Abstract
Venous thromboembolism (VTE) is frequently encountered across various specialties. The management of VTE has become more nuanced, requiring consideration of several factors when deciding on the choice and duration of anticoagulation. This evidence-based review article summarises the current practice and evidence behind anticoagulation in VTE, incorporating national and international guidelines. Factors influencing decision-making around the choice and duration of anticoagulation, along with special circumstances such as cancer and antiphospholipid syndrome, are discussed. The clinical utility of thrombophilia screening is also addressed.
Keywords
Introduction
Venous thromboembolism (VTE) is a common condition, affecting 1 in 12 people in their lifetime in the Western world. 1 VTE occurs when there is dysregulation to natural haemostasis, typified through Virchow’s triad: abnormalities in the vessel wall, blood flow and blood constituents (Figure 1). 2 VTE presents as deep venous thrombosis (DVT) in two-thirds of patients and as pulmonary embolism (PE) in one-third of patients (occasionally with concomitant DVT). VTE has a range of complications including immediate (pain, hospitalisation, death) and delayed (recurrence, post-thrombotic syndrome and chronic thromboembolic pulmonary hypertension). 3
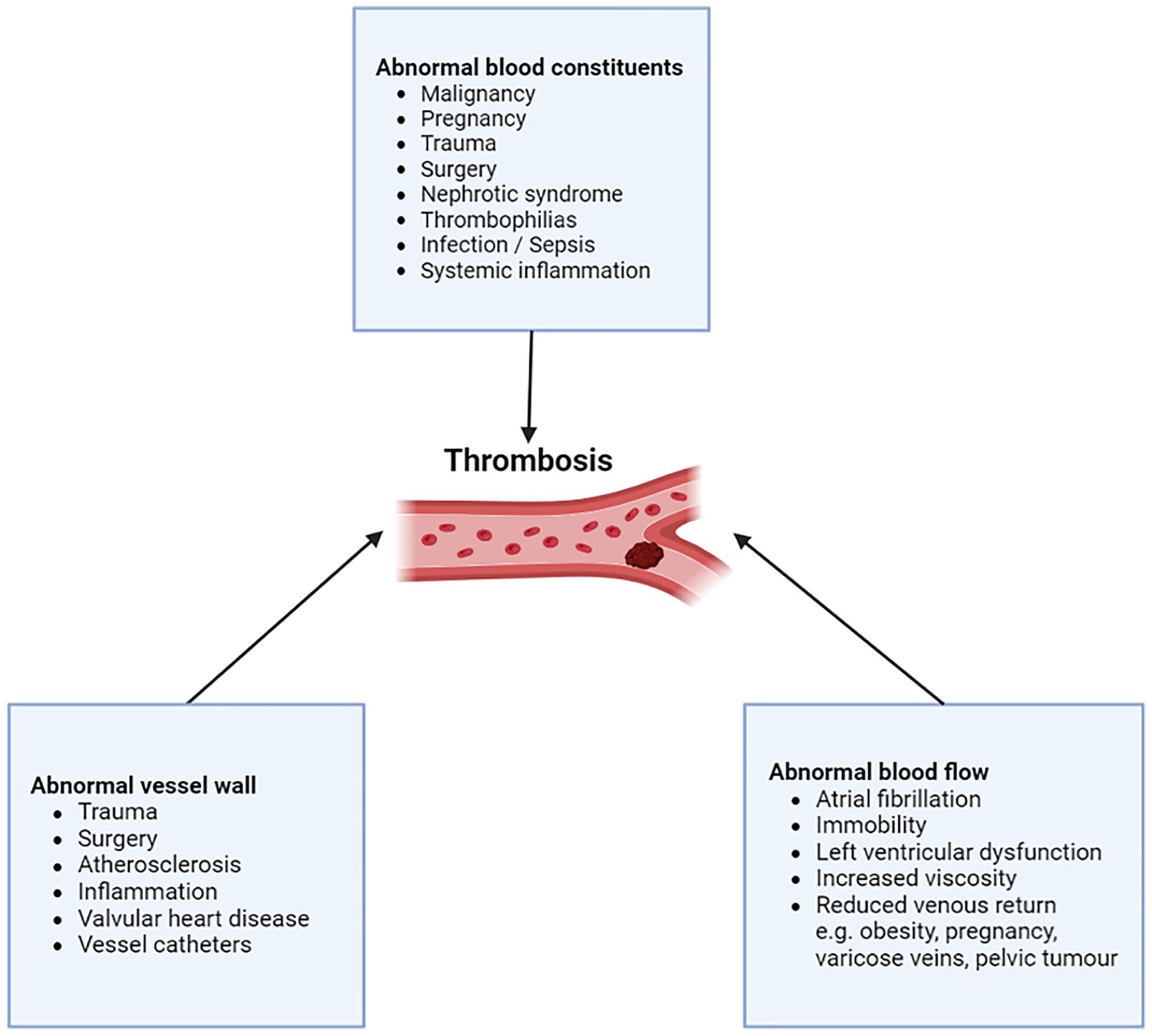
Virchow’s triad describes the three categories of factors that contribute to abnormal thrombus formation. Image created using Biorender.com.
