Abstract
This study aims to develop a system approach for modeling and simulation of marine scrubbers, which are devices that reduce sulfur oxides (SOx) emissions from ships powered by heavy fuel oil (HFO). Marine scrubbers are a viable option to comply with the new regulations issued by the International Maritime Organization (IMO) for SOx control. This system model of a marine scrubber was developed using the bond graph method, which simulates the interactions between exhaust gas and seawater within a marine scrubber, incorporating the physical and chemical processes involved, and supports modular modeling concepts. The model was validated by comparing the simulation results with the data collected from a scrubber installed on a real ship under operational conditions. The comparisons show that the model is robust and accurate, and can be used to simulate cases under real operational conditions, and hence can be used for optimization and integration studies.
Introduction
Background
With its extraordinary carrying capacity and exceptionally low unit cost, maritime shipping outperforms other transportation methods in international trade, particularly for long-distance commodity shipment in terms of both cost and emissions. It has played an important role in the history of human economic and cultural exchange and is projected to grow steadily in future global trade. 1 Currently, maritime shipping remains the dominant way of global goods transport, accounting for approximately 80% of internationally traded goods by volume. However, this prominence makes it a significant contributor to global exhaust gas emissions. 2
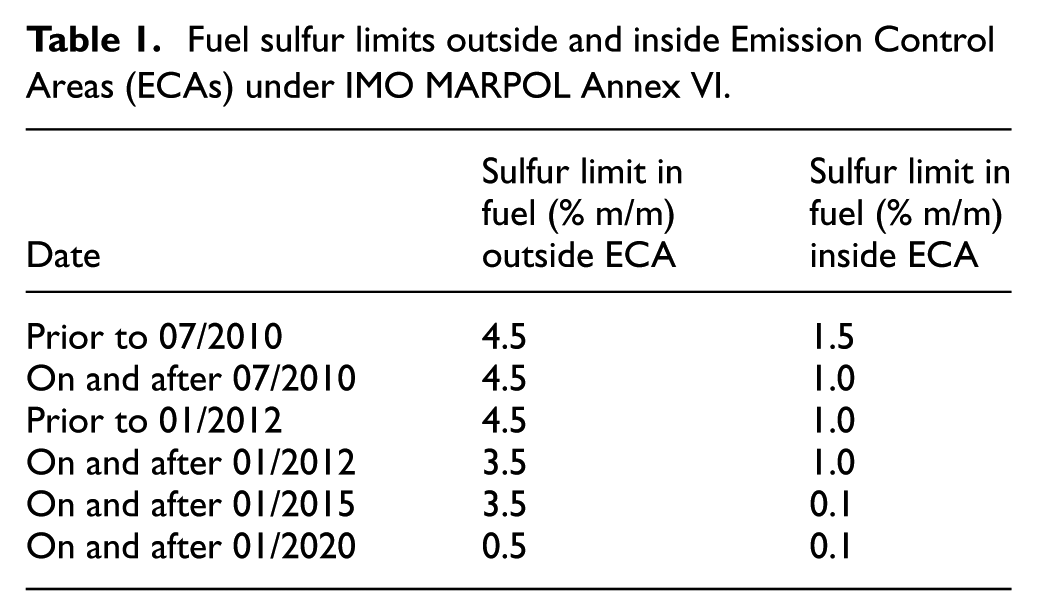
The exhaust gases from ships contain greenhouse gases (GHG), nitrogen oxides (NOx), particulate matter (PM), and sulfur oxides (SOx), which contribute to global warming, acid rain, and adverse human health effects. To address these environmental impacts, the International Maritime Organization (IMO) established the International Convention on the Prevention of Pollution from Ships (MARPOL 73/78), with subsequent amendments introducing increasingly stringent regulations. MARPOL Annex VI 3 specifically targets air pollutants, including SOx, through progressively stricter emission limits (See Table 1).
Fuel sulfur limits outside and inside Emission Control Areas (ECAs) under IMO MARPOL Annex VI.
Exhaust gas cleaning systems (EGCS) have emerged as a practical retrofit technology for existing vessels to comply with IMO regulations. EGCS includes subsystems designed to remove NOx, SOx, PM, and potentially GHG emissions. The SOx-removal subsystem, commonly termed a “scrubber,” is the focus of this study.
Marine scrubber
A marine scrubber removes SOx from the exhaust gas by spraying an alkaline scrubbing liquid (e.g. seawater or freshwater with additives) into the gas stream, allowing ships to continue using heavy fuel oil (HFO), despite its high sulfur content. While the maritime industry is gradually transitioning to greener alternatives—such as liquefied natural gas (LNG), hydrogen, or ammonia, these technologies face challenges in scalability, infrastructure readiness, or economic viability. Therefore, marine scrubbers are a vital transitional solution for HFO-powered vessels under current regulations, providing scalable efficiency to meet future potential regulatory tightening.
Marine scrubbers have demonstrated SOx removal efficiencies exceeding 95% under optimal design and operational conditions. Additionally, they exhibit a secondary benefit of reducing PM emissions by over 60%, further enhancing their environmental value. 4
Scrubbers are classified based on design principles and operational parameters:
– –
Wet scrubbers are further categorized by:
– – –
Despite these variations, all marine scrubbers share a core advantage: they allow vessels to continue using cost-effective heavy fuel oil (HFO) while complying with IMO 2020 standards. This positions scrubbers as a critical retrofit solution for existing vessels transitioning toward long-term decarbonization goals.
Marine seawater scrubber performance hinges on the physical and chemical interactions governing sulfur dioxide (SO2) absorption in seawater. Early studies established foundational insights into solubility and reaction kinetics: Douabul and Riley 5 studied the solubility of SO2 in distilled water and decarbonated seawater. The experiments were set with a pressure of 1 atm of SO2 at pH 0.8, with a temperature range of 5.8°C–30°C, and salinity of 0‰–40‰. They found that the solubility of SO2 in seawater decreases slightly with increasing salinity, and decreases greatly while the seawater temperature increases. In pure water and seawater, the solubility ranges from about 3.065 mol/L at 5°C to 1.198 mol/L at 30°C, depending on the salinity (range from 0.0 to 40.0 g/kg). Al-Enezi et al. 6 and Rodríguez-Sevilla et al. 7 quantified SO2 solubility in seawater. Caiazzo et al. 8 conducted experiments to study the efficiency of seawater spray scrubber removing SO2. The experiments tested real seawater from the Naples coast and studied SO2 absorption efficiency by varying exhaust gas flow rates, seawater flow rates, and SO2 concentrations. The study concluded that seawater spray scrubbers can achieve SO2 removal efficiencies of up to 93%. These studies offer foundational insights for SO2 absorption mechanisms in marine scrubbers.
Building on the studies of SO2 absorption in seawater, many studies have explored different options and fidelity for modeling.
Darake et al. 9 studied SO2 absorption in seawater spray scrubbers, identifying several critical parameters for scrubber efficiency. Mestemaker et al. 10 developed advanced dynamic models for closed-loop wet scrubbers using freshwater with caustic soda as scrubbing liquid. The model has demonstrated its adaptability to simulate the SO2 removal efficiency under transient loads in ships.
The studies mentioned above all focus on the mathematical modeling of marine scrubbers. However, these models are generally difficult to integrate into other system-level models. With increasing demands for digitalization, holistic system models of the entire EGCS, or even the full ship propulsion system, have become necessary. This creates a need for more flexible modeling approaches capable of representing such comprehensive systems. Bond graph modeling11,12 is a first-principles-based methodology that supports modular model construction and clear interface definitions between subsystem components, enabling the development of integrated, holistic ship system models.
Bond graphs provide a unified graphical framework for representing chemical kinetics, fluid dynamics, and energy transfer. 13 Their versatility in maritime engineering has been demonstrated in several studies. Yum et al. 14 used bond graphs to model a two-stroke diesel engine and capture its transient response in waves. Nielsen and Pedersen 15 applied bond graphs to selective catalytic reduction (SCR) reactors, developing SCR models with three levels of fidelity and comparing their performance under dynamic conditions. A bond graph model for simulating fuel cells in marine power plants was developed by Bruun. 16 Together, these studies highlight the method’s ability to model complex maritime systems involving coupled physical and chemical processes.
Although the use of bond graphs in the maritime field is still limited, the approach shows strong potential for system integration in holistic simulations. Developing a scrubber model using the bond graph method therefore addresses an important gap in system-level modeling of de-SOx systems.
Objectives
The main objectives of this study are: (1) To develop and validate a system-level model of a marine scrubber using the bond graph method, which can simulate the physical and chemical processes in the SOx removal process. (2) To conduct a careful convergence test for the system model to ensure reliable simulation results. (3) To compare the simulation results with the data collected from a scrubber installed on a real ship under operational conditions.
Scrubber modeling
Figure 1 shows a schematic diagram of the modeling object we have chosen for this study, a marine spray scrubber. In this model, scrubbing liquid (e.g. seawater) is pumped through pipelines and dispersed within the scrubber via multiple layers of nozzles in the upper part of the scrubber to optimize contact with exhaust gas. The exhaust gas enters from the lower part of the scrubber, mixing with the seawater spray as it rises. The seawater spray creates numerous droplets that facilitate physical interactions (mass and heat transfer) between the exhaust gas and droplets. SOx is transferred from the exhaust gas in gas phase to the droplets in liquid phase. Concurrently, a portion of the liquid SOx undergoes a chemical reaction with the alkaline components of seawater inside the droplet, enhancing the efficiency of SOx elimination during the process. Afterward, seawater droplets fall to the bottom, and is drained out from the scrubber.
