Abstract
Whipple’s disease is a multisystemic chronic infectious condition caused by Tropheryma whipplei (T. whipplei). Though characterised often by insidious weight loss, diarrhoea, and arthralgia, three other distinct manifestations can be observed, namely localised disease, acute infection and asymptomatic carriage. The diagnosis relies on histopathological examination of duodenal biopsies and polymerase chain reaction analysis of the 16S rRNA gene for T. whipplei. We report the case of a middle-aged man admitted for etiologic investigation of prolonged, migrating, and inflammatory arthralgias and subsequent development of gastrointestinal symptoms. Despite its reputation as a great mimicker of many different illnesses, the difficulty in diagnosis probably lies with its rarity rather than its masking.
Introduction
Whipple’s disease is a multisystemic chronic infectious condition caused by the bacterium Tropheryma whipplei (T. whipplei) with an estimated prevalence of 1.1 per million.1,2 It was initially described in 1907 by George Whipple in a patient with malabsorption associated with mesenteric lymphadenopathy, arthritis and skin pigmentation. 3 In 1952, it was first thought to be of infectious aetiology as a patient with Whipple’s disease responded to treatment with chloramphenicol, but only in the year 2000, the bacterium was grown and isolated from the heart valve of a patient with blood culture negative endocarditis.1 –3 T. whipplei is present in stool samples of 1%–11% of healthy individuals and in 12%–26% of sewage plant workers; 2 nonetheless it is estimated that less than 0.01% of carriers will develop Whipple’s disease, 4 and that susceptibility depends on host genetic factors, specifically the HLA (human leucocyte antigen) alleles DRB1*13 and DQB1*06, the latter potentially correlated with the immune reconstitution inflammatory syndrome.1,2,4 Four distinct presentations could be observed: classical Whipple’s disease, localised infection, acute infection and asymptomatic carriage. The diagnosis is based on histopathological examination of small bowel biopsies and polymerase chain reaction (PCR) analysis of the 16S rRNA gene for T. whipplei.
Case
A 58-year-old male worker as an inspector at a local water company, was admitted for arthritis of large joints, diarrhoea, and elevated acute phase reactants. He reported inflammatory arthralgias of insidious onset, initially in the left hip joint and later in both knees and elbow joints, which had been going on for 18 months and thought to be rheumatoid arthritis despite no improvement after treatment with non-steroidal anti-inflammatory drugs and corticosteroids. In the last 6 months he had developed an intermittent watery diarrhoea, without blood or mucus, associated with anorexia and 9% weight loss. He denied other personal medical history but had a familial history of colonic polyposis, and a colonoscopy performed 4 months prior to admission, was unremarkable. On physical examination he had arthritis of the referred joints and generalised abdominal tenderness on palpation. Initial laboratory work-up showed mild hyponatremia (133 mmol/L), hypoalbuminemia (3.1 g/dL), folate deficiency (2.4 ng/mL), elevated reactive C protein (11.09 mg/dL) and leucocytosis with neutrophilia (13.7 × 109/L and 11.66 × 109/L, respectively). Stool multiplex PCR assay was negative. HIV and tuberculosis were excluded. Thyroid hormones, serum protein electrophoresis and immunoglobulins were within normal range. Rheumatoid factor, antinuclear antibodies and anti-citrullinated peptide antibodies were also negative. A computed tomography (CT)-scan of the abdomen and a positron emission tomography with 2-deoxy-2-[fluorine-18] fluoro-D-glucose integrated with computed tomography (18F-FDG PET/CT) performed to exclude underlying malignancies showed multiple para-aortic enlarged lymph nodes with mild 18F-FDG uptake and associated mesenteric fat densification. Given the history of arthralgia, diarrhoea, and weight loss, and facing the possibility of Whipple’s disease, an upper gastrointestinal endoscopy was carried out showing marked villous hypertrophy of the second portion of the duodenum (Figure 1), whose biopsies revealed macrophages with periodic-acid Schiff-positive inclusions, suggestive of the disease (Figure 2). Subsequent confirmation by PCR targeting T. whipplei genome sequences confirmed the diagnosis. Treatment was started with intravenous ceftriaxone 2 g od for 2 weeks and then oral trimethoprim/sulfamethoxazole 160/800 mg bid, according to patient’s preference for shorter duration of treatment. Despite initial clinical improvement, follow-up upper gastrointestinal endoscopy performed after 18 months of initiating treatment still had typical findings of Whipple’s disease, therefore treatment was changed to combination doxycycline and hydroxychloroquine.