The effectiveness of anticoagulant therapy in managing VTE is well established. Nonetheless, the decision-making on anticoagulant use has become more complex, due to rising co-morbidities and an ageing population. Anticoagulants are initially recommended for at least 3 months. Subsequently, a decision should be made on whether anticoagulation can be safely discontinued or be continued indefinitely for secondary prevention. Accurately predicting the risk of VTE recurrence is at the heart of this decision and is balanced against bleeding risk. This is important since both VTE recurrence and bleeding related to inappropriate anticoagulation are associated with significant morbidity and mortality. 3 The case fatality rate from one prospective registry study for recurrent VTE and major bleeding were 12.1% and 19.7%, respectively, during anticoagulation. 4 Notably, the advent of direct oral anticoagulants (DOACs), which have considerably lower bleeding rates, 5 has altered this risk–benefit balance.
In this evidence-based review article, we discuss various aspects of anticoagulation in the management of VTE for the general physician (Figure 2). We focus on decision-making around the duration and choice of anticoagulation, and how to assess risks of VTE recurrence and bleeding. We also discuss when thrombophilia testing should be considered since this may alter the choice of anticoagulation. In preparing this article, we reviewed numerous national and international guidelines.6 –11 We summarise and present key points for a general audience.
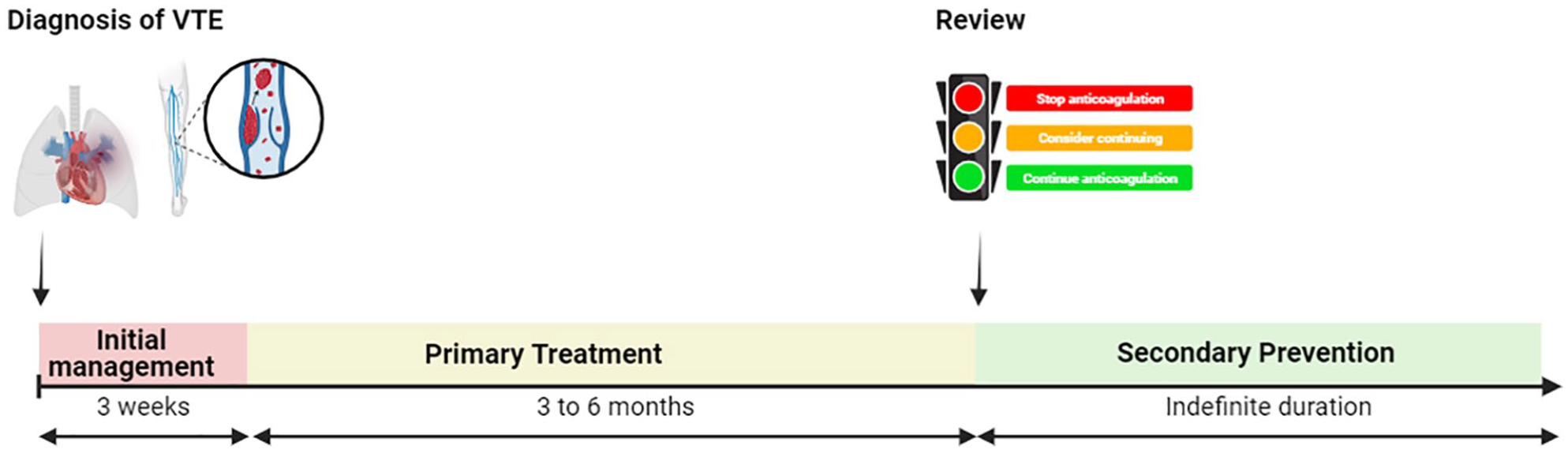
Key decision time-points in management of venous thromboembolism. Image adapted from reference. 8 Image created using Biorender.com.
Initial management
The initial management of VTE is aimed at addressing life- or limb-threatening complications and preventing thrombus propagation. Treatment considerations here include the following:
Place of care – both National Institute of Health and Care Excellence (NICE) 6 and British Thoracic Society 12 guidelines support outpatient management of DVT and low-risk PE after appropriate risk stratification.
Thrombolysis – patients who have PE with haemodynamic instability or DVT with significant compression may require systemic or local catheter-directed thrombolysis. This has been reviewed elsewhere. 13
Inferior vena cava (IVC) filter placement – in acute VTE, placement of a temporary retrievable IVC filter can be considered if there are contraindications to adequate therapeutic anticoagulation (such as a significant bleeding risk). This has been reviewed elsewhere. 14
Anticoagulation – with consideration of route of administration, need for monitoring, drug–drug interactions, patient comorbidities and reversibility.
Primary treatment
Primary treatment represents the minimal duration of anticoagulation for VTE. Duration and choice of anticoagulant need to be carefully considered.
Choice of anticoagulant
Warfarin, a vitamin K antagonist (VKA), was once considered the oral anticoagulant of choice. Anticoagulant prescribing trends have since changed 15 due to warfarin’s several drawbacks and the availability of the DOACs. These limitations include a slower onset of action, requirement for preceding parenteral anticoagulation, narrow therapeutic window, frequent blood international normalised ratio (INR) monitoring, and many drug–drug and drug–dietary interactions. National 6 and international 8 guidelines recommend DOACs as the initial choice of anticoagulation in patients with VTE, assuming no contraindications. This recommendation is based on several randomised controlled trials (RCTs) and meta-analyses 8 showing non-inferior efficacy and a lower major bleeding risk compared to low molecular weight heparin (LMWH) and warfarin. In one pertinent meta-analysis, the incidence of major bleeding during extended anticoagulation was 1.74 per 100 patient-years for VKAs compared to 1.12 per 100 patient-years for DOACs. 5 There has also since been the development and increasing availability of DOAC reversal agents, such as idaracizumab and andexanet alpha.