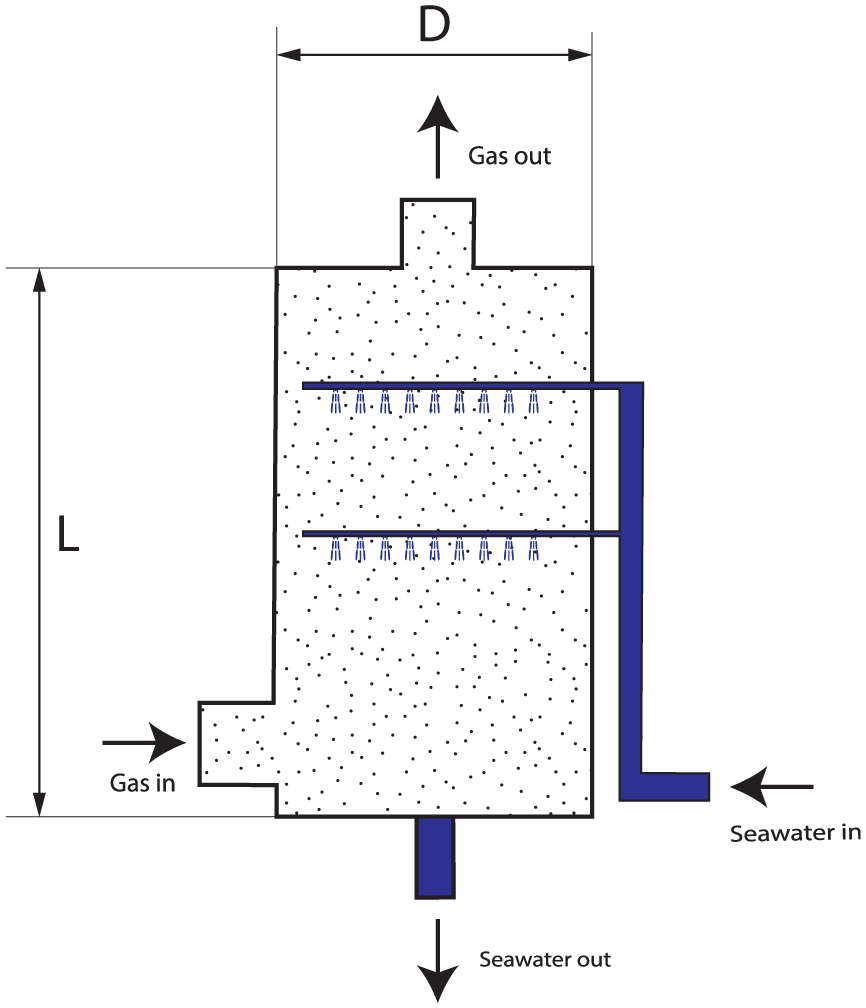
Schematic design of a marine scrubber. L is the length of the scrubber column, D is the diameter of the scrubber column.
Referring to Figure 1, the axially symmetric cylindrical scrubber column allows a one-dimensional (1D) simplification along its height, neglecting lateral variations. Despite this simplification, the model remains multi-domain, involving fluid dynamics, heat transfer, and chemical reactions. A 1D distribution of the process variables along the scrubber height is still necessary to obtain accurate simulations.
Bond graph structure
Due to the presence of the thermal, chemical and flow domains in a marine scrubber, a pseudo-bond graph formulation 17 is adopted to properly describe all energy and mass transfers in the whole system. As Figure 2 demonstrates, three pairs of effort and flow variables representing mass, mass fraction and thermal energy exchange between two systems A and B 18 are chosen for this bond graph model:
Effort variables:
– P: Pressure – – T: Gas temperature
Flow variables:
– – –
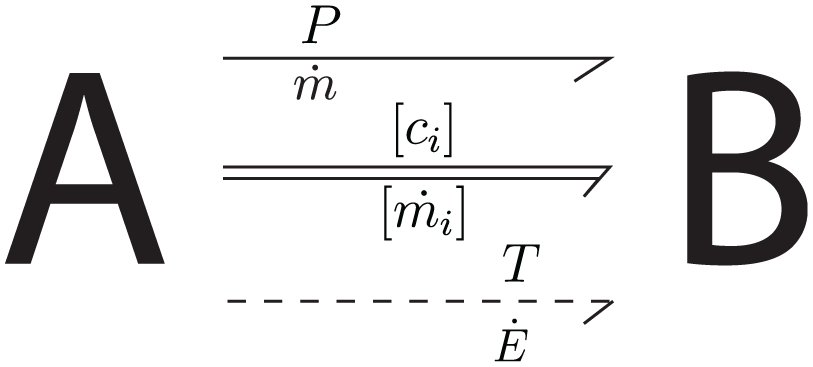
Effort and flow variables were chosen for modeling marine scrubber in this study.
The basic bond graph elements used in this study are described below based on their physical functions 17 :
For the system model in this study, the boundary conditions are the exhaust gas composition, pressure and temperature at the scrubber inlet, and the pressure at the scrubber outlet. These values can normally be easily calculated or measured for each scrubber. Since the exhaust gas does not travel at a very high speed inside the scrubber, it can be assumed as incompressible, that is, with constant density throughout the scrubber. This density can be calculated from the average pressure at the scrubber inlet (P in ) and outlet (P out ), and the inlet temperature (T in ), as given in equation (1).
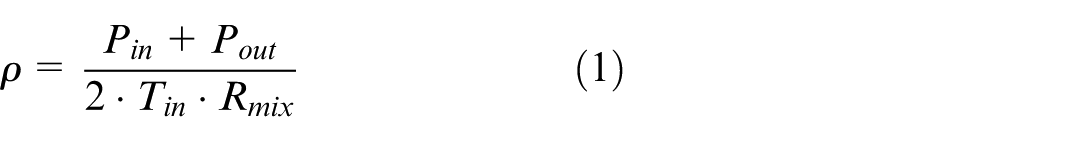
where R mix is the specific gas constant of the exhaust gas mixture.
The exhaust gas velocity, u
g
is determined by the pressure drop across the scrubber

Previous work by Nielsen and Pedersen 15 examined the impact of assuming constant gas density and velocity, and concluded that this simplified approach yields predictions reasonably close to those of more advanced variable-density models, while substantially reducing computational cost. Although assuming a constant mass flow rate may appear to conflict with conservation laws, the very low concentration of SO2 in marine exhaust gases means that this simplification has minimal influence on the simulation accuracy. For example, for heavy fuel oil containing 3 mass % sulfur, the resulting SO2 concentration in the exhaust is only about 600 ppm (about 0.3 mass %). 19 Even complete removal of SO2 would therefore change the total exhaust mass flow only marginally.
Figure 3 outlines how the scrubber model is discretized into multiple control volumes in this study. Figure 4 shows the bond graph model of the i-th control volume together with the models of its neighbors (the i−1-th and
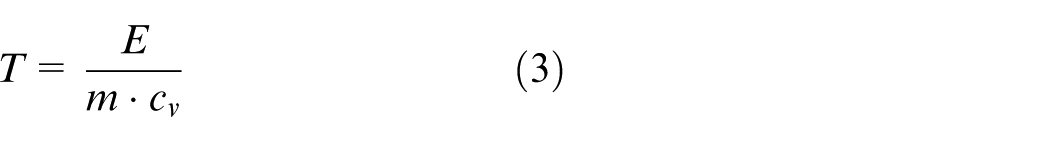
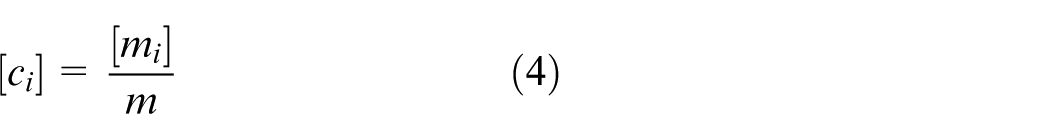
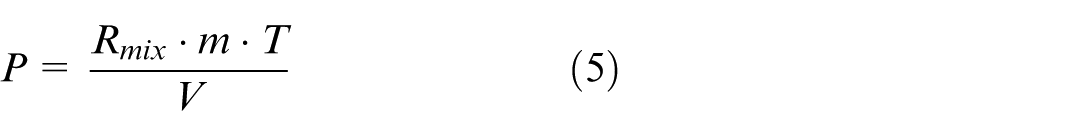
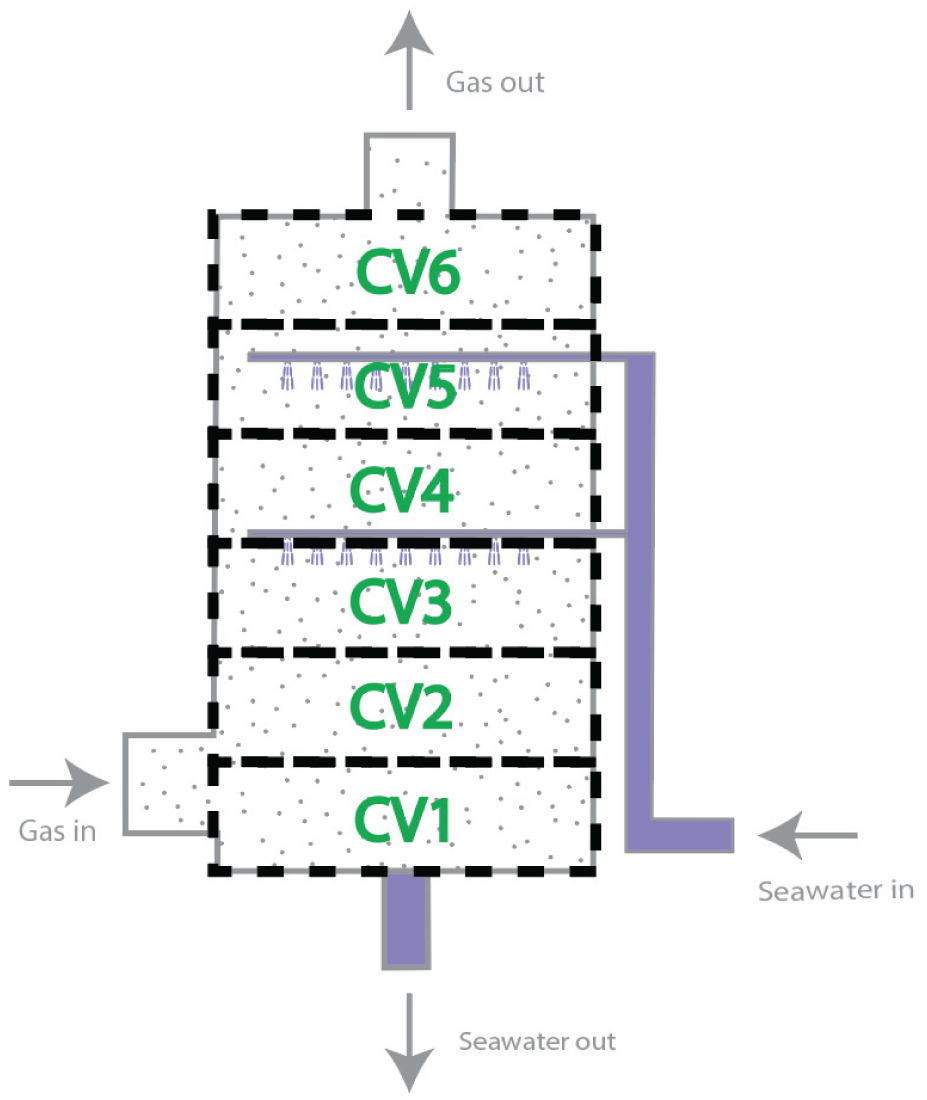
Discretization of scrubber into multiple control volumes.
