Abstract
Polyethylene terephthalate (PET) is a versatile material with a wide range of applications, and the demand for PET products has been steadily increasing in recent years. This has generated large quantities of non-biodegradable PET waste. To reduce environmental pollution, recycling and reuse of waste PET is an economical solution. Different PET products vary greatly in their impurity contents, processing technology, and performance, which poses certain challenges for the recovery, purification, and reuse of PET waste in a high value-added manner. Various recycling and reutilization methods are compared, potential issues with different PET products in the recycling process are analyzed, and viscosity enhancement modification techniques and applications of recycled PET are introduced. This paper provides a reference for the recycling of various waste PET products.
Keywords
Introduction
Polyethylene terephthalate (PET), which is polymerized from purified terephthalic acid and ethylene glycol, is a thermoplastic polymer with a linear molecular structure, symmetrical dense chain stacking, strong intermolecular bonding, high glass transition temperature and high melting point. However, because of its conjugated structure, the crystallization rate is slow, which facilitates the production of amorphous PET products with excellent light transmittance. PET products typically have high tensile strength, abrasion resistance, optical clarity, chemical resistance, dimensional stability, and recyclability. They are also cost-effective, light weight, resistant to breakage, and reusable. Major PET products include bottle grade PET (used for packaging various beverages, especially carbonated beverages), film grade PET (used for packaging materials, films, and tapes), and fiber grade PET (used for producing PET textiles).
PET is one of the most widely used plastics in the world. According to statistics, the global annual output of PET exceeds 70 million tons, of which 66% is used for fiber and 30% for packaging. Only 14% of waste PET is recyclable. 1 PET has a stable chemical structure and is not readily degraded in the natural environment. Large quantities of PET waste have exerted environmental and resource pressure. The treatment of PET waste has become a global concern. Recycling technology has evolved from simple recycling to high-value utilization of modified waste PET.
However, there are still big gaps and great challenges that need to be overcome in the field of PET recycling and applications of recycled PET. Firstly, there is a pressing need to develop highly efficient and economically viable recycling techniques for PET since the efficiency and feasibility of current technologies and processes are rather low. Secondly, the physical and mechanical properties, chemical stability, and reliability of the recycled PET are usually inferior to that of the virgin PET. For example, the impurity content of the recycled PET could be higher due to the impurities introduced during the recycling process and degradation byproducts. As a result, the performance of recycled PET products may not always meet the stringent requirements of certain high-performance applications, limiting its full potential as a sustainable substitute for virgin PET.
To address these issues, there is an urgent need for improvements in PET recycling technologies. Advanced sorting techniques, enhanced cleaning processes, and improved solid-state polymerization are crucial in enhancing the recyclability of PET waste and producing high-quality recycled PET. This paper summarizes and compares existing PET recycling and reutilization methods, highlights the advantages and disadvantages of various methods, and discusses developments in this field.
Recycling methods for waste pet
PET is rarely used in composite materials in daily life, which makes it easier to recycle than some other polymers.2,3 Waste PET products are mainly recycled using physical, energy, and chemical recovery methods. Other methods, such as biological methods, can also be used to recycle and degrade PET.
Physical recovery
Physical recycling methods generally involve processing waste PET products into new products through simple mechanical treatment (e.g., crushing or melting) either directly or after reprocessing. The biggest advantage of physical recycling methods is their simplicity, and the major disadvantage is that the products obtained by physical recycling methods can usually only be downgraded. Physical recovery methods are generally be divided into mechanical, melting, and dissolution. 4 The mechanical method does not involve destruction of the original material and only requires simple crushing for direct processing into new products. The melting method involves simple mechanical crushing, followed by using various separators or suspension separation technology to remove impurities and then producing the recycled materials through techniques such as melt granulation. The dissolution method is similar to the melting method except that different solvents are used to dissolve crushed waste PET products, separate them, and then granulate them to obtain the recycled materials.
Energy recovery
Energy recovery is generally applied to waste polyester products that have been recycled multiple times or cannot be recycled. Waste PET products can be burned for energy conversion and used as a raw material for thermal energy generation. However, only waste PET with a high energy value can be used. Therefore, the problem of low energy value waste PET disposal needs to be addressed in the future.
Chemical recovery
Chemical recycling involves breaking down the waste PET products into lower molecules or monomers, such as ethylene terephthalate (BHET), terephthalic acid, dimethyl terephthalate (DMT), and ethylene glycol, through a series of chemical reactions.
5
The most used methods for chemical recovery are hydrolysis, alcoholysis, aminolysis, and ammonolysis. (1) Hydrolysis
In hydrolysis, ester bonds in waste PET are broken down to terephthalic acid and ethylene glycol monomers under the appropriate reaction conditions (pH, temperature, pressure, and catalyst). The hydrolysis can be classified as acidic, neutral, or basic according to the pH. While neutral hydrolysis is environmentally friendly but requires extreme reaction conditions, which limits its application.
Technological advancements have enabled the development of emerging techniques such as microwave-assisted neutral hydrolysis and supercritical hydrolysis. Unlike traditional heating methods, microwave pyrolysis provides non-contact, instant, and rapid heating with high precision. 6 Microwaves can accelerate processing by up to 1000 times. Microwave-assisted neutral hydrolysis not only speeds up PET hydrolysis but also enables the production of 2-pyridone-4,6-dicarboxylic acid, a valuable monomer for biodegradable polymers, from terephthalic acid using an integrated chemical-microbial process. 7
Research shows that controlling the temperature and pressure to achieve supercritical water greatly accelerates PET hydrolysis. Under these conditions, it takes just 2 min to reach 95% decomposition and 5 min to reach 100% decomposition compared with at least 30 min for conventional high-temperature hydrolysis.
8
Subcritical water also efficiently hydrolyzes PET. For example, with zinc acetate as a catalyst in a molten silica capillary reactor at 240°C, terephthalic acid recovery increases to 90.5% in the same reaction time.
9
Moreover, subcritical water can directly vaporize PET microplastics,
10
which offers a new solution for microplastics pollution. However, supercritical water hydrolysis shares the same downsides as neutral hydrolysis
11
and the extreme conditions (400°C, 40 MPa) and low ethylene glycol yields (10%) hamper industrialization.