As there are no head-to-head trials comparing the different DOACs in VTE, no evidence-based recommendation can be made on the use of one over another. Parallels can be drawn from studies reviewing DOAC use in atrial fibrillation (AF). One systematic review, network meta-analysis, and cost-effectiveness analysis indirectly compared a range of DOACs for stroke prevention in AF. 16 Although comparisons between different trials were inherently difficult due to different patient selection criteria, the authors report that apixaban ranked highest on the balance of efficacy, safety, and cost. 16 A recent large international retrospective cohort study compared the efficacy and safety of different DOACs among AF patients and found that apixaban was associated with the lowest rates of gastrointestinal bleeding. 17 The recent FRAIL-AF RCT assessed whether switching from patients established on warfarin therapy to DOACs was safe and effective for older patients with AF. 18 Surprisingly, DOACs were associated with significantly greater bleeding, without any significant reduction in thrombotic risk in this cohort. While the American Society of Haematology (ASH) 2020 guidelines do not favour any DOAC in VTE, 8 NICE guidelines recommend using either apixaban or rivaroxaban. 6
DOACs are prescribed at fixed doses and do not require routine monitoring. However, this has led to uncertainty about the efficacy and safety of using DOACs in patients with obesity as well as abnormal renal and liver function – as each of these scenarios represents altered pharmacokinetics (volume of distribution and clearance). These groups of patients were not well represented in DOAC clinical trials. Real-world outcome studies and pharmacokinetic data have now permitted more evidence-based recommendations in each situation. 19
Renal impairment and chronic kidney disease (CKD)
Patients with renal impairment and CKD are paradoxically at increased risk of both recurrent thrombosis and bleeding. 20 Furthermore, since all DOACs are partially renally cleared, there is an increased half-life in renal impairment and CKD. Patients with a creatinine clearance (CrCl) <30 ml/min are not well represented in clinical trials, particularly those with dialysis-dependent end-stage renal disease (ESRD). 19 There is also limited real-world data published in this specific cohort.
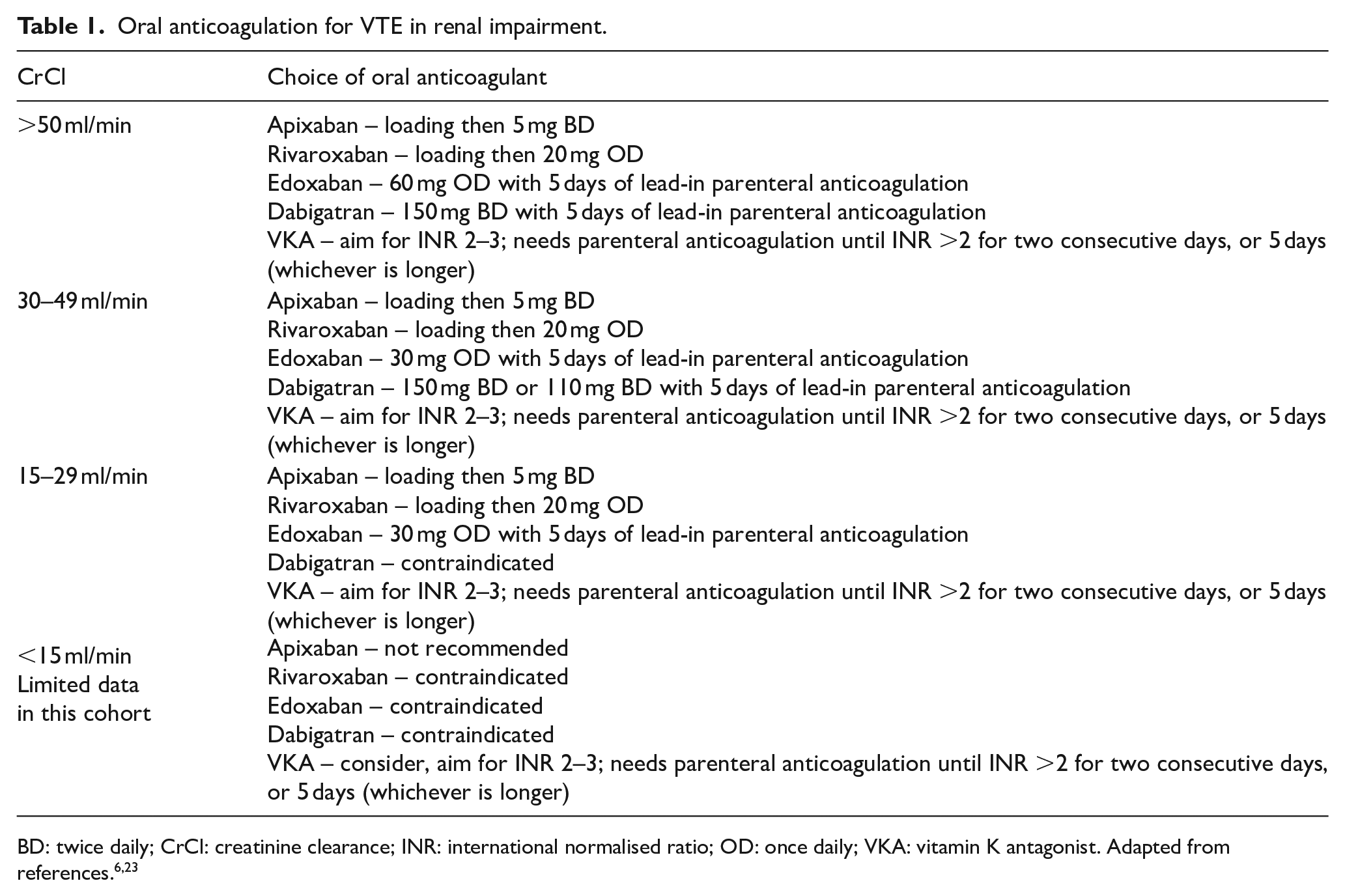
A recent systematic review found DOACs to be safer than warfarin in terms of bleeding risk and as effective for minimising VTE recurrence. 21 However, the majority of these patients had a CrCl of 30–50 ml/min. There is a growing use of apixaban in ESRD patients on dialysis with AF. One retrospective study reviewed the use of apixaban and warfarin for AF in patients with ESRD on dialysis. Apixaban was associated with a lower risk of major bleeding compared with warfarin, and standard apixaban doses were effective in reducing thromboembolic events and mortality risk. 22 The Summary of Product Characteristics (SPC) for each DOAC provides information on the dose adjustments and indications in renal impairment (summarised in Table 1).
