Abstract
The principle of clinical equipoise has been variously characterized by ethicists and clinicians as fundamentally flawed, a myth, and even a moral balm. Yet, the principle continues to be treated as the de facto gold standard for conducting randomized control trials in an ethical manner. Why do we hold on to clinical equipoise, despite its shortcomings being widely known and well-advertised? This paper reviews the most important arguments criticizing clinical equipoise as well as what the most prominent proposed alternatives are. In the process, it evaluates the justification for continuing to use clinical equipoise as the gold standard for randomized control trials.
Background
Conducting randomized clinical trials in an ethical manner can be a delicate balancing act for physician–researchers. On the one hand, as physicians, they cannot compromise on their duty of care toward patients, but on the other hand, as researchers, they must conduct the clinical trial as to achieve their research goals. The first involves providing individualized care; the second involves testing for the effects of an intervention across a population of test subjects.
The history of biomedical research illustrates how, at least in the past, these two objectives have clearly come apart, 1 and since end of the Second World War the rights of test subjects have been strongly protected. 2 Hence, one of the central challenges for research ethics has been to find the conditions for conducting randomized clinical trials such that scientific goals can be pursued without compromising care.
An early attempt at finding these conditions was the concept of “equipoise,” introduced by Fried in 1974. 3 Equipoise refers to a condition of personal and genuine uncertainty of the physician–researcher about which treatment arm can most benefit the patient. Unlike in placebo-controlled trials, where the active therapeutic intervention is simply withheld from the control group, meeting the condition of equipoise allows randomized control trials (RCTs) to involve two active arms, thus providing data on the efficacy of the novel treatment without compromising on what Fried called the “duty of personal care.”
Fried's concept of equipoise has widely been argued to be flawed. For instance, equipoise implies that as long as a physician–researcher lacks a firm conviction in the effectiveness of an intervention, they are ethically justified in embarking on a clinical trial of that intervention. This could even be the case if the lack of conviction was based on outdated knowledge. In fact, any firm conviction about the efficacy of a novel treatment – justified or not – would ethically preclude conducting a randomized clinical trial, since the condition of personal and genuine uncertainty would be lacking. Such problems motivated Freedman's subsequent introduction of “clinical equipoise” in 1987. 4 Under clinical equipoise (CE), there exists an honest, professional community-level disagreement regarding the best possible treatment. Enrolling patients in a randomized clinical trial is structurally like patients going to different physicians who have different views on the best treatment. Hence, it is not unethical for a clinician to give a patient the standard treatment during a clinical trial even though they may personally believe the standard treatment to be inferior.
Today, the concept of CE can be regarded as one of the most influential principles guiding the ethics of clinical trials. A concrete indication of this status of CE is how it implicitly informs the Declaration of Helsinki. In particular, in article 33 of the 2013 version of the Declaration of Helsinki, it is stipulated that clinical trials of new interventions should be compared against the best available treatments. 5 Furthermore, placebo-controlled trials are permitted only in exceptional circumstances, and the Declaration cautions that “extreme care must be taken to avoid abuse of this option.” 5 In this way, even though the exact phrase “clinical equipoise” is not explicitly mentioned, despite some arguing that CE should be explicitly mentioned in the Declaration,6,7 it is clear that the concept of CE has made its way into one of the most important regulatory documents in clinical ethics.
Despite this influence, the principle of CE has been subjected to striking criticism over the decades. Some criticisms problematize the scientific legitimacy of CE, calling it “fundamentally flawed” 8 or “a myth.” 9 Other criticisms go further and cast doubt on the ethical intentions underlying CE. Thus it has even been suggested – albeit in the relatively informal context of letter – that CE mainly functions as “a moral balm to sooth the conscience of research sponsors and researchers alike” and as a tool to entice poor but healthy individuals to enroll in randomized clinical trials. 10
Although the formulation of such criticisms may not be entirely fair, as will be reviewed in this paper, the criticisms are often cogent and often pick up on genuine shortcomings facing the principle of CE. The severity of the criticisms are also surprising given the dominance of CE. For if the criticisms were to be taken at face value, they would imply the need for a fundamental redesign of how clinical research is to be conducted. The principle of CE would need to be dropped or amended. However, that has not happened, despite the criticisms of CE going back decades. It is puzzling why CE, despite the intense criticism, continues to be the de facto gold standard for RCTs.
The purpose of this paper is to raise this question and chart some of the main ethical issues at stake. The question has been touched on before, for instance, in a debate article with prominent contributors taking sides on CE. 11 However, the present article shifts the focus away from questions “for” or “against” CE and aims at foregrounding the question why the arguments “against” the principle, even if correct, may still not be sufficient to reject it.
Why does CE continue to be taken as the gold standard for RCTs despite its well-advertised flaws? Note that an ethical review as this one cannot provide a full, interdisciplinary discussion. Understanding why clinical trials are designed in certain ways rather than others would require looking at them from multiple angles: sociological, legal, and financial. It is reasonable to presume that all these factors may play at least some role in fully explaining why CE continues to be endorsed despite (alleged) ethical flaws. However, this review narrows its focus to the ethical dimension of CE.
We will especially be interested in the hypothesis that CE could perhaps be considered the least ethically flawed principle available to ethically design RCTs. Hence, to address the research question, we further operationalize it into two further subquestions: (1) what have been the main arguments against CE? (2) what viable alternatives for the replacement of CE on the academic literature in the design of RCTs are available?
