Abstract
This research produced textile pigment printing paste using expanded polystyrene (EPS) as a binder, sodium alginate as a thickener, and ultramarine pigment. The printing paste production was optimized through response surface methodology, using central composite design (CCD). The dynamic viscosity of the produced printing paste samples ranges from 5200 to 5550.50 cP (Centi Poise). The Fourier infrared transform (FTIR) spectroscopy revealed the functional groups present in the printing paste.
The result of the thermogravimetric analysis (TGA) conducted shows a first step decomposition of 94.14% that occurred at the range of 30.96°C–127.75°C. Differential scanning calorimetry (DSC) showed an endothermic transition at 119.44°C due to the absorption of energy during the breaking of printing paste bonds. Textile characterization such as wash fastness, croak fastness, and light fastness was carried out to determine the suitability of the print paste on cotton and polyester fabrics. The results of the fastness tests range from 4 to 5 for wash fastness, 3 to 4/5 for croak fastness, and 4 to 6 for light fastness. The optimum values of the model variables are thickener (50%), binder (40%), and pigment (5%). The performance of the printing paste as revealed from the results is moderate and comparable with textile printing paste suitable for commercial application.
Keywords
Introduction
Textile pigment printing formulation involves four main components thickener, binder, pigment, and auxiliaries. 1 Pigment printing has an advantage over the conventional dyeing method of fabric coloration because it applies to all fabrics and gives proper fixation. 2 There is practically no chemical affinity or bonding between the pigment and the substrate; therefore, bonding is achieved by the polymerization of monomers in binders and cross-linking agents. 3 On a global scale, 50% of all printed textiles are produced using pigment printing because the process is easier and universally applicable to all textiles. 3
For a successful print with good rubbing fastness to be achieved, the selection of a good binder system is very pertinent. 4 N-methylolacrylamide was among the most frequently used self-cross-linking binders for textile printing, but the problem associated with N-methylolacrylamide was the release of formaldehyde at high temperature due to the presence of hydroxymethyl amide group. 5 The release of formaldehyde is not environmentally friendly; successful substitutes for N-methylolacrylamide binder which has no hydroxymethyl amide group that releases formaldehyde at high temperatures include hydroxyl ethyl acrylate, carboxylic monomers, and glycidyl methacrylate. 6 The binders used in the early stage of printing paste formulation development require high-temperature cure and may release formaldehyde; to avoid this, mild cross-linking agents were introduced to complement the binder but excessive use of cross-linking agents causes clogging of screens and nozzles during printing. 7
Recent researches are focused on the development of more environmentally friendly binders for textile printing paste. Water-based polyacrylate resin has shown some positive prospect as a binder system and research are geared toward exploring it as a textile binder. 8 Researchers have tried to develop an eco-friendly binder system due to the release of Volatile Organic Compounds (VOC) that characterizes conventional binder system. For instance, Zhang, Zhong, Sui, Xu, Zhang, and Mao 2 synthesized a low-temperature self-cure core-shell acrylate binder containing acetoacetoxyethyl methacrylate. Their result shows that self-cross linking occurred at low temperatures without releasing formaldehyde. They achieve better results by using an amino silicon softener. Furthermore, Haggag, Elshemy, Hashem, and Mohamed 9 synthesized a colored dual-purpose recipe that serves as both binder and pigment from unsaturated pigment 2-anthracene triazole acrylate using click chemistry. Their result reveals that the unsaturated pigment contained a functional group that can be polymerized with monomers such as acrylic acid and butyl acrylate to form a colored binder used for printing paste formulation. They used a synthetic thickener based on polyacrylic acid for this formulation. Additionally, Yang, Li, and Fu 10 prepared a novel polymer colorant based on polyglycidyl methacrylate (PGMA) to achieve dual functionality of both coloration and binding action. They achieve this by modifying the PGMA microsphere using polyethyleneimine (PEI) to obtain aminated (PGMA-NH2) microspheres which were then bonded with reactive dye (X-3B). This recipe was then applied to citric acid-treated cotton fabrics. Their result shows good croak fastness and softer hand feel were obtained but the process is sophisticated and has a high-cost implication.
On the other hand, Hakeim, Haroun, Trif, and Feczkó 11 synthesized pigment with some binding ability by a method of encapsulation of phthalocyanine pigment blue into citric–acrylate oligomer in the presence of glutaraldehyde using phase separation. Their research highlighted that encapsulating pigment facilitated pigment fixation on cellulosic fabrics while reducing the quantity of binder requirement. Also, they added cross-linker glutaraldehyde which helped to stabilize the encapsulated pigment on the cellulosic fabrics. Going further, in a rather more sophisticated process, Hakeim, Abdelghaffar, and Haroun 12 developed UV-curable hyper-branched polyester acrylate also by encapsulation of phthalocyanine pigment adopting the liquid phase separation method. This served as a binder as well as a pigment; their result shows that fabric printed with encapsulated pigment gave good color strength and a better hand feel. However, El-Molla, El-Sayad, El-Kashouti, and El-Khawaga 13 took a different route based on polyurethane; they synthesized water-borne polyurethane acrylate oligomers as an aqueous ultraviolet–curable binder for pigment printing. They pointed out that better color strength and fastness were obtained compared to commercial binders.