Oral anticoagulation for VTE in renal impairment.
Some patients may require a brief period of parenteral anticoagulant prior to oral anticoagulation for example, if the oral route was temporarily not available or peri-procedurally where bridging anticoagulation is used. The choice usually lies between LMWH or unfractionated heparin (UFH). While UFH has been considered generally safer in patients with CKD (due to reticuloendothelial clearance), monitoring and dose adjustments remain challenging due to a small degree of renal clearance. 23 Dalteparin and enoxaparin are licensed at reduced doses in CrCl <30 ml/min and tinzaparin is licensed at a CrCl up to 20 ml/min. UFH may be the safer option in patients with ESRD. 21
Liver impairment and chronic liver disease/cirrhosis
Liver disease is associated with complex pathophysiological changes that ultimately ‘re-balance’ haemostasis. 24 All oral anti-Factor Xa inhibitors are partially metabolised through the liver and therefore in hepatic impairment, there is a risk of impaired clearance. 19 Although dabigatran, which is primarily renally cleared, may be the preferred anticoagulant in hepatic disease – there is little in the way of published experience. Unfortunately, patients with liver enzyme derangement or liver disease were excluded from DOAC clinical trials in VTE and this has led to the current recommendations of prescribing from the SPC (Table 2). One systematic review of retrospective studies compared DOACs with VKAs in patients with AF and cirrhosis. The study concluded that DOACs were more efficacious and safer but that RCTs were required. 25
Oral anticoagulation for VTE in hepatic impairment.
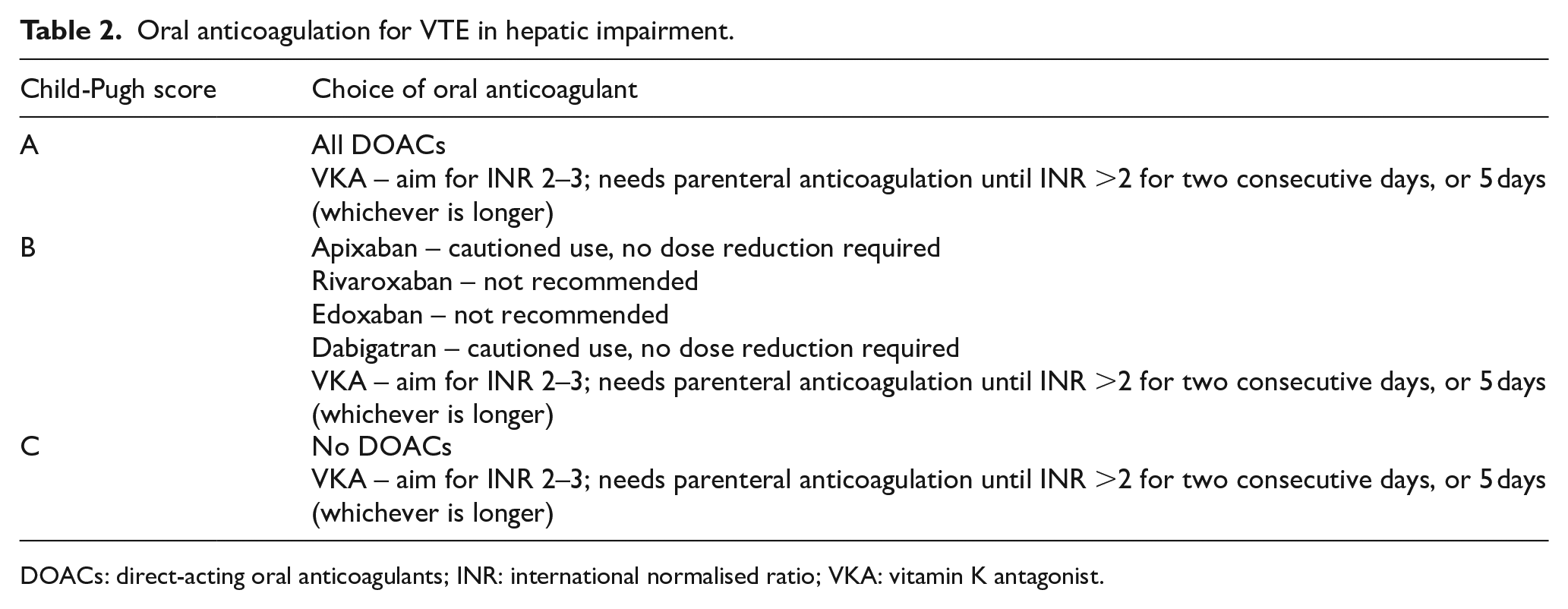
DOACs: direct-acting oral anticoagulants; INR: international normalised ratio; VKA: vitamin K antagonist.
