Abstract
Findings on female sexual motivation across the ovulatory cycle are mixed. Some studies have reported increased female sexual desire on fertile days or midway through the ovulatory cycle, whereas others have reported increased sexual desire on nonfertile days. We postulated and tested the hypothesis that the pattern of the cyclical change of female sexual desire is associated with women's life history. Female participants completed life-history measures and rated their levels of sexual desire on the survey day and reported the first day of their current and subsequent cycle, respectively (Study 1), or recorded their sexual desire throughout an entire cycle by submitting daily reports (Study 2). Results indicate that women with a fast life history experienced peak sexual desire midcycle, whereas women with a slow life history experienced two peaks of sexual desire midcycle and around their menses. These findings suggest that, consistent with the underlying life history, cyclically differential peaking of sexual desire may serve different reproductive functions.
In contrast to the female sexuality of most mammals, which is confined to the temporal state of estrus, human women have concealed ovulation and are sexually receptive throughout the ovulatory cycle (Thornhill & Gangestad, 2008). Studies on cyclical changes in sexual behaviors have reported inconsistent results, with findings indicating that sexual activities throughout the ovulatory cycle vary from being flat to peaking immediately after menstruation, midcycle (Crowley, 2020), or in the luteal phase (Grebe et al., 2013). However, whether women experience genuine sexual desire that motivates them to continually have sex throughout the cycle remains unclear. The ovulatory shift hypothesis posits that sexual interest peaks during ovulation (Gangestad et al., 2005), suggesting that increased sexual behavior midcycle is motivated by genuine physiological and psychological needs. By contrast, the extended sexuality hypothesis asserts that women remain sexually active during the nonfertile phase of the ovulatory cycle as a strategic measure to retain their long-term partners (Grebe et al., 2013). However, few studies have examined female sexual desire on nonfertile days. We speculated that sexual desire motivates sexual behaviors, which in turn become a component of adaptive reproductive strategies that enhance the reproductive success (RS) of women. Because fast–slow life history strategies focus on the energetic trade-offs among various life-history traits (including the trade-offs between present-oriented mating and future-oriented parenting) and emphasize the optimization of RS, we adopted a life-history framework to integrate the ovulatory shift and the extended sexuality hypotheses. Relative to the ovulatory shift hypothesis, which focuses on immediate mating outcomes, the extended sexuality hypothesis emphasizes future parental and spousal investment. The objective of the present study was to examine spontaneous female sexual desire throughout the ovulatory cycle and how life history influences the cyclical changes of sexual desire.
Reproductive Strategies in Relation to Fast–Slow Life History
Life history strategies along the fast–slow continuum are highly correlated with mating and sexual behaviors (Lu, 2021). Organisms with fast life-history strategies sexually mature and reproduce early, mate frequently, and produce a relatively large number of offspring who survive by relying on a concealed but favorable genetic endowment over committed parental investment. By contrast, organisms with slow life-history strategies exhibit slow development patterns, late sexual maturation, less sexual activity, and a greater investment in parenting over mating (Del Giudice et al., 2015; Lu, 2021). Life history strategies are shaped by the levels of environmental predictability and resource scarcity (Chang & Lu, 2021). When resources are plentiful, organisms adopt fast life-history strategies and use available resources to mature early and produce a large number of offspring that can survive and thrive in a resource-abundant environment with limited parental investment (MacArthur & Wilson, 1967; Pianka, 1970). When the high reproductive rates of organisms increase their population density to the point of resource depletion, they adopt slow life-history strategies by producing fewer offspring and teaching them the skills required to compete for limited resources. Similarly, environmental unpredictability due to frequent and myriad extrinsic risks (e.g., predation, war, and disease) compel organisms to adopt fast life-history strategies to accelerate their development and reproduction before these risks affect their mortality and morbidity (Promislow & Harvey, 1990). When extrinsic risk is high, investment in parenting is a maladaptive strategy because its benefits are nullified by high mortality (Del Giudice & Belsky, 2011). By contrast, when the extrinsic risks of mortality and morbidity are low, organisms have a predictable future that enables them to realize the benefits of parental investment in offspring. Thus, the determination whether to adopt a mating-oriented or parenting-oriented reproductive strategy by an organism is an adaptive response to how its environment promotes fast–slow life history.