12
(2) Alcoholysis
Alcoholysis is the most popular PET recycling method. Compared with hydrolysis, alcoholysis uses milder conditions, causes less equipment damage. The most common depolymerizing agents are methanol, ethanol, and ethylene glycol. Diols depolymerize PET more readily than monohydric alcohols, are safer, and provide better pathways for industrial application. 13 Scientists have explored the use of complex diols for PET depolymerization. Zhou et al14,15 used neopentyl glycol, dipropylene glycol and polypropylene glycol for the depolymerization of waste PET, and analyzed the effect of reaction temperature, catalyst and PET concentration. The recovered products showed good stability and were able to be used as raw materials for the preparation of aqueous polyurethanes.
Supercritical alcoholysis shows more promise than supercritical hydrolysis. The conditions in methanol are relatively mild and PET can be degraded to DMT and ethylene glycol in 30 min at 300°C and 923 MPa, with a yield of approximately 80% for DMT.
16
Homogeneous catalysis boosts PET degradation. Ultrafine ZnO nanoparticles in methanol and ethylene glycol give 97% PET conversion and 95% DMT yield in 15 min.
17
Pudack et al.
18
optimized supercritical methanol degradation, removing non-PET segments and impurities by distillation and crystallization to obtain high-purity monomers. Gases also accelerate PET degradation. For example, CO2-catalyzed supercritical alcoholysis effectively increases the monomer yield.
19
(3) Aminolysis and ammonolysis
The amination method uses methylamine, hexylamine, morpholine, ethanolamine, and polyamides to depolymerize PET into terephthalic acid and ethylene glycol. The conditions are easy to achieve and reactions are mild, but reaction times are long and the product diversity high, which has limited research in this area. Aminolysis often uses ionic liquids, simple salts (e.g., acetic acid or sodium acetate), metal-doped oxide nanoparticles, and microwave catalysis.
Ammonolysis uses anhydrous ammonia glycol to dissolve waste PET and produce terephthalamide and ethylene glycol. These products can then be used to obtain p-phenylene dimethyl diamine, 1,4-diamine, or cyclic amines. Ammonolysis rapidly achieves 99% conversion but has not been used in large-scale applications.
Other methods
Biological methods, glycolysis, and catalytic degradation have also been investigated to recycle waste PET. (1) Biological method
The biological method uses hydrolytic enzymes isolated from microorganisms for the degradation and recycling of waste PET (Figure 1). Microorganisms capable of producing keratinase-like hydrolases include the bacteria Thermobifida fusca, Thermobifida celllosilytica, and Ideonella sakaiensis, the fungi Humicola insolens, and Fusarium spp. Additionally, an enzyme, called LC keratinase.20–25 The conditions for PET degradation using hydrolytic enzymes are mild (typically below 100°C, at atmospheric pressure, in a buffer solution with a neutral to slightly alkaline pH).
26
The depolymerization reaction is usually carried out at a temperature of approximately 30–80°C. The substrate selectivity shown by the enzymes may enable them to selectively depolymerize specific PETs in materials containing multiple hydrolysable polymers without affecting the other polymers. Enzymatic degradation process.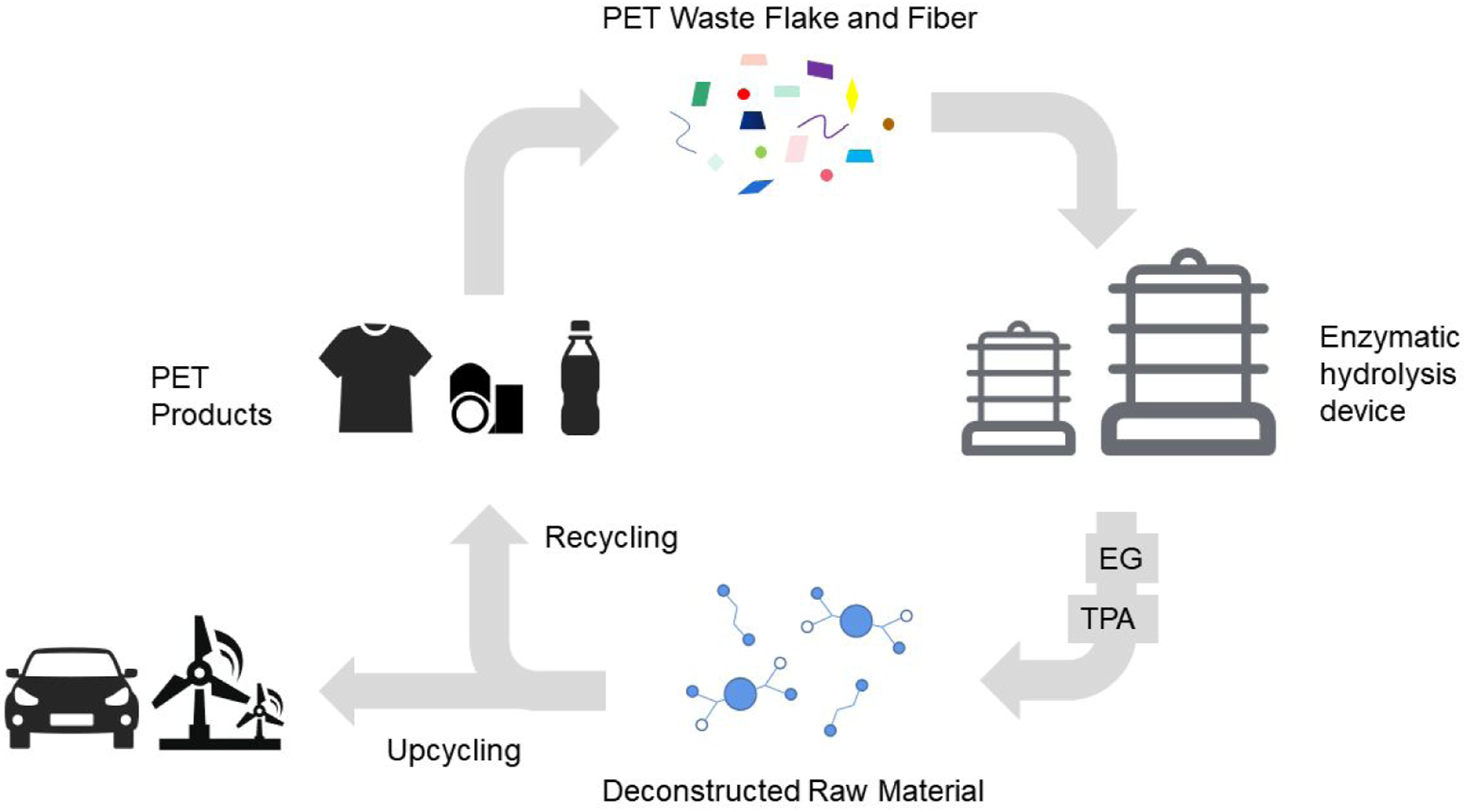
The enzymatic method has drawbacks. Most studies use low crystallinity PET, while PET fibers with higher crystallinity or polymorphic blends may require harsher hydrolysis conditions, producing PTA and oligomers. 27
Carbios, a French biotech company, commercializes an enzymatic PET recycling process. In their largest reaction, they treated 20 kg of pretreated waste PET in a 150 L reactor using engineered LC keratinase at 72°C and pH 8 for 12 h, achieving 90% recovery.