Several attempts have been made by researchers towards reclaiming EPS into useful products for various applications. Myint, Zakaria, and Ahmed 14 formulated a paint based on an EPS waste binder intending to replace alkyd resin. They reported an optimum performance at 27% EPS incorporation in the paint formulation which was comparable with commercial alkyd resin paint. Researchers like Gutierrez-Velasquez, Monteiro, and Colorado 15 degraded EPS with a co-solvent of acetone and ethyl acetate for the application of binder in asphalt. Their results showed asphalt with better mechanical and rheological properties when compared with conventional asphalt. Also, Pczieczek, Schackow, Effting, Dias, and Gomes 16 combined EPS waste and waste tire rubber for application in a mortar; they reported that the use of fine rubber powder and EPS gave mortars with lightweight and more workable. Other notable applications include adhesive bonding applications Uttaravalli, Dinda, Gidla, Kasturi, Kasala, and Penta 17 ; aggregate material in constructions Koksal, Mutluay, and Gencel 18 ; wood plastic composites Poletto, Dettenborn, Zeni and Zattera, 19 and fiber applications. 20
This research developed a sustainable binder additive from expanded polystyrene (EPS) to reduce the importation of binders and usage of synthetic additives while encouraging the reuse and recycling of EPS for textiles printing paste. EPS was used because of its inherent adhesive properties, so the present research hopes to explore its applicability as a binder in printing paste. Sodium alginate was used as a thickener, and the pigment was Ultramarine blue. The produced printing paste was characterized by FTIR, Rheological properties, DSC, and TGA as well as textile characterizations such as wash fastness, croak fastness, and light fastness.
This research turned expanded polystyrene waste into a useful product; the printing paste production process is anticipated to be localized. This will lead to an increase in its use, profitability, and entrepreneurial growth.
Experimental method
Expanded polystyrene waste was sourced from Albertina Electronics located at Enugu-Onitsha Expressway, Awka, Anambra state, Nigeria. Sodium Alginate thickener (Analytical purpose grade) was supplied by Kem Light laboratories; Ultramarine blue pigment, Toluene, Calgon, Glycerin, and other chemicals (analytical grades) were supplied by chemical dealers. All chemicals bought were used as supplied without further treatment.
Experimental design
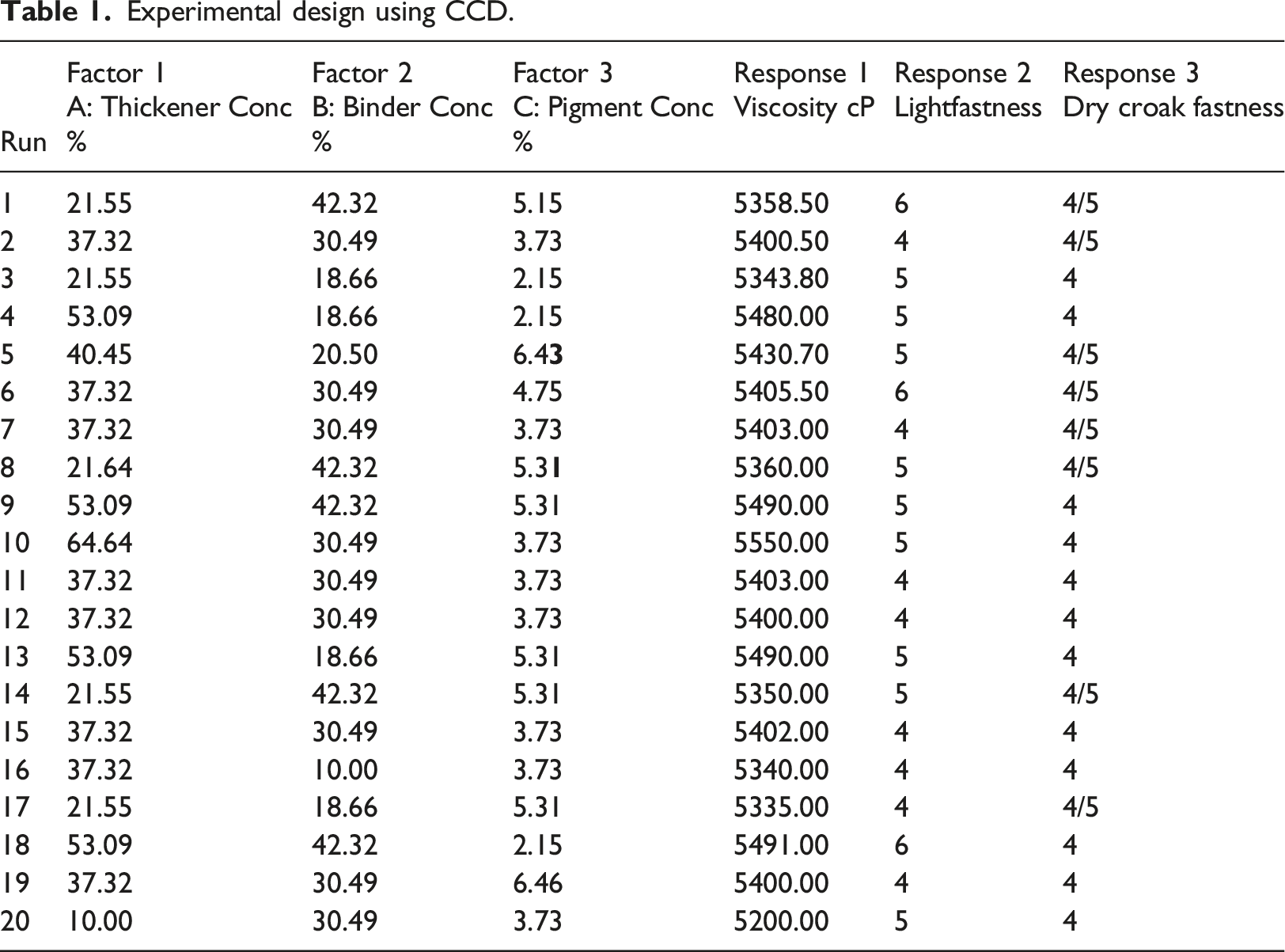
Design Expert version 10.0 by Stat-Ease was used to design this experiment. Response Surface Methodology 21 was employed to optimize textile printing ink sample production using central composite design (CCD). For printing ink production optimization, the response variable: viscosity was influenced by three (3) input variables: Concentration (x) of sodium alginate thickener, Concentration (y) of expanded polystyrene binder, and quantity (z) of pigment concentration. Other factors such as auxiliary concentration and volume of solvent will be kept constant. The result from the experimental design gave rise to twenty (20) samples of different formulations and ratios for thickener concentration, binder concentration, and pigment concentration.