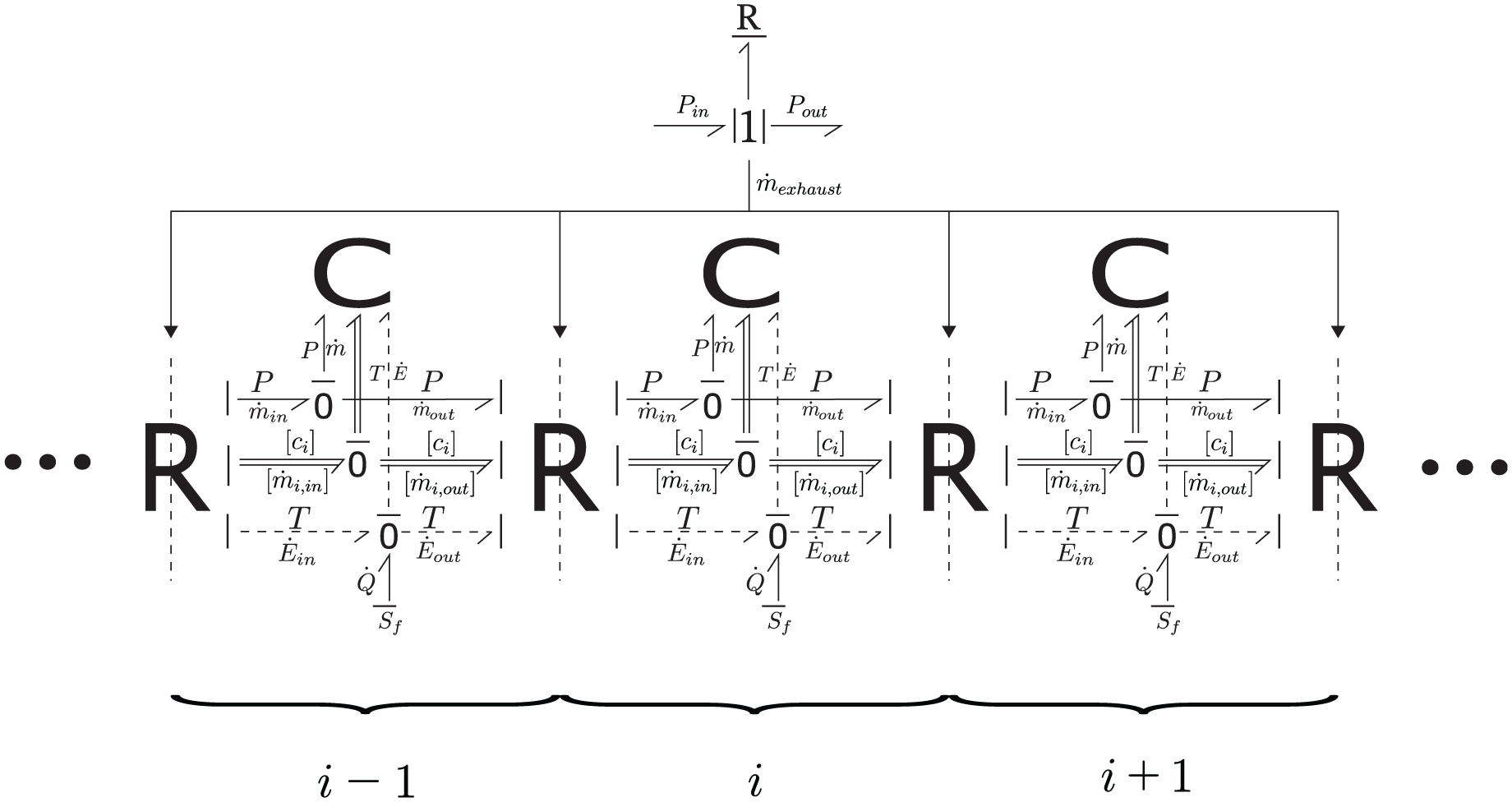
Bond graph presentation of control volumes.
In equations (3)–(5), m and c v are respectively the mass and specific heat capacity of the exhaust gas in a control volume. V is the volume of the control volume.
As also shown in Figure 4, the interfaces between adjacent control volumes are represented by multi-port R-elements, through which mass and energy are exchanged. Each interface boundary is predominantly influenced by upstream flow, given the known exhaust gas velocity. The exhaust gas mass flow rate (
Physical and chemical reactions in the scrubber model
When the exhaust gas meets the seawater droplets in the scrubber, a physical dissolution process happens, and a certain amount of SOx will dissolve through the gas-liquid interface to become an aqueous form. The efficiency of this dissolution process is not uniform throughout the scrubber, because as a droplet gradually falls to the bottom, the absorption rate becomes lower due to saturation. After the gaseous SOx is dissolved, its aqueous form will start a chemical reaction with the alkaline part in the seawater droplets, thus to enhance a de-SOx process.
By further implementing the gas dissolution process and chemical reactions into the bond graph structure, this study will be able to comprehensively account for the physical and chemical processes taking place within a marine scrubber. We note that SOx usually stands as a composition of SO2 and SO3, but the predominant species present in ship emissions is SO2, so the chemical reactions described in this section focus only on SO2.
Dissolution of SO2 happens while the exhaust gas passes through the scrubber and being in contact with the seawater droplets. The solubility of SO2 is augmented in seawater relative to pure water. 6 Mass and heat transfer between the gas and seawater take place during this process. After dissolution, SO2 will be present in both molecular and dissociated forms within the solvent. This process of SO2 dissolving in seawater droplets is explicated by the chemical model provided below.6,7,9
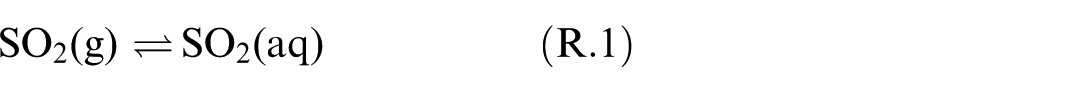
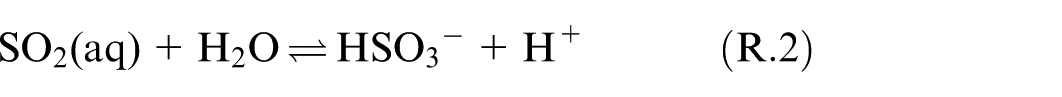

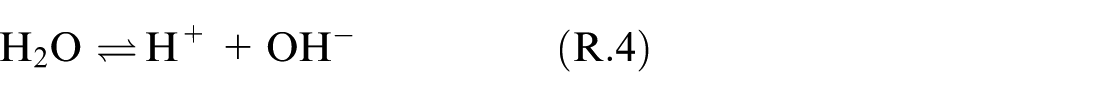
The equilibrium solubility of SO2 in seawater, as an important factor in the absorption process of SO2 by seawater in a marine scrubber, can be predicted using this chemical reaction model.
Based on Reactions (R.1)–(R.4), the main molecular species in the solvent are:
For Reaction (R.1), the amount of non-reacted

where C gas is the molar concentration (mole fraction) of a gas species in the aqueous phase. There is also a more practical expression for k H , as given in equation (7).