28
In 2020, Carbios partnered with Novozymes for PET-degrading enzymes. Carbios plans a 40,000 t/y production line in 2024.
29
(2) Glycolysis method
Glycolysis degrades PET chains at 180°C–240°C using excess diol solvent and a transesterification catalyst. The product is an α,ω-dihydroxy compound. Common solvents include ethylene glycol, propylene glycol, and diethylene glycol. Catalysts such as metal salts, ionic liquids, and hydrotalcite can be used to improve the reaction efficiency. The product of glycolysis is not necessarily monomers. Because the products or intermediates can be reacted with maleic anhydride to give unsaturated PET.
27
(3) Catalytic degradation
Improving catalytic methods is a priority for addressing the shortcomings of PET recycling methods. Kratish et al. 30 used a Hf(OTf)4 homogeneous catalyst and Pd/C non-homogeneous catalyst under hydrogen to achieve solvent-free selective hydrogenolysis of PET (Figure 2(a)). Liu et al. 31 used carbon nanotube (CNT)–PDA to convert solar energy into heat to drive PET glycolysis with an ionic liquid catalyst (Figure 2(b)). This achieved selective, cost-effective PET recovery from a waste mixture at three times the efficiency of thermal catalysis. Jiang et al. 32 used a polyionic liquid PIL–Zn2+ for PET methanolysis and obtained DMT in 89.1% yield in 60 min at 170°C, which shows potential as an alternative to PET methanolysis (Figure 2(c)). Gopal et al. 33 synthesized kaolinite and bentonite loaded with 5% Zn2+, Al3+, and heteropolyacids by wet impregnation. The heteropolyacids gave better glycolysis and amino acid depolymerization of PET at 150°C–210°C than Zn2+ and Al3+ (Figure 2(d)).
Figure 2 summarizes and compares the mechanisms of different catalysts for PET recycling. All these catalysts act on PET ester bonds, making them more susceptible to breakage and thus degradation under readily achievable experimental conditions. However, the catalyst preparation cost is generally high, which makes large-scale implementation difficult.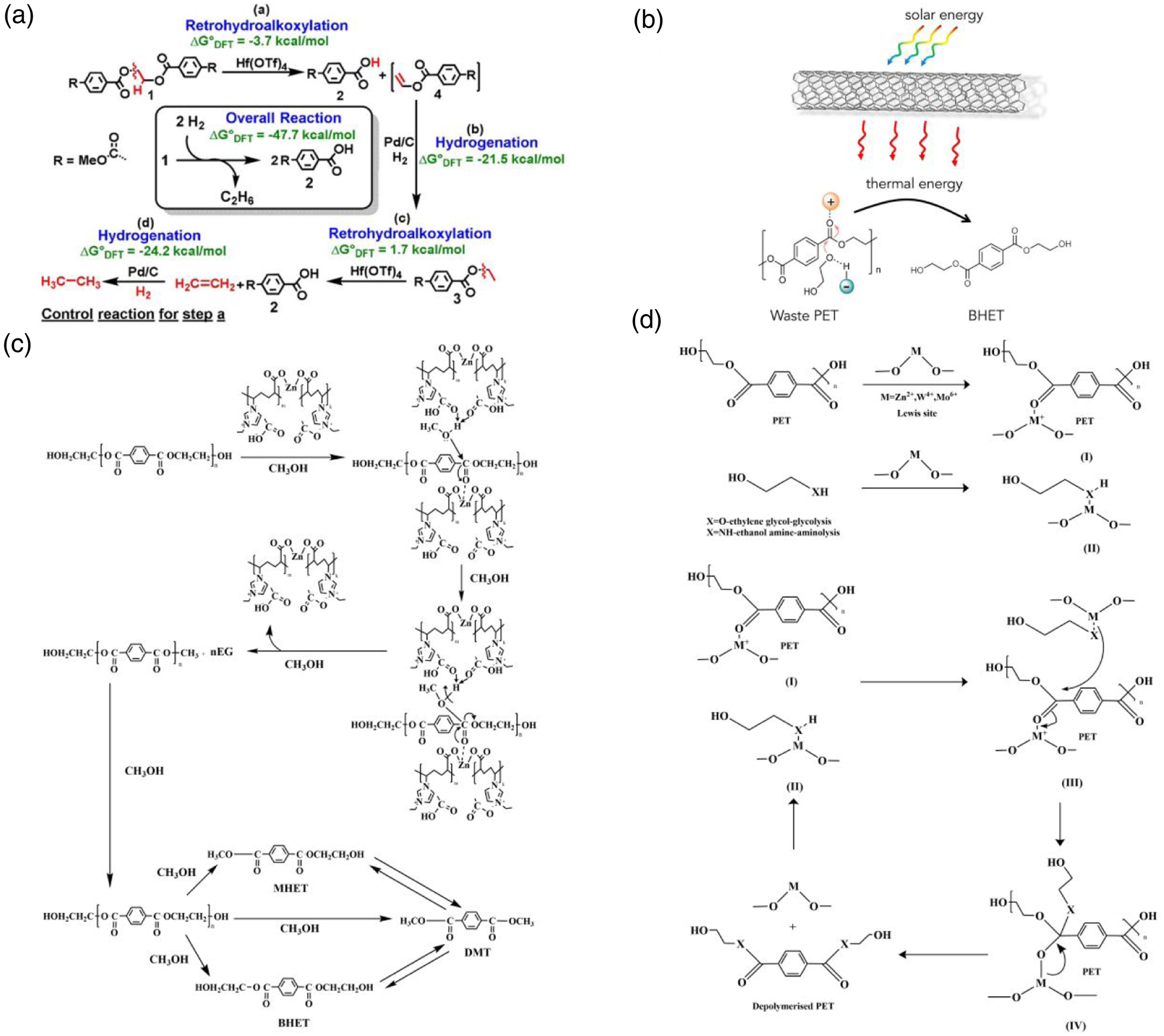
Recycling of different kinds of waste pet
Common PET products include textiles, flakes, and films, which all differ in their viscosities and physical and chemical properties. Because of these differences, PET products have different recycling method requirements, issues, and results. The recycling methods for each product type are discussed in this section.