Experimental design using CCD.
Procedures
The expanded polystyrene (EPS) waste was collected, washed, and dried for 24 h under the sun. Size reduction was carried out in a ball milling machine. 43 g of expanded polystyrene was measured and dissolved in 100 mL of the toluene solvent and stirred for 30 min using a magnetic stirrer at an average speed of 60 r/min to obtain the binder solution. Also, 200 mL of water was treated with 2 g of Calgon (a preconditioner) and divided into two parts in a ratio of 1:9. The smaller part was used to dissolve 5 g of ultramarine pigment and stirred for 20 min. The remaining part of the water was then used to dissolve 21 g of sodium alginate powder and stirred vigorously for 1 h to obtain the thickener solution. The homogenized thickener solution was then mixed with the already prepared pigment solution under constant stirring for another 30 min. This was followed by the binder solution. Continuous stirring was carried out on the entire printing paste solution. 10 mL of glycerin (which helps in fluidity and elongation of cure time) was added to the recipe. All steps were carried out at room temperature.
This procedure was followed for the 20 samples while keeping the volume of solvents and other auxiliary constants.
The printing paste that was produced was then used to print on cotton and polyester fabrics using screen printing. The samples were oven cured at a temperature of 70°C overnight.
Characterization techniques
Fourier transform infrared spectroscopy (FTIR)
The FTIR measurement was carried out using the Thermo Scientific 4700 FT-IR model. There was no uncertainty in measurement.
Viscosity measurement
The viscosity of printing paste is measured using a viscometer, NDJ-5S viscometer model in Centi Poise units. The viscometer operated at a maximum speed of 100 r/min and has six spindles.
Thermal analysis
Thermal analysis was conducted to ascertain the heat behavior of the printing paste; the two thermal analyses that were conducted were differential scanning calorimetry (DSC) and thermo gravimetric analysis (TGA). DSC and TGA were carried out at PGE Applied Resources, Orlu, Imo State, Nigeria. The Ramp method was used to conduct the TGA using the 2950 TGA HR V5.4A model of the TGA machine; the size is 12.5520 mg. Also, the Ramp method was used to conduct the DSC using the 2920 MDSC V2.6 model of the DSC machine.
Wash fastness
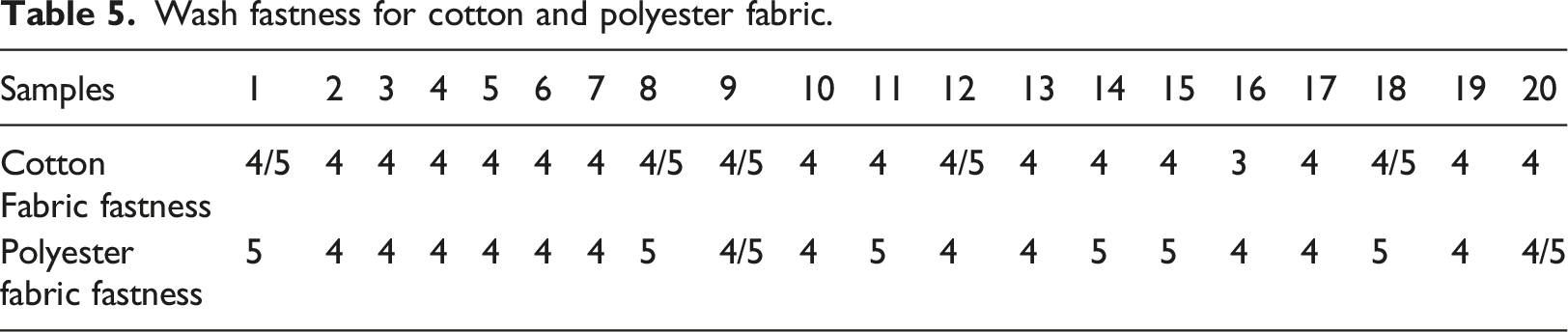
The wash fastness was carried out on the 20 samples (the formulated printing paste was used to print on cotton and polyester fabrics) using the GT-D07 model fabric washing fastness machine; the fabrics were conditioned for 24 h at a temperature of 27°C and 65% relative humidity. The result is shown in Table 4
Croak fastness
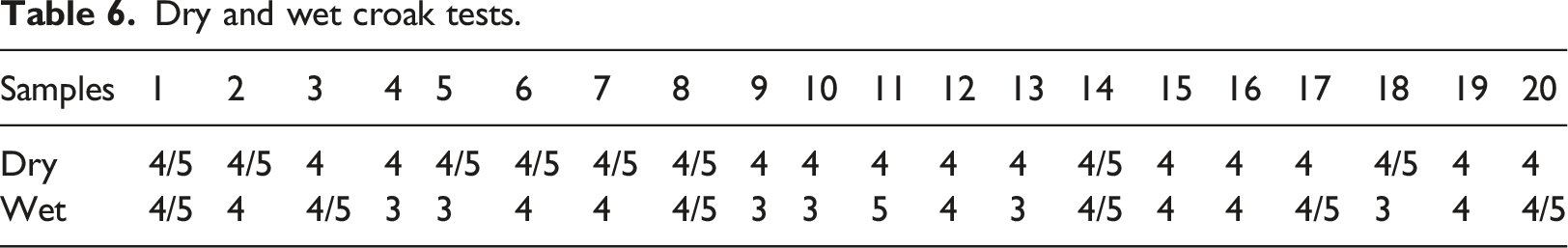
The dry and wet croak fastness was conducted using the VGO Tech Croak meter. The fabrics were conditioned at 27°C and 65% relative humidity. The Croak fastness was carried out according to ISO 105 following a similar process reported by Solangi, Noonari, Channa, Khan, and Siyal; 22 the equipment has a 16 mm croaking finger and a 9 N downward force (to and fro movement). The result of the test is shown in Table 6
Light fastness
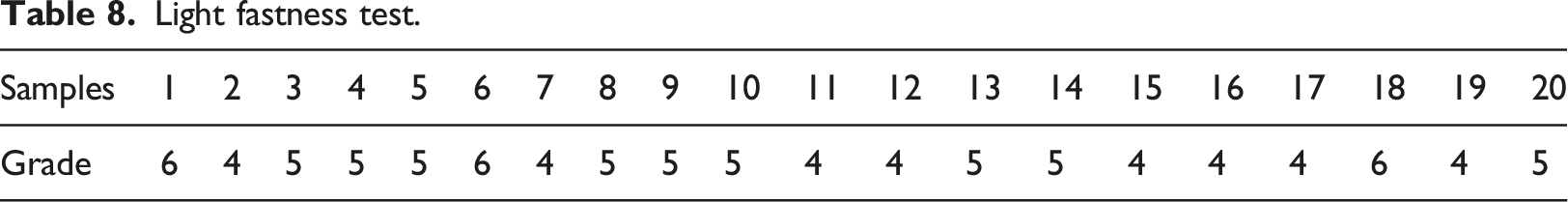
ISO 105-B02:2013 procedure reported by Kabir and Koh, 23 was followed in the light fastness test. The results of the test for the 20 samples are shown in Table 7.
