Abstract
The ventriculoperitoneal shunt (VP shunt) is one of the most common neurosurgical procedures performed in daily practice. Various complications following a VP shunt are as follows: post-procedure shunt-related infections, shunt block, shunt displacement or exteriorisation and haemorrhage associated with it. Delayed intraventricular haemorrhage is a relatively uncommon complication following the aforementioned procedure. Here we present an atypical case of a 72-year-old male who presented with subarachnoid haemorrhage with hydrocephalus and underwent a VP shunt, following which the patient had early intraventricular haemorrhage (eIVH) with an unfortunate outcome. Here, we propose pathophysiology and risk factors for eIVH.
Keywords
Introduction
A ventriculoperitoneal (VP) shunt is a procedure done to divert cerebrospinal fluid from its normal course. VP shunting is considered the most common procedure done in neurosurgery. As with any other surgical procedure, a VP shunt can also be associated with consequences in the form of various complications like post-shunting infection, subdural haemorrhage, seizures, shunt exteriorisation, blockage or malfunction. 1 All of these complications have been extensively reported in the literature, of which post-ventriculoperitoneal shunt intracerebral (ICH) and/or intraventricular haemorrhage (IVH) is very rare. Amongst these, cases of delayed haemorrhage (dICH/dIVH), which is defined as 5 days after a VP shunt, are reported; however, early intracerebral or intraventricular haemorrhage (eICH/eIVH) following the procedure is a rare entity that needs to be considered and defined. eICH/eIVH is associated with a high mortality rate. The purpose of this case report is to highlight eICH/eIVH as a distinct entity.
Case
A 72-year-old male presented in the emergency department with complaints of headache, vomiting and altered sensorium for 1 day. Headache was typical thunderclap in character and holocranial in distribution. Headache was associated with vomiting, which was projectile, non-bilious and non-blood-staining. There was no history of trauma, fever or abnormal body movements preceding these symptoms. There was no history of loss of consciousness. There was no history of prior brain surgery, antiplatelet or anticoagulant intake, or history of prior intracranial bleeding. His family history was also unremarkable for a bleeding diathesis or cerebrovascular accident. Physical examination was suggestive of neck rigidity; pupils were equal, 2 mm sluggishly reacting to light. The Cushing triad was seen in the form of bradycardia, hypertension and irregular breathing. The patient was irritable and not oriented to time, place or person.