Recycling of waste PET textiles
There are two primary sources of waste textiles: (1) Post-consumer textiles from daily life, including old clothing, shoes, socks, bedding, and curtains. (2) Industrial textile waste from manufacturing, such as silk, garment, and chemical fiber remnants.
PET textile waste can be recycled by physical recycling via simple methods, such as cutting old clothes into strips of cloth to be used as table cloths, thermal insulating materials for buildings, or carpet padding. 34 Chemical recycling breaks down PET fibers into monomers to produce other polymers or improve the PET properties. For instance, Cho et al. 35 produced highly hydrophilic polyester fabrics in under 3 min using microwave-assisted, which greatly reduced the energy requirements and cost.
The use of catalysts has been explored to improve the PET recycling efficiency and reduce the cost and environmental impact. For instance, Bartolone et al. 36 used recoverable magnetic γ-Fe2O3 nanoparticles but the method required a temperature of 300°C. Al-Sabagh et al.37,38 deposited Fe3O4 on CNTs and obtained a 100% yield when the reaction was conducted at 190°C for 2 h. Guo et al. 39 usedMg-Al-O@Fe3O4 microparticles to obtained BHET in 80% yield in 90 min at 240°C with 0.5% catalyst. The deactivated catalyst could be regenerated via heat treatment and performed in a similar manner. Catalysts and processes are constantly being refined to improve PET recycling rates in a cost-effective, scalable manner.
Waste PET textiles often contain polyester–cotton blends, which require separation for recycling. The following methods are commonly used for separation: 40 alcoholysis, solvent method, enzymatic hydrolysis, ionic liquids, hydrothermal method and so on. These methods all achieve separation by selectively dissolving or degrading PET over other fibers based on their difference in solubility, chemical bond stability, and susceptibility to degradation.
Separation of dyes is also important when recycling waste PET textiles. Common methods include high-speed kneading, kneading and rinsing, melt shear dispersion, and three-roller grinding.
41
Dye destruction and dye extraction methods are commonly used to achieve decolorization. However, neither of these methods can provide sustainable, complete color removal without damaging the polymer. Dye destruction degrades the polymer and alters dyeability. Dye extraction does not fully remove color and requires harsh conditions like high temperature (Figure 3(a)), long reaction time, and caustic solutions, which can damage the polymer. Mu et al. used solvent systems with different solubility parameters, like DMSO, ethyl carbonate, glycerol, and tetramethyl urea.
42
This achieved high decolorization without polymer damage (Figure 3(b)).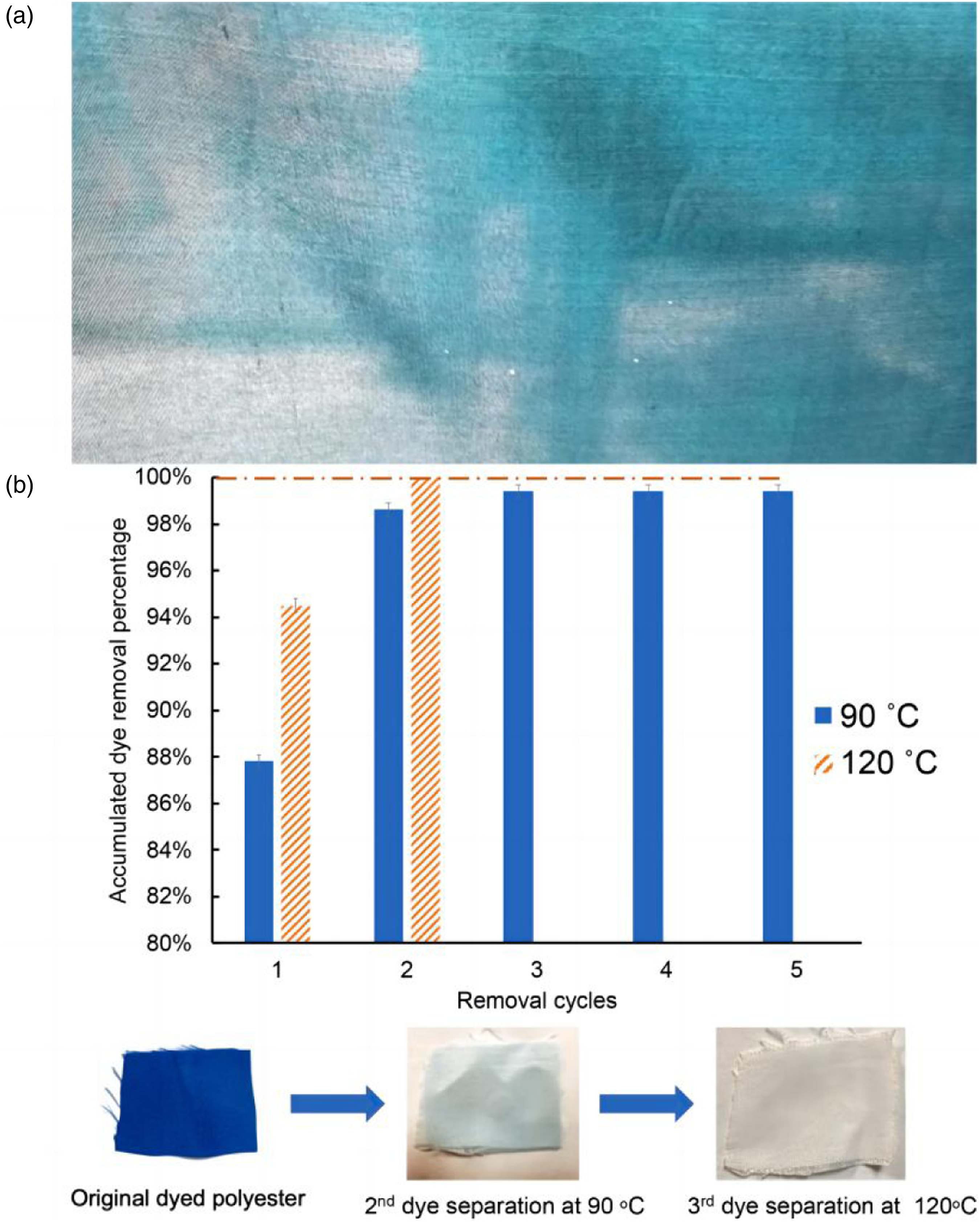
Although recycling of waste textiles has been investigated extensively, recycling of waste PET textiles still faces limitations. 43 Fibers often contain additives like pigments, UV stabilizers, and flame retardants, or harsh treatments are required that adversely affect the fiber properties. Consequently, further progress is needed to recycle waste PET textiles.