Methods 1
The main methodology used for this review was that of the “critical interpretive literature review.” 12 The main reason for this choice is that the concept of CE is discussed, to varying degrees, across biomedical literatures – not just in the bioethical and research ethics literatures. Hence, it would not have been possible to capture all contributions regarding the research questions. However, insofar we set out to capture the “key ideas” regarding the arguments on CE (McDougall, 12 Table 1), and to “put forward an argument about the literature” on CE (McDougall, 12 p. 527), McDougall's method was well-suited. Further, for the extraction and synthesis of information, we followed the quantitative methodology of the “systematic review of reasons.” 13 The reason or argument types were coded and subsequently we quantified how often each was mentioned.
The following steps were followed: identifying a focused research question, conducting a literature search in the relevant databases, selecting the relevant papers for the research question, analyzing the selected articles, and drawing conclusions. Criteria for selecting and analyzing articles were predefined before the research started. These included which databases to be included, the method of including articles and how various articles were to be analyzed. The final decision to include or exclude any article was unanimously agreed by both authors.
An initial search was conducted using combinations of Boolean operators and various truncations of relevant terms such as “clinical equipoise,” “equipoise,” “randomization,” or “trials.” This was done in several databases: PubMed, Web of Science, Google Scholar, and Scopus. Several databases were searched to avoid missing important articles since different databases return varying search results. Table 1 describes the search strings used for the various databases.
The search strings used for the initial search.a

Different databases require different search strings to identify various articles. The exact string used is conveyed here.
After this initial search, articles were excluded from consideration based on the following criteria:
Only peer-reviewed, English-language journal articles published after 1987 (the year of the introduction of CE) were included. Textbooks and letters to the editor(s), such as Wells et al.,
14
were not included. We only included articles dedicated to a conceptual critique of “clinical equipoise” or “equipoise.” Since the principle of CE has been and continues to be very influential, this meant discarding many articles referring to CE. For instance, many preclinical studies report on whether CE was achieved or not. A second category of studies referring to CE is review studies on the extent to which equipoise conditions are met in clinical studies in a certain medical field (e.g. Fries and Krishnan
15
) reviews industry-sponsored RCTs in rheumatology. A third category charts attitudes toward CE (e.g. Johnson et al.
16
). For purposes here, we only included those articles which focused on evaluating the conceptual or methodological foundations of CE. This included articles suggesting possible alternative or replacement option for CE.
For some articles, the title was sufficient to judge that the inclusion criteria were not met. For others, abstract and main text needed to be read. This was sufficient to exclude all except 38 articles. Following the recommendation of, e.g. Cooper,
17
the next step was to screen the bibliographies of these selected articles in order to double check that relevant articles were not missed in the initial search. In this way, 40 articles were selected for final inclusion in the study (Figure 1). Endnote citation software was used to manage the various references that were used in the review.
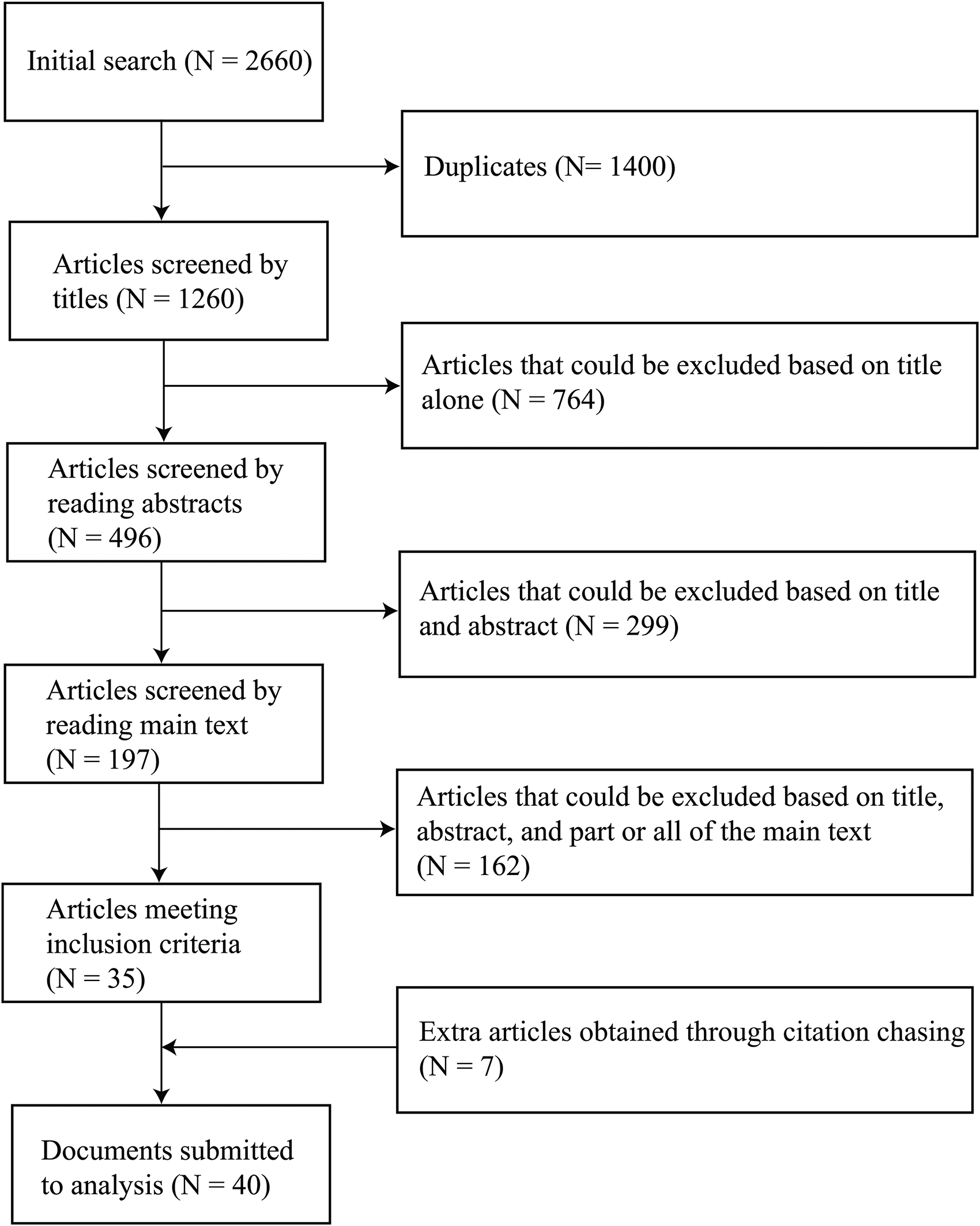
Flowchart illustrating the search and selection process.
