Reported Edmonton Frail Scale (m-REFS) – removes the question which asks the patient to draw a clock and instead replaces it with a question around a history of cognitive impairment, shown to be equivalent to the Reported Edmonton Frail Scale in a prospective cohort study. 14 The original Edmonton frail scale is valid and reliable 15 ; this had been modified slightly to allow it to be answered by next of kin and trialled successfully. Respondents are classified as ‘not frail’/‘apparently vulnerable’/ ‘mildly frail’/‘moderately frail’/‘severely frail’. 14
For the sake of simplicity, the scores were divided into ‘frail’ and ‘non-frail’/‘robust’ as shown in Table 1:
Division of frailty scores into frail versus non-frail/robust according to numerical score.

CFS: clinical frailty scale; FIND: Frail Non-Disabled Questionnaire; FRAIL: Fatigue/Resistance/Ambulation/Illness/Loss of weight scale; FRESH: FRESH-screening instrument; GFI: Groningen Frailty Index; M-REFS: Modified Reported Edmonton Frail Scale.
The following data were collected at baseline and at discharge from the rehabilitation unit:
Functional Independence Measure – a global measure of disability combined with the Functional Assessment Measure to create FIM+FAM which is designed to measure disability in people with brain injury, where a higher score equates to more independence 16
Northwick Park Nursing Dependency Scale, which is designed to measure patient care needs/nursing requirements in rehabilitation settings, where a lower score equates to more independence 17
Rehabilitation Complexity Scale – an overall measure of medical, nursing, therapy, equipment and basic care needs, where a lower score indicates more independence 18
Northwick Park Therapy Dependency Assessment – a measure of therapy intensity required in a rehabilitation unit, where a higher score indicates higher need 19
Additional data collected at discharge comprised length of stay, discharge destination and whether a care package was required.
Further follow-up was conducted at 12 months to collect data on mortality and readmission to the same hospital over the year since recruitment. The primary outcome was readmission over 12 months post-discharge to the hospital where the patient had undergone rehabilitation for any reason.
A full schedule of measurements is shown in Supplemental Appendix 1.
Power calculation, data handling and statistical analysis
Readmission was chosen as the primary outcome because readmission is common in neurorehabilitation, occurring in up to 1/3 of patients in the year after discharge. 20 In non-rehabilitation studies, readmission is reported to be 2.8 times more common in frail than non-frail patients. 21 Estimating a 1-year readmission rate at 24% and estimating this to be three times higher in a frail than a non-frail group, and assuming equal numbers of frail and non-frail patients, for a power of 0.8, alpha (type i) error of 0.05 and beta (type 2) 0.2, a sample size of 228 was required (calculated using http://clincalc.com/stats/samplesize.aspx). Excel and GraphpadPrism 9 for Windows, GraphPad Software, Boston, Massachusetts USA, www.graphpad.com were used for statistical analysis.
Baseline data were reported using descriptive statistics, with central tendency reported using mean (SD) and median (IQR) for parametric and non-parametric variables, respectively.
Agreements between frailty indices were assessed using the kappa statistic, with level of agreement classified according to the recommendations of Landis and Koch. 22
Differences between groups for outcomes were reported using Mann-Whitney U test for non-parametric linear or ordinal variables and Fisher’s exact tests for categorical variables.
Results
Seventy-eight participants were recruited (50 and 28 from the level 2b and level 2a units, respectively). Median age was 57 years.
Recruitment rates
It was possible to collect details regarding admissions from the 19-bedded 2b unit. There were two active recruitment periods – 15 February 2019–20 March 2020 (halted by COVID), then restarted 2 June 2020–20 November 2020. During this time, 56 potential participants were approached and 50 patients were recruited. During this period, there were 205 admissions for neurorehabilitation to the unit, so 56/205 = 27% of those admitted were approached and 50/56 = 89% of those approached consented to participation.
The potential participants/consultees who were approached but declined, declined for the following reasons:
4 no reason given
1 too busy
1 didn’t want to talk about pre-morbid level of function
Similar details were not available from the other site.