Obesity
Obesity can theoretically lead to under treatment of patients with VTE due to changes in the volume of drug distribution and clearance. 19 The International Society on Thrombosis and Haemostasis (ISTH) has guided the use of DOACs in obesity. 26 Briefly, they recommend that rivaroxaban or apixaban can be used in any upper body limit but that evidence is significantly lacking in patients >150 kg. In this group, shared-decision making is important and warfarin should be considered. Edoxaban and dabigatran are not recommended.
Duration of anticoagulation
NICE guidelines recommend that primary treatment for VTE should be for a minimum of 3 months (or 3–6 months when the patient has concurrent active cancer). 6 Similarly, ASH recommends that primary VTE treatment should be for 3–6 months. 8 This time frame represents the time taken for both the thrombotic state associated with the thrombosis itself to resolve and for the individual risk to return to baseline. These recommendations are based on meta-analyses and RCT evidence.
Secondary prevention
On completion of primary treatment, a clinical assessment is required to decide whether anticoagulation can be discontinued or is required indefinitely for secondary prevention of recurrent VTE. Anticoagulation reduces the risk of recurrent VTE only for the duration of treatment; there is no longer-term benefit once therapy is complete. 27 These consultations usually occur within haemostasis and thrombosis clinics with a haematologist or by general or respiratory physicians with specialist interests in thrombosis. Both recurrence of VTE and bleeding related to inappropriate anticoagulation are associated with significant morbidity and mortality and are considered. A recent study investigated statistical models to identify the optimal ratio between bleeding and recurrence risks. 28 The authors highlighted that clinical equipoise still exists in some but not all these decisions and emphasised shared decision-making.
Thrombosis recurrence risks
The circumstances at the time of the initial VTE event are crucial in determining the risk of VTE recurrence. The ISTH classifies VTE into different categories based on the provoking aetiology of the initial thrombus (Figure 3). 29 Provoking aetiologies may be transient (major or minor) or persistent. There may also be unprovoked VTE events when no clear provoking aetiology is identified. Classifying VTE in this format has been adopted by several guidelines6,8 and is clinically relevant for identifying patients at higher risk of VTE recurrence and those who would benefit from indefinite anticoagulation. For example, in patients with transient provoked factors, primary treatment with 3 months of anticoagulation would be sufficient, however, in patients with persistent provoking factors (such as thrombophilia or inflammatory conditions), indefinite anticoagulation should be considered.
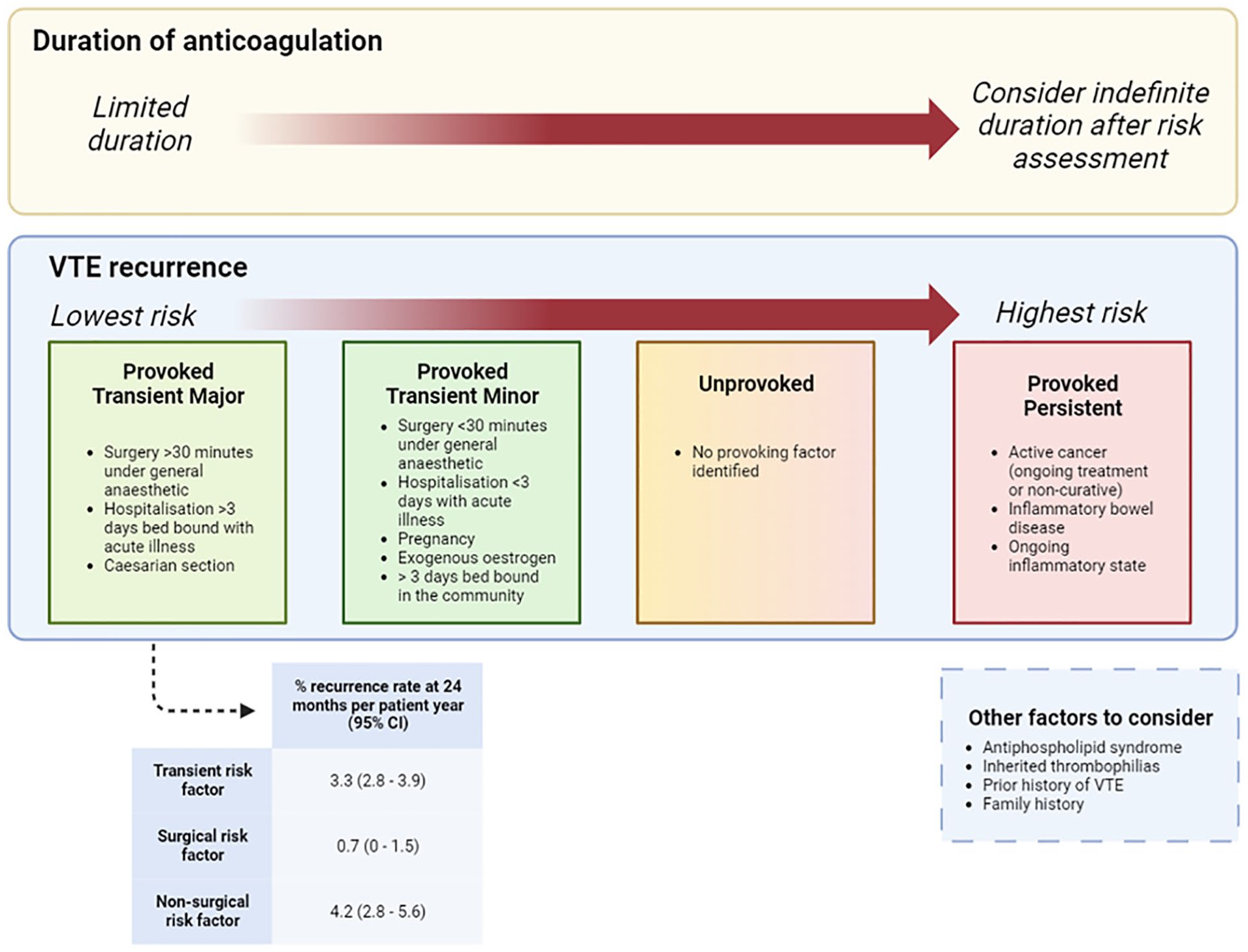
Classifying venous thromboembolism based on risk factors helps stratify the risk of recurrence and guides decisions on the duration of anticoagulation. Adapted from reference. 27 The data for the risk of recurrence at 24 months per patient-year are from the systematic review by Iorio et al. 30 Image created using Biorender.com.