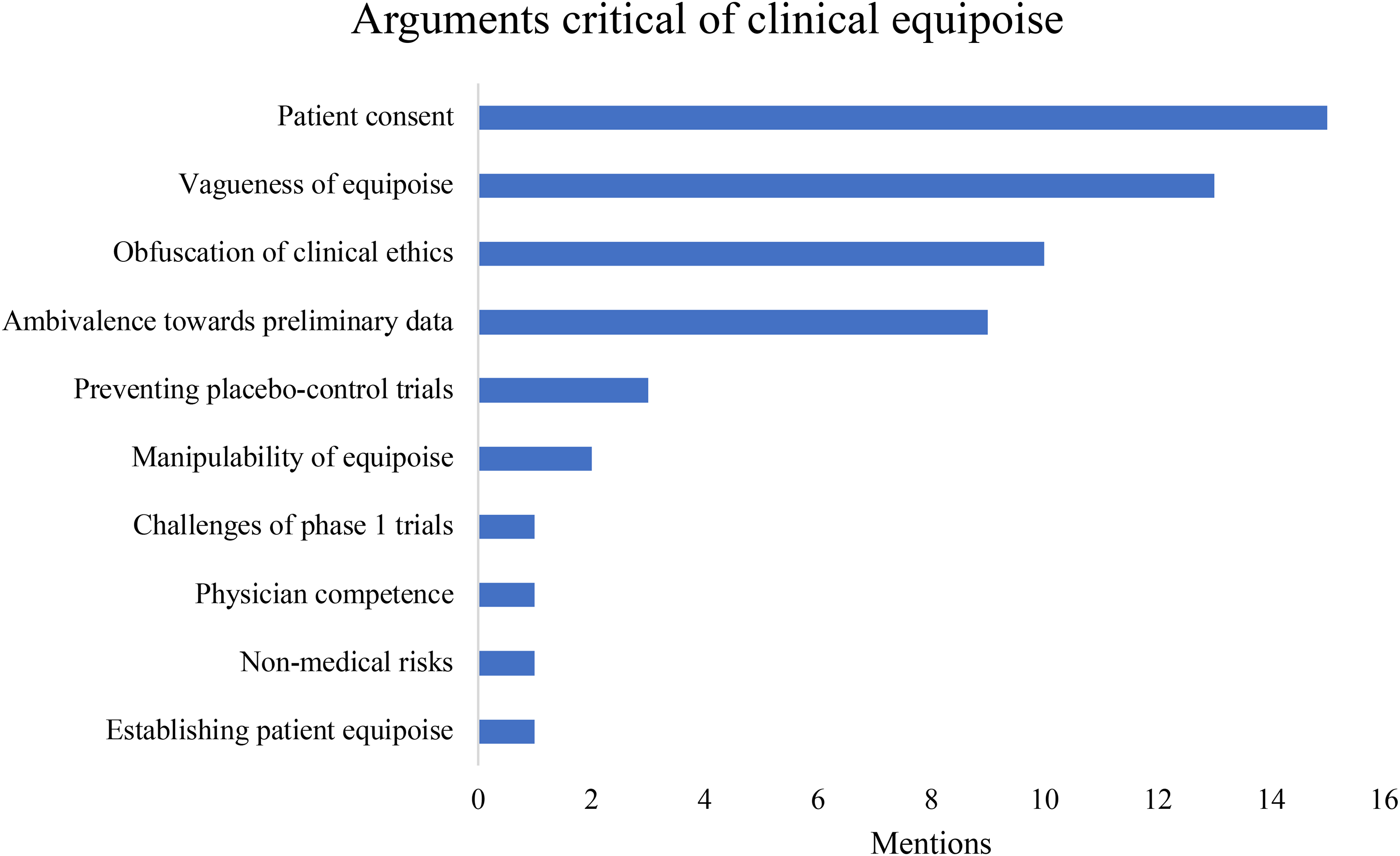
The arguments against clinical equipoise, plotted against the number of times each argument was used among articles reviewed here.