The life history and corresponding reproductive strategies that women adopt should optimize their survival and RS. To enhance their RS, women who adopt a mating-oriented strategy should focus on securing mates with good genes that benefit their RS at insemination. The attributes of good-gene mates include physical and behavioral features such as masculinity, symmetry, physical attractiveness, robust health conditions, creativity, and a sense of humor. These attributes suggest favorable genetics that maximize the quality of offspring when parenting investment is limited (Lu et al., 2015). Having a good-gene mate increases the likelihood of conceiving offspring with high genetic quality in terms of strong immune functioning and a low probability of harmful mutations (Gildersleeve et al., 2014). By contrast, the parenting-oriented strategy focuses on retaining mates who can provide resources that ensure a favorable quality of life, direct paternal care, or both to improve offspring quality and competitiveness (Chang et al., 2017; Trivers, 1972). However, male provisioning and paternal care occur only through postmating events that are long term in nature. Thus, the time frames for realizing the benefits of mating- and parenting-oriented reproductive strategies correspond to the fast and slow LH, respectively, which in turn are present- and future-oriented, respectively (Del Giudice & Belsky, 2011; Figueredo et al., 2006).
Ovulatory Shift and Extended Sexuality in Ovulatory Cycle
In contrast to men whose every ejaculation during intercourse has the potential to result in pregnancy, conception for women is highly dependent on the ovulatory cycle because they are fertile only during the short period of time before and after ovulation. The ovulatory shift hypothesis posits that ovulating women experience increased sexual desire, which manifests as physiological, cognitive, and behavioral responses (Gangestad et al., 2005). This phenomenon occurs because increased sexual behavior during the fertile window, which resembles the estrus of female primates, enhances RS. Compared with women in the low-fertility phase, those in the fertile window are more likely to experience sexual desire (Haselton & Gangestad, 2006) and accept invitations to dance in nightclubs (Guéguen, 2009). The ovulatory shift aligns with the reproductive strategy of women, which emphasizes mating while fertile and mating with men who possess good-gene attributes that increase the likely fitness of offspring right from the time of insemination. Therefore, women with a fast life history are theoretically more likely to exhibit an ovulatory shift. By contrast, the ovulatory shift should be weaker in women who have a slow life history and are unlikely to be highly concerned about immediate sex (Gildersleeve et al., 2014).
In contrast to the ovulatory shift hypothesis, the extended sexuality hypothesis asserts that women exhibit increased sexual behavior on both fertile and nonfertile days because sex during the nonfertile period enables women to obtain nongenetic benefits such as financial or parental investment from men (Grebe et al., 2013; Rodríguez-Gironés & Enquist, 2001). Nonhuman mammals have explicit estrus, whereas women benefit from concealed ovulation that confuses men in terms of paternity. Because of their imperfect knowledge of female ovulation timing and fertility status, men are likely to maintain sexual relationships with their long-term partners even on nonfertile days to increase the likelihood of reproduction and minimize the potential of their female partners engaging in extrapair copulation (Grebe et al., 2013). Women may cooperate with their long-term partners by agreeing to and even initiating nonfertile copulation, which serves to confuse paternity and to elicit paternal care, protection, and investment from their long-term partners (Thornhill & Gangestad, 2008). Therefore, extended sexuality aligns with reproductive strategies that emphasize parenting, and slow life-history women, who prioritize long-term relationships and parenting over mating, should exhibit more extended sexuality relative to fast life-history women.