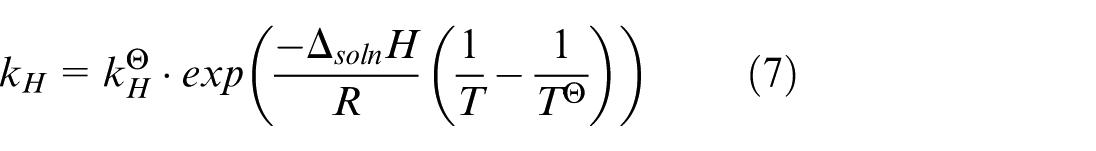
where
From equation (7), we see that the value of k
H
changes with the temperature of the solvent, types of the solvent, as well as types of the gas species. Note that
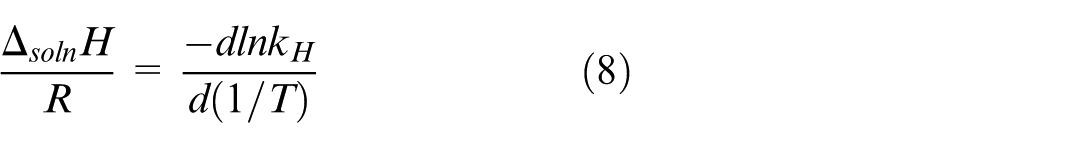
In this study,
The equilibrium constants of reaction (R.2–R.4) can be expressed as the corresponding concentration of chemical products divided by reactants, as presented in equations (9)–(11).

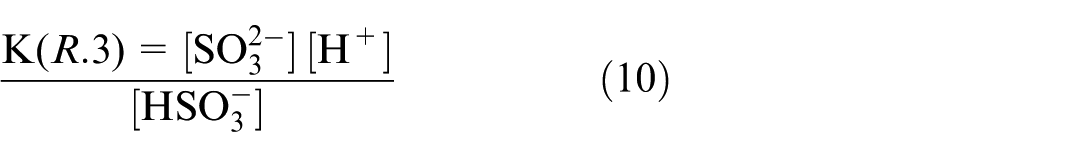

The equilibrium constant K is dependent on the water temperature and can be calculated by:
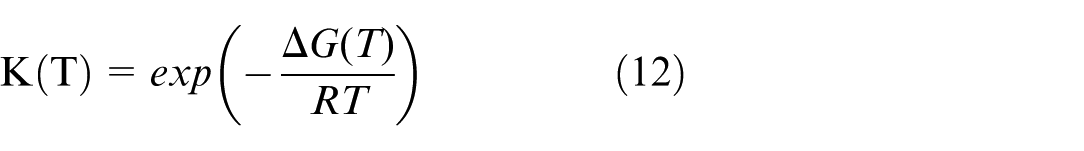

where
By combining the equations above, the concentration of
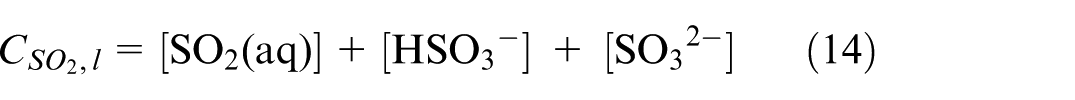
This could be further expressed as:

where
Absorption of SO2 by seawater droplets, which are generated by nozzle sprays, is the main physical process inside a marine scrubber. There are several ways to model the mass transfer process of SO2 from gas mixture to the droplets. One commonly used absorption model is based on two-film theory, one film on the gas side and the other on the liquid side. Figure 5 shows the concept of this theory.
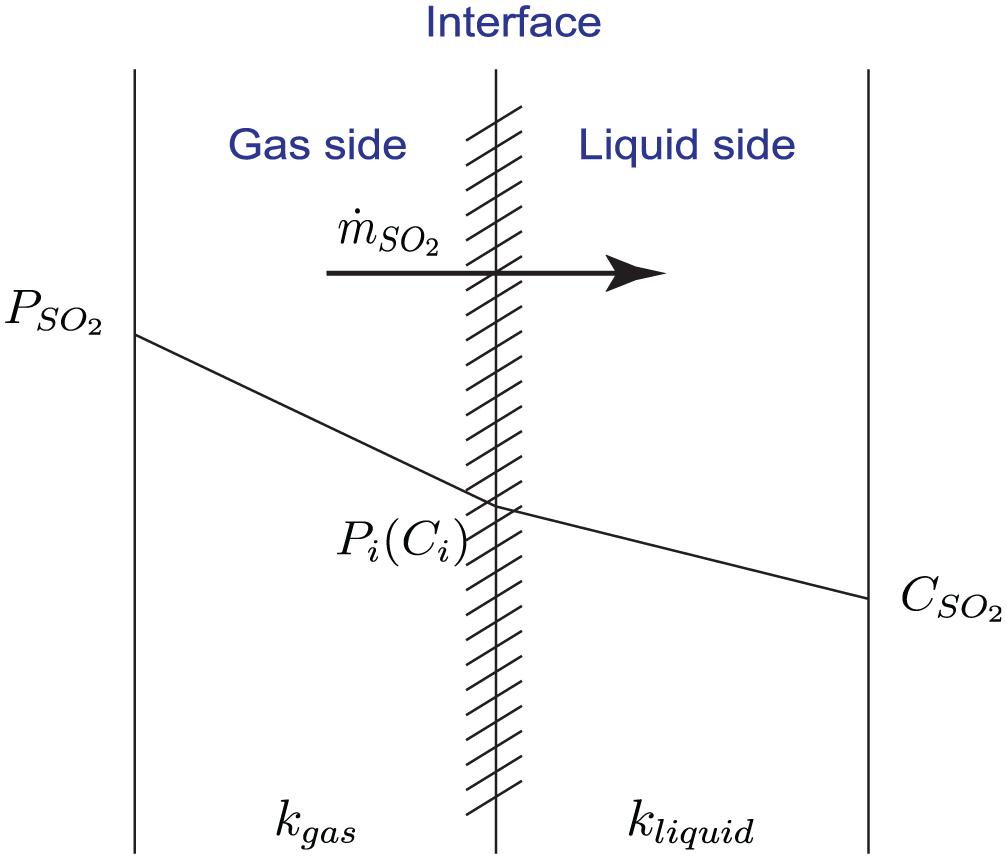
Diagram of SO2 mass transfer from gas to liquid based on two-film theory.
On the gas side, the driving force of SO2 mass transfer is the difference between the partial pressure of SO2 on the gas side (denoted as
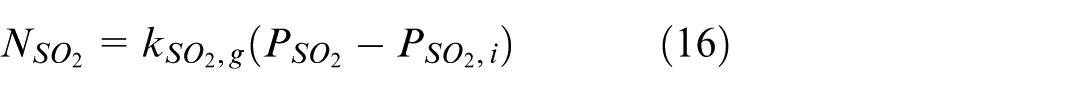
where
On the liquid side, the driving force of SO2 mass transfer is the difference between SO2 concentration at the gas-liquid interface (
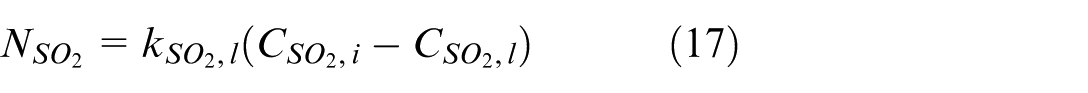
where
The relationship between
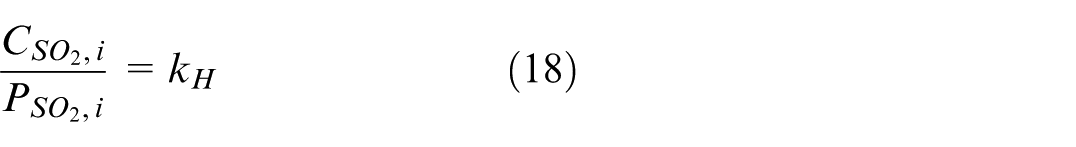
Combining equations (15)–(18), an expression of
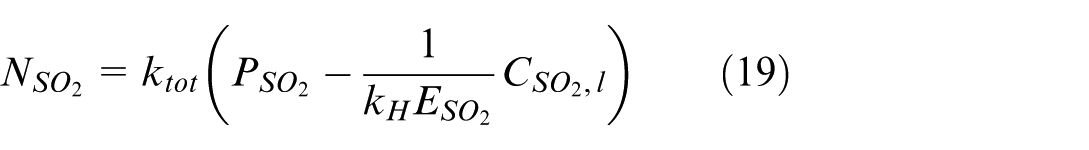
where k tot is introduced to represent the total mass transfer coefficient and expressed as:
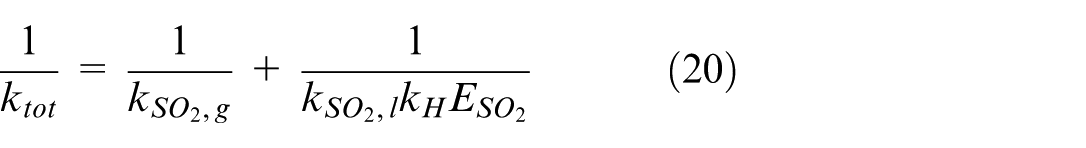
This SO2 mass transfer process is modeled using a bond graph in this study as shown in Figure 6.
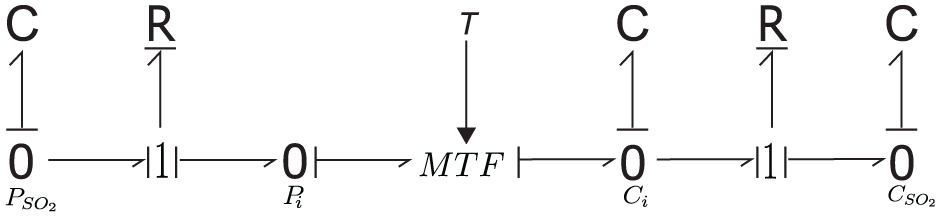
Bond graph presentation of SO2 mass transfer from gas to liquid.