On admission, his non-contrast computed tomography (NCCT) head was suggestive of subarachnoid haemorrhage (SAH) in basal cisterns with hydrocephalus (Figure 1). The patient was in Hunt and Hess Grade 3 and modified Fischer Grade 3 on SAH grading. Computed tomography (CT) angiograms of brain vessels were done to look for aneurysms or vascular anomalies that were suggestive of mild ectasia in the short segment of the distal aspect of the inferior division of the right middle cerebral artery (MCA), measuring approximately 2.6 mm in maximum diameter. There was no aneurysm or arteriovenous malformation. Blood investigations revealed a normal complete blood count, including normal platelet counts, prothrombin time, international normalised ratio, activated partial thromboplastin time, and liver and renal functions.
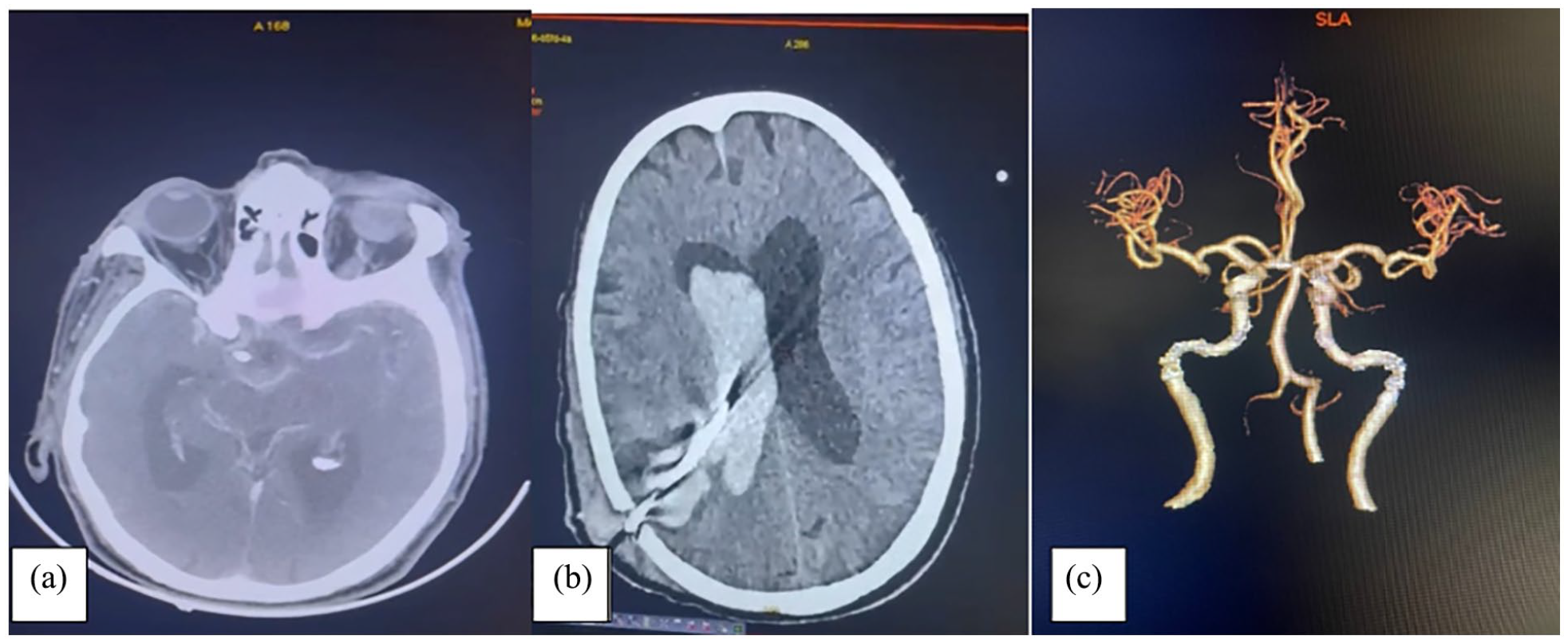
(a) NCCT image (axial) suggestive of SAH in basal cistern and peri-mesencephalic cistern and hydrocephalus. (b) NCCT image (axial) suggestive of tract site haemorrhage with extension into right ventricle causing IVH. (c) CT angiography of brain vessels (reconstructed) was suggestive of a normal angiogram showing no aneurysm or arteriovenous malformation.
The patient was admitted and managed surgically for hydrocephalus. Subsequently, medium-pressure ventriculoperitoneal (MPVP) shunting was done on the right side. The patient’s sensorium improved after the MPVP shunt. Within less than 48 h of the procedure, the patient’s sensorium deteriorated again. A repeat NCCT head was done, which was suggestive of areas of haemorrhage seen along interhemispheric fissures, bilateral sylvian fissures and basal cisterns predominantly on the right side. A V-P shunt was seen in situ on the right side with no significant decompression of ventricles and IVH was seen in both lateral, third and fourth ventricles. An intraparenchymal haemorrhagic component was also seen in the right parietal lobe adjacent to the right lateral ventricle along the path of the VP shunt with mild perifocal oedema. The ICH score was 5. Given the neurological worsening and the presence of eICH/eIVH, the left frontal external ventricular drain was inserted. The patient was managed conservatively in the form of antiepileptic drugs, anti-oedema measures, antibiotics and other supportive measures. However, the patient had a deteriorating course. The patient had a fall in GCS from E4V5M6 to E1V(T)M1 with fixed dilated and non-reacting pupils, absent brainstem reflexes, no spontaneous breathing efforts, a negative caloric test and no spontaneous limb movements. The patient had a subsequent fall in blood pressure, which warranted inotropic support. Despite all the supportive measures, the patient could not survive and succumbed to death.