Existing studies have focused on the relationship type (intra- vs. extra-pair) and male mate attributes (good genes vs. good provisioning) that evoked increased midcycle sexual desire (Gangestad & Haselton, 2015). These studies have yielded mixed results (Jones et al., 2019; Welling & Burriss, 2019). For relationship type, early studies report an increased midcycle extra-pair sexual desire but only in women self-reporting weak pair-bond with (Eastwick & Finkel, 2012) or low sexual attraction toward their primary partner (Haselton & Gangestad, 2006; Pillsworth & Haselton, 2006). Recent studies report a midcycle increase of sexual desire for both intra-pair and extra-pair partners (Arslan et al., 2021; Roney & Simmons, 2013), and do not reveal any effect of primary partner's sexual attraction or relationship quality on extra-pair sexual desire (Arslan et al., 2021; Shimoda et al., 2018). For mate attributes, meta-analysis reviews either confirm (Gildersleeve et al., 2014) or refute (Wood et al., 2014) an ovulatory shift of preference for good-gene attributes, whereas primary investigations report similarly inconsistent findings (e.g., shift to masculinity [Gangestad et al., 2019] vs. shift to male bodies in general [Jünger et al., 2018; Stern et al., 2021]). Fast and slow life history strategies are each associated with different mating characteristics and preferences (Chisholm et al., 1993; Figueredo et al., 2006). Specifically, a fast strategy is associated with a mate preference for good genes and the related mating behavior aimed at securing instantaneous endowment of high-quality genes. A slow strategy prioritizes parenting over mating to achieve RS by nurturing competitive offspring with high parental efforts that include paternal care and investment. By investigating across-cycle sexual desire between fast and slow life-history strategists, the present study should shed light on the aforementioned controversies both in terms of findings and interpretations.
Studies on extended sexuality have focused on the receptivity and proceptivity of women with respect to sexual behaviors on nonfertile days as strategic actions for retaining a long-term partner and soliciting paternal and spousal investment (Grebe et al., 2013). However, studies examining sexuality during the nonfertile phase have mostly overlooked sexual arousal and fantasies, which are the fundamental drivers of sexual behavior and are generally included in the construct of sexual desire (Spector et al., 1996). Female sexual desire varies among individuals (Clark, 2006), but the life-history framework has yet to be used to conceptualize individual differences to provide a comprehensive explanation of female sexuality.
Arms Race Between Two Sexes in Concealment and Detection of Ovulation
Extended sexuality plays a key role in female RS. Among nonhuman primates, female primates promiscuously mate with resident males, and the evolutionarily adaptive function of extended sexuality is to confuse paternity of all copulation targets, which enables female primates to protect their offspring from attacks and harm instigated by surrounding male primates (Hrdy, 1981). In ancestral and contemporary human society in which monogamy is a common practice, the evolutionary function of extended sexuality that benefits offspring is maintained, but its focus has shifted from the protection of offspring to the material and emotional support of offspring (Gangestad & Thornhill, 2008).
In species in which parental investment is essential for the survival of offspring, the conflict between the two sexes in terms of investment in offspring is always present even at the genomic level (Badcock & Crespi, 2008; Haig & Trivers, 1995). Each sex tends to invest less and let the other sex invest more in their offspring. In human evolutionary history during which pair-bonding or monogamy prevailed, the conflict between sexes is more evident at the behavioral level. In the case of women, they attempt to assure paternity and maximize paternal investment through extended sexuality and by preventing their male partners from mating with other women and investing in other women's offspring (Wakano & Ihara, 2005). In the case of men, they attempt to maximize maternal investment in their offspring and minimize their own paternal investment because of paternal uncertainty (Chang et al., 2010) and a drive to mate with other fertile women as much as possible. The fundamental male–female conflict in parental investment causes both sexes to experience evolutionary pressure. Women conceal their ovulation by expressing signs of estrus and maintaining sexual receptivity and proceptivity throughout their ovulatory cycle, whereas men look for signs of estrus and attempt to prevent extrapair copulation during the fertile days of women (Gangestad & Thornhill, 2008; Welling & Puts, 2014).