Results and discussions
Table 1 shows that the viscosity of the 20 samples of the printing paste ranges from 5200.00 cP to 5550.00 cP. There was also an observable direct relationship between the percentage of thickener concentration and the viscosity of the sample. For instance, 10% thickener concentration gave the least viscosity and 64.64% thickener concentration gave the highest viscosity. A similar observation was reported in Reference 24. 24
Statistical analysis
Model summary Statistics.
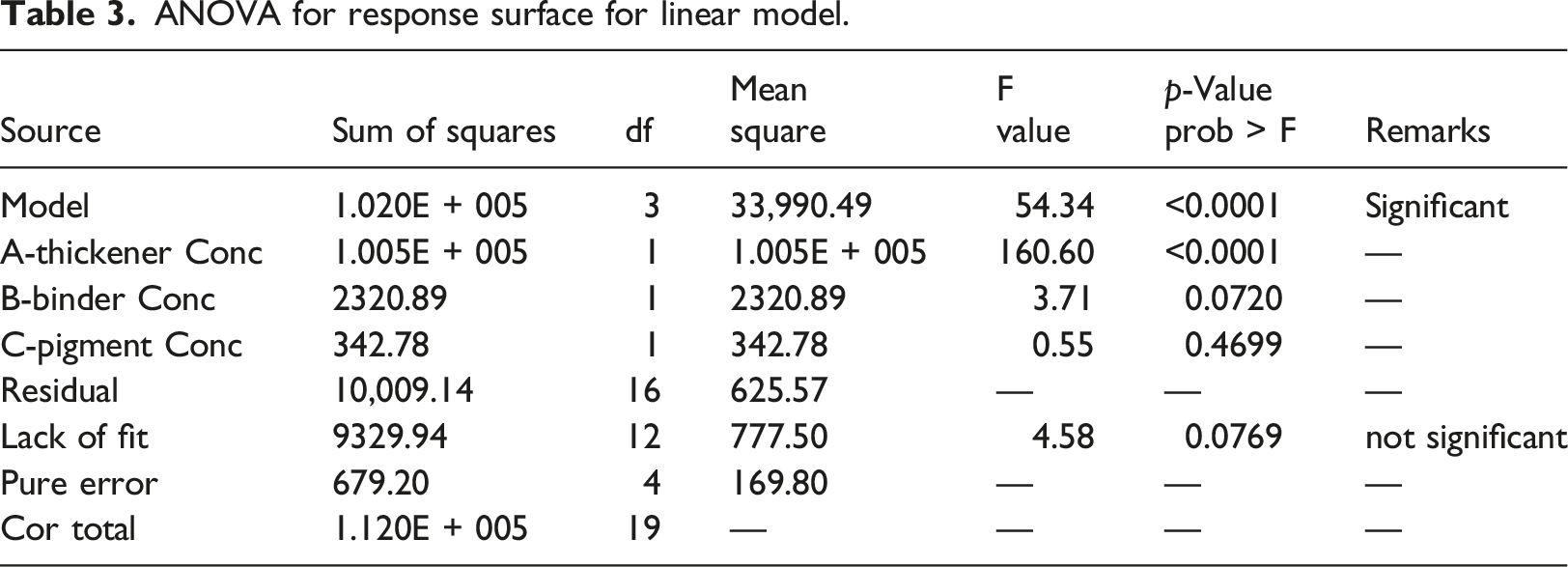
ANOVA for response surface for linear model.