Equations (19) and (20) show that several parameters influence the absorption of SO2 in seawater when the partial pressure of SO2 is known. These include the total solubility of SO2 in seawater, (represented by
The mass transfer coefficient on the gas side, that is

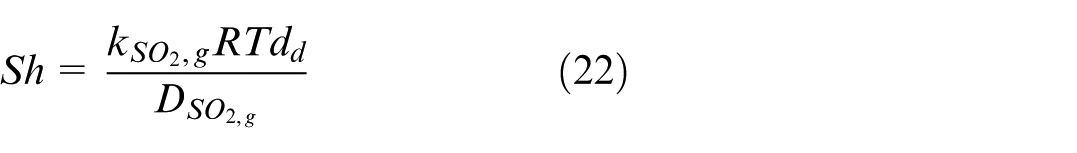
Where Sh is the Sherwood number, Re is the Reynolds number, and Sc is the Schmidt number. All these are non-dimensional numbers. R is the universal gas constant, T is the gas temperature, d
d
is droplet diameter, and
Reynolds number represents the ratio between inertial and viscous forces, and can be calculated based on the droplet reference length and the relative velocity between a droplet and the surrounding gas. While Schmidt number is the ratio of gas viscosity to molecular diffusion. They can be written as:


where:
μ
g
is gas viscosity in
d d is droplet diameter in m
u d and u g are the droplet’s and gas’s velocity, respectively, both in m/s.
Combining equations (21)–(24),
For spherical droplets, its transient velocity u d can be dynamically calculated based on forces acting on it, that is, drag force F d , buoyancy force F b and gravitational force F g , as expressed by equation (25).
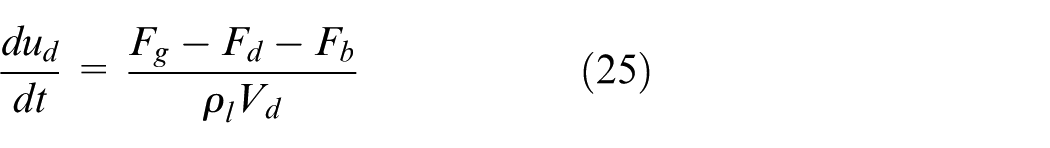
where F g , F d , F b can be calculated as follows:
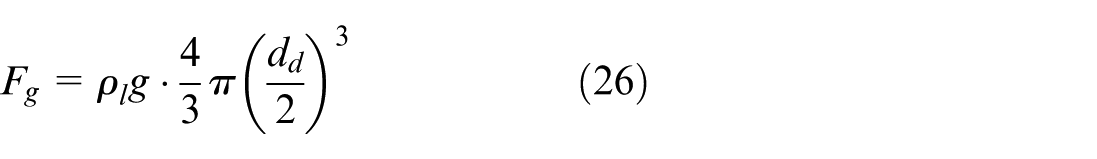
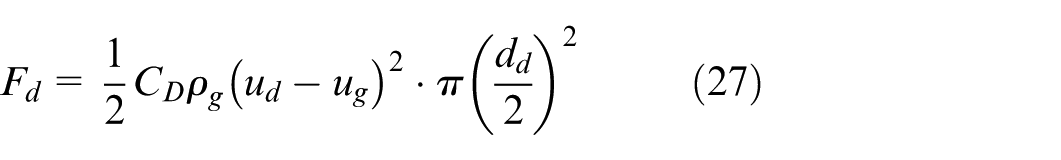
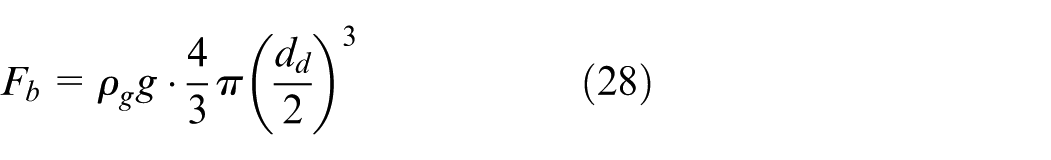
The velocity of the droplet u
d
exiting the nozzle is high, but rapidly decreases until it reaches a constant value known as terminal velocity and denoted as U herein. By letting
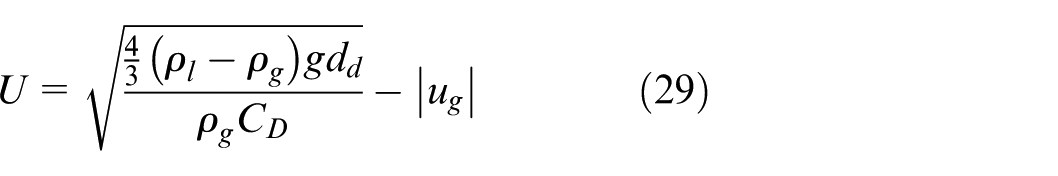
When calculating the R e , the terminal velocity U is used in place of u d . This simplified form allows efficient estimation of droplet motion without solving the full transient equation.
For calculating mass transfer coefficient on the liquid side, that is


where σ is surface tension of liquid in N/m,
ρ
L
is density of liquid in
f d is the frequency of drop oscillation in Hz,
Another widely used approach for determining the value of

Where t denotes the penetration time. In the study by Brogren and Karlsson 24 the penetration time is assumed to be between 0.01 and 0.1 s.
Several other semiempirical expressions can be used to calculate
Once the gas and liquid mass transfer coefficients of
Besides mass transfer between exhaust gas and seawater, there is also heat transfer between them due to temperature differences. The energy transfer rate can be calculated using equation (33).
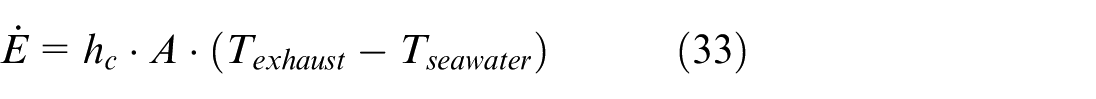
where A represents the total contact area between the exhaust gas and seawater droplets in a control volume, and h c is the convective heat transfer coefficient between these two substances.
The bond graph structure representing the heat transfer process between the exhaust gas and seawater is shown in Figure 7. When combined with the mass transfer bond graph shown in Figure 6, the bond graph model of a marine scrubber can be extended to the model in Figure 8. This extended model uses two multi-port C-elements (Ci,exhaust, Ci,seawater) to represent the exhaust gas (in gas phase) and the seawater droplets (in liquid phase) in control volume i, respectively. The “Mass & Heat Transfer” block in this model contains the details regarding mass and heat transfer between these two phases. This extended model enables the accurate modeling of these processes within a single control volume.
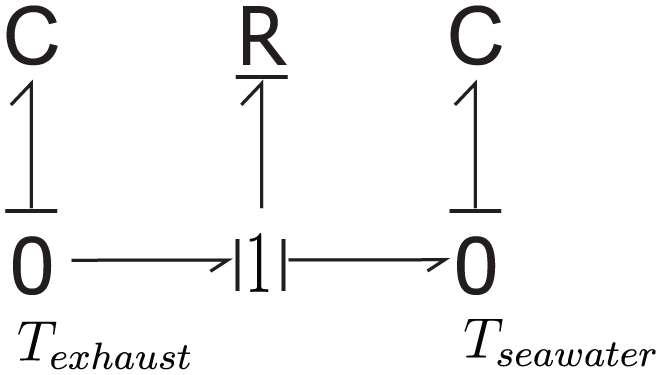
Bond graph illustrating heat transfer exchange between exhaust gas and seawater.
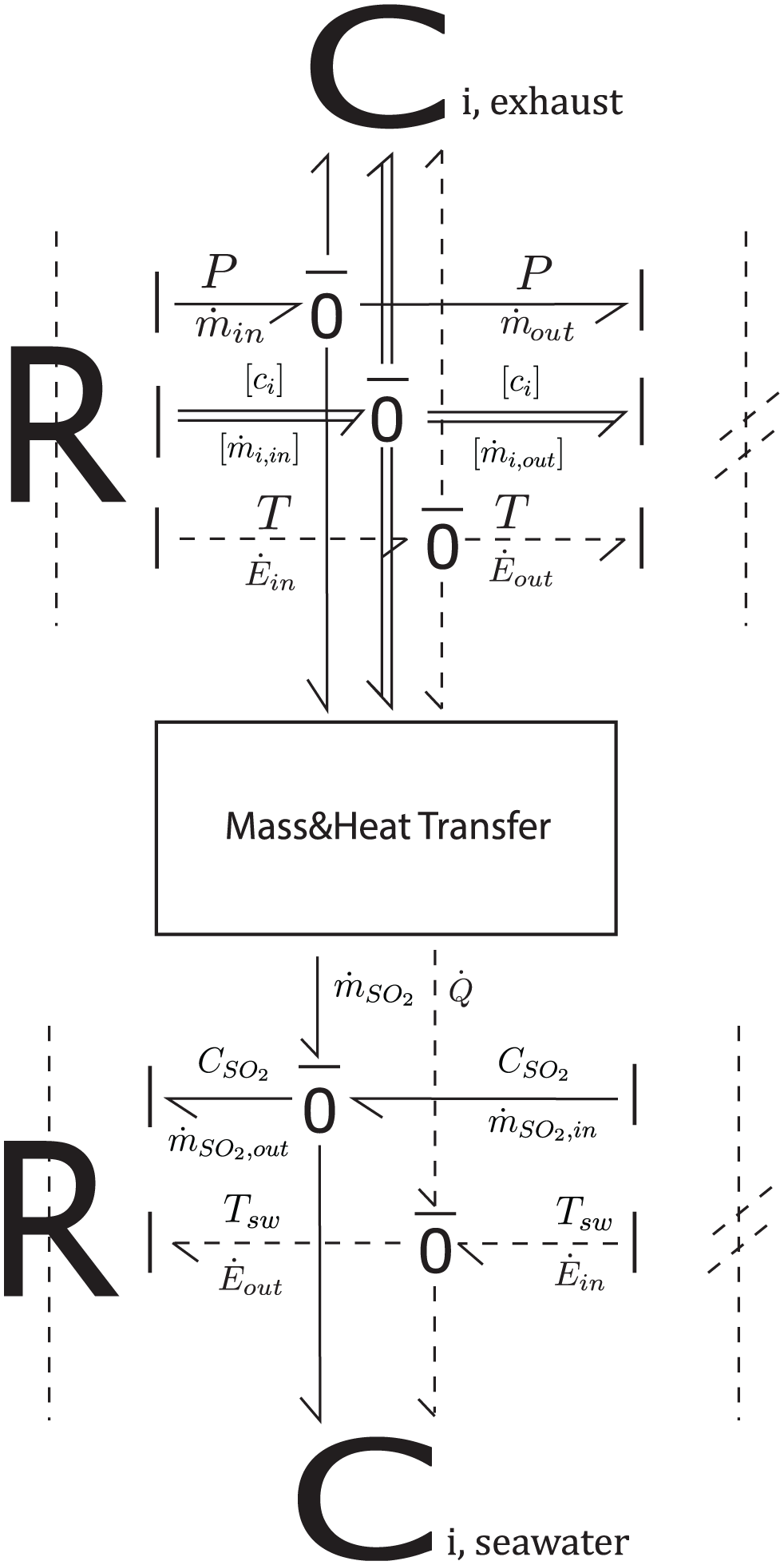
Bond graph model illustrating heat and mass transfer between exhaust gas and seawater in control volume i.
