Abstract
Pancreatic exocrine insufficiency (PEI) is an under-diagnosed condition. Untreated PEI can result in developing gastrointestinal symptoms and long-term complications including weight loss, nutrient deficiencies, sarcopenia and osteoporosis. Current best practice recommends testing for PEI in certain disorders including chronic pancreatitis, cystic fibrosis, pancreatic cancer and post-pancreatic surgery. However, there is increasing evidence that PEI is associated with a number of conditions in addition to the aforementioned diseases. These ‘at-risk’ conditions are a heterogeneous group of diseases, for example, diabetes mellitus, people living with human immunodeficiency virus, high alcohol intake, and coeliac disease. The pathophysiology of some of ‘at-risk’ conditions is becoming increasingly recognised; therefore, the list of associated conditions are in evolving process. We present a case of a 60-year-old male with Parkinson’s disease and persistent abdominal pain who was found to have low faecal elastase levels indicative of severe PEI. His past medical history included none of the known risk factors for PEI. After examining the literature, we report a similar pathophysiological process underlying the development of pancreatitis and Parkinson’s disease which is dysfunction of the Unfolded Protein Response. We suggest further research to assess the prevalence of PEI in the population of patients with Parkinson’s disease.
Introduction
The pancreas is responsible for the secretion of several key enzymes for digestion, including amylase, lipase and proteases. 1 Pancreatic exocrine insufficiency (PEI) is defined as inadequate production or availability of pancreatic enzymes resulting in maldigestion and malabsorption. PEI is caused by a range of disease processes which affect the parenchyma or ducts of the pancreas. These include chronic pancreatitis, cystic fibrosis, pancreatic surgery and pancreatic cancer. There is emerging literature showing other conditions, termed ‘at-risk’ conditions such as inflammatory bowel disease, coeliac disease and human immunodeficiency virus, may also cause PEI, Table 1. Despite the known association, the pathophysiology linking PEI and these conditions is not yet well understood.
Prevalence of PEI in ‘at-risk’ conditions.
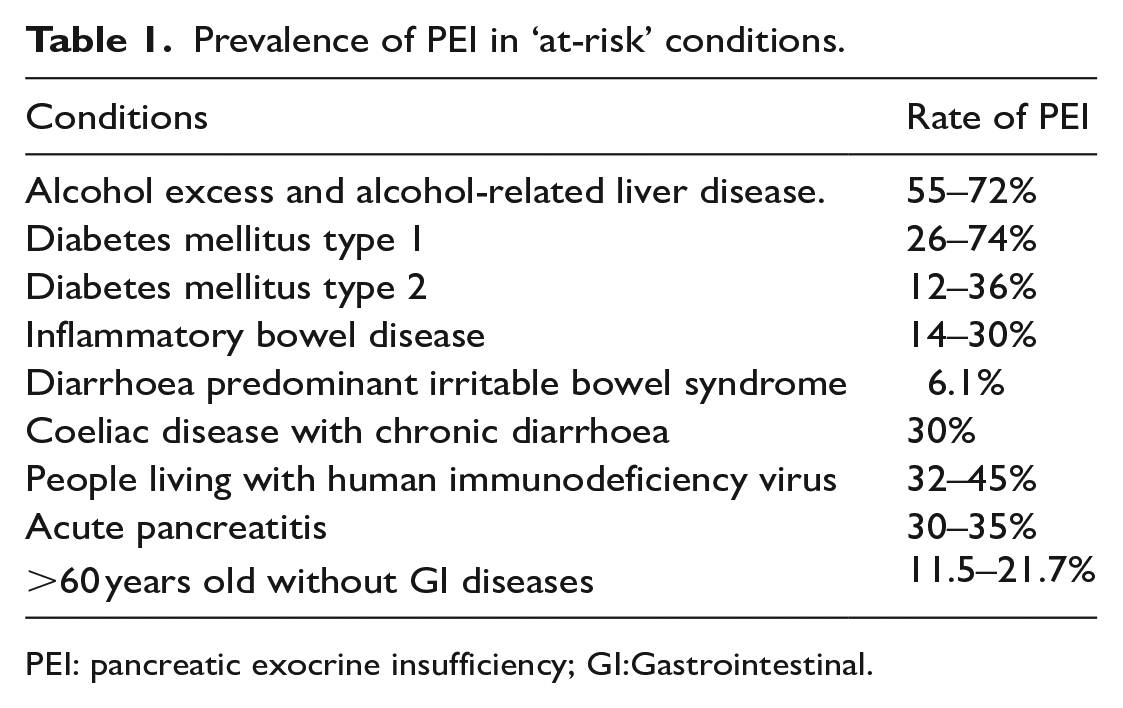
PEI: pancreatic exocrine insufficiency; GI:Gastrointestinal.