Recycling of waste PET bottle pieces
Compared with recycling of waste PET textiles, recycling of used PET bottles is more advanced. Bottles contain fewer additives and pigments than textiles and can be easily sorted using automated Near-Infrared Reflectance Spectroscopy (NIRS) technology. Wu et al. 44 compared the properties of recycled PET fibers, bottle flakes, and virgin PET pellets. The recycled PET fibers had lower molecular weights and inferior properties and contained more impurities.
Recently, PET bottle recycling has been expanded beyond traditional chemical methods. Studies have used renewable catalysts to depolymerize bottles into monomers in 2 h. 45 Metal–organic frameworks like UiO-66 and MIL-140A have been used to effectively degrade PET under H2 or Ar. The pigments contained in colored PET can be directly removed by activated carbon during the degradation process, and these catalysts are reusable, which means that bio-based materials can be used as catalysts for PET degradation.
The recycled PET bottle flakes can be used in other applications. Chan K
46
et al. converted waste PET bottle flakes into nitrogen-doped carbon dots with significantly enhanced fluorescence (Figure 4(b)) by eight different treatments (Figure 4(a)). It has also been shown that PET waste bottles can be used as a raw material to synthesize graphene using hot press tanks (Figure 4(c)),
47
which can significantly reduce the emission of harmful gases and is less harmful to the environment.
With increases in the capacity for production of PET bottle flakes and demand, it is crucial to improve and optimize the technology for recycling waste PET bottle flakes. Achieving green closed-loop recycling of PET bottle flakes and large-scale application, and enhancing automation technologies such as crushing and sorting, are the future priorities of this industry.
Recycling of waste PET film
PET film is often used as part of a laminated film, along with other materials, which means that recycling of PET film faces the same material separation problems as recycling of waste PET fibers. Guclu et al. 48 used DMF to separate and recover waste Thin Film Composite (TFC) based reverse osmosis (RO) membrane flakes with a reactive polyamide top layer, a polysulfone middle layer, and a nonwoven PET bottom layer. The nonwoven PET bottom layer and reactive polyamide top layer did not dissolve, while the polysulfone middle layer dissolved completely and was then recovered as flakes to prepare a clean PET product that could be reused in new applications. There have been few reports on composite membrane separation and recovery, and there are many technical challenges in this field.
PET films can be treated with microorganisms to increase the degradation rate. Huang et al. 49 used stenotrophomonas pavanii JWG-G1, for bacterial pretreatment of PET films. This was followed by degradation of PET using thermobifida fusca cutinase. The highest degradation rate of the PET film was approximately 91.4% after pretreatment with the JWG-G1 strain for 8 days. This degradation rate was 14% higher than that of the untreated PET film. This approach is promising for the industrialization of plastic biodegradation. These soluble products can be recycled into new plastic products. Subsequently, the soluble residue is mineralized by microorganisms into CO2 and H2O. This method not only improves the recycling rate of waste plastics, but also reduces environmental pollution.
Research on waste PET films is relatively limited and further investigations are required.
Different techniques for extension of pet chains to increase viscosity
PET is exposed to oxygen, light, high temperatures, shear, and water during use and recycling, all of which can cause degradation through chain breakage and a decrease in the molar mass of the polymer, which impedes the recycling of used PET. 50 To improve the molecular weight and characteristic viscosity of waste PET and thus improve its mechanical properties, PET can be tackified by melt polycondensation, solid-state polymerization (SSP), liquid-phase polymerization, and chemical tackification to meet the production requirements of different PET products.
Melt polycondensation
Melt polycondensation is an industrial manufacturing process that is used to improve the intrinsic viscosity of PET by adding an additional reactor after the PET polycondensation kettle to increase the reaction time. This method is rapid, clean, and is currently the most economical industrial route. However, because melt polycondensation needs to be carried out at high temperature, pyrolysis occurs and affects the molecular weight of the PET (the molecular weight of the polycondensed product is generally approximately 23 000) and may cause yellowing of the product. Secondly, melt polycondensation usually uses a metal catalyst to facilitate the polymerization process and increase the molecular weight of PET. 51 However, most metal catalysts pose a non-negligible risk to human health and the environment. 52 Additionally, there is a need to reduce the acetaldehyde content of the resulting product through deacetalization technology. Consequently, there have been very few studies on melt polycondensation.
SSP
SSP is currently the most widely used method for increasing viscosity in industrial production. This method involves heating the solid prepolymer to molten in an inert gas purged or vacuum environment. According to the two-phase model (Figure 5), in SSP, the end groups and low molecular weight substances (condensates and oligomers) are excluded from the crystalline region and polymerization occurs only in the amorphous region. SSP is usually divided into three stages: crystallization, preheating, and polycondensation.
53
The two-phase model in the SSP process. Reproduced with permission from ref.
