Abstract
Digoxin toxicity can be life-threatening. Digoxin-specific antibody (DSA) fragments are used in severe digoxin toxicity, binding to serum-free digoxin and enabling increased renal excretion. In severe renal impairment, clearance of these complexes is prolonged, leading to rebound toxicity. Digoxin and DSA complexes are not dialysable. We present a case of a gentleman with severe digoxin toxicity and acute kidney injury (AKI). Despite receiving DSA doses, his digoxin levels rebounded and symptoms persisted. Based on published case reports, plasma exchange (PEX) after further dosing was arranged. PEX facilitated the removal of digoxin–DSA complexes, bypassing renal excretion. During PEX, clinical signs improved and were sustained. He did not require further dialysis or PEX, renal function recovered and he was discharged. This case highlights challenges in the management of severe digoxin toxicity in patients with a concurrent AKI. The use of PEX enabled digoxin–DSA complex removal and should be considered in these circumstances.
Introduction
Digoxin toxicity of varying degrees complicates 1% of patients taking the medication. 1 Severe toxicity is life-threatening and can be treated with the administration of digoxin-specific antibody fragments (DSA). These have a high affinity for digoxin and bind with free drugs to create complexes which are then renally excreted. If the patient has renal impairment and reduced renal clearance, there is a risk of subsequent rebound of the free digoxin level as the DSA effect wears off. Whilst there are no formal guidelines, combining plasma exchange (PEX) to remove these complexes in patients with severe renal impairment should facilitate the removal of the drug in the plasma and prevent this rebound phenomenon, and there have been previous case reports highlighting this approach.2 –5 We present a further case of the use of PEX to prevent the rebound of digoxin levels in a patient with an acute kidney injury (AKI) and life-threatening digoxin toxicity.
Case
An 80-year-old gentleman presented to the Emergency Department with a reduced consciousness level, nausea, bradycardia, hypotension and a severe AKI. On arrival, he had a heart rate of 30–40 bpm, a Glasgow Coma Score (GCS) of 11 and a systolic blood pressure of 70 mmHg. Serum creatinine was 580 µmol/l and he was oligo-anuric, passing 0–10 ml/h of urine.
This demonstrated that he had clinical evidence of digoxin toxicity (bradycardia, hypotension, altered mental state and nausea).
His notable past medical history included chronic kidney disease (baseline serum creatinine 140 µmoll/l), ischaemic heart disease, atrial fibrillation, hypertension, type 2 diabetes and gout. His regular medications were warfarin, digoxin, carvidelol, allopurinol, metformin and atorvastatin and he had continued to take all medications prior to his admission.