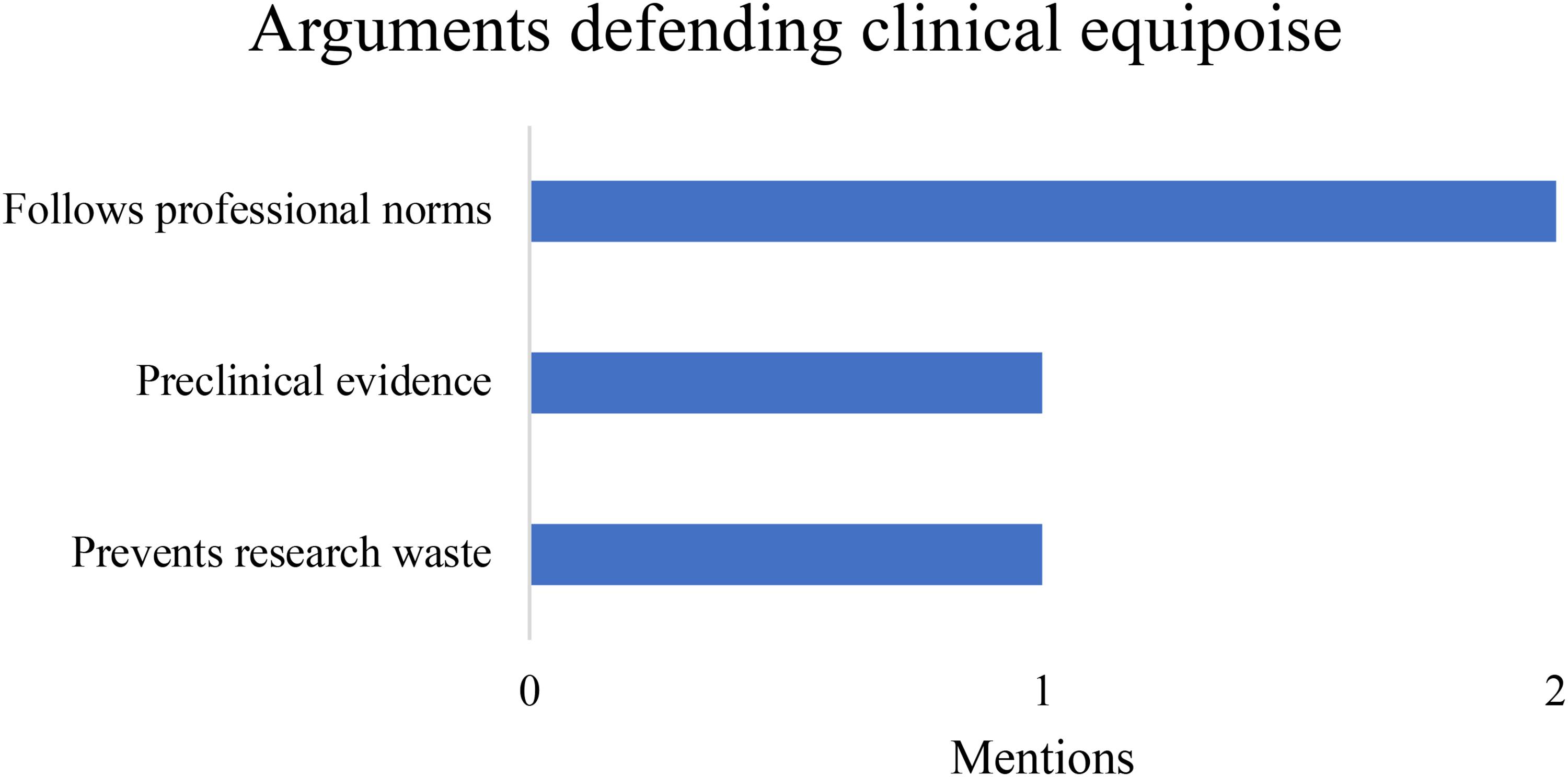
The arguments defending clinical equipoise, plotted against the number of times each argument was used among articles reviewed here.
In the following step, we then used a data-driven thematic analysis method 18 in order to identify the important reasons or arguments criticizing CE, as well as to identify the important alternatives to CE. For this, selected articles were read fully and multiple times to identify the important arguments advanced by the authors concerning the strengths and weaknesses of the principle of CE, and to identify which alternatives to CE were advanced. We categorized the different arguments (and/or reasons) and constructed a taxonomy of arguments (and/or reasons) types. Since the purpose of the interview was to identify key ideas, reason types were defined relatively broadly. This also allowed some counting of the relative frequencies of reason types (Strech and Sofaer, 13 Table 2). Finally, once the taxonomy of arguments was in place, we counted the number of authors who endorsed the argument. Here, the mere exposition of others’ arguments was not sufficient to merit inclusion: it was necessary for inclusion that the authors of a study posited and endorsed the argument or the alternative to CE.
Results
In our analysis, we identified nine separate arguments criticizing CE. Some of these – for instance, the criticism concerning patient consent – were repeated by many authors. Arguments that explicitly supported the principle of CE were much less varied and were also mentioned much less often. What lessons are to be drawn from these criticisms? Eight proposals to amend CE and six proposals to replace CE altogether were identified. Here, these results are detailed with a brief description of each argument or proposal (for the in-depth discussions, we refer to the original articles).