The built scrubber model is a framework that can be set up by modifying model parameters to represent different scrubber designs and configurations. For instance, the geometry of the scrubber, seawater properties, droplet size, etc. all parameters that vary with scrubber design and operation area.
Lastly, we remark several simplifications made during the implementation of the scrubber model described above:
The exhaust gas is treated as an ideal gas, and with constant mass flow through the scrubber.
Heat loss to the environment is neglected.
The seawater droplets are homogeneously distributed and in a complete mixture with the exhaust gas in each control volume.
The diameter of seawater droplets is considered uniform in the scrubber and does not change during the falling process.
Uncertainty and fidelity
The proposed scrubber model in this paper includes some sources of uncertainty, mainly related to the equations and parameters used. For example, there are several empirical correlations available in the literature to calculate SO2 mass transfer on the gas and liquid sides. In this study, one well-established set of equations is chosen, while the alternative correlations could also be applied and may lead to small differences in results. In addition, some parameter values are taken from published data or general engineering assumptions, which may introduce minor variations. As for limitations, the model is developed and validated for a spray-type scrubber, and its performance for other scrubber types has not yet been evaluated. In terms of fidelity, the model can be integrated with other ship subsystems, which makes it suitable for system-level simulations. Overall, these uncertainties do not affect this model’s ability to represent the key physical and chemical processes inside the scrubber. The fidelity of the model fits the intended purpose of a system-level modeling and simulation.
Simulation and results
The physical and chemical processes within marine scrubbers are encapsulated within a complete marine scrubber model in the above section. This section presents the verification and validation of this model. A convergence study is first conducted to examine the validity of the numerical discretization, followed by model tuning and calibration. Then use two groups of experimental data measured from different sea area to validate the model. Simulation results are directly compared with the experimental data.
Discretization convergence study
The discretization of the entire scrubber volume, allows dynamic representation of the system’s behavior. However, it also necessitates a careful examination of the model’s sensitivity to the level of discretization to minimize uncertainties from the numerical implementation. In other words, it is crucial to ensure that the simulation outcomes are not significantly influenced by the number of control volumes utilized in the discretization process. This is achieved through a convergence test. This test is conducted under representative inflow/boundary conditions, which are chosen to reflect typical operating scenarios for the system.
As depicted in Figure 9, a series of simulations were conducted, wherein the scrubber model was divided into 2–10 control volumes, respectively. The results, specifically the SO2 mass flow rate and SO2 concentration (in ppm), along the scrubber height were plotted for comparison. It is noteworthy that the system behavior is qualitatively well represented, irrespective of the number of control volumes used. This is evident from the fact that all curves in the figure show a smooth decrease in the SO2 mass flow rate along the scrubber’s height. This decrease is attributed to the absorption of SO2 as the exhaust gas interacts with seawater droplets within the scrubber.
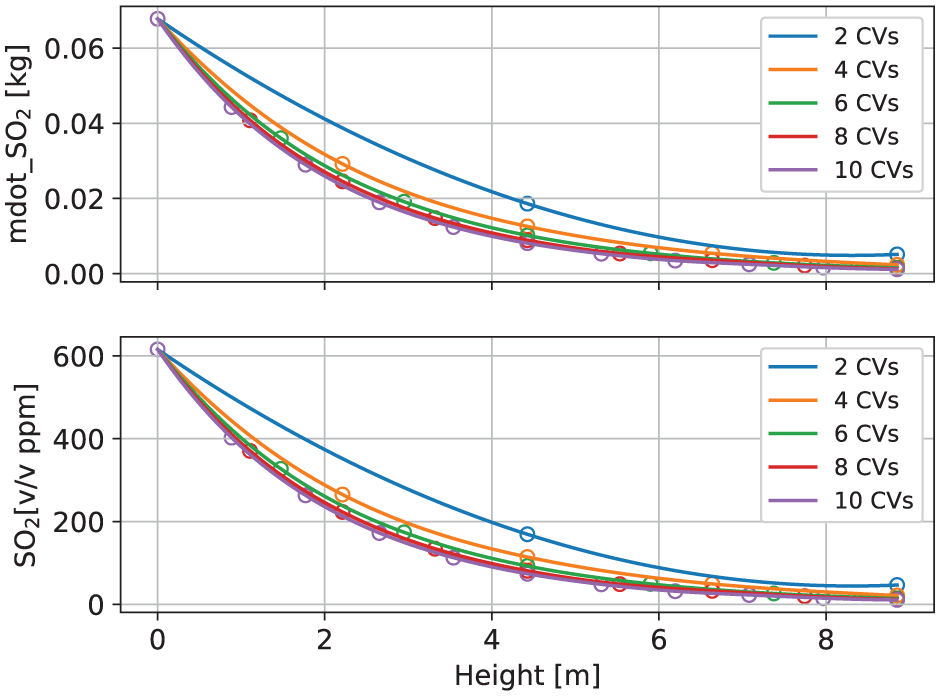
Sensitivity analyses regarding the number of control volumes (CVs). The first subfigure is SO2 mass flow rate (
Further examination of Figure 9 reveals that altering the number of control volumes from 2 to 10 noticeably impacts the distribution of SO2 mass flow rate along the scrubber’s height, particularly when the number of control volumes ranges from 2 to 6. However, when the number of control volumes exceeds six, additional control volumes have almost negligible effects on the results, indicating a convergence at six control volumes. These observations support that a model with six control volumes is sufficient for accurate representation.
Model tuning
As mentioned in the previous section, the model was built with several simplifications, either because some processes or parameters have less pronounced influence to the performance of the system, or because some processes include complex nonlinearity that introduces too much difficulties in properly modeling them. Anyways, due to the existence of simplifications and assumptions, we cannot expect the model to perfectly reflect all details of the real-life processes in a marine scrubber, model tuning therefore becomes necessary.
For instance, in reality, the speed of a droplet changes with time. The droplet obtains an initial velocity after being sprayed from the nozzle, and this initial velocity is affected by various factors, such as droplet size, injection condition, nozzle size, spray angle, etc. So, it is difficult to get this initial velocity. Also, depending on its location, droplet speed distribution is not uniform along the radial direction of the nozzle. 28 Nevertheless, the droplets experience a deceleration process after being sprayed out from the nozzle, under the joint effect of drag and buoyancy forces, until their speeds decrease to the terminal velocity, as expressed in equation (29). For the preliminary simulation set, the terminal velocity of droplets is defined as their average velocity in the model. This assumption is recognized as a source of uncertainty and can be improved.
A calibration of this parameter was therefore performed by introducing a slightly modified droplet average speed. This has been a process through trial-and-error, but the target was clear, that is, the average speed should be higher than the terminal speed while smaller than the initial speed. The resultant droplet average speed after calibration, which meets the above target, is shown in equation (34).
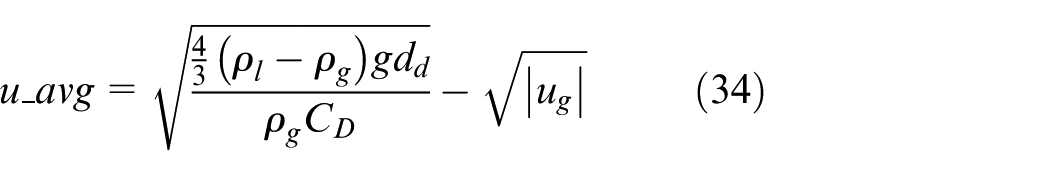
Equation (34) represents a corrected form of the droplet terminal velocity, introduced to approximate an average droplet speed between the initial spray velocity and the final terminal velocity. In this study, a single representative average velocity was adopted within the bond graph framework. Although Payri et al. 28 observed that the spatial velocity distribution of spray droplets follows a Gaussian profile, such detailed radial variation is beyond the resolution of the present system-level model, where the terminal velocity is used as an average representative value. This simplification is noted as a source of uncertainty and possible improvement for future work.