It is known that VTE recurrence risk after an unprovoked event is significantly higher than that following a VTE provoked by a major transient risk factor.27,29,31,32 Identifying patients who can safely discontinue anticoagulation after a first unprovoked VTE remains challenging. The current expert consensus opinion is that if the predicted annual recurrence rate of VTE is <5% after an unprovoked VTE, discontinuing anticoagulation after primary treatment is reasonable. 31 Several scoring systems have been developed to quantify the VTE recurrence risk after an initial unprovoked VTE:
The HERDOO2 score assesses for hyperpigmentation; oedema; lower limb erythema; D-dimer level; BMI ⩾ 30; or age ⩾65 years. 33 Women with a first unprovoked DVT and a HERDOO2 score of ⩽1 may stop anticoagulation after primary treatment, without an increased risk of recurrence. This score is however limited to women and cannot be used in patients with PE.
The DASH score predicts recurrence risk based on: D-Dimer level 3 weeks after completion of anticoagulation; age; sex and hormone therapy at the time of VTE occurrence.34,35 Individuals with a DASH score of ⩽1 had a low risk of VTE recurrence (<5%) in the initial and external validation study and could safely discontinue anticoagulation. Recently, its application in clinical practice has changed after a validation cohort study reported that the VTE recurrence risk in patients ⩾65 years was >5%, even in those with the lowest DASH scores. 35
The Vienna Prediction Model bases VTE recurrence risk on sex, VTE location and D-Dimer level. A recent recalibration has provided an updated model to inform decision-making around the duration of anticoagulation and identify individuals at low risk of recurrence. 36
These scoring systems are not used in isolation. Factors including clinician gestalt and patient preference are important, as well as accounting for bleeding risks and identifying any associated thrombophilias.
Bleeding risks
Real-world studies highlight that many clinicians favour limiting the duration of anticoagulation after VTE due to concern for bleeding complications. 37 The most common site of major bleeding while on anticoagulation is from the gastrointestinal (GI) tract, but intracranial haemorrhages (ICH) also occur. The rate of major bleeding is lower with DOACs compared with VKAs, but significant in both groups. 5 Guidelines recommend incorporating a formal assessment of bleeding risk into decision-making. The NICE guidelines favour the HAS-BLED score and recommend that anticoagulation may be stopped if the score is 4 or more and cannot be modified. 6 It should be noted however that the HAS-BLED score was developed for AF patients – an older patient cohort with higher bleeding risk. Various other bleeding risks exist, including the VTE-BLEED score which has been validated in assessing bleeding risk in anticoagulated patients. 38 These scores may also be used as an aide memoire to screen for modifiable bleeding risk factors (e.g. hypertension) and thus to guide the optimal anticoagulant duration and dose. However, these risk scores do not account for the dynamic nature of bleeding risks which requires routine review.