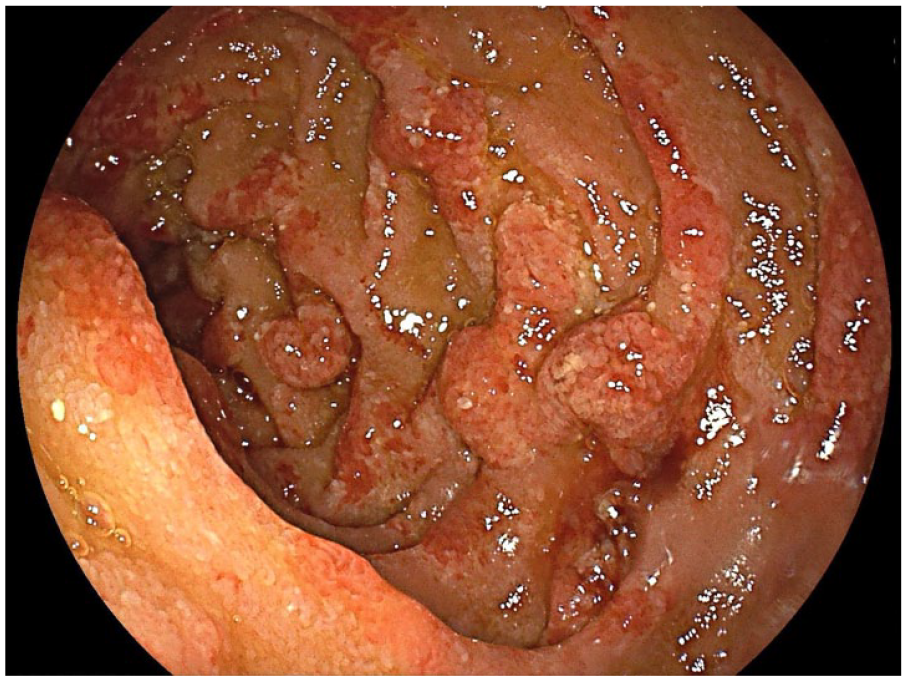
Upper gastrointestinal endoscopy with prominent folds and white punctate mucosal patches with an overlying yellowish exudate in the second part of the duodenum.
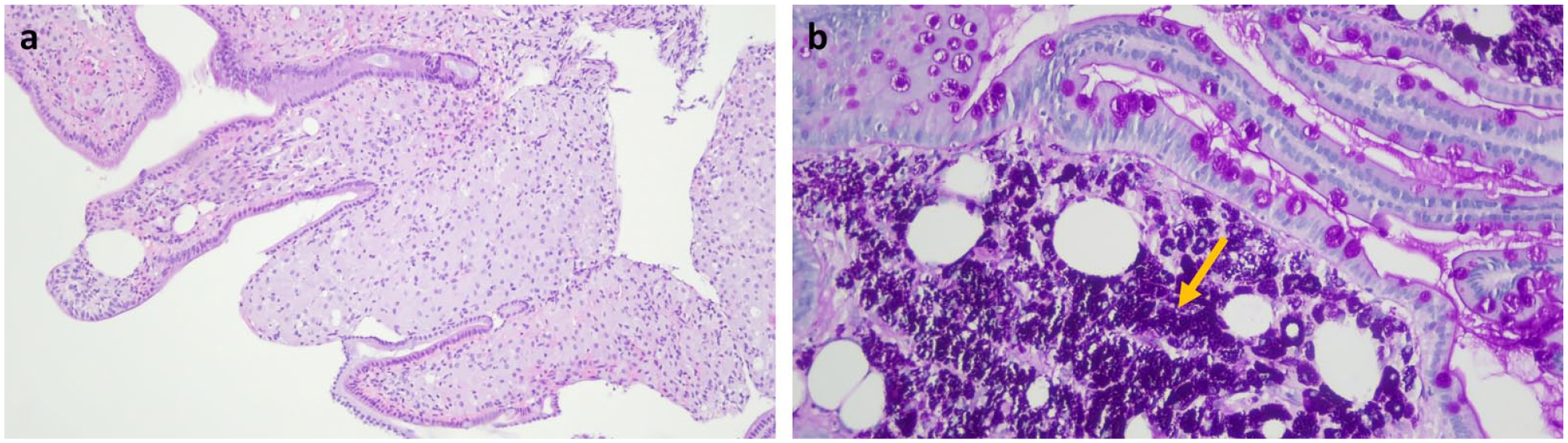
Duodenal villi distended by an infiltrate of foamy macrophages, haematoxylin-eosin, original magnification ×100 (a); periodic-acid Schiff staining showing macrophages with intense positivity (arrow), ×200 (b).