Arguments criticizing CE
Nine main arguments against CE were identified. Some arguments target the scientific methodology and the empirical standards involved in establishing equipoise (b, d, g, h). Another group of arguments target CE on more explicitly ethical grounds, for instance regarding patient consent or standards of care (a, e, f, i). However, we caution against sharply distinguishing between these two types of argument. Methodological limitations have ethical consequences: for instance, vagueness (argument b) sets the conditions for manipulation to occur (argument e). Moreover, one of the most influential arguments in the literature – that CE involves a confusion of medical care and scientific research (c) – criticizes CE on both scientific and ethical grounds. Figure 2 summarizes the results of this subsection.
Patient consent. According to this argument, CE is in and of itself insufficient to guarantee the moral justifiability of clinical trials since CE fails to acknowledge the importance of the informed consent of patients to be part of one of the treatment arms.8,19–29 In Freedman's original formulation, the only consideration of importance is whether the community of clinicians is indifferent to the efficacy of the various treatment arms. However, this leads to the potential of abuse, since it allows patients to be exposed to risky or unsafe trials as long as there is sufficient disagreement within the medical community.30–32 The vagueness of equipoise. This argument criticizes CE for not stipulating how much disagreement among clinicians is necessary to establish equipoise. Could a single maverick advocating an alternative treatment be sufficient to establish CE? Clearly not, but neither does it seem necessary that the possible treatments have an equal number of adherents. So, at what point does the disagreement among clinicians become sufficient to qualify as the collective indifference necessary for CE? This argument criticizing CE for its vagueness is often linked to questions about who must be doing the disagreeing. Medical personnel, researchers, ethical review boards, or pharmaceutical sponsors: all these groups of persons are involved in conducting clinical trials. Is it only the indifference between clinician–researchers that matters? Or must there also be equipoise in the ethical review boards, and so on? The criticism of vagueness charges the principle of CE with being silent on such questions.9,19,28–30,32–39 Confusion of care and research. The principle of CE obfuscates the fact that there is a genuine ethical dilemma at the heart of every clinical trial: how to weigh the potential benefits of research against the potential costs for the individual patients enrolled in the trial. Providing care involves deliberation on what specific treatment would be best for an individual patient, regardless of whether the course of treatment would lead to compromising the research protocol. Conversely, pursuing scientific research involves abstracting away from individual patients’ particular needs, and randomly assigning them to a small number of nonpersonalized treatment arms. This criticism is one of the most fundamental criticisms of the principle of CE, because it claims that CE simply turns on a misunderstanding of how fundamentally different providing care is from conducting scientific research.9,11,24,26,27,30,39–42 Ambivalence toward preliminary data. As preliminary data come in during a clinical trial, one treatment arm may be established as the superior one. Does this mean that equipoise no longer holds? In other words, should the clinician stop the trial once one treatment arm seems to be superior, or should the trial continue to follow protocol and seek to definitively establish the outcomes of the treatment arms? The principle of CE is silent on whether equipoise can only be established before the start of the trial, or whether equipoise should be updated as more data come in. This has important consequences for the ethics of the trial, as well as approval by regulatory agencies such as the Food and Drug Administration. We found this argument (with small variations) to be repeated across.11,23,24,28–30,34,37,43 Manipulability of equipoise. Given the vagueness of equipoise, there are many strategies by which equipoise could, in principle, be manufactured or manipulated. For instance, financial incentives could push a small minority of clinicians to advocate or withhold endorsing a certain treatment, tilting the community as a whole toward equipoise.9,30 Preventing placebo-control trials. The norm of CE often precludes placebo-control trials from being conducted. For some this is a strong reason in support of CE and have in fact advocated a strengthening of the Declaration of Helsinki to include a reference to CE.6,7 However, for others, this is a ground for criticizing CE, since they argue that placebo-control trials remain crucial for the goals of clinical research.8,9,44 Challenges of phase 1 trials. In phase 1 trials, CE typically fails to hold. There are little data concerning the novel treatment, and hence there is a risk for greater adverse outcomes compared to the established treatment. It is not possible to conclusively establish the novel treatment as in equipoise with the established treatment. This is reflected in very structure of phase 1 trials: they involve a small group of healthy participants. Instead, a utilitarian calculation is made where the potential benefits for the wider population outweigh the potential adverse consequences for the initial participants.
9
Nonmedical risks. In establishing equipoise, only medical risks are taken into consideration. This fails to address nonmedical adverse consequences of a clinical trial: religious risks, moral risks, and community-based risks which are subjected to a particular community. The latter risks constitute potential harm because of a relation to a broader community that is in disagreement with the conducting of a clinical trial.
45
Establishing patient equipoise. This argument, strictly speaking, is not a criticism of CE as originally formulated, but a criticism of a modification of CE (to take patient consent into consideration). Some have pointed to fundamental obstacles for the clinician to evaluate whether the patient consent is genuine and informed. For instance, patient consent presupposes that the patient is indifferent to which of the treatment arms they are enrolled in. However, clinicians rarely if ever can know and evaluate patients’ true preferences, as this can depend on a range of nonmedical values, including cultural, economic, legal, religious, familial, or aesthetic interests. The clinician thus cannot be certain that the patient's consent is genuine and adequate.