Thrombophilia screening
Hereditary and acquired thrombophilias are risk factors for VTE. Whether testing guides management in patients who would otherwise be anticoagulated regardless of the thrombophilia result is controversial. 8 As a rule of thumb, the majority of VTE patients do not require thrombophilia testing. Seminal studies have shown that testing for heritable thrombophilias only predicts the first VTE but not for the risk of VTE recurrence in unselected patients,32,39 supporting the argument that there is a lack of clinical utility in decision-making in patients who have already had VTE. Selected patients should undergo thrombophilia testing only if this changes management and after completion of primary treatment. Notably, anticoagulants interfere with clotting-based laboratory thrombophilia assays and if these pre-analytical variables are not recognised then false-negative or false-positive results could lead to inappropriate decisions. Ideally, thrombophilia screening should therefore be performed under the supervision of a haematologist or a thrombosis specialist. The British Society of Haematology (BSH) 2022 7 and ASH 2023 guidelines 8 provide recommendations on when thrombophilia screening should be considered after VTE:
– Testing for heritable thrombophilias (e.g. protein C or S deficiency, antithrombin deficiency, Factor V Leiden) is generally not recommended.
– The ASH guidelines recommend testing for thrombophilias in patients with VTE provoked by non-surgical major transient risk factors, pregnancy and oral contraception. The BSH however favour a more conservative approach.
– Screening for antiphospholipid syndrome (APS) is recommended after an episode of unprovoked VTE or VTE provided by a minor factor, as this may change the choice and duration of anticoagulation.
– Screening for myeloproliferative neoplasms (MPN) should be considered in patients with abnormal full blood count (FBC) indices for example, thrombocytosis or basophilia. Note that JAK2 positivity can occur even with normal FBC but screening should be targeted.
– In patients with thromboses at unusual sites (e.g. splanchnic vein or cerebral venous sinus), testing for MPNs and paroxysmal nocturnal haemoglobinuria should be considered.
Special circumstances
Cancer-associated thrombosis (CAT)
Management of CAT has been reviewed extensively. 40 Nonetheless, it is commonly encountered by physicians in various disciplines and there are certain important considerations.
Choice of anticoagulation
LMWH has been the favoured anticoagulant in the management of CAT. The CLOT trial found LMWH to be superior to warfarin in reducing the risk of VTE recurrence. 41 However, several RCTs have now evaluated the safety and efficacy of DOACs compared to LMWH in cancer. Two recent meta-analyses of these RCTs support the efficacy of DOACs in the treatment of CAT but highlight the greater rates of bleeding in patients with GI and genitourinary tumours.42,43 Drug–drug interactions are particularly important in CAT. For example, targeted treatments, like tyrosine kinase inhibitors, may interfere with DOAC pharmacokinetics through P-glycoprotein interactions. 40
Thrombocytopenia
Thrombocytopenia may be common in cancer patients undergoing active treatment and will increase the bleeding risk while on anticoagulation. In this instance, LMWH may allow greater flexibility for dose adjustments. Both BSH and ISTH have published clinical practice guidelines for this scenario44,45:
A platelet count of 50 × 109/L or more is considered safe for full-dose anticoagulation.
If platelet count is <50 × 109/L, platelet transfusion support should be considered to permit therapeutic anticoagulation during primary VTE treatment.
If the platelet count is between 25 and 50 × 109/L, 50% of the therapeutic LMWH dose should be administered
If the platelet count is <25, then anticoagulation should be interrupted.
Duration of anticoagulation
Malignancy is one of the greatest risk factors for VTE recurrence, with a twofold to ninefold greater risk when compared to those without active cancer. 27 There is good evidence to support a minimum duration of anticoagulation of 3–6 months for the primary management of CAT.6,40 However, there is limited good-quality evidence beyond this period to guide therapeutic decision-making. Several factors need to be considered when deciding if further anticoagulation is required (Figure 4).