The arms race between women concealing ovulation and men detecting ovulation leads to subtle forms of deception from women and a high sensitivity to ovulation signals in men. An ideal form of deception is self-deception, in which the deceiver is also unaware of the truth while deceiving (Lu & Chang, 2011, 2014). Because of the possible discovery by men that women involuntarily exhibit high sexual interest and desire during ovulation, women attempt to conceal their ovulation by also exhibiting sexual interest on nonfertile days. Women may accept sexual invitations and even initiate sex on days that are outside their fertile window (Grebe et al., 2013). However, feigned sexual pleasure without a genuine desire for sex may leak signals for men to detect feigned sexual interest and, accordingly, detect nonovulation. Therefore, to thoroughly confuse men about the timing of ovulation, women should increase their self-deceptive behavior to experience genuine sexual arousal and intention when they initiate sex with their primary partners during their nonfertile phase.
Present Study
Because women with a fast life history focus on the present and tend to achieve an early RS through immediate insemination, they should theoretically exhibit a clear ovulatory shift on fertile days. By contrast, slow life-history women should theoretically focus on offspring development by strategically using extended sexuality to retain paternal investment. The literature has reported that women experience increased sexual desire on fertile days (e.g., Pillsworth et al., 2004) and engage more frequently in sexual behavior with their primary partners on nonfertile days (e.g., Grebe et al., 2013). However, the research on the sexual desire that motivates sexual behaviors on nonfertile days is limited. To assure men of their paternity in exchange for relationship investment and parental investment, genuine experiences of sexual desire that drive sexual behavior is a more adaptive strategy than copulation without genuine sexual interest. Therefore, on nonfertile days, women with a slow life history are theoretically more likely to experience sexual desire than are those with a fast life history. In two studies, female participants were subjected to life-history measures, and they reported their sexual desire levels on the survey day and also reported the first day of their current and subsequent cycle, respectively (Study 1), or recorded their sexual desire throughout an ovulatory cycle by submitting daily online reports (Study 2). We hypothesized that fast life-history women would undergo a clear ovulatory shift of sexual desire midcycle when their conception risk is high and that slow life-history women would exhibit a high sexual desire on nonfertile days.
Study 1
Participants
In total, 753 heterosexual women (mean age [Mage] = 31.41, standard deviation [SD] = 7.02) were recruited from communities in Hong Kong. The sample size exceeded the minimum required sample size of 110, which was determined through a power analysis in which 95% power was applied to identify a small-to-medium effect size at the 0.05 probability level for multiple regression with seven predictors. The sample size also exceeded the minimum requirement of 456 for achieving an 80% power to detect a medium effect size with a generally high validity of 0.7 adopted by studies of ovulatory cycle that use the backward-counting method with a known accurate onset of the next ovulatory cycle (Gangestad et al., 2016). Women with irregular menstrual cycles and those using hormonal contraceptives were excluded from the present study. Among women who participated in the present study, the duration of their cycles ranged from 18 to 49 days (Mean = 31.40, SD = 4.50). Because the duration of a normal cycle is 21–35 days (applicable for >85% of women in the Hong Kong population; Chan et al., 2009) and 105 participants (13.94%) had a cycle duration of 36–40 days, we excluded the participants with a cycle < 21 days (four women, 0.53%) or > 40 days (more than two standard deviations from the mean, 22 women, 2.92%). The final sample size was 727 with a mean cycle duration of 31.07 days (SD = 3.93). Among the participating women, 69.46% were unmarried, 27.79% were married, and the remaining were either divorced (2.34%) or widowed (0.41%). If a participating woman was married or unmarried but had a long-term partner, she was regarded as being in a long-term relationship (59.28%).