Response surface optimization
Figure 1 shows the effects of the independent variables (thickener, binder, and pigment) on viscosity. Two factors were considered at a time (thickener and binder) while keeping the third factor (pigment) constant. This was because thickener was a significant model term (a major contributor to viscosity). 3D plot showing the impact of thickener and binder on the viscosity of the print paste.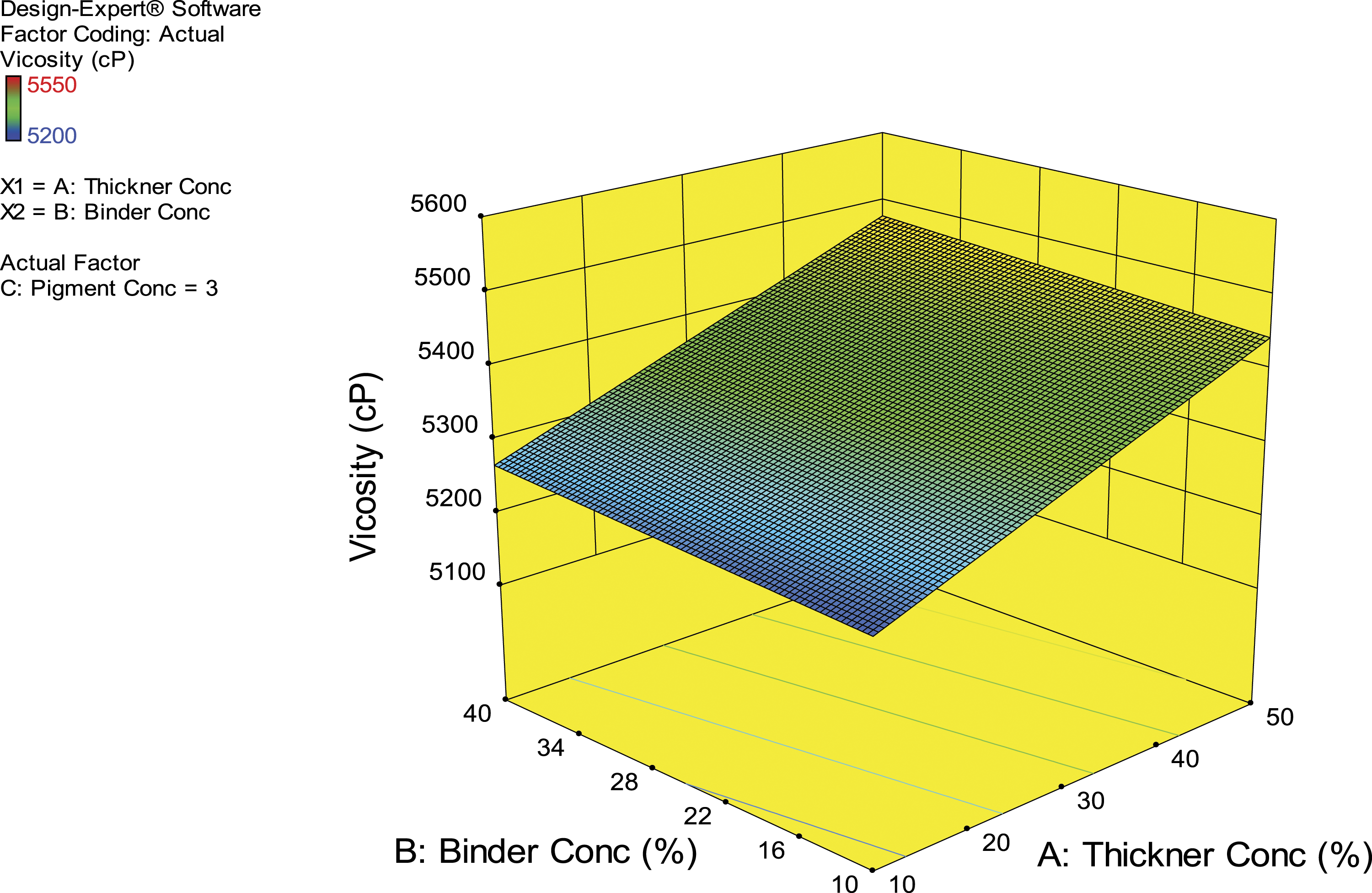
Experiment optimization
Figure 2 shows the optimal values of thickener concentration, binder concentration, pigment concentration, and viscosity. Optimal values of both the independent variables and viscosity.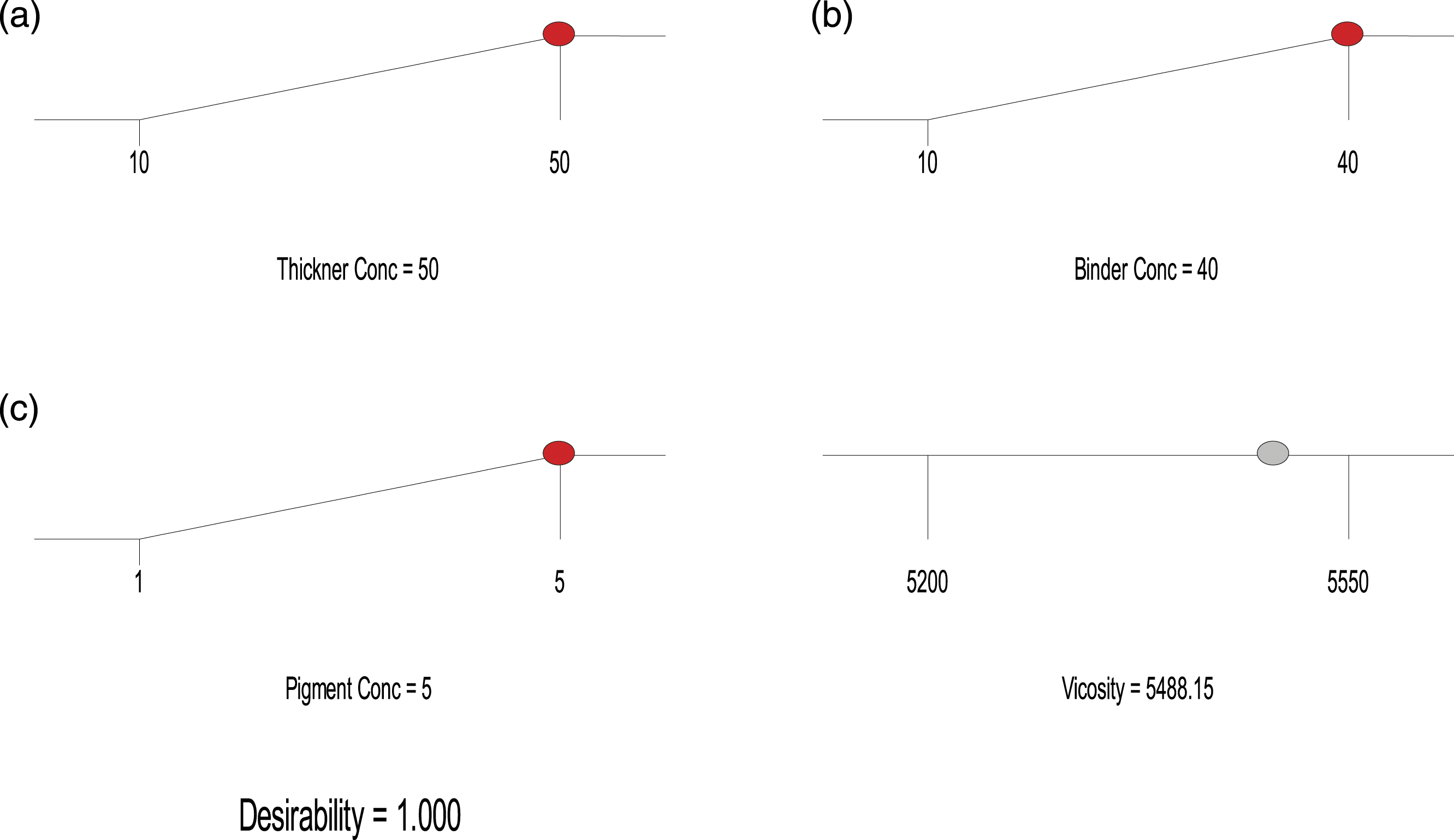
Printing paste chemical composition
Figure 3 shows the Fourier transform infrared spectroscopy analysis which was conducted to determine the chemical composition of the printing paste. A peak of 3818 cm−1 identifies the hydroxyl group present in water Biswas, Priyadarsini, and Mallik,