Literature search
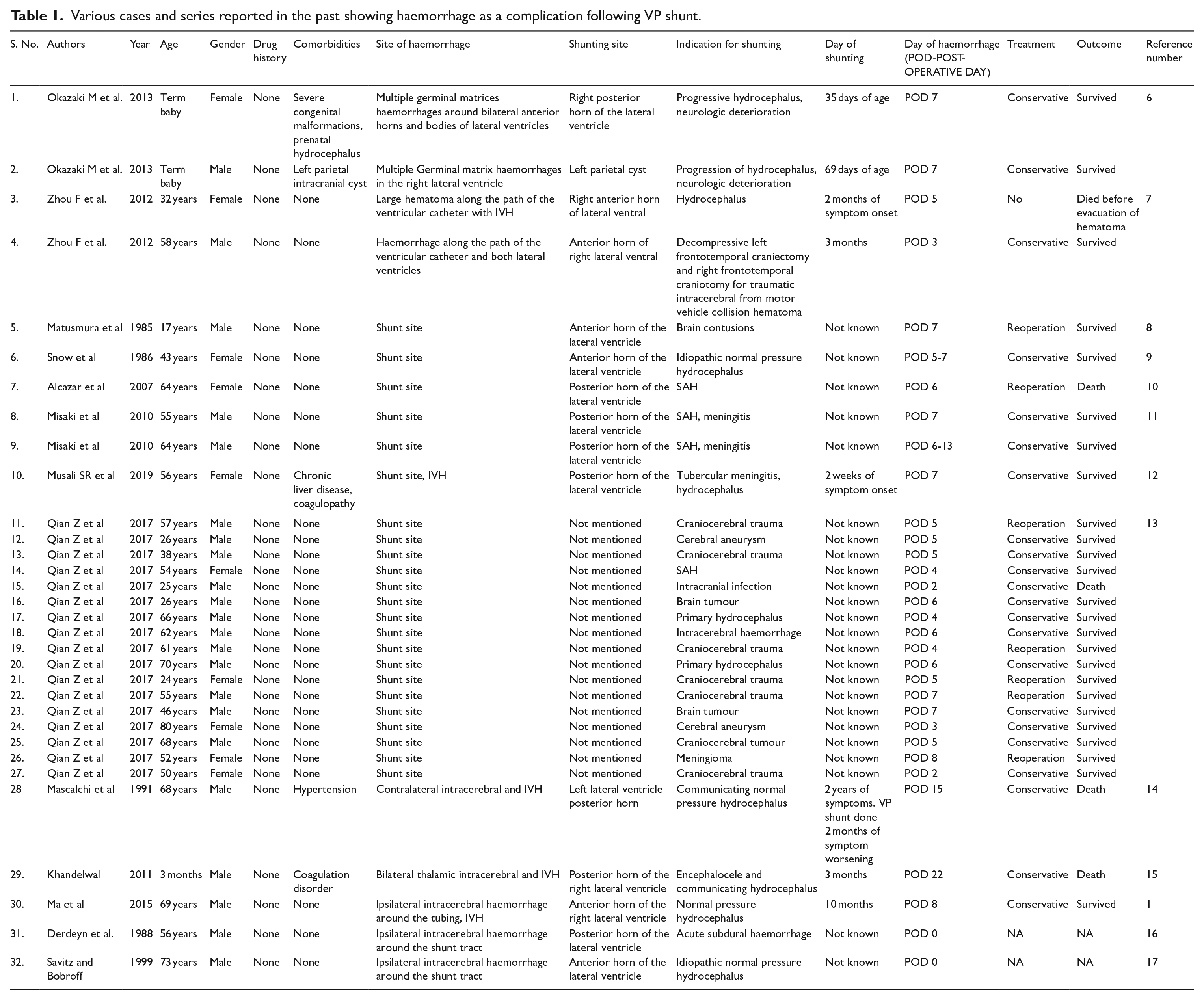
The literature was searched by two reviewers. ‘PUBMED’ and ‘Google Scholar’ databases were searched from 1985 to 2023 using keywords ‘Ventriculoperitoneal shunt AND Intraventricular hemorrhage’ OR ‘VP Shunt AND Intraventricular hemorrhage’ OR ‘Ventriculoperitoneal shunt AND Intracerebral hemorrhage’ OR ‘VP shunt AND Intracerebral hemorrhage’ OR ‘Post VP Shunt Intracerebral hemorrhage’ OR ‘Post VP Shunt Intraventricular hemorrhage’ OR ‘Haemorrhagic complications of VP Shunt’. A total of 1040 studies were found. These were reviewed independently by two reviewers. Case reports and case series were included in the results. Articles that were not in English, duplicate studies or those for which full text was not found were excluded from the review. After a careful review of these articles, 32 cases were found in the literature with available text. These are summarised in Table 1.
Various cases and series reported in the past showing haemorrhage as a complication following VP shunt.
Discussion
Immediate, early and repeated haemorrhages after a VP shunt are very rare. There are only 32 cases (Table 1) of post-VP shunt haemorrhage worldwide. The age of presentation varies amongst the various cases reported, ranging from 35 days of life to 80 years. The present case has an age of 72 years. Various modifiable factors are responsible for haemorrhage following a VP shunt, including coagulation and platelet disorders (primary or secondary). Age is itself a non-modifiable factor. Both extremes of age are vulnerable to post-VP shunt haemorrhage owing to the brain friability and friable choroid plexus, which, if severely injured, causes early or delayed IVH. Usually, minimal haemorrhages can occur after the procedure of VP shunting, but large, life-threatening ICH/IVH is rare.