Participants from the two sites were comparable in terms of age, gender, diagnosis, comorbidities, frailty and rehabilitation outcomes (all p values >0.05), so the two units’ data are combined hereafter.
Table 2 gives details of participant characteristics on admission.
Participant characteristics with primary reason for admission and details of comorbidities.

SD: standard deviation.
Questionnaire completion
Four hundred fifty-nine (of a total of 468 = 98%) questionnaires were completed in full. Data were missing from one GFI, one FRESH screening and seven FRAIL questionnaires. The most common reason for incomplete questionnaires was missing data about weight loss.
Several participants and/or consultees became briefly tearful or upset when discussing things that they had previously been able to do but were no longer able to. Fortunately, this resolved quickly in all affected participants, and in no cases did data collection have to stop because of participant or consultee distress.
Prevalence of frailty and agreement between questionnaires
Depending on the questionnaire used, between 23% and 46% of respondents were classified as frail (Figure 1 and Supplemental data).

Representation of frailty scores by diagnosis. Left column indicates diagnosis (GBS: Guillain Barre syndrome; ICH: intracranial haemorrhage; MS: multiple sclerosis; TBI: traumatic brain injury). Each individual row represents a patient with the specified diagnosis. The green, red and grey squares indicate the outcome of each frailty assessment tool – red indicates that that patient was assessed as being frail by the specific tool; green indicates non-frail and grey indicates that the questionnaire was not completed.
There was ‘substantial’ (kappa 0.61–0.80) agreement between
FRESH-scale/CFS
GFI/m-REFS;
‘moderate’ (kappa 0.41–0.60) agreement between
FRESH-scale/FRAIL scale
FRESH-scale/FIND
FRESH-scale/m-REFs
FRAIL scale/CFS
FRAIL-scale/FIND
CFS/FIND
CFS/GFI
CFS/m-REFS
FIND/GFI
FIND/m-REFS
(see Supplemental data for details).
No associations were seen between frailty and age or gender (see Supplemental data for details)
Diagnosis was associated with frailty status. Higher rates of frailty were seen in people with multiple sclerosis (MS – 75–100% prevalence depending on questionnaire used) than with other conditions. Lowest rates were seen in people with traumatic brain injury (TBI; 0–42%) (see Supplemental data for details).
Rehabilitation outcomes showed wide variation between participants. Median length of stay was 55 days (range 9–246 days); most people completed their inpatient rehabilitation episode without being transferred off the unit (median interruptions to stay = 0) and the most common discharge destination for participants was to their own home, but frequently with a package of care (62% of participants). (Further details available in Supplemental data).
No statistically significant differences were seen between frailty categories and length of stay, interruptions to stay, discharge destination, changes in rehabilitation measures or 1-year mortality. Chance of readmission was higher in those who scored as frail by the FRESH scale, the CFS and the FIND Index (Table 3). However, allowing for multiple comparisons in the analysis, these should be interpreted with caution.
Readmission rates by frailty status and questionnaire used. Example: data were available from 76 participants who had the FRESH-screening questionnaire completed. Of those, 31 had been readmitted in the 12 months post-discharge; 15 of those readmitted were classified as frail and 16 were classified as non-frail/robust. Forty-five participants were not readmitted in the year following discharge: 8 frail and 37 robust/no-frail participants.