25
which was the used solvent for the printing paste. While peaks of 3687.80 cm−1 and 1857.57 cm−1 correspond to the carboxylic acid OH group and C = O anhydride present in sodium alginate respectively. The peak of 2975.20 cm−1 is a stretching vibration due to the C-H bond in the expanded polystyrene binder; this is similar to the result obtained in Reference 26.
26
The peaks at 1622.07 cm−1 and 1386.13 cm−1 are the C = C aromatic group and CH3 group that is present in both the expanded polystyrene and toluene solvent. FTIR spectrum of printing paste.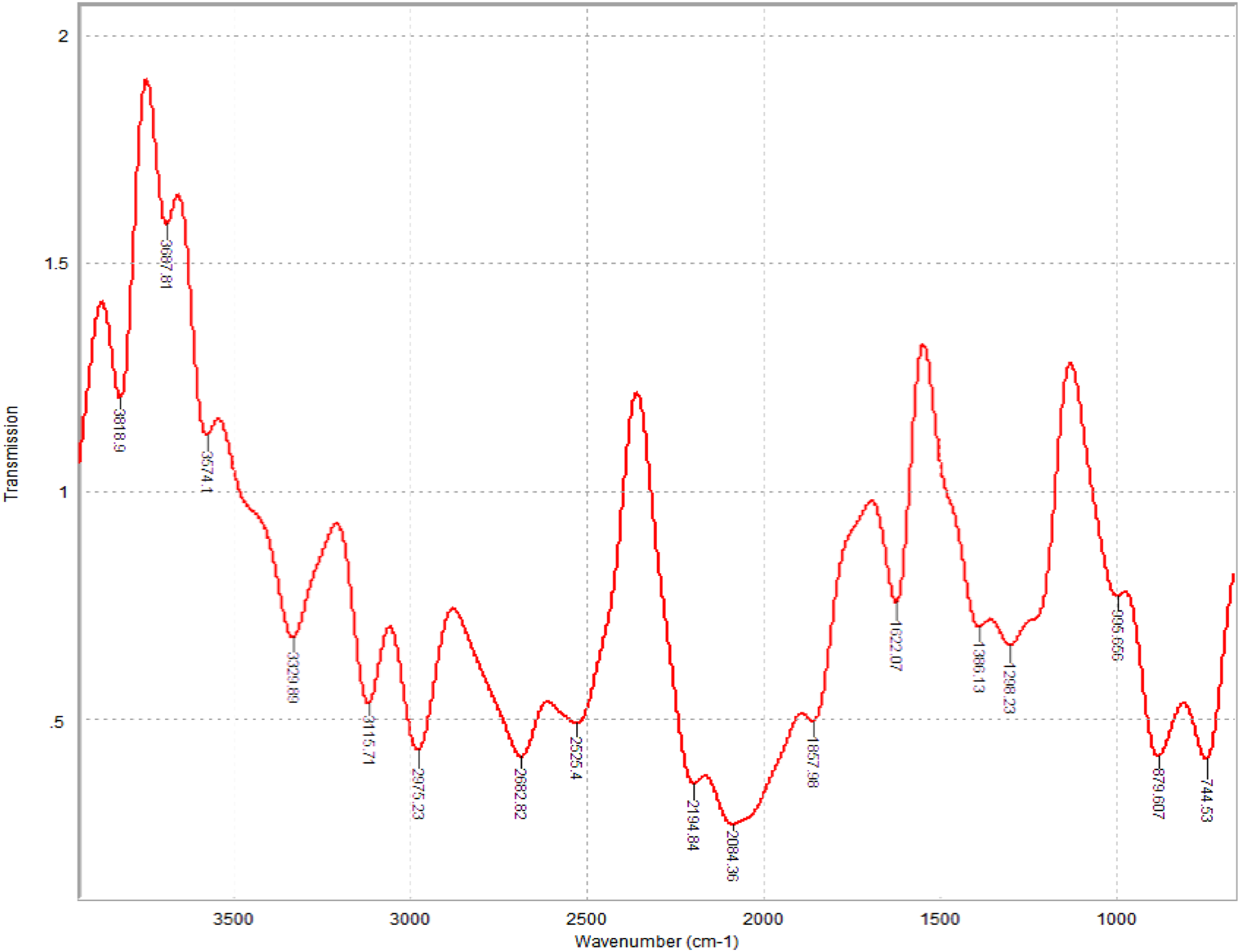
Thermo gravimetric analysis (TGA)
From Figure 4, there is a 94.14% (approximately 11.82 mg) weight loss at the first decomposition temperature range between 30.96°C and 127.75°C. This is due to the evaporation of volatiles (toluene molecules) and water components in the printing paste.