Detecting PEI can be challenging due to its non-specific symptoms. Faecal elastase (FEL-1) testing is the first-line investigation for PEI due to its non-invasive nature, and wide availability. FEL-1 has sensitivity between 54–90% and specificity of 80% in mild-moderate PEI.2,3 In severe PEI, FEL-1 sensitivity increases to 100% and specificity to 93% compared to a faecal fat collection (gold standard). 4 Screening for PEI is only performed in a limited number of conditions where the association with PEI has been well-established; for example, cystic fibrosis where it occurs in up to 85% of patients. The British Society of Gastroenterology recommends screening for PEI in all patients with chronic diarrhoea. However, as steatorrhoea is a late stage of PEI, this means potentially a large number of cases of PEI are unidentified. The significance of this is PEI can lead to malnourishment with a number of important substances including fat-soluble vitamins (A, D, E, K), magnesium, selenium, zinc and prealbumin. 5 Importantly, there is a widely accessible and effective treatment available for PEI in the form of Pancreatic Enzyme Replacement Therapy (PERT), commonly known by the brand name CREON. PERT has been shown to improve gastrointestinal symptoms and quality of life in patients with PEI.
Case
A 60-year-old male patient presented to gastroenterology clinic with a 4-year history of epigastric pain. The pain was not associated with food. He had no other gastrointestinal symptoms, apart from constipation which had now resolved following regularly ingesting Flax seed. His past medical history included Parkinson’s disease (PD) diagnosed in the preceding year. He denied ever experiencing pancreatitis. His regular medications were co-beneldopa and fludrocortisone. There was no family history of gastrointestinal or neurological symptoms. He had no smoking history and minimal alcohol intake. He weighed 76.2 kg with body mass index of 23.8. Examination was unremarkable with a soft, non-tender abdomen and no palpable organomegaly.
Blood tests including amylase, B12, magnesium, vitamins A and E were unremarkable. His coeliac serology was negative. An Oesophageogastroduodenoscopy was also unremarkable. An ultrasound abdomen showed biliary sludge but no signs of ductal dilatation or biliary pathology; furthermore, due to the pain not being related to food this was deemed very unlikely to be the cause. A FEL-1 test was requested showing a level of 44 µg/g. This test was repeated 22 months later, with a result of 37 µg/g. No other possible cause for PEI was identified in the patient’s history. A Computed tomography scan of abdomen and pelvis was performed as per guidelines showing no morphological changes in the pancreas. 6 The patient’s abdominal pain was attributed to PEI due to the array of normal investigations, excluding the consistently low FEL-1 levels. The patient was started on regular CREON. The patient was followed-up 6 months following the initiation of CREON; his epigastric pain was found to have completely resolved following this intervention.