Methods
Life History. The 20-item Mini-K scale was adopted to measure the behavioral and cognitive aspects of life-history strategies (Figueredo et al., 2006). The scale is scored on a single continuum in the direction of a slow life history. The sample items of this scale include “When I was growing up, I had a close and warm relationship with my biological mother” and “I do not give up until I solve my problems.” The items were rated on a 7-point scale ranging from − 3 (strongly disagree) to 3 (strongly agree), and a higher score indicates a slower life history. For the present study, the internal consistency of this scale was 0.85. In addition, the 18-item fast–slow scale (FSS, Lu et al., 2017) was adopted to measure behavioral and cognitive manifestations of LH during childhood. Sample items of this scale include “When I was growing up, I was the type of person who focused on the present instead of the future” and “When I was growing up, I was the type of person who was physically more mature than my peers.” The items were rated on a 6-point scale ranging from 1 (strongly disagree) to 6 (strongly agree), and the scores were reverse coded with a higher score indicating a slower life history. For the present study, the internal consistency of this scale was 0.80. The Mini-K and FSS scores were revealed to be highly correlated (r = .37, p <.00001). An aggregate life-history indicator was established by averaging the standardized scores of the two scales.
Sexual Desire. The participants reported their levels of general sexual desire in the preceding 24 h relative to other days (Haselton & Gangestad, 2006). They answered questions in the format of “Relative to other days, in the preceding 24 hours, to what degree did you … ,” with the items, “have sexual thoughts,” “have sexual fantasies,” “have sexual desire,” “have sexual intention,” and “have sexual arousal” ending the questions. The items were rated on a 9-point scale ranging from 1 (far less than usual) to 9 (far more than usual), and a higher score indicated a greater level of sexual desire. For the present study, the internal consistency reliability of this scale was 0.98.
Ovulatory Cycle. Each participant reported on the first day of her current menstrual cycle, completed a questionnaire that comprised the aforementioned scales, and subsequently reported the first day of her next cycle. For each participant, we calculated her actual cycle duration and recorded the date on which she completed her questionnaire. We then considered her actual cycle duration (sample Mean = 31.07 days, SD = 3.93) and mapped her to a 31-day cycle to estimate her day in her cycle. Because the duration between a normal ovulation day and the next menses is typically 14 days and this duration is stable regardless of cycle duration (Lenton et al., 1984; Reed & Carr, 2018), we applied the estimation method used in other studies (Gangestad et al., 2004; Gangestad & Thornhill, 1998). Specifically, if a participant was in the last 14 days of her cycle when she completed the questionnaire, she was mapped to the last 14 days of her 31-day cycle. Otherwise, she was proportionally mapped to the first 17 days of her cycle. After estimating which day of her cycle a participant was on, we applied the actuarial table of conception risk to estimate the probability of conception for each participant (Baker & Bellis, 1995; Jöchle, 1973).
Results and Discussion
The relationship status of a participant was coded as 1 for a stable relationship and 0 for the absence of a stable relationship. Zero-order correlations among relationship type, life history, conception risk, and sexual desire are presented in Table 1. Relative to other participants, those with a slow life history were more likely to be currently engaged in a stable relationship (r = .08, p = .03) and to have lower sexual desire (r = − .07, p = .04). Relative to the participants who were not in a stable relationship, those who were in a stable relationship experienced less sexual desire (r = − .09, p = .02). All participants exhibited a positive correlation between conception risk and sexual desire (r = .12, p = .002).
Correlations Among Variables in Study 1.

*p <.05, **p <.01.
We performed multiple regressions to examine the main effects of relationship type, life history, conception risk, and the interactions among these variables on sexual desire. Our results revealed the main effect of relationship type (β = –.08, p = .04) and conception risk (β = .11, p = .004). We did not detect a three-way interaction (β = .02, p = .51) or a two-way interaction between relationship type and conception risk (β = .04, p = .27) or between relationship type and LH (β = − .03, p = .48). However, we detected a two-way interaction between life history and conception risk (β = − .10, p = .005). A simple slope analysis revealed the absence of any relationship between conception risk and sexual desire (β = .02, p = .68) at + 1SD of the aggregated life history scale (slow life history) and a positive association between conception risk and sexual desire (β = .22, p <.001) at −1SD of the scale (fast life history). Figure 1 presents the distribution of daily sexual desire as a function of conception risk for fast and slow life-history women identified by a median split on the aggregated life-history scores.