53

Compared to the melt polycondensation method, the thermal degradation and other side reactions in the solid phase polycondensation process can be effectively suppressed. Therefore, the SSP method has the following advantages: better thermal stability of the sample, and the polymer crystallinity and crystal perfection are gradually improved. Additionally, the oligomer content is very low throughout the process, the process can be operated continuously with little environmental pollution, and it is less costly. 54 However, the solid-phase polycondensation method also has relatively obvious disadvantages, such as longer chain reaction time and slower reaction rate, more equipment is required for the production line, and high production costs. Moreover, it is not conducive to the discharge of small molecule products and also triggers bonding. The reaction rate is influenced by parameters such as the temperature, particle size, crystallinity, nature and flow rate of the carrier gas, and catalyst concentration. 55
Reaction temperature
The reaction temperature is the most important factor affecting SSP. The reaction temperature can influence the rate-limiting steps of the process and may lead to changes in the mechanism, which can affect the activation energy of the reaction. 56 Chemical reactions play a controlling role when the effect of the reaction temperature is relatively strong. 53
Increasing the SSP temperature accelerates the chemical reactions, makes functional end groups more mobile, and increases the rate of by-product diffusion. 57 The appropriate SSP temperature should be approximately 20°C below the final Tm to prevent particle agglomeration. 58 To solve the problem of polymer particle agglomeration in SSP, a phased approach can be used, where the solid phase condensation is carried out continuously at different temperatures, and this approach can also reduce the formation of oligomers.
Carrier gas properties
The use of inert gases in SSP systems serves three main purposes: removal of condensate, inhibition of polymer oxidation by removing oxygen from the reaction system, and heating of the reaction material. The most used inert gases in SSP are nitrogen (N2), carbon dioxide (CO2), helium (He), superheated steam, and supercritical carbon dioxide. Helium has a smaller molecular size and higher diffusion coefficient than both N2 and CO2, which means that higher free volume growth rates and higher DPs can be obtained using He. CO2 is more soluble in polymers, induces plasticizing effects, and greatly increases local mobility compared with N2. The reaction activation energy of super critical CO2 is approximately half that of N2. Additionally, super critical CO2 can induce a plasticizing effect that increases the end-group mobility and facilitates SSP. 59 SSP proceeds faster under vacuum than under nitrogen 60 because of faster removal of by-products. However, the return to atmospheric pressure may lead to oxidation and discoloration of the polymer.
The presence of small amounts of water in the carrier gas can have a large effect on SSP and can greatly decrease the molar mass of the final product. With increases in the water content, reaction temperature, and reaction time, the degradation reaction caused by water can greatly decrease the quality of the final polymer. 61
Other factors
The intrinsic viscosity also affects the SSP process. The intrinsic viscosity is affected by the end groups and the number average molecular weight. Because the end-group concentration is effective in limiting the amorphous phase, the concentration of the reaction chain ends in the amorphous region of a prepolymer with higher Mn0 content is higher and more uniformly distributed, and the tendency to crystallize during polymerization is lower, which means that end-group migration is less inhibited. This inhibits the movement of end groups. Therefore, the lower the end group concentration (lower the intrinsic viscosity), the lower the number average molecular weight at the end of the SSP reaction. 62
Another factor influencing the SSP is the particle size of the prepolymer. Under certain reaction temperatures, the SSP rate increases when the polymer particle diameter decreases, which indicates that the diffusion of by-products in the solid polymer (internal mass transfer) is rate controlled. The reduction in particle size may lead to a change in the mechanism, where the reduction in polymer particle size undergoes a shift from internal diffusion control to surface diffusion control.
Finally, the degree of crystallinity also affects the SSP rate. The effect of crystallinity on the SSP rate is bidirectional. On the one hand, an increase in crystallinity leads to a higher concentration of expelled end groups in the amorphous phase, which increases the reaction rate. On the other hand, as SSP proceeds, the polymer chain mobility decreases because of increased crystallinity, which also hinders the release of by-products from the reaction mass. Thus, the diffusion rate decreases with increasing crystallinity. 63
Liquid-phase polymerization
Similar to the solid-phase condensation viscosification method, the liquid-phase viscosification method also uses inert gas purging or a vacuum environment. However, the liquid-phase viscosity-enhanced prepolymer is in the molten state, and the reaction temperature is above the melting point. Under these conditions, the reactive end groups start to move, which increases the reaction rate.
The advantages of the liquid-phase viscosification method are numerous. The reaction time can be greatly reduced from more than 30–40 h for SSP to just a few minutes, and the resulting product can be processed directly without the need for re-granulation. Overall, this method has a short reaction time and process flow, and can be used to treat PET using existing polycondensation equipment at low cost. 64
Chemical tackification
Chemical tackification is a modified liquid-phase tackification method in which a chain extender is added to the liquid-phase polycondensation to boost the molecular weight of PET.