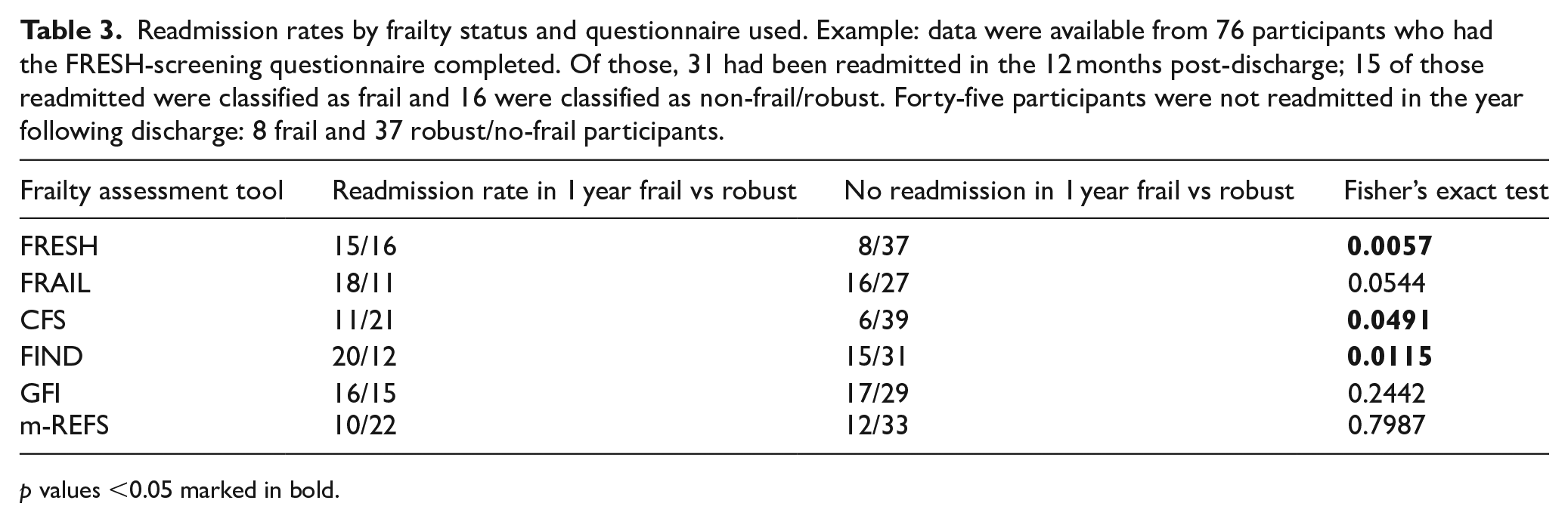
p values <0.05 marked in bold.
Discussion
This UK-based cross-sectional study examined use of frailty assessment tools in a neurorehabilitation inpatient setting. Our main findings were a high prevalence of frailty in patients admitted for neurorehabilitation, ranging from 23% to 46%, depending on the scale used. There was no association between age or gender and classification as frail or not frail. Three frailty indices – FRESH, CFS and FIND – were associated with increased risk of readmissions at 1 year. No associations were seen between frailty and rehabilitation outcomes at discharge or at 1 year.
While we endeavoured to conduct a pragmatic study that could be delivered within a busy, often short-staffed and time-pressured NHS environment, there were some weaknesses in this study. We were only able to recruit around 1/3 of our target despite extending our end date twice. The COVID-19 pandemic was, of course, a key limiting factor, with further reductions in staffing levels, restrictions to patient visiting, changes in lengths of stay and pause on research by the two participating NHS Trusts. It is, therefore, difficult to draw definitive conclusions, particularly around associations with 1 year readmission. We were only able to collect data around readmission to the hospital where the patient had undergone their rehabilitation and did not collect data around reason for readmission, which is also a limiting factor, as patients may have had admissions to other hospitals, and reasons for admission may not have been related to their neurological diagnosis.
We included participants with all diagnoses undergoing neurorehabilitation in our study. Amalgamating ‘all neurorehabilitation inpatients’ may not be appropriate for this sort of work, and future work could perhaps differentiate between those with exacerbations of pre-existing conditions (notably MS, where there was a high level of frailty) versus acute onset for example, trauma or first episode of Guillain Barre syndrome (GBS). Some factors associated with frailty may also be associated with people being admitted for neurorehabilitation – for example, falls are a common cause of TBI 23 ; adults who fall are likely to be older 24 and taking multiple medications. 25 The same associations are not seen in some other conditions requiring neurorehabilitation for example, GBS. However, the individual case numbers in this study were too small for such subgroup analysis.
