Abstract
Background:
Preliminary data suggest that the prevalence of pulmonary hypertension (PH) in patients with COVID-19 is around 13%, but its prognostic role remains unclear. Approximately 3% of patients develop chronic thrombo-embolic pulmonary hypertension (CTEPH) following diagnosis of acute pulmonary embolism (PE). It is recommended that patients are screened for CTEPH if they remain symptomatic 3 months following diagnosis of PE. The primary aim of the study was to assess the chances of persistent PH following PE secondary to COVID-19.
Methods:
We conducted a retrospective cohort study at a District General Hospital (DGH) in the United Kingdom. All patients diagnosed with COVID-19 and PE between April 2020 and October 2021 were examined. Patients were divided into two groups:·COVID-19 and PE with comorbidities (excluding pre-existing PH) and·COVID-19 and PE without comorbidities. We compared the ECHO features suggestive of PH between the two groups at the time of diagnosis of PE and at 3 months following treatment.
Results:
80 patients were included in the study (49 with comorbidities and 31 with no comorbidities). Average age of comorbidities and no comorbidities groups were 73 years and 70 years, respectively. Average PaO2/FiO2 ratio for comorbidities and no comorbidities groups were 170 and 195, respectively. Fourteen patients (13 with comorbidities and 1 with no comorbidities) died in total. Results showed that risk of persistent PH and subsequent mortality following PE in COVID-19 is 4.17 times and 1.32 times more in comorbidity group as compared to no comorbidity group, respectively (p < 0.001).
Conclusion:
Patients with comorbidities are at high risk of persistent PH and mortality due to PE secondary to COVID-19.
Keywords
Introduction
Acute respiratory distress disease caused by coronavirus 2 (SARS-CoV-2) has spread rapidly across the sphere causing over six million deaths to date. 1 Pharmacological and nonpharmacological actions have lessened the burden of disease and death to some extent. 2 Although, vaccines played a big role in controlling this pandemic, but for more than 2 years into the pandemic we are seeing the long-term effects of COVID-19.
Although it predominantly affects the respiratory system, multi-organ involvement has been reported with a multitude of manifestations, including impacts on the neurological, renal and gastrointestinal systems amongst others. 3
COVID-19 is noteworthy in its propensity to cause long-term multisystem sequelae, the epidemiological impact of which is yet to be declared. COVID-19 is a recognised risk factor for venous thrombo-embolic disease. 4
Data from the national registry in Sweden showed a relative risk increase of up to 46.40 for pulmonary embolism (PE) in the second week following COVID-19 infection. 5 The relative risk remained elevated compared to a control population for up to 110 days. The risk of PE is increased in those with severe COVID-19 infection and is associated with worse outcomes. Several mechanisms of hyper-coagulability have been postulated and it is thought that immune-thrombosis in addition to conventional thrombo-embolism plays a key role. A study by Han et al. confirmed the hypercoagulable state in COVID-19. 6 High levels of D-dimer, fibrinogen and fibrinogen depravity amount are associated with worse outcomes. 7 Previous studies on the incidence of venous thromboembolism in COVID-19 have proved contradictory results. A meta-analysis concluded that occurrence of venous thromboembolism in COVID-19 is about 13%. 8 Incidence of PE in patients with COVID-19 was 21% in another study. 9
There is sparse literature on the longitudinal outcomes of patients diagnosed with pulmonary embolism in the context of COVID-19 infection. It is thought that approximately 2–3% of patients develop chronic thrombo-embolic pulmonary hypertension (CTEPH) following a diagnosis of PE. 10
CTEPH is defined as mismatched perfusion defects associated with evidence of organised clots on imaging. The primary aim of the study was to assess the likelihood of developing persistent pulmonary hypertension (PH) following PE in COVID-19 patients. We also looked at the risk factors which pre-dispose to persistent PH in this cohort.
Methods
Study design and study time
We conducted a retrospective, cohort study at a District General Hospital (DGH) in the United Kingdom. All patients who were admitted with a diagnosis of COVID-19 and subsequently developed PE between April 2020 and October 2021 were examined. Patients’ ECHO findings were recorded at diagnosis of PE and at 3 months follow-up (normally ECHO is repeated in patients who had evidence of PH in their initial ECHO).
Data collection
Data was collected for age, gender, PaO2/FiO2 ratio, past medical history, ECHO findings at diagnosis and at 3 months interval.
Inclusion and exclusion criteria
Patients were divided into two groups.
First group comprised COVID-19 and PE with comorbidities (excluding pre-existing PH). These comorbidities include:
Hypertension
Diabetes
Renal failure
We did not include patients with conditions known to cause PH, for example, cardiac disease, lung disease, malignancy and previous PE.
We also excluded those patients who did not have a follow-up ECHO.
Second group comprised COVID-19 and PE without comorbidities.
We compared the ECHO features suggestive of PH between the two groups at the time of diagnosis of PE and at 3 months following treatment.
Statistical analysis
Statistical analysis was done using SPSS version 21. Chi square relative risk of persistent PH and subsequent mortality following the diagnosis of PE between two groups were calculated. p-Value of 0.05 or below was considered significant.