27
The weight remained constant between 130°C–200°C. Further heating resulted in the second step decomposition of 3.937% (0.4942 mg weight loss) which occurred between 200°C–309.15°C which is a result of the breaking of the polymer main chain.
28
The third step decomposition occurred at 350°C leading to a weight loss of 1.856% (0.2330 mg). The leftover at a temperature above 350°C is the residue in the form of ash (0.00003258% and 0.000004089 mg). TGA curve for the print paste.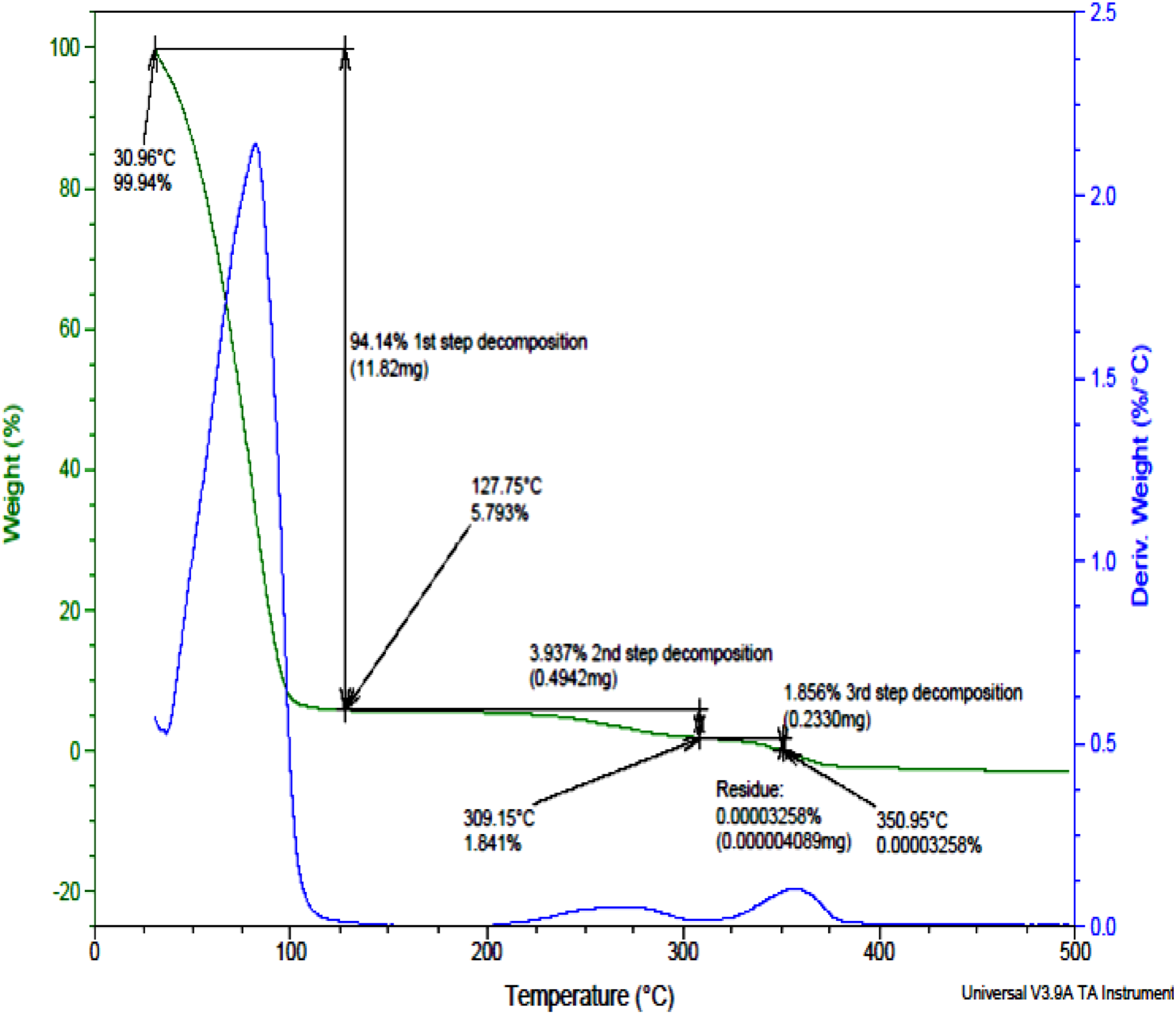
Differential scanning calorimetry (DSC)
From Figure 5, the maximum degradation of the printing paste occurred at the temperature range of 134.44°C which is similar to that obtained in the TGA analysis above. There was also an endothermic transition at 119.44°C. DSC curve for printing Paste.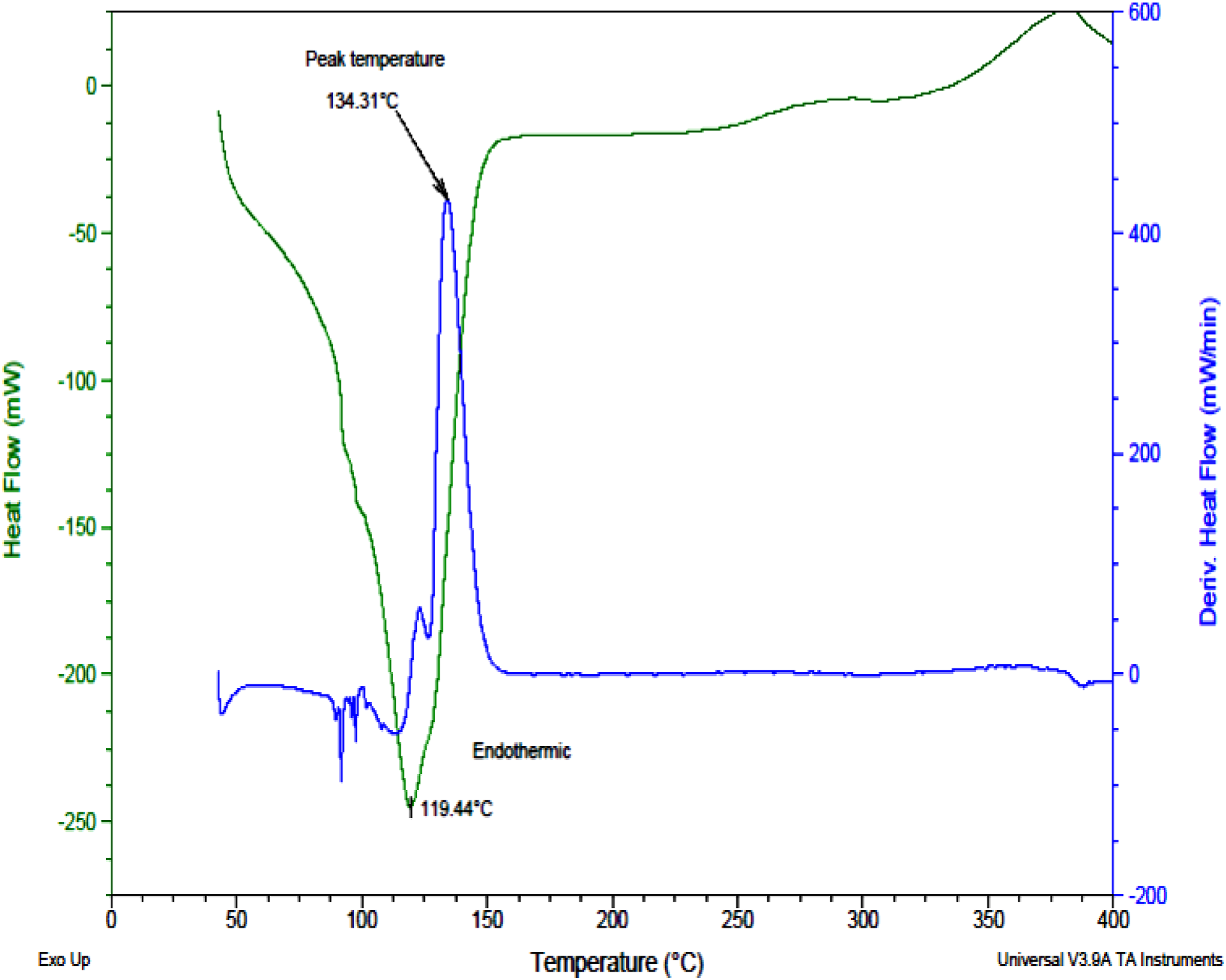
Textile characterization
Wash fastness
Washing and crocking fastness test key.
Wash fastness for cotton and polyester fabric.
Croak fastness
Dry and wet croak tests.
Light fastness
Light fastness scale/grad/rating.

Light fastness test.
Summary
This report has successfully used expanded polystyrene waste to develop a binder used for textile printing paste formulation using sodium alginate thickener and Ultramarine pigment. An experimental design using the Central Composite Design (CCD) was employed in the printing paste formulation which gave 20 runs that were used in the formulation of the printing paste samples.
Conclusion