Common chain extenders include epoxy resins, isocyanates, oxazolines, and acid anhydrides. These chain extenders react with the terminal carboxyl groups of PET to lengthen the chain. The ring-opening reaction of epoxy resin (Figure 6(a)) with PET can occur between the epoxy ring and PET carboxyl group or hydroxyl group. The -NCO group of the isocyanate molecule is highly chemically reactive and all -NCO groups can react with PET end groups (Figure 6(b)). Oxazolines (usually bisoxazolines) undergo ring-opening reactions with two PET carboxyl end groups to form certain byproducts (ester amide groups) (Figure 6(c)). 1,2,4,5-Benzenetetracarboxylic anhydride (PMDA) is the most common chain extender containing anhydride functional groups. The anhydride functional group of the dendritic branching structure of PDMA reacts with the two hydroxyl groups of PET to produce a double-branched arm of the trans structure. Subsequently, PMDA reacts with two new PET molecules, with the first reaction occurring faster than the second one. At the end of the reaction, a four-armed star-like structure is formed (Figure 6(d)). Reaction mechanisms of various chain extenders (a) Epoxy, (b) Isocyanate-based molecules, (c) Dioxazoline, (d) 1,2,4,5-homophthalic dianhydride (PMDA).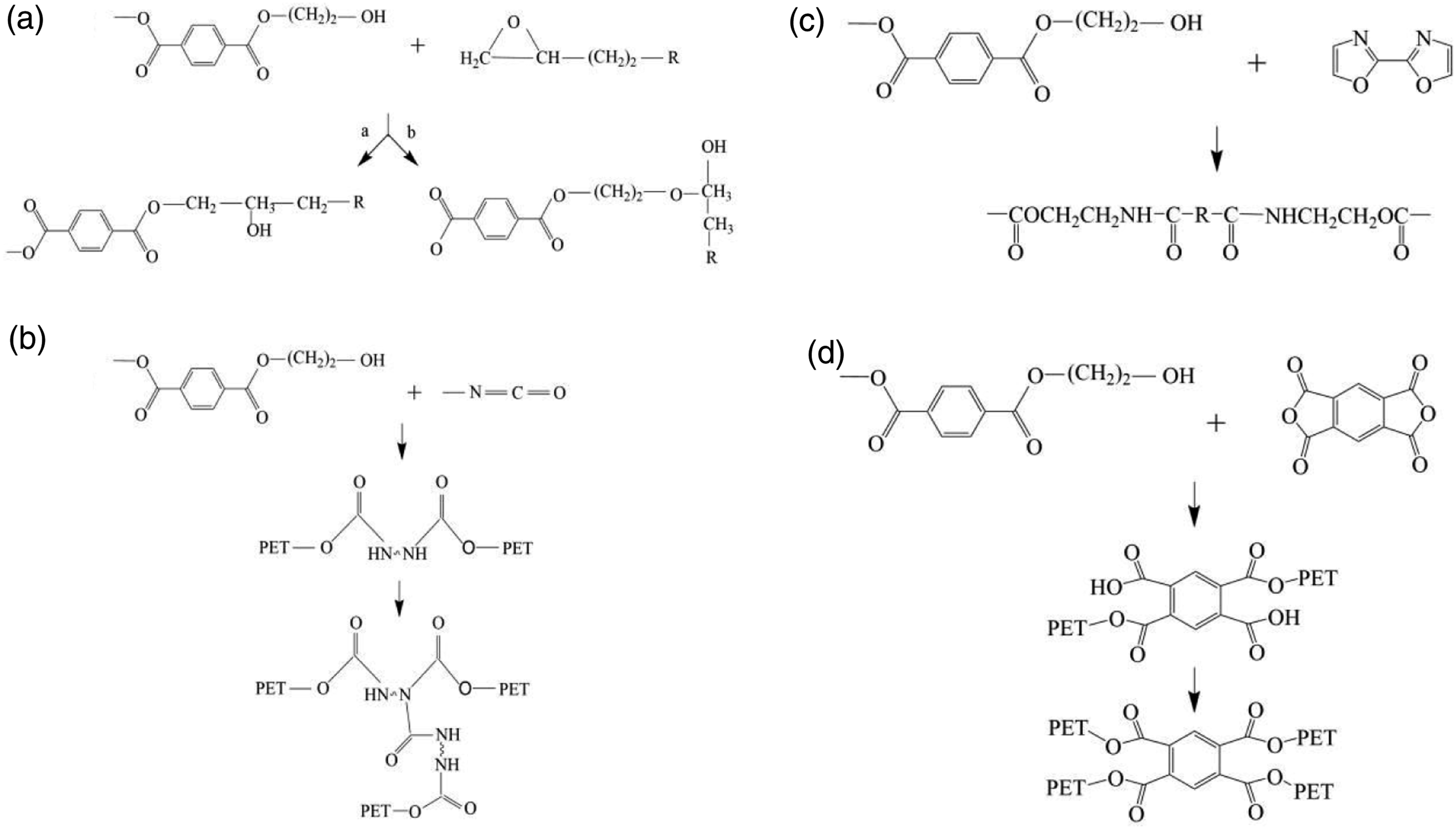
In addition to the commonly used chain extenders mentioned above, scientists are constantly searching for other polymers that can be used as chain extenders with higher efficiency. Zhao et al.65,66 used isocyanate trimer as a trifunctional chain extender to lengthen the chain and increase the viscosity of PET. When the chain extender content was increased from 0.6% to 1.4%, the viscosity-averaged molecular weight of PET effectively increased from 2.36 × 104 g·mol−1 to 5.46 × 104 g·mol−1. Many studies have looked at multifunctional oligomeric chain extenders, which are represented by Joncryl. Duarte et al. 67 found that the molecular weight of PET increased with increasing Joncryl concentration, and that higher rotor speed was favorable for PET chain expansion and increased viscosity. The use of Joncryl as a modifier is accompanied by gel generation and the modified PET is more elastic than when using PMDA as a chain extender. Additionally, Joncryl plays a role in controlling the degradation of nanoparticle-containing PET nanocomposites. 68
Chain extension and crystallization
PET has very low crystallinity and needs a long cycle time to achieve the required crystallinity because of its high structural rigidity. The level and morphology of crystallization during processing has a large impact on the final properties of the polymer. In the case of recycled PET, the addition of chain extenders during the recycling process can reduce the crystallinity of the product. Additionally, when nucleating agents are added to improve crystallinity, the PET matrix undergoes “nucleation-induced degradation”, resulting in severe PET chain breakage, which leads to a decrease in the mechanical and rheological properties of PET. 69
To solve this problem, a nucleating agent has been combined with a chain extender to modify PET. Huang et al. 70 used a synergistic approach combining chemical nucleation (sodium linoleate) and chain expansion (glycidyl methacrylate/trimethylpropane triacrylate) to modify PET, achieving a joint increase in the characteristic viscosity number and crystallinity. Liu et al. 71 used poly(phenylmethylene isocyanate) as a chain extender and Surlyn as a nucleating agent to increase the viscosity and crystallinity of PET.
Currently, PET tackifying methods have advantages and disadvantages, and research on PET tackifying chain extenders is ongoing. Identification of economical and efficient viscosity enhancement methods and environmentally friendly chain extenders with high activity is crucial to PET viscosity enhancement. Methods to concurrently increase the viscosity of spun-grade recycled PET chips and enhance the overall performance of the resulting fibers are also required.