Measurement campaign
Voyages
A marine scrubber, designed by Wärtsilä Moss and installed on a Wilh. Wilhelmsen ship, is chosen as the modeling target. With a diameter of 4.50 m and a height of 8.85 m, this scrubber was intended to handle a maximum SO2 mass flow rate of 265 kg/h.
Measurement campaigns were carried out by Marintek (now SINTEF Ocean AS) with the target scrubber. The measurements were made while the target ship was sailing in different areas, respectively in Europe, across the Pacific from Manzanillo, Panama, to Brisbane, Australia, and across the Atlantic from Baltimore to Zeebrugge, reflecting the real-world performance of the chosen scrubber. The data from the measurement campaign across the Atlantic will be the main focus due to its relatively high data quality. Worth noting that measurements taken during actual operation present practical challenges, primarily because the test conditions cannot be as controlled as those in laboratory tests.
Measurement setup
Figure 10 illustrates the setup of the measurement equipment used in the campaigns. The measurement campaign produces a collection of data recorded under various engine load levels with varying seawater temperatures and properties, including the mass flow rate and concentration of various gas species (SO2, CO2,
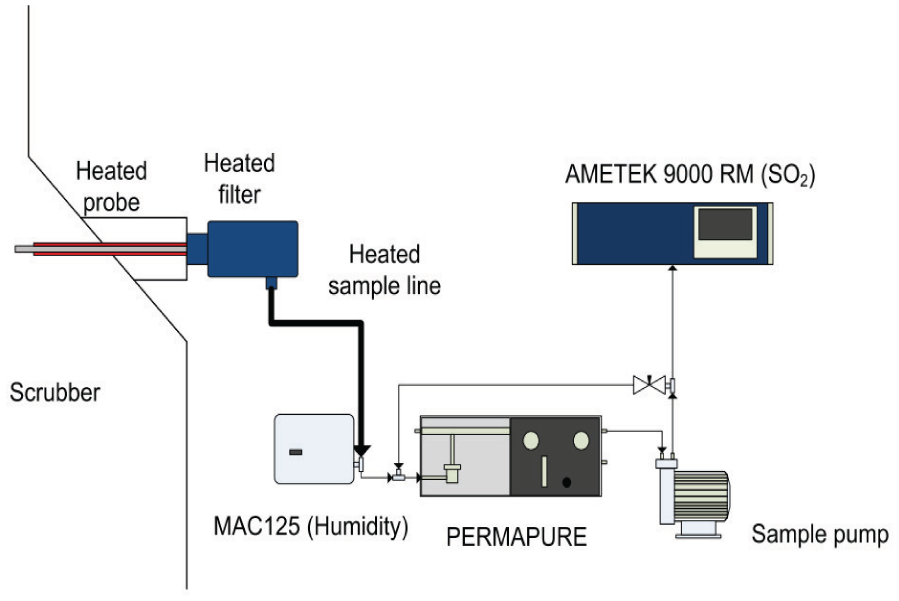
SO2 and exhaust humidity measurement setup in measurement campaigns.
Relevant parameters
The values of several parameters used in the model verification are listed in Table 2. These parameters are estimated based on a reasonable range for each parameter. Notice that the values of these parameters may change by various of scrubber and shipping locations.
Parameter values used in this model.
Table 3 shows the fuel composition used in the Wilh. Wilhelmsen ship. The sulfur mass percent is known, but the hydrogen and carbon mass percent are assumed based on a fixed hydrogen-to-carbon ratio.
Assumed fuel compositions.

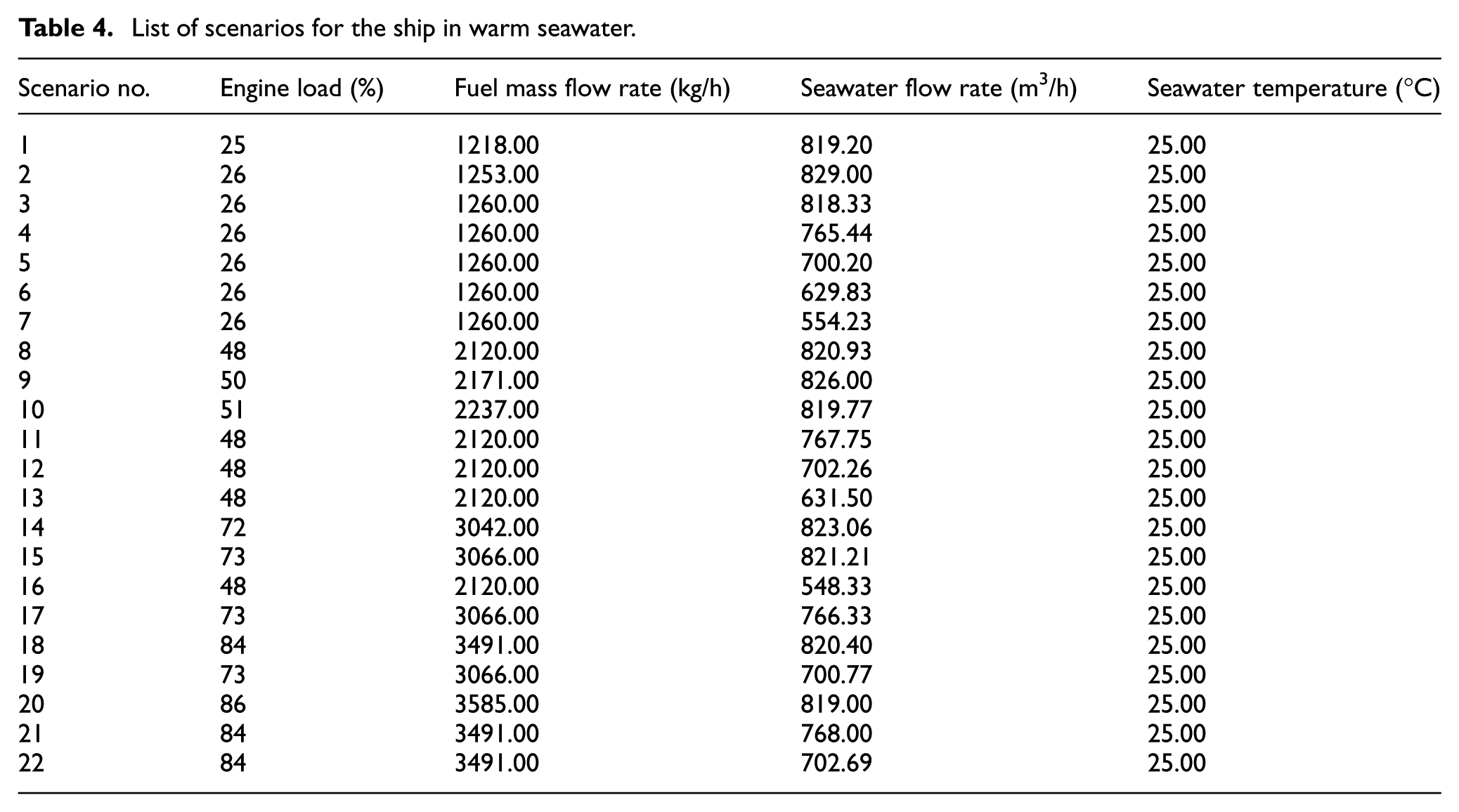
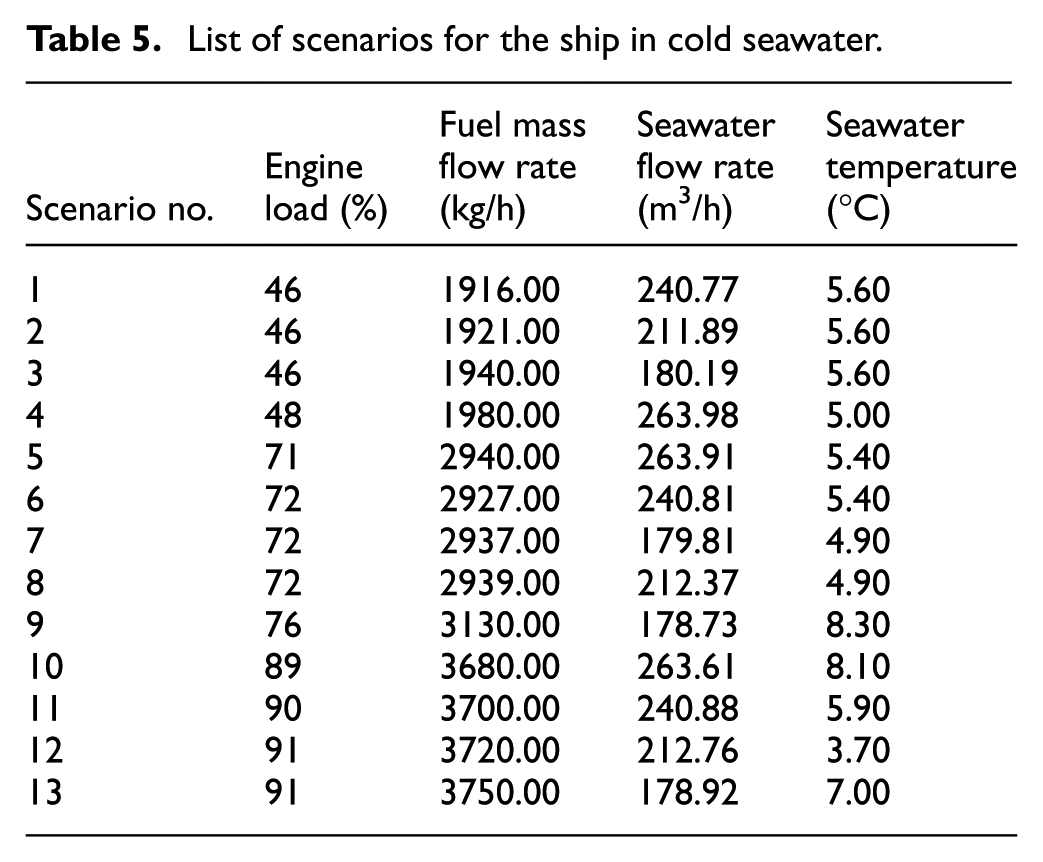
The measurement data for the onboard scrubber were presented in two different scenarios: warm seawater (with temperatures exceeding 297.15 K) and cold seawater (with temperatures below 283.15 K). These measurements were taken in different areas where the engine load, fuel mass flow rate, and seawater flow rate varied accordingly (detailed in Tables 4 and 5).