Discussion
Whipple’s disease characteristically affects middle-aged men. However, with improved awareness, Whipple’s disease is increasingly being recognised in women also leading to a decrease in this gender divide of prevalence of the disease. 4 Classical presentation generally manifests as intermittent arthralgia, arthritis, or other joint problem accompanied by chronic digestive symptoms (diarrhoea and abdominal pain).1 –4 Although these symptoms are frequent, Whipple’s disease is a rare disease, and a few patients might manifest without gastrointestinal symptoms. 5 Cardiac and pulmonary involvement have also been described in patients with Whipple’s disease; however, neurological involvement is the most complicated and feared within T. whipplei localised infections. 3
The natural course of classic Whipple’s disease is characterised by two stages,2,3,6 a first long-lasting prodromal stage, where arthritis is the first and most common manifestation followed by a second stage where gastrointestinal manifestations predominate. Manifestations such as weight loss, nausea, and diarrhoea can lead to hospitalisation due to dehydration, electrolyte imbalance or incapacitating symptom. A retrospective multicentric observational study by Tison and colleagues reported that 91% of patients with classical Whipple’s disease had rheumatological symptoms. 7 They usually present with a prolonged, migrating seronegative arthritis (most commonly of the knees, ankles and wrists) and associated with elevated acute-phase reactants that worsens with administration of immunosuppressive medications. Early arthritis was found to be detectable for an average of 6.7 years before the final diagnosis of infection. 8 Notably, it rarely leads to severe arthritis or deformities. Our patient had been followed for over a year and a half for polyarthralgia, presumed to be rheumatoid arthritis despite no improvement with immunosuppressive therapy. Although in line with everything said, an initial diagnosis of Whipple’s disease, based solely on polyarthralgia is, at least, far-fetched. Even after gastrointestinal symptoms started, Whipple’s disease was not our first consideration, as there are still other more common diseases with similar presentation, such as inflammatory bowel disease, human immunodeficiency virus or tuberculosis.
Localised T. whipplei infection frequency has been increasing over the years, probably due to the improvement of diagnostic techniques and consequent earlier diagnosis. 1 Cardiovascular disease is a common symptomatic presentation of systemic T. whipplei infection, second only to classic Whipple’s disease. 4 Typically seen in very late phases, neurological involvement by T. whipplei is the most feared complication of Whipple’s disease.1,3,8 Although cognitive changes such as dementia or pre-dementia and psychiatric signs (personality changes and depression) are frequently reported, the triad of dementia, supranuclear ophthalmoplegia and myoclonus is considered highly specific for Whipple’s disease. 3
There is no optimal regimen for treatment. Intravenous ceftriaxone for 2 weeks followed by oral trimethoprim with sulfamethoxazole (TMP-SMX) for 1–2 years or doxycycline and hydroxychloroquine for 12–18 months followed by lifetime treatment with doxycycline are both acceptable strategies.1 –3,6 However, the later treatment regime has been preferred in recent years based on recent evidence 9 showing no treatment failure in 13 patients who received combination of doxycycline plus hydroxychloroquine while a high failure rate (including on-treatment failure and relapse) with TMP-SMX was seen, similar to what we observed in our patient, even if induction intravenous therapy was given. Our patient preferred treatment with TMP-SMX because of the shorter treatment duration, but after the relapse he was initiated on treatment with doxycycline plus hydroxychloroquine.
PCR for T. whipplei becomes negative soon after antibiotic therapy has commenced, and patients should be followed up with duodenal biopsies at 6 months and 1 year after diagnosis to ensure complete remission.2,3 Relapses usually arise shortly after the cessation of antibiotics, but they can present after many years (average interval 4.2 years) even in patients in clinical remission and without treatment.2,6
Conclusion
Whipple’s disease has a reputation as a great mimicker of many different illnesses; however, the difficulty in diagnosis probably lies with its rarity rather than its masking. In patients with arthralgias, diarrhoea, abdominal pain, and weight loss, Whipple’s disease must be considered after exclusion of most common causes. In our patient’s case we first excluded HIV infection and tuberculosis, then other gastrointestinal diseases that present with diarrhoea or abdominal pain, such as inflammatory bowel disease, malabsorption syndrome or infectious diarrhoea. Seronegative polyarthritis was excluded after laboratory confirmation of Whipple’s Disease. A high level of suspicion is essential given that nowadays the diagnosis can be easily confirmed with upper gastrointestinal endoscopy, duodenal biopsy, and PCR-testing.
Footnotes
Author contributions
BTC and JM: drafted the main manuscript text. BTC, JM and AS: treated the patient in ward. AT: performed upper endoscopy. JIM reviewed histology. BTC, AT, AS and JM: provided important intellectual input and critically revised the manuscript. All authors reviewed the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
CARE guidelines were followed, and written consent was given by the patient.