12
Arguments supporting CE
The main argument in support of CE is that it offers a superior alternative to the placebo-controlled trials where giving a placebo to a patient would amount to withholding care.6,7 Since this is simply the basic rationale for CE, it is widespread enough as to not be explicitly advanced as an original contribution to the debate in journal articles. By contrast, the following arguments were explicitly advanced among the selected articles (see Figure 3 for summary):
Avoiding negligence. Physicians are licensed only after demonstrating professional knowledge: this knowledge represents the consensus of the expert community. A subsequent failure to adhere to professional standards is thus potentially a form of culpable negligence. Some defend the principle of CE as a natural extension of this professional ethics: when consensus fails to hold concerning the relative efficacy two treatments, then there is simply no established professional standard with regard to those two. The physician is thus not bound by any standard to offer one treatment rather than another. Conversely, if equipoise would fail to hold, a clinical trial would be tantamount to pressing ahead with treatment despite the weight of medical opinion being against it. This could qualify as culpable negligence.4,29 Prevention of research waste. Conducting a trial despite evidence that one treatment is superior is not only just potential negligence but also a waste of research resources.
11
Preclinical evidence. As a response to worries about CE in phase I trials, some point to the importance of establishing the biological plausibility of a medical intervention (obtained through animal experiments). This can help to establish CE even in the early phases of clinical trials.
46
Proposals for reforming CE
Following the large number of criticisms that CE has received over the years, many authors have also given various positive suggestions. These range from reforming or modifying the principle of CE, to abandoning it altogether and reorganizing clinical trials according to different principles. Here, we list the proposals for reform or modification that were present in the reviewed literature:
Patient Consent and Patient Equipoise. Clinicians should be conceived as an equipoise when not only the community of medical practitioners is indifferent to treatment arms but also the individual subjects to be enrolled in the trial. Clinical equipoise should thus be amended to include patient equipoise as a precondition. This is a way of respecting patient consent, because if patients are indifferent, they can be assumed to consent to being part of a randomized trial. Conversely, if they would prefer one treatment arm over the other, that would invalidate consent.24,26,28,47–50 Probability distribution. Some authors such as Hansson
51
or Chard and Lilford
20
propose that the outcomes of treatment arms are presented by probability distributions over possible outcomes, instead of simply a single expected outcome. This probability distribution could allow for a much more precise judgment on whether the two treatment arms are equivalent from the point of view of the standards of care. Integrating nonmedical risks. To address the criticism that CE does not address nonmedical risks (religious, cultural, and social), it has been proposed that researchers, institutional review boards (IRBs), and patients collaborate to identify nonmedical risks and prior to trial.
45
Moreover, as these risks can change during trial, patients should be informed at every stage of the trial. This is a “hybrid” approach since the burden is not only entirely on researchers and IRBs to know of nonmedical risks in advance but also on the researcher–patient relationship since patients and communities can alert researchers and IRBs to nonmedical risks they may not have been aware of. Collective decision-making. A similar proposal to the previous, but with more emphasis on the importance of consent: it has been proposed that patients should be able to participate in research decisions at certain points, including study design, review board decision-making, and evaluation of ongoing studies.
24
As a mid-level principle. Furthermore, some researchers such as Chiapperino and Nardini
52
have suggested that CE should be adopted as a mid-level ethical principle, in between ethical theory and clinical practice. Thus, the vagueness that CE appears to have at the level of clinical reality simply means that, as a mid-level principle, individuals must use their judgment and knowledge of particulars in applying that principle. This suggestion is tantamount to changing the expectations we should have of CE. Early halting of trials. Trials should be halted once evidence emerges that the treatment arms may not be in equipoise. This provides better protection of the needs of research subjects.
53
A hypothetical retrospection (HR) approach. This approach was initially developed by Hansson
54
to deal with judgments of uncertainty and moral permissibility and has been suggested as an alternative approach to CE by Chiffi.
55
Evidence-based equipoise and research responsiveness. Halpern
56
suggests distinguishing between evidence-based equipoise and research responsiveness. Evidence-based equipoise simply refers to the uncertainty regarding which of two or more interventions is to be preferred based on existing evidence. However, Halpern argues that “research responsiveness” must additionally determine whether it is ethical to conduct a clinical trial: whether the goal of clinical research is if it serves a legitimate public health interest.