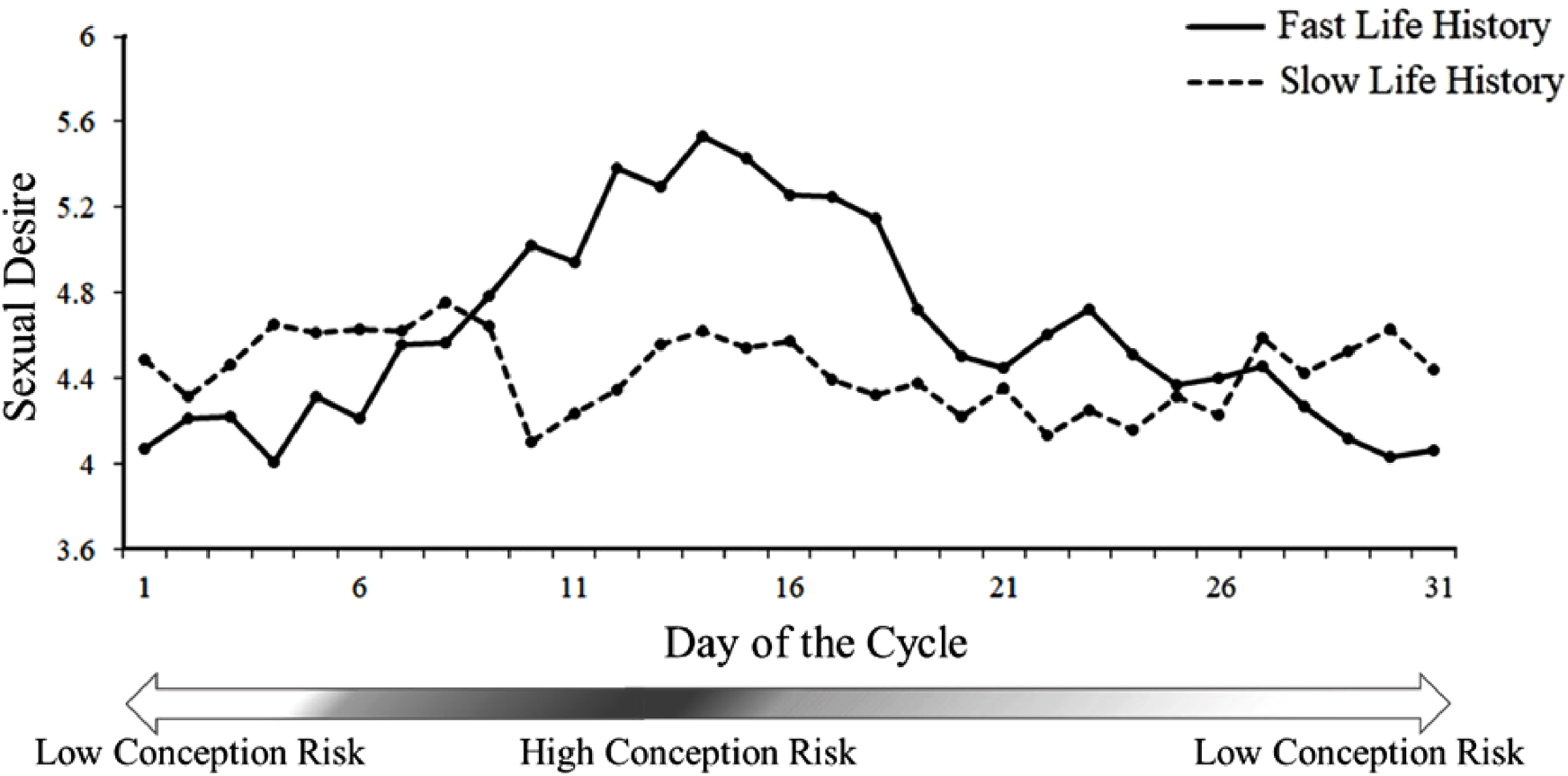
Sexual desire as a function of the day of the cycle in Study 1. Points are 3-day moving averages.