These ICH/IVH can be classified further as early or delayed based on the time of onset of the complication post-procedure. The entity dIVH is defined in the literature as IVH, which occurs 5 days after the VP shunt procedure. However, there is no consensus on the entity known as eIVH. Moreover, IVH is most of the time associated with ICH; hence, these terms need to be redefined as eICH/eIVH and dICH/dIVH. If we consider 5 days as the cut-off for dIVH, any ICH or IVH that occurs within 5 days should be classified as eICH or eIVH. In the present case, the haemorrhage and complication occurred within 48 hours of the procedure, making this entity ‘very early’. So, the timelines need to be defined based on the onset of complications. Four cases out of 32 cases (Table 1) were in the very early category, 17 were in the delayed category and only 11 were in the early category. Most of the patients with dICH/dIVH survived, whereas the patients with early and very early onset died. Along with the time of onset, other factors are also required for predicting the outcome. One of them is the site of ICH in association with IVH. Most of the haemorrhages are located along the catheter path, and these hematomas can extend from the tract site to the parenchyma with or without intraventricular extension. In our case, the right parietal intracerebral hematoma extended predominately into the ventricular system, increasing the hydrocephalus.
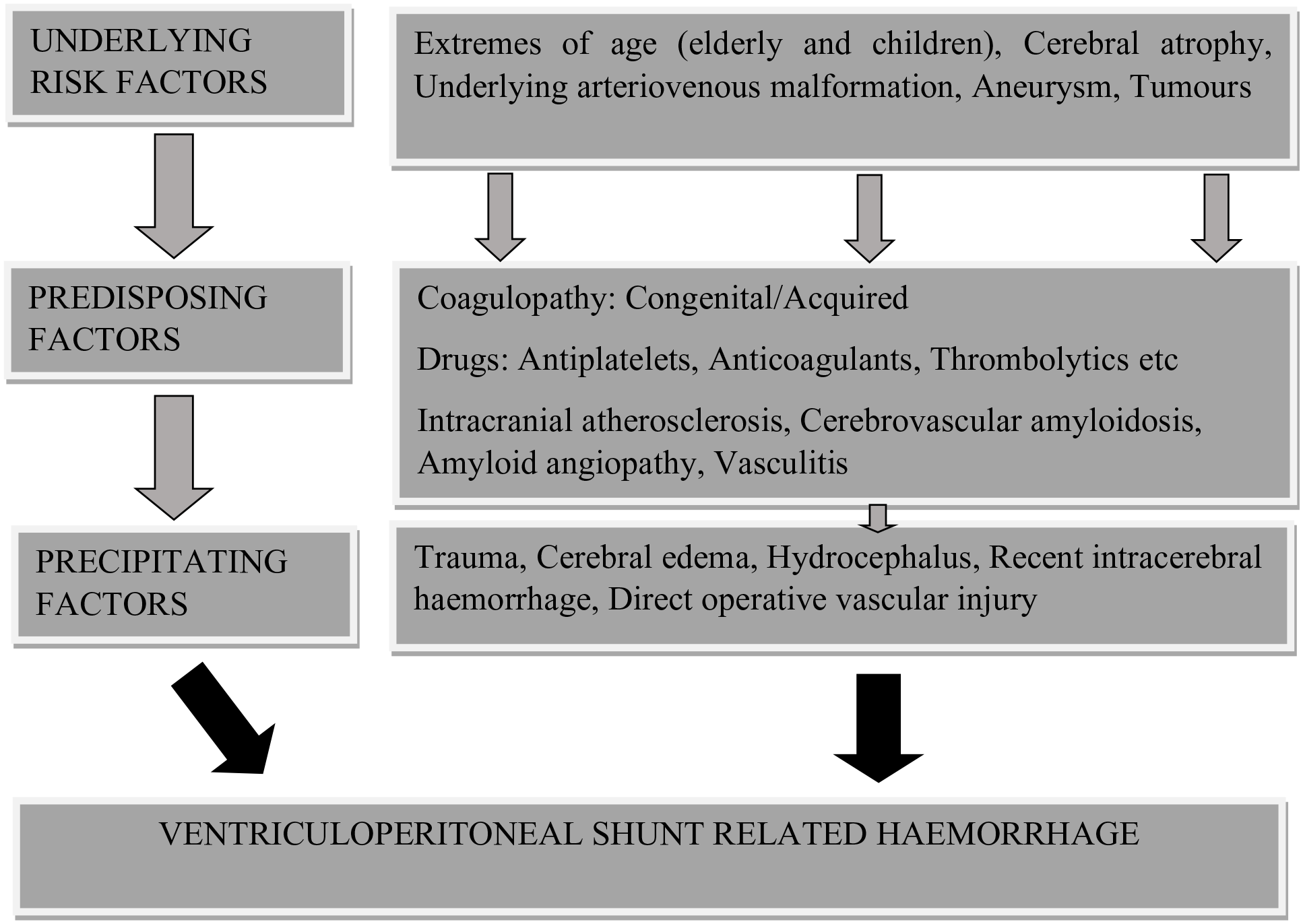
There are various proposed mechanisms for such complications after a VP shunt. One proposed mechanism is the pre-existing bleeding or clotting disorder, disseminated intravascular coagulation induced by surgical procedures and ongoing anticoagulant therapy. Another important mechanism could be the mechanical disruption of the cerebral blood vasculature, which is attributed to direct catheter insertion, indirect erosion of adjacent vessels by the catheter itself, fragile brain tissue or abnormal cerebral vessels (oedema, vascular malformation, atherosclerosis, cerebrovascular amyloidosis and moyamoya disease), head trauma, or intracranial tumour haemorrhage after the VP shunt. All the preoperative laboratory tests excluded coagulation defects, and CT angiography did not find any obvious vascular malformations. The haemorrhage in the present case occurred less than 48 h after the VP shunt and was located along the catheter pathway extending into the ventricles. The proposed mechanism may be the disruption of adjacent vessels and the choroid plexus by the catheter itself. The intracerebral hematoma progressed subsequently. Another proposed mechanism, in this case, could be degenerative vasculopathy in elderly people. Also, fragile brain tissue can be one of the reasons for haemorrhage (Table 2 and Figure 2). Unfortunately, we could not find out the exact reasons resulting in the extensive haemorrhage in our case. However, our case alludes to the fact that an overall thorough pre-operative examination to screen for this rare but disastrous potential complication should be performed for all, but most importantly, for elective VP shunt candidates. For instance, blood examination, transfusion of platelets or appropriate coagulation factors are needed for pre-existing coagulation disorders. Long-acting anticoagulant therapy should be stopped or replaced by short-acting anticoagulant drugs.