Proposed replacements for CE
Other authors have proposed alternative frameworks to replace the principle of CE. In the reviewed literature, we found six distinct proposals:
The nonexploitation ethical framework. In response to the confusion between care and research, Miller and Brody suggest acknowledging that the basic calculus involved in clinical trials is utilitarian: i.e. the potential benefit for health outcomes for a large number of persons outweighs the potential risk for a small number of test subjects. The goals of scientific research, since they lead to better care over the longer run, can potentially justify compromising on the standards of care in the short run. However, this utilitarian calculus can lead to obvious abuse, and needs to be constrained by a nonutilitarian principle, namely the norm of nonexploitation.8,26 The net-risk framework. An expansion of the previous proposal, the net-risk framework (also “systematic evaluation of research risks” (SERR)), proposes that clinical trials should be subjected to a systematic risk–benefit analysis, where not only societal gains in health are weighed against the interests of participants but also the professional integrity of researchers and public trust in science. This framework proposes seven main steps by which to evaluate randomized clinical trials, including evaluating and enhancing the potential benefits for participants, evaluating if the net risks are justified by the potential benefits of other interventions, and judging whether the remaining net risks are justified by the study's social values.57–59 The integrative approach. The integrative approach42,60 rejects the idea that clinical trials depend on utilitarian reasoning, where common welfare is thought of as a function of individual welfare. Instead, individuals have different kinds of interests. One kind is basic interests, which permit humans to pursue a life plan and regulated their behavior by considering what they believe to be good and right. The other is personal interests. Clinical trials must give priority to “basic interests” of patients, as these also constitute the common good. Such prioritization can justify compromising on “personal interests.” Kantian Universalizability Test. In response to problems regarding patient consent and the tension between care and research, a universalizability test has been proposed
48
: physicians can compromise on care (in favor of conducting a trial), if that is a decision process that all physicians would subscribe to. The main basis for universalizability is that, by compromising somewhat on the level of care for current patients, one benefits future patients. Rehabilitation Fried's theoretical equipoise. Some have advocated a return to Fried's initial concept of equipoise, and to give renewed weight to the personal uncertainty of the physician–researcher.34,47,61 Multifactorial approaches. Something in between an amendment and an alternative, Emanuel and collaborators
49
proposed seven ethical requirements for clinical trials: (1) scientific or social value; (2) scientific validity; (3) fair subject selection; (4) favorable risk–benefit ratio; (5) independent review; (6) informed consent; and (7) respect for enrolled research participants. This framework integrates CE (as an element of scientific validity) and acknowledges the importance of nonexploitation (via respect).
Discussion and conclusion
From the reasons and arguments reviewed in the results section, it seems fair to say that the concept of CE suffers from several fundamental limitations. One important limitation is vagueness: no clear criteria are available to judge when disagreement among expert physician–researchers reaches “equipoise.”9,19,28–30,32–39 This vagueness is not an abstract, theoretical problem: as several authors indicate, the lack of clear criteria means that equipoise can be manufactured through the influence of undue financial incentives.9,30 Another important criticism is that CE, at least in its original formulation, did not acknowledge the importance of getting consent from patients.
CE and informed consent
One common response strategy has been to amend the principle of CE through additional criteria. This response strategy is most clearly applicable to issues regarding informed consent, and in fact, one of the most frequently mentioned suggestions in the reviewed literature was to make clinical trials conditional on obtaining informed consent from patients.23,25–27,32,38,39 Not only does this respect patient autonomy but also educating patients on the trial increases their willingness to participate. 62
However, the concept of “informed consent” faces its own issues which are at least as widely known as those facing CE (see e.g. Beauchamp and Childress, 63 chapter 4). For instance, patients’ lack of relevant training and knowledge may make it difficult for physicians to avoid exerting undue influence on patient choice and thus to obtain genuine consent. 63 Patients may also be very vulnerable to the therapeutic misconception, especially when CE seems to hold, since CE can give the impression that (individualized) care need not be compromised in the course of research. Hence, the condition of informed consent should entail explaining both the science and the medical ethics of the clinical trial they are about to enroll in. However, even then it is unclear that genuine consent could be obtained in all situations that can arise in the context of clinical trials (disadvantaged patients, desperate patients, external pressure on researchers to conduct a specific trial, etc.). Thus, CE inherits all the general difficulties associated with the concept of genuine consent. There remains a fundamental asymmetry between physician–researcher and patient, both in terms of medical knowledge and understanding of the nature of scientific research. Such asymmetries, in another context, are precisely the rationale for standards of professionalism, which professionals are expected to follow, on pain of negligence, according to their best judgment and relatively independently of the wishes of clients, patients, etc.64–66
CE and vagueness
Other criticisms of CE seem difficult to address by means of a strategy of introducing additional criteria. Vagueness, for instance, opens up on to larger issues: (1) how to clearly delineate “experts” from “non-experts” in a subdomain, and (2) how to decide when there is “no scientific consensus” about some intervention. The first issue becomes particularly difficult when a proposed intervention is viewed differently through different medical backgrounds (for instance, interventions on cognitive function may be viewed very differently by neurologists and psychiatrists). The second issue tends to arise when a lot is at stake. It has long been documented how scientists may be unduly influenced by financial incentives in order to express uncertainty about scientific questions. 67 In clinical research, financial incentives may provide motivation for emphasizing the downsides of established treatments or for emphasizing the potential benefits of proposed treatments. In this way, clinical equipoise could easily be manufactured.
In sum, the principle of CE inherits many larger difficulties surrounding other concepts: in this case, the concepts of “expertise” and “consensus.” It is hard to see how CE could be amended to avoid the charges of vagueness, especially with regard to defining “lack of consensus". The presence of “scientific consensus” is, at least in principle, definable and is often proposed as an indicator of trustworthiness of a scientific statement. 68 Ironically, CE requires – by design – that there is no scientific consensus. In this sense, it is not clear whether CE could be amended to address issues regarding this aspect of vagueness without in fact rejecting CE entirely. One could speculate whether this explains why vagueness was not the target of any constructive response in the reviewed literature, despite being one of the most frequently mentioned criticisms of CE (see Figure 2).