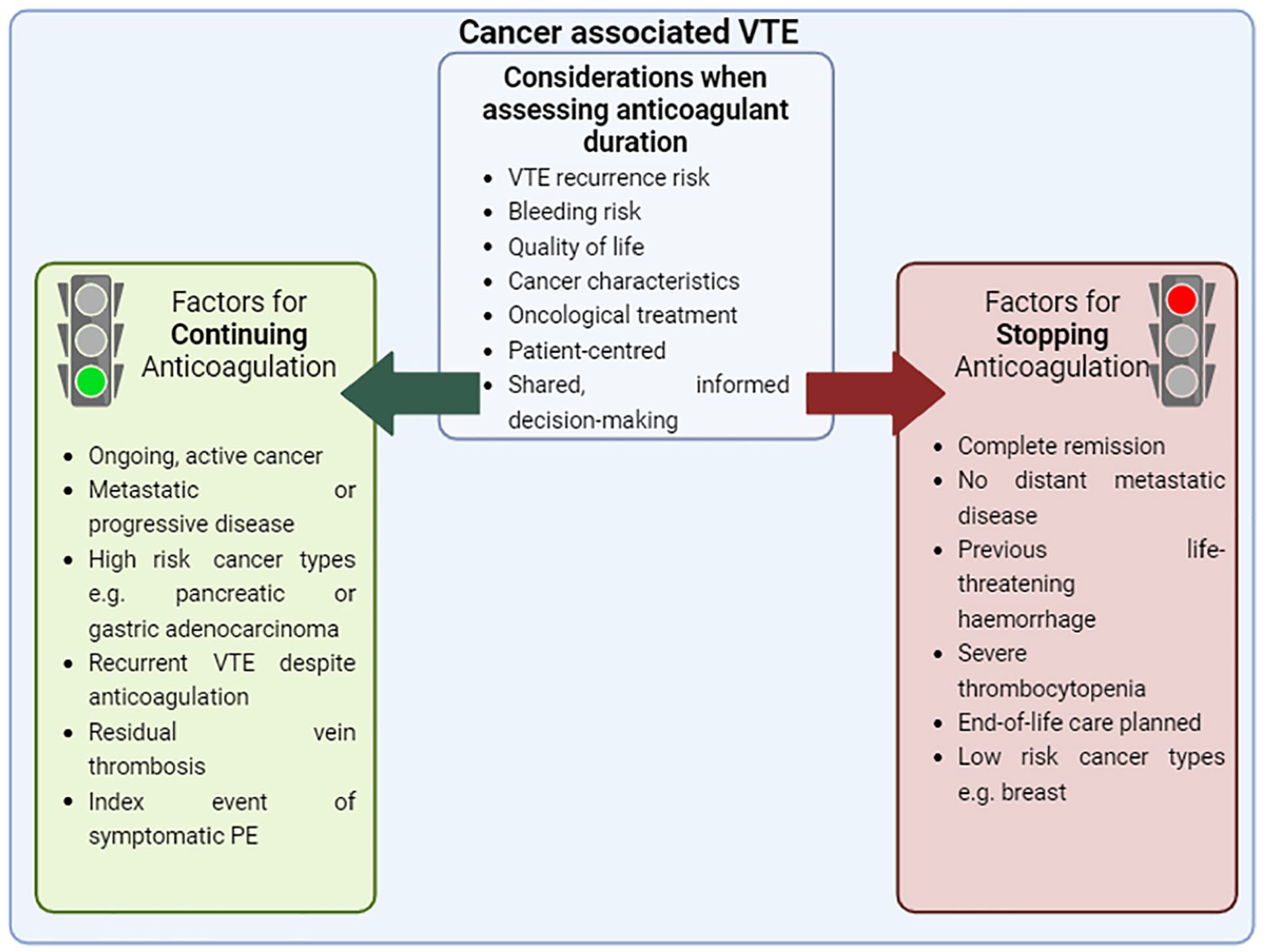
Factors favouring continuing or discontinuing anticoagulation for cancer-associated venous thromboembolism after initial therapy of 3–6 months. Figure adapted from reference. 40 Image created using Biorender.com.
Antiphospholipid syndrome
APS is an acquired autoimmune condition that requires both laboratory and clinical criteria for diagnosis. 11 ‘Triple-positive APS’ is defined as the presence of lupus anticoagulant, beta-2 glycoprotein-1 and anticardiolipin. It is associated with a particularly high risk of recurrent arterial and venous thromboses. Studies report that around 10% of patients with VTE will have a diagnosis of APS. 46 Certain clinical features increase the likelihood of APS, including history of autoimmune disorders; livedo reticularis; recurrent thromboses; VTE at unusual sites; recurrent miscarriages or severe pre-eclampsia; and cardiac valve abnormalities without any alternative explanation.
APS is considered an indication for indefinite anticoagulation. The role of DOACs in treating thrombotic APS was initially reported through meta-analyses of several case reports. These had identified a relatively high rate of recurrence (~15%) in patients on Factor Xa inhibitors. 47 The TRAPS RCT compared rivaroxaban to warfarin in triple-positive APS patients. 48 The trial was terminated early after an excess of thrombotic events in patients in the rivaroxaban arm. A 2-year follow-up of the TRAPS study produced the same outcome. 49 Following the initial publication of the TRAPS study, the Medicines and Healthcare Products Regulatory Agency issued recommendations advising healthcare professionals against using any DOAC in patients with APS. 50 The ASTRO-APS RCT assessed apixaban against warfarin in single-, double- and triple-positive APS patients. Despite small numbers, this trial also reported inferior outcomes of DOACs compared to warfarin. 51 The BSH recommends the following for managing thrombotic APS 10 :
– Warfarin should be used for the treatment and secondary prophylaxis of arterial thrombosis.
– In patients with triple-positive APS and VTE, DOACs are not preferred over warfarin; however, if patients were already on DOACs then these should continue over no anticoagulation.
– In patients with non-triple-positive APS and VTE, there is insufficient evidence to make firm recommendations but DOACs are not preferred over warfarin.
Warfarin is initially prescribed with a target range INR of 2–3 for APS. There can be a discrepancy between point-of-care (POC) and laboratory-based INR testing in APS. Although the laboratory-based INR is more accurate, this does not preclude the use of POC if appropriate quality assurance is in place. Our local approach has been to recommend paired POC and laboratory samples once established on anticoagulation to assess the correlation between INRs and more importantly whether this discrepancy would not result in a dose change. This should be at least every 6 months and every time that the POC machine strips or reagents batch is changed.
Conclusions and learning points
▪ Management of VTE can be considered in three phases: initial management, primary treatment and secondary prevention.
▪ The choice and duration of anticoagulation for managing VTE needs to account for several factors including creatinine clearance, liver function and body weight.
▪ Several scoring systems exist to assist decision-making on secondary prevention for VTE. These scores can assess the risk of thrombosis recurrence and bleeding risk. An annualised risk of thrombosis recurrence of <5% is the generally accepted threshold for limited duration anticoagulation.
▪ Thrombophilia screening is commonly not recommended for the majority of patients and should be done only in selected patients where the management will be altered.
Footnotes
Authorship statement
KS wrote the first draft. All authors contributed to the editing and writing of the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