The viscosity of the formulated printing paste samples ranges from 5200 cP to 5550 cP. The statistical analysis reveals that the thickener concentration term of linear model Response Surface Methodology is significant; this means that thickener concentration contributes most to the viscosity of the printing paste. 24 The optimization values were thickener concentration (50%); binder concentration (40%); pigment concentration (5%), and viscosity (5488.15 cP). The wash fastness score of the printing paste samples (20 samples) ranges from 4 (very good) to 4/5 (excellent) on cotton fabric; and 4 (very good) to 5 (outstanding) for polyester fabric. Also, the dry crock fastness test score ranges from 4 (very good) to 4/5 (excellent); however, the wet crock fastness showed lower performance ranging from 3 (moderate) to 5 (outstanding). Furthermore, the printing paste samples showed a light fastness score ranging from 4 (moderate, which is appreciable fading) to 6 (very good, which is slight fading).
The results of the wash fastness, croak fastness, and light fastness conducted show appreciable values that were comparable with those obtained in the literature.22,29,30 This means that the printing paste produced using expanded polystyrene (EPS) waste can be used for textile printing of cotton and polyester based on the values of fastness obtained. However, the performance of the printing paste could be improved especially for light fastness by use of a co-binder; finding a suitable co-binder will be an area of future research.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Nnamdi Azikiwe University, Awka, Nigeria, through the 2018/2020 merged TETFUND Research Project Intervention Funds.
Data availability statement
The raw/processed data required to reproduce these findings cannot be shared at this time as the data also forms part of an ongoing study. However, data are available from the corresponding author on reasonable request (Shadrack. C. Ugwu- ![]() ).
).