Ethical committee approval
The study complies with the Declaration of Helsinki. The study protocol was discussed with local R&D Department. A formal ethical approval was exempted as the study does not fall under the criteria of research as per health research authority guidelines, England. It falls under the category of service evaluation.
Results
From April 2020 to October 2021, 156 patients with COVID-19 and PE were admitted to our hospital. 80 patients fulfilled the inclusion criteria.
Forty two patients were male and 38 were female. 49 patients had comorbidities and 31 had no comorbidities. Average age of the group with comorbidities and the no comorbidities groups were 73 years and 70 years, respectively
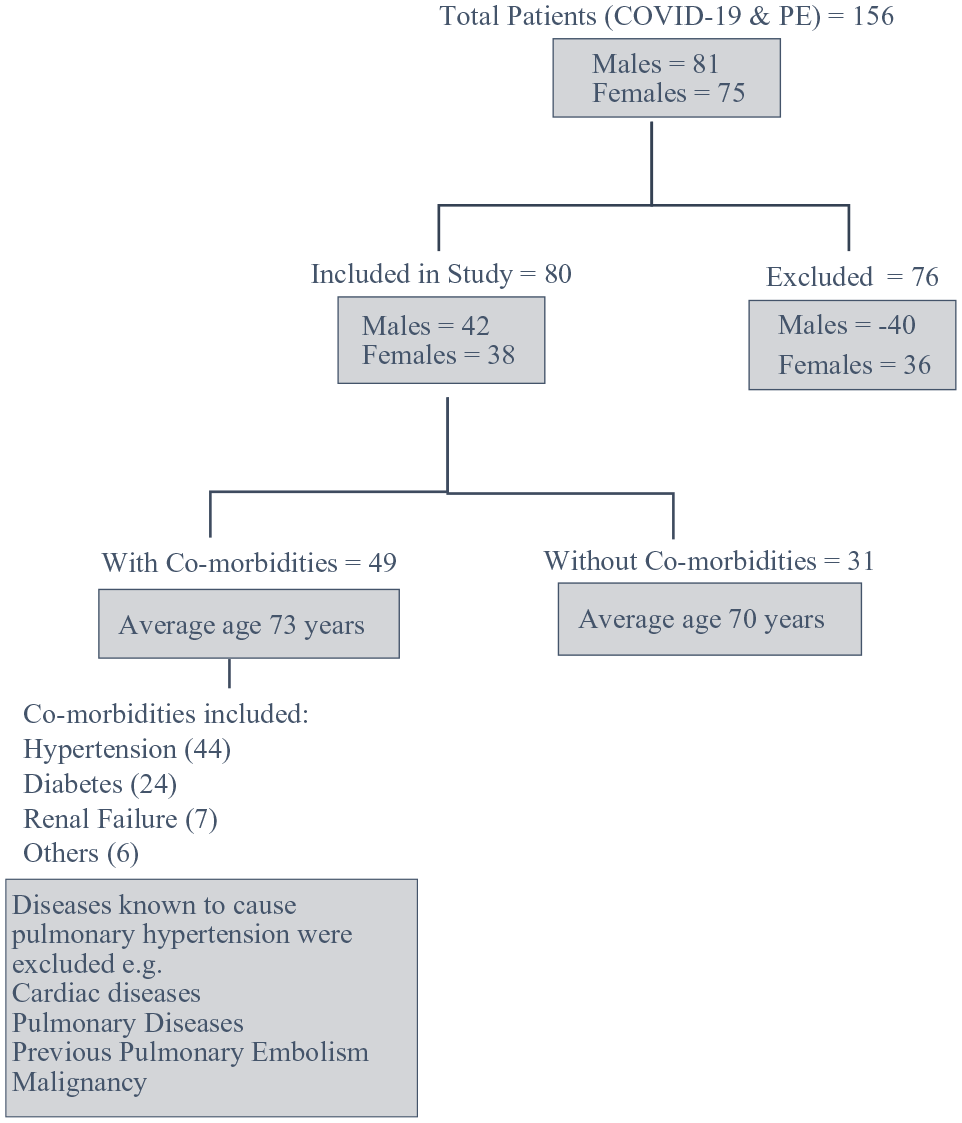
Showing study inclusion and exclusion individuals.
Average PaO2/FiO2 ratio for comorbidities and no comorbidities groups were 170 and 195, respectively.
Upon initial echocardiography pulmonary arterial hypertension (PAH) was present in 52 (24 with comorbidities and 24 without comorbidities). PAH was still persistent among 40 (38 with comorbidities and 2 without comorbidities) individuals on repeat ECHO imaging 3/12 after the initial study.
Chi square test showed significant association between persistent PAH and comorbidity status (p value < 0.001). Risk estimates showed relative risk of 4.17 (95% CI 2.45–7.07) which can be interpreted as risk of persistent PAH is 4.17 times more in those who have comorbidities as compared to those who do not have any comorbidity.
The risk of death was estimated in individuals with comorbidities and PAH in one-on-one relationship. This showed a relative risk of 1.32 (95% CI 1.10–1.57) which means that the risk of death is 1.32 times more in those who have comorbidities as compared to those who do not have it.