Limitations of this review
At this point, we should discuss the main risks associated with the chosen methodology of this review study. The study excluded a number of potentially relevant discussions of CE: textbooks, clinical studies that include some discussion about CE, empirical bioethical studies on attitudes toward CE, letters to the editor, and journal articles published before 1987. Therefore, it remains possible that some important arguments regarding CE were missed. Nonetheless, insofar this study sought to extract key ideas from the literature, and insofar this study included many influential conceptual discussions of CE, it is unlikely that the global results from the review – e.g. which criticisms receive most weight or attention in the literature – would be affected.
Another limitation to the current study is that the various replies to the objections to CE were not surveyed. Such replies provide indirect support to CE, but are not covered by the review since they usually do not constitute direct, positive arguments supporting CE, nor do they tend to be constructive proposals to amend or replace CE. Some replies were exceptionally included in this review: for instance, the replies to criticisms regarding informed consent constituted an amendment to CE, and hence they were included “Patient Consent and Patient Equipoise.” Nonetheless, replies (and replies to replies) were not systematically charted: not only is it doubtful that this argumentative depth could be surveyed in a review such as this one, but it is unlikely that the argumentative landscape charted in the results section would dramatically change.
In sum, the limitations inherent to the critical-interpretive methodology mean that not all relevant contributions to the debate about CE were included. However, despite these limitations, it seems possible to draw some reasonably robust conclusions, with the most important being that three decades of criticism of CE have failed to provide a conclusive refinement of CE or an uncontroversial superior alternative.
Shifting the expectations of CE?
Does our review then corroborate the suspicion that CE may simply be the least flawed available principle for designing ethical RCTs? Not necessarily. We would like to suggest that one could view CE's many “failures” in a very different light. How so? In general, a feature can only be deemed a “failure” with respect to a goal or expectation. Failure is a relative concept, and the many shortcomings of CE could point to misguided expectations of what an ethical concept can deliver. Exploring this in detail could be a subject of a separate study, but we wish merely to point to the implicit but widespread expectation that CE can deliver a collection of fail-safe and universal conditions for conducting ethical RCTs. Such conditions would guarantee physician-researchers that their planned RCT is ethically justified -- as long as the conditions are met. Against this expectation, the discovery of the vagueness of what counts as "honest, professional community-level disagreement", or CE's sensitivity to ambiguous preliminary data appear as "failures". However, it may be misguided to expect this type of clarity from a principle such as CE.
An alternative expectation would be to view the primary function of the principle of CE as guiding the reasoning and judgment of individual physician–researchers. From this perspective, it is a feature – not a bug – that CE is vague and easily upset by preliminary data. The limitations imply that the physician–researcher must use individual judgment to decide on the state of equipoise in the relevant community of experts. They must take into account the particulars of the clinical situation: whether or not to ignore a dissenting colleague or not, or whether to nonrandomly assign a patient to the novel treatment arm. Chiapperino and Nardini 52 speak of CE functioning as a mid-level principle; another way of categorizing this line of reasoning is that it views CE as a principle for the professional ethics of physician–researchers. The limitations of CE may not necessarily be evidence that CE needs to be “fixed,” nor that CE needs to be replaced, but rather that physician–researchers should be acutely aware of the limitations of CE and not take CE as a means to entirely outsource ethical deliberation to a research ethics committee.
It is interesting to note that, in the original Declaration of Helsinki, it was incumbent on individual physician–researchers to judge the appropriate course of action. The researcher needed to conduct a “careful assessment of inherent risks in comparison to foreseeable benefits to the subject or others” (Principle 4). 69 It was only later, in the first revision in 1975, 70 that the individual judgment of the physician-researcher was subordinated to that of an external committee. At one level, the principle of CE supports this development: judgments about lack of consensus in a medical community about treatment options are arguably better made by a diverse committee than by an individual. However, at another level, given that it can be difficult to judge whether equipoise holds in particular circumstances, the limitations of CE can be interpreted as pointing to the role that individual judgment must play. In this view, CE may remain a frequently used principle because it is useful – and perhaps more so than other proposed alternatives – as a guide for the reasoning of individual physician–researchers. What precisely this usefulness consists of is a question beyond the scope of this discussion. The suggestion here is that the continued popularity of CE need not be interpreted as its being the “least flawed” principle, but rather as being a “maximally useful” principle in physician–researchers’ efforts to combine therapy and research.
Conclusion
Although the principle of CE suffers from many limitations, it remains an elegant and influential approach to resolving trade-offs between therapy and research. In many ways, it simply seems to be the least flawed principle available; however, it is also possible that its bugs are actually design features. In that case, CE's problems with vagueness or informed consent mean that establishing CE requires an individual, professional judgment that is mindful of the particulars of the trial and of the patients involved.
Supplemental Material
sj-pdf-1-cet-10.1177_14777509221121107 - Supplemental material for Clinical equipoise: Why still the gold standard for randomized clinical trials?
Supplemental material, sj-pdf-1-cet-10.1177_14777509221121107 for Clinical equipoise: Why still the gold standard for randomized clinical trials? by Charlemagne Asonganyi Folefac and Hugh Desmond in Statistical Methods in Medical Research
Footnotes
Authors’ contribution
Charlemagne Folefac designed the study, carried out the search, conducted the analysis, extracted the data, and wrote the initial drafts of the paper. Hugh Desmond designed the study, rewrote the successive drafts of the paper, and wrote the background and discussion sections.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Hugh Desmond's work on this article was supported by the Horizon 2020 Framework Programme (grant number 741782).
Notes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