Serum digoxin level at the time of presentation was elevated at 3.6 nanograms/ml and as he had clinical evidence of digoxin toxicity he received one dose of DSA in the emergency department. However, despite this, his level rose to 8 nanograms/ml (see Figure 1). He then developed signs of peripheral and pulmonary oedema (consistent on chest x-ray) with a new oxygen requirement after receiving intravenous fluid resuscitation with minimal urine output. He transferred to the Renal High Dependency Unit to commence haemodialysis for the management of fluid overload. He had received a reversal of warfarin with Beriplex and vitamin K prior to the insertion of a temporary dialysis central venous catheter. A further dose of DSA was given with improvement in his digoxin level. Unfortunately, this rebounded again 24 h later, and he remained bradycardic and hypotensive. Discussions were undertaken with both the National Poisons Information Service and the local Haematology department (who take referrals and facilitate PEX) to discuss the use of PEX for the removal of digoxin, based on evidence from published case reports.2 –5
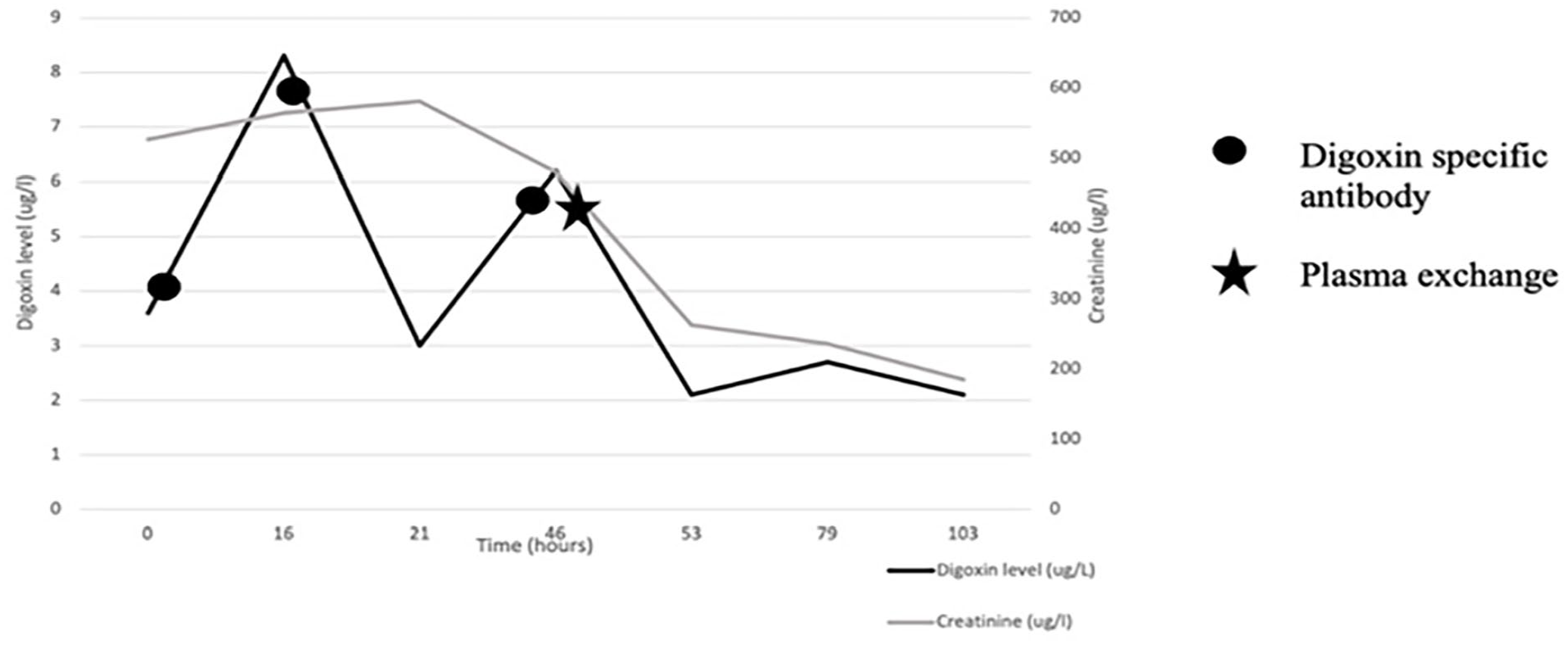
Serum digoxin levels and creatinine over time.
Neither department had previously used PEX for this indication, however agreed with the mechanistic principles. Based on information regarding the time to peak formation of digoxin–DSA complexes, PEX was undertaken 4 h after a third dose of DSA was administered.
This gentleman received a single session of PEX consisting of one total blood volume of therapeutic PEX. The plasma was replaced with Gelofusine for the first 500 ml and then Alburex thereafter with a total of 3,605 ml replacement. It was well tolerated and uneventful. During the procedure, staff could see clinical improvement in GCS. After the procedure, he was considerably brighter and sat up and chatting to his family. His heart rate and blood pressure also improved. Interestingly, the removed plasma was noted to have a slight green sheen (see Figure 2).

Plasma was removed after DSA administration.
Over the next 24 h, his clinical condition continued to improve and stabilised, with a sustained heart rate of >60 bpm and blood pressure >110/50 mmHg, and his confusion improved. Over the next few days, his urine output increased, and he did not require further dialysis or PEX sessions. His renal function subsequently returned to baseline, and he was discharged home. Serum creatinine at the time of discharge was 115 µmol/l.
Discussion
This case highlights the difficulties faced when dealing with drug toxicities in someone with an AKI. Renal impairment affects the clearance of both digoxin and DSA. To understand why PEX was considered in the management in this case, we must consider why the conventional treatment was unsuccessful.