Discussion
Parkinson’s disease is the second most prevalent neurodegenerative condition following Alzheimer’s disease. 7 It is caused by the development of intracellular structures called Lewy bodies which are associated with increased death of dopaminergic neurones in the substantia nigra. This leads to the classical symptoms of resting tremor, bradykinesia and rigidity seen. 8 Dysfunction of the Unfolded Protein Response (UPR) of the endoplasmic reticulum (ER) has been implicated in the development of these pathophysiological changes. The ER is an organelle which controls cellular protein synthesis, folding and modification. In periods of cellular stress, for example due to oxidative stress, the ER can malfunction causing the accumulation of misfolded proteins within it. This ultimately leads to cellular dysfunction and death, Figure 1. The UPR is a cellular mechanism which aims to restore ER homeostasis in response to this dysfunction. It helps to reduce the synthesis of proteins, improve the folding of proteins, and increase destruction of misfolded proteins. There is significant data showing dysregulation of the UPR is associated with PD development. For example, Ryu et al showed that administration of the molecules 6OHDA, MPP+ and rotenone, which are commonly used to mimic Parkinson’s due to their induction of dopaminergic cell death, correlated with increased levels of proteins associated with the UPR. 9
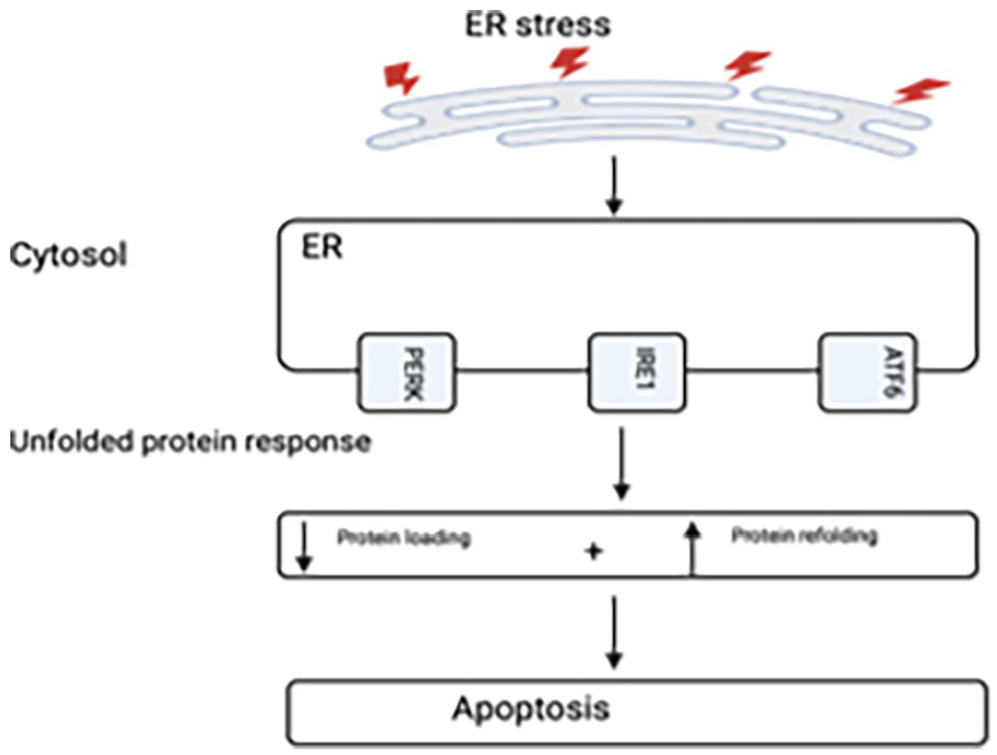
The unfolded response protein pathway.
There is also evidence demonstrating the UPR may also be implicated in the development of pancreatitis. Pancreatitis can result in long-term consequences including chronic pancreatitis and/or PEI. In mice which were chronically fed alcohol, pancreatic oxidative stress was induced and consequently the UPR activated. In mice where there was a genetic defect in the UPR, for example, X-box binding protein 1, there was an associated increase in pancreatic acinar cell death; cells which form the excretory part of the exocrine pancreas. 10 There is currently a paucity of studies establishing a correlation between the UPR dysfunction and pancreatitis in humans.
Although the finding of PEI in our patient with PD could be merely a coincidence, recent evidence showed that PEI can be under-diagnosed. PEI can result in maldigestion, weight loss, osteoporosis, and nutrient deficiencies. Similarly, weight loss is a common feature of Parkinson’s disease, with one study showing weight loss to be four times more likely to be present in patients with PD compared with controls. 11 Also patients with PD are found to be malnourished, with one meta-analysis showing the prevalence of malnutrition in the PD population to be 8.8%. 12
Therefore, untreated PEI in patients with PD can result in complications including malnutrition. Correcting malnutrition in these patients has been shown to improve quality of life and even slow disease progression. 13
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Patient consent
The authors declare that the patient has been anonymised.