Application of recycled pet fiber
Both recycled PET and virgin PET can be used to produce bottles; however, recycled PET is more often used as fibers for consumer applications because of differences in its impurity content and viscosity compared with virgin PET. These fibers are inexpensive and can be produced with low energy consumption and low carbon emissions. The recycled PET fibers are mainly applied as spinning materials and textiles, industrial filaments, functional fibers, and composite materials. 72
Spinning materials and textiles
Many textiles in daily life are made from recycled PET fibers, such as carpets, blankets, and garments. Fabrics produced with recycled PET fibers have different properties than those produced with virgin PET fibers, for example, the breaking strength of 100% recycled PET fabric is lower than that of 100% virgin PET fabric. 73 However, the blended fabric prepared using recycled PET and other raw materials (especially cotton fibers) is of similar quality to that prepared using virgin PET. 74 Yüksekkaya et al. 75 studied the performance of blended fabrics produced from virgin PET and recycled PET with cotton. They used rotor spinning machines to produce yarns containing 100% primary, 100% recycled, and 50% primary/50% recycled fibers and found that the yarns produced with recycled fibers were of better quality. There are significant limitations in using recycled PET to produce partially oriented yarns, stretch-deformed yarns, and fully stretched yarns for textiles, microfilaments, and cordage. 76 These differences may be caused by the spinnability, molecular weight, and rheology of recycled PET. 77
Industrial wire
PET fiber cord is a critical tire reinforcement material. Previously, it was challenging to apply recycled PET in cord because of its low mechanical strength. However, several companies worldwide have used recycled PET fibers made from waste PET in tires and recycled PET cord from scrap tires. 78 The first company to realize the application of recycled PET tire cord was Teijin, who introduced Eco Circle fiber (made from recycled waste PET) to the European market in 2009. In 2021–2022, Michelin and Continental also achieved this technology. Michelin, in collaboration with the French biotechnology company Carbios, used PET degradation enzymes to recycle recovered PET and successfully produced recycled PET fibers that met the performance criteria for tire cord. 79 Continental used ContiRe.Tex technology (a new recycling process that does not require chemical decomposition) to prepare recycled PET fiber cord from PET bottles and compounded the cord with dandelion-based rubber or natural rubber to manufacture green tires, which were launched in the European market in June 2022. In 2022, Goodyear announced they were developing an environmentally friendly tire containing 90% sustainable materials using a chemical process that converts PET recovered from plastic bottles and other plastic waste into monomers, which are then synthetically spun to obtain technical grade PET fibers for tire cord applications.
Functional fibers
Recycled PET fibers can be modified to obtain functional fibers with special properties. 80 Laoutid et al. 81 combined wrapped red phosphorus with additives to enhance the mechanical and refractory properties of recovered PET. Rajamanickam and Vasudevan 82 investigated the antibacterial activities of yarns containing lyocell and recycled PET at different blending ratios. Their results showed that the lyocell/recycled PET blended yarn had high strength and elongation, exhibited good antibacterial activity, and could be used as a medical textile. With further improvements in technology, additional roles for recycled PET functional fibers will be discovered in other fields. Currently, recycled PET can be utilized to produce nanofiber membranes through electrospinning. These nanofiber membranes have significant implications in water and air filtration, as well as smoke control, contributing to the mitigation of water and air pollution.83,84
Composite materials
Besides being widely used in the textile field, recycled PET fibers also excel as construction, automotive, and acoustic materials. 85 The fatigue deformation resistance of asphalt is greatly improved by addition of recycled PET fibers because of the presence of recycled fibers in both liquid and semi-crystalline states. The liquid portion fills the gaps in the asphalt mixture and the semi-crystalline portion disperses the energy applied to the asphalt mixture. 86 However, further research is needed to address the issue of decreased flexural strength of asphalt mixes. 87 Recycled PET fibers can also be used in geosynthetics. 88 Mixing nanoclay with recycled PET fibers affects the mechanical behavior of the soil and increases its friction angle and cohesion, which can effectively enhance the strength of the soil.89,90 Furthermore, recycled PET products can also be utilized as value-added raw materials for paints and coatings. 91
Summary and outlook
Waste PET products, as a primary source of plastic pollution, are receiving increasing attention. This paper presents a systematic review of waste PET recycling technologies and explores the future prospects and opportunities in the field of recycled PET. With continuous development, there will be a rise in the efficiency for PET waste sorting and recycling, improvement in re-polymerization techniques, and advance in process control to ensure the quality and performance of recycled PET. Technological innovation will drive the improvement of recycled PET quality, expand its applications, and reduce production costs.
The future development of recycled PET will be centered around enhancing its quality and sustainability. This includes improving its physical properties, chemical stability, and reliability to broaden its use as a substitute for virgin PET. Emphasizing sustainability will become a key focus in the recycled PET field, involving reducing energy consumption, lowering carbon emissions, and promoting the development of a sustainable economy. Strengthening the collaboration among governments, businesses, and society at large is crucial to establish robust recycling infrastructure, formulate effective policies and regulations, and raise public awareness and acceptance of recycled PET. A comprehensive circular economy system, encompassing recycling, reuse, and widespread application of recycled PET, will contribute to the long-term sustainable development of the field.
High-quality recycled polyethylene terephthalate (PET) exhibits significant potential for a wide range of applications beyond traditional polyester products. Its exceptional thermal stability and processability render it highly suitable for diverse 3D printing techniques, facilitating the production of intricately shaped components and products. Moreover, through its integration with materials such as glass fibers or carbon fibers, recycled PET has the capability to generate high-performance composite materials characterized by enhanced strength, rigidity, and chemical resistance. These composite materials hold promise for various sectors, including automotive, aerospace, electronics, and sports equipment. By expanding the scope of application domains for recycled PET, valuable business prospects can be created while simultaneously stimulating market growth.
In conclusion, future work should focus on overcoming challenges in PET recycling, identifying environmentally friendly and cost-effective methods to degrade waste PET products, and enhancing the performance of recycled PET. It is imperative for governments, businesses, and society at large to strengthen collaboration and collectively drive the advancement of the recycled PET field, ultimately achieving a more sustainable plastic circular economy.
Footnotes
Acknowledgements
This work was financially supported by the National Natural Science Foundation of China, Basic Science Center Program (Grant No. 51988102), the Beijing Nova Program (Grant No. 20220484213), and the Innovation Teambuilding Program of Beijing Institute of Fashion Technology (Grant No. BIFTTD201904). We thank Gabrielle David, PhD, from Liwen Bianji (Edanz) (![]() ) for editing the English text of a draft of this manuscript.
) for editing the English text of a draft of this manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the National Natural Science Foundation of China, Basic Science Center Program (Grant No. 51988102), the Beijing Nova Program (Grant No. 20220484213), and the Innovation Teambuilding Program of Beijing Institute of Fashion Technology (Grant No. BIFTTD201904).