The relative risk for mortality with persistent PH was found to be 1.44 (95% CI 1.15–1.80) which means that the risk of death with persistent PH is 1.44 times more in those who have comorbidities as compared to those who do not have it.
14 (13 with comorbidities and 1 without comorbidity) patients died during the study time interval.
Discussion
Our study showed that if you have underlying health problems, PE secondary to COVID-19 is a massive risk for persistent PH and mortality.
PH definition and classification
PH is defined as a mean pulmonary arterial pressure of more than 20 mmHg at rest on right heart catheterisation. 11 PH is divided into five major groups with PH due to lung problems in Group 3 and CTEPH in Group 4. 12
COVID-19, PH and CTEPH
COVID-19 can have an effect on brain causing brain fog, inflammation and brain atrophy and can also affect cerebrospinal fluid volume.13,14
Studies showed that the frequency of acute kidney injury in COVID-19 patients could be as high as 25%. 15 COVID-19 can damage the kidneys and increase a patient’s risk of developing end-stage renal disease leading to ones requiring dialysis. Acute tubular necrosis in relation to COVID-19 is not uncommon either. 16
Gastrointestinal (GI) symptoms in COVID-19 are not infrequent and can occur in 50% of COVID-19 patients. The symptoms can range from diarrhoea and nausea on one extent to abdominal discomfort, colitis and hepatitis on the other. 17
COVID-19 can affect cardiovascular system enormously.18,19 PH and right ventricular dysfunction were frequently recorded leading to acute respiratory distress syndrome and respiratory failure.20,21 The proposed mechanisms of PH in COVID-19 are widespread pulmonary damage (due to interstitial and alveolar inflammation) as in PH of Group 3 and variations of pulmonary vasculature (induced by thrombotic/thromboembolic methods, endothelial injury, or, at least, hypoxic vasoconstriction), like in the PH of Group 4.
CTEPH is a distinctive form of PH. CTEPH is understood to evolve from one or multiple endothelialised pulmonary thrombi that lead to chronic obstruction of the pulmonary artery tree, causing high pulmonary vascular resistance leading to PH and progressive right heart failure. 22 The curative treatment choice is surgery by pulmonary endarterectomy.
A study by Joseph Newman et al. 23 showed a fall in incidence of CTEPH referral by 32% to PH centres and proposed that physicians should be wary of this rare condition, as untreated disease have a 5-year survival of only 53%. 24
A meta-analysis reported that the incidence of CTEPH following acute PE is 0.79%. 25
Our study showed that in high-risk patients, relative risk of persistent PH remained high 3 months after an episode of PE and these patients should be investigated thoroughly for possible CTEPH. This is in accordance with previous studies that emphasise that CTEPH should be considered in three categories of PE patients: (1) those with symptoms signifying for CTEPH; (2) those with signs suggestive of CTEPH at the moment of the acute PE diagnosis; and (3) those with risk factors for CTEPH 3–6 months after an episode of PE. 10
A study by Bonderman et al. 26 showed that splenectomy, infected pacemakers, and previous and recurrent venous thromboembolism (VTE) are strongly associated with CTEPH. Our cohort did not include any patients who were at high risk for CTEPH and that finding gives a school of thought for future research in that regard.
Diabetes was a common comorbidity in our cohort. Patients with diabetes frequently have reduced lung volumes. Various studies have demonstrated that patients with diabetes have a 1.6-fold greater risk for restrictive lung function patterns as compared with those who do not have diabetes. Poor lung reserve can lead to recurrent respiratory infections, chronic hypoxia and volume overload which can cause PH. Diabetes is also a big risk for ischemic heart disease and can also lead to diastolic heart failure which can lead to PH.
Six patients in our cohort had renal failure with a mean creatinine clearance of 45 millilitres per minute (mL/min). Renal failure is currently listed in Group 5 of PH and the prevalence of PH in one study was found to be up to 80% in end stage renal disease (ETRD) and has been linked to increased mortality.
Strengths and limitations of study
To our knowledge, this is the first study that has looked at PH and possible CTEPH after PE in COVID-19 patients. Findings of this study will also enable physicians to look and investigate possible CTEPH in patients presenting with persistent PH. Underlying health problems of hypertension, diabetes etc. are not known to cause PH, but our study has clearly depicted that these COVID-19 patients should be kept under close surveillance of medical teams. It has also depicted that mortality in COVID-19 is increased further if you have persistent PH on ECHO. The age and PaO2/FiO2 ratios of two groups were comparable which takes out the confounding factor out from the study.
The study has many limitations. The sample size is small and the study design is retrospective. Some patients were left out of the study as follow-up ECHO was missing. None of the patients were referred for right heart catheterisation and a diagnosis of PH was made only on basis of ECHO. None of the patients had a V/Q scan.
Conclusion
Patients with comorbidities are at high risk of persistent PH and mortality due to PE secondary to COVID-19.
Footnotes
Author contributions
NM and IN contributed equally as combined first authors. AB is the supervising consultant for the study.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