The tools we selected had some limitations. There were no assessments of current (or even ‘on admission to rehabilitation unit’) physical or cognitive function. This was by design, as we wished to employ measures that could be used fairly universally, irrespective of cognitive or physical impairment at the time of completion. However, by excluding physical, cognitive or laboratory assessments, we may be missing valuable data. We selected measures to focus on patients’ frailty in the immediate period before their hospital admission, believing this to be most representative of their baseline. However, many patients who are admitted to neurorehabilitation units have had lengthy hospital inpatient stays before they reach the rehabilitation unit – they may have had weeks or months in the Intensive Care, Major Trauma or Neurosurgical Units, and they may have accumulated a significant pathophysiological burden during that time, with repeated infections, surgeries and nutrition issues to name only a few. It may, therefore, be relevant in the future to assess frailty on admission to the rehabilitation unit, as well as before admission.
Some of the items within the questionnaires may not have been answered accurately by respondents or their consultees. Recall and self-report can be inaccurate, with self-reports tending to over-estimate physical activity 26 and under-estimate weight. 27 Furthermore, three of the questionnaires (FRAIL scale, FIND, GFI) ask about specific quantities of weight loss. Self-weighing is not universal (25% of adults in the USA reported never weighing themselves 28 ), so the responses to these questions may not be entirely accurate.
We chose to dichotomise the scales (frail versus robust/non-frail) for ease of comparison, but the scales are not all designed to be binary in this way, so this artificial dichotomisation may have added a further error.
Patients with MS were most likely to be classified as frail, which is in keeping with previous work.29,30 However, the majority of scales used to include questions around mobility and fatigue, both of which are commonly impaired in people with MS31,32, and this may reinforce the need to develop tools to distinguish between disability and frailty while recognising their complex interactions.33,34
The poor correlation between individual frailty scales is compatible with previous research, which found heterogeneity between scales. 35
The increased rate of readmission in frail individuals, although needing to be interpreted with caution from our own findings, has been previously described in medical and surgical patients.36,37
While we recognise that frailty is an important concept, one of the reasons why it may not perhaps be as immediately relevant in the neurorehabilitation population may be due to the ‘accumulated deficits’ model – which could be conceptualised as multiple small factors interacting to increase physiological burden and risk. Many participants in this study, and being admitted to neurorehabilitation units, had experienced a major, often acute event (trauma, infection, stroke etc.) and one might conceptualise that the overall ‘weight’ of one such incident may ‘outweigh’ the smaller accumulated deficits in frailty. This is speculation but a potentially interesting area for future work.
Supplemental Material
sj-docx-1-rcp-10.1177_14782715241242509 – Supplemental material for The use of frailty questionnaires in inpatients in two neurorehabilitation units in the East Midlands – A cross-sectional cohort study with follow-up to 1-year after discharge from inpatient rehabilitation
Supplemental material, sj-docx-1-rcp-10.1177_14782715241242509 for The use of frailty questionnaires in inpatients in two neurorehabilitation units in the East Midlands – A cross-sectional cohort study with follow-up to 1-year after discharge from inpatient rehabilitation by Laura Edwards, Vina Tesorero, Fattaneh Zonouzi, Piera Santullo, Phoebe Owen and Adam L Gordon in Journal of the Royal College of Physicians of Edinburgh
Footnotes
Acknowledgements
All of the patients and their families and consultees who took part in the study.
University of Nottingham as sponsor, University Hospitals of Derby and Burton NHS Foundation Trust and Nottingham University Hospitals NHS Trust Research and Development/Innovation Departments.
Members of the multi-disciplinary teams on King’s Lodge and Linden Lodge who were able to assist in areas such as capacity assessment and communication.
Morag Sime for all her hard work.
Margaret Phillips and Mary Antoun for local support.
Jacqueline Middleton for help with UKROC data.
Author contributions
Design: LE, ALG; Acquisition of data: LE, PS, VT, FZ, PO; Analysis of data: LE, ALG.
All authors have critically revised the manuscript and approved the final version for publication.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Adam Gordon is part-funded by the NIHR Applied Research Collaborations for East Midlands (ARC-EM) and is an NIHR Senior Investigator. The views here are those of the authors and not necessarily those of the NHS, the NIHR or the Department of Health and Social Care.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