Digoxin and DSA fragments
Digoxin is a commonly used drug in the management of atrial fibrillation. 6 It is a cardiac glycoside that increases contractility of the myocardium through inhibition of Na+-K+ exchange activity. 7 Digoxin toxicity is potentially life-threatening, and the most common cause of death is cardiac arrhythmia. Toxicity often presents with non-specific symptoms such as lethargy, confusion, nausea and vomiting. A therapeutic digoxin serum level range is relatively narrow: 0.7–2.0 nanograms/ml. In health, digoxin is excreted unchanged, primarily by renal clearance (75%), with a half-life of 30–40 h. 7 Clearance is prolonged with those with renal impairment as elimination is correlated to creatinine clearance, with terminal elimination rising to up to 100 h in anuric patients. 8
DSA fragments (trade names: Digibind/Digifab) are polyclonal anti-digoxin immunoglobulin fragments obtained from sheep that have been immunised with digoxin dicarboxymethoxylamine 9 and are licensed for use in the management of severe digoxin toxicity. 10 The fragments bind to digoxin molecules forming complexes that then mobilise into the extracellular space. The bound digoxin–DSA complexes cannot bind to Na+-K+ ATPase reducing the clinical effects of digoxin. These complexes are then eliminated primarily by renal excretion but also partially through the reticuloendothelial route. In patients with normal renal function, the half-life of these complexes is 12–20 h. This half-life increases up to 10-fold in renal failure. 8 The mean time for digoxin to bind to DSA is 2.6 h and each vial of DSA costs £750. 6 Dosing is based on the patient’s weight and serum digoxin concentration. In this case, each dose of DSA required four vials (£3,000 per dose).
Therefore, severe renal impairment not only increases the risk of acute and chronic digoxin toxicity due to reduced clearance but also limits the efficacy of DSA. Ineffective clearance of digoxin–DSA complexes risks rebound of digoxin levels as the complexes dissociate and free digoxin re-emerges. This can happen as soon as 12–24 h after initial DSA treatment, or much later for those in cases of severe renal impairment. 8
The role of dialysis
Although some drugs and toxins can be removed through the process of dialysis, neither digoxin nor DSA complexes are effectively cleared. The extent to which a drug is removed through dialysis is influenced by several physiological factors including molecular weight, protein binding, volume of distribution and water solubility.10,11 Modern high-flux dialyser membranes were effectively clear small molecules (<500 Daltons). 12 The molecular weight of digoxin is 780.9 Daltons, it is 25% protein bound and 50–75% is renally excreted. 13 Furthermore, digoxin has a large volume of distribution (Vdss = 5,10l). It is estimated that only 3% of the drug is removed during a 5-h session of haemodialysis due to a large amount of tissue distribution. 13 DSA alone is too large at 46,000 Daltons to be removed and molecular weight increases when DSA complexes are formed. 8
Plasma exchange
PEX is a mechanical process of in vivo separation and removal of plasma from whole blood. 14 The volume of discarded plasma is replaced with an appropriate physiological alternative. It is generally used in situations where a plasma component is driving a disease process. In this case, PEX was carried out at a time when peak DSA binding to digoxin was thought to have occurred. These complexes were then predicted to be removed within the exchanged plasma. This management plan is supported by a handful of previously published case reports.2 –5 Little change in the clinical condition of the patient in this case was observed until after PEX and DSA were combined.
Conclusion
The use of DSA in the management of digoxin toxicity is well described. However, in the context of anuric AKI, there is a significant risk of treatment failure and subsequent rebound toxicity due to complex dissociation. The use of PEX to facilitate complex removal after DSA administration (timed with peak digoxin–DSA complex formation) allows renal clearance to be bypassed and should therefore be considered for use in the management of digoxin toxicity in severe renal impairment. Understanding the physiological principles of drug removal both in health and through renal replacement therapy prompted the use of PEX which facilitated efficient and effective management, alongside helping reduce hospital stay, morbidity and mortality of the patient.
Footnotes
Acknowledgements
We would like to thank Carol Terry, Charge Nurse in the Apheresis Unit for providing details of the plasma exchange carried out in this case.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