List of scenarios for the ship in warm seawater.
List of scenarios for the ship in cold seawater.
Compare simulation with measurement data
The mass flow of exhaust gas varies with changes in engine load, which in turn affects the mass flow of SO2 exiting the scrubber. To assess the validity of the scrubber model, simulation results for the mass flow rate of SO2 under different engine loads are examined. In the scenarios presented in Table 4, the engine load ranges from 25% to nearly 85% within the same shipping area. The corresponding exhaust gas mass flow rates are calculated and used as inputs to the scrubber model. Subsequently, the concentration of SO2 in ppm exiting the scrubber is compared with experimental data, as illustrated in Figure 11. From this figure, one notices that the simulation results follow a clear and smooth trend, while the experimental data are quite scattered, mostly due to influences of uncontrollable operational conditions in the real sea environment, and to slight variation in seawater flow rate, where in the simulation, it assumes a constant seawater flow rate. Nevertheless, a best fit curve is made for the scattered experimental data, which is in the same trend as the simulation outlines, only with a slight offset.
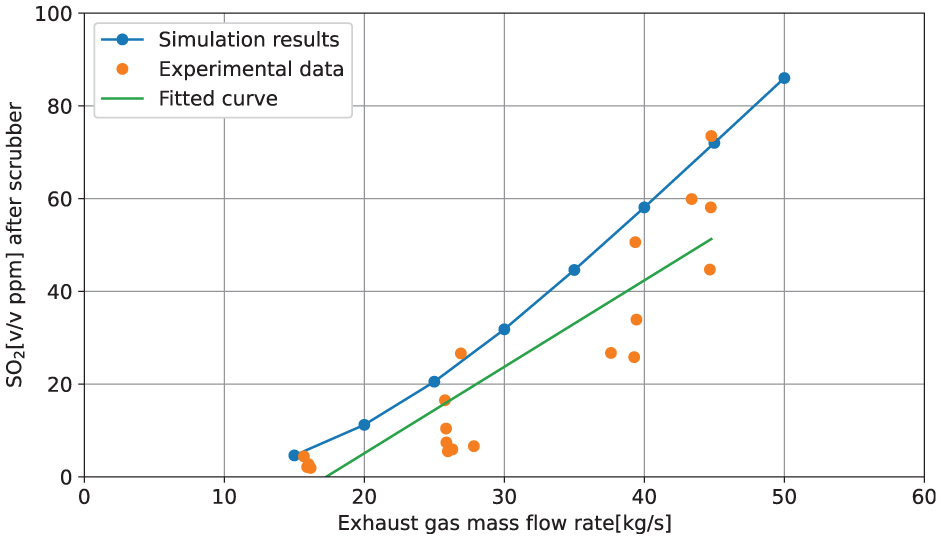
Simulation results of SO2 [v/v ppm] after scrubber versus exhaust gas mass flow rate, plotted together with data from measurement campaign.
The scrubber’s performance in the measurement campaign was evaluated using a specific metric: the concentration of SO2 (in v/v ppm) after passing through the scrubber, relative to the ratio of the mass flow of SO2 in the exhaust gas before the scrubber to the mass flow rate of seawater in the scrubber.
This special metric offers a comprehensive understanding of the scrubber’s effectiveness under different operational scenarios. Figure 12 shows the scrubber performance using the metric. The x-axis presents the ratio of mass flow of SO2 before entering the scrubber to the mass flow rate of seawater entering the scrubber, while the y-axis represents the concentration of SO2 after passing through the scrubber. These measurements correspond to the scenarios presented in Tables 4 and 5.
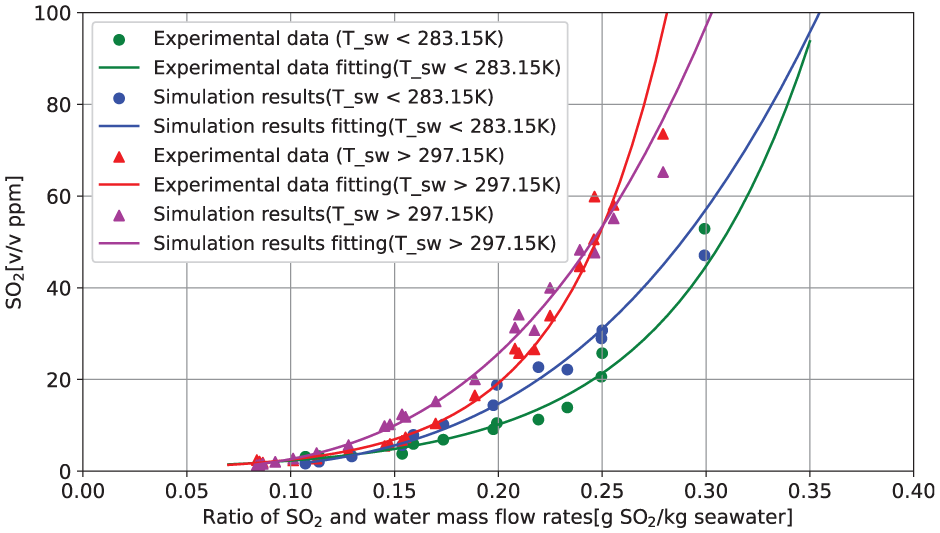
Comparison of simulated and measured SO2 concentrations at scrubber outlet as a function of the ratio of SO2 at scrubber inlet to seawater mass flow rate.
From Figure 12, we see that the scrubber’s performance varies under different seawater temperatures. There is a noticeable trend: as the ratio of the mass flow rate of SO2 to seawater increases, the concentration of SO2 at the scrubber’s outlet also increases, and the results indicate an exponential tendency.
A series of simulations reflecting the full list of scenarios included in Tables 4 and 5 are conducted, where 22 simulations were in the warm seawater category and 13 in cold seawater category. The comparison of the simulation results and the experiment data are shown in Figure 12. We see that the trends of the experimental data are nicely captured by the simulation results. These observations suggest that the scrubber model represents the complex physical and chemical processes in a marine scrubber with high accuracy.
The comparisons between the simulation results and measurement data in Figure 12 are overall promising. Yet, some deviations are still observed. These deviations can be attributed to several factors. Firstly is still simplifications in the mathematical model, which resulted in some relations being linearized, even though certain calibration work has been carried out. As such, it is expected that this model may not handle situations where strong non-linearities are present, which are quite likely to occur among some scenarios considered in this study. Secondly, maintaining a controlled measurement environment similar to that in a laboratory is challenging when measuring data under actual operational conditions on a sailing ship, therefore certain uncertainties are also expected from the measurement side.
Taking these uncertainties into account, we consider the simulation results to be in reasonable agreement with the measurements. We are confident that this validation demonstrates that the marine scrubber model performs well.
Concluding remarks
In conclusion, this study presents an approach to the system modeling of marine scrubbers. Firstly, we have constructed a system model of a representative marine scrubber using the bond graph method. This model carefully incorporates the physics and chemical reactions occurring within the scrubber.
Secondly, we conducted a convergence test on the discretization of the scrubber volume, ensuring the numerical robustness of our model, and carried out model calibration accordingly.
Lastly, we performed a series of simulations for a scrubber installed on a Wilh Wilhelmsen ship. The simulation results were then compared with measurements taken under operational conditions while the ship was sailing in the ocean. The validation showed promising comparisons, indicating that our scrubber model is both robust and reliable.
In light of these results, we believe that our scrubber model serves as a valuable tool for understanding and predicting the performance of marine scrubbers in realistic operations. Despite some deviations from measured data due to inherent complexities and uncertainties in real-world conditions, our model shows good agreement with measurements and captures key trends accurately.
Future work could focus on refining the model by incorporating more complex relations or exploring other influential parameters, as well as on making efforts to integrate the scrubber model with other components in ECGS and eventually achieve a comprehensive ECGS model. The modular framework of marine scrubbers allows seamless integration with existing engine 14 and SCR models, 15 enabling holistic simulations of ship exhaust systems. Furthermore, the model’s adaptability extends beyond SOx removal: particulate matter (PM) reduction, a known scrubber byproduct, 4 can be incorporated by adding particulate transport dynamics. Similarly, carbon capture systems, such as Wärtsilä’s pilot onboard SOLVANG ships, 32 which shares structural parallels with scrubbers, could be modeled by adjusting chemical reactions. Thus, this scrubber model can also help develop future systems that can control multiple pollutants, supporting maritime decarbonization.
Footnotes
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was carried out at SFI Smart Maritime, which is mainly supported by the Research Council of Norway through the Centres for Research-based Innovation (SFI) Funding scheme, project number 237917.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