Proposed risk factors for haemorrhage following VP shunting.

Proposed pathophysiology of ventriculoperitoneal shunt-associated haemorrhage.
Magnetic resonance imaging is another important preoperative procedure. Magnetic resonance angiography (MRA) can exclude marked vascular abnormalities, including moyamoya disease and arteriovenous malformation. However, Husson and Lasjaunias 2 reported that anomalies of intracranial arteries could be detected by angiography but not by MRA in 34% of their patients, particularly in the branches of the MCA. Second, T2*-weighted gradient-echo MR imaging could reflect hemosiderin deposits, which were related to prior intracerebral haemorrhages. 3 The presence of such positive T2* signals may be a risk predictor for intracerebral haemorrhage. 4 For elderly patients, SWI (Susceptibility Weighting Imaging) is highly recommended, especially when amyloid-angiopathy is suspected. 5 The development of hydrocephalus after aneurysmal SAH is not unusual. CT angiography or digital subtraction angiography may be needed before a VP shunt in cases of SAH. The sudden change in intracranial pressure may trigger the rupture of residual or recurrent aneurysms, which should be avoided before the shunt surgery. Patients with chronic hydrocephalus usually have a much thinner cerebral cortex compared with patients with acute hydrocephalus. So, the gradual decline of the intracranial pressure may be an appropriate method to minimise the sudden collapse of the cortex after the VP shunt, which may result in intracranial haemorrhage.
The present study is one of its kind from the region, contributing to its strength. Here, a compilation of studies is done that were found in the literature. Previous studies did not focus on the risk factors for haemorrhage related to the VP shunt. We tried to predict the various risk factors for haemorrhage related to the VP shunt based on the literature. An attempt has been made to predict the pathophysiology of haemorrhage related to the VP shunt. Moreover, this article will serve as the basis for further research on this entity. Nonetheless, this article will add to the existing literature as an update. These are the strengths of this study. However, no study is beyond its limitations. Being a single-centred study and single-case report, it is required for a larger sample size to better predict the underlying mechanisms and risk factors for eICH and eIVH.
Conclusion
eICH/eIVH is a distinct entity from dICH/dIVH associated with a high rate of mortality. Pre-procedure risk screening includes age, underlying disease, brain friability, drugs, comorbidities and bleeding diathesis, which adds to the pre-procedure probability assessment of complications of VP shunt. Regular monitoring post-procedure, early detection of deterioration in a patient’s condition after the procedure, early neuroimaging and early suspicion of eICH/eIVH are important in addressing such a complication of a normal routine procedure and decreasing mortality in such cases.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