The above-mentioned life history was based on an aggregate of the mini-K scale and the FSS scale. We further examined the two scales as separate indicators of life history. Results were similar to those from the aggregates. While using the mini-K, we detected a marginal interaction between life history and conception risk (β = − .07, p = .07). A simple slope analysis revealed that conception risk was unassociated and positively associated with sexual desire for participants with slow life history (β = .06, p = .23) and fast life history (β = .17, p = .001), respectively. While using the FSS, we detected a significant interaction between life history and conception risk (β = − .09, p = .01). The simple slope analysis showed similar patterns as those of the mini-K. Specifically, for participants with slow life history, conception risk was unassociated with sexual desire (β = .05, p = .37), whereas for participants with fast life history, conception risk was positively associated with sexual desire (β = .19, p <.001) in the ovulatory cycle.
Our results suggest that fast life-history women experienced a single peak of sexual desire midcycle, which is the peri-ovulatory period of high conception risk, whereas slow life-history women experienced the same peri-ovulatory peak midcycle as well as a second surge consisting of two equal-magnitude rises immediately before and after menstruation. Because sexual desire represents an individual's physiological and psychological needs for sexual activity, the findings of the present study suggest that at least for the slow life-history group, sexual behaviors initiated by women in the nonfertile phase, as depicted in the extended sexuality hypothesis, are motivated by genuine sexual interest. The present study relied on the participants’ retrospective report of the first day of their current cycle, and each participant submitted a one-off report on their sexual desire. Memory bias and individual differences in rating standards may reduce the precision of findings. In addition, how sexual desire changes across a cycle remains unclear. The objective of Study 2 was to replicate the results of Study 1 by using participants’ daily reports of their sexual desire across a full ovulatory cycle.
Study 2
Participants and Methods
In total, 130 participants (Mage = 30.91, SD = 6.13) with a cycle duration of 21–35 days were recruited from Study 1. Half of these participants were recruited from the fast and the slow life-history groups, respectively, which were identified based on a median split of the aggregated LH score in Study 1. The sample size was larger than the minimally required sample size of 110, derived from a power analysis with 95% power, .05 probability, and a small-to-medium effect size for multiple regression involving three predictors. The sample size also reached required group size of 55–71 for ovulatory studies with multiple within-subject measures (Jones et al., 2019). The participants signed consent forms and were briefed about their tasks in the study. Starting from the first day of her subsequent cycle, each participant was asked to rate her sexual desire every day for the duration of the cycle. From the first day of reporting, each participant was reminded every evening through an email or text message to complete the daily questionnaire before going to sleep. The questionnaire was submitted online. The participants rated their level of sexual desire for a given day by rating items that were identical to those used in Study 1. After their menses, the participants self-assessed their midcycle ovulation everyday by using an ovulation predictor kit, which is frequently used to detect luteinizing hormone surges. The test kit is the DAVIDR one-step urine ovulation test produced by Runbio Biotech, China (http://www.runbio-bio.com). The test kit of this brand is sensitive (at 10mIU/ml) and has been adopted in previous studies (e.g., Lobmaier et al., 2018; Probst et al., 2017). The test's threshold of luteinizing hormone is 25mIU/ml, which can present the best prediction of ovulation within 24 h (Leiva et al., 2017). A luteinizing hormone surge indicates ovulation, which typically occurs within 24–48 h after the start of a surge (Cahill et al., 1998; Dunson et al., 2001; Kerin, 1982). Following the test kit instruction, the participants self-tested each day between 10:00 and 20:00 and reduced liquid intake 2 h prior to the test. During testing, participants were instructed to compare the color of two lines, the test line and control line, and to record the day as having a positive result (i.e., surge of luteinizing hormone) when the test line became as red as or redder than the control line. Following the positive result, participants were asked to continue to perform the test twice a day for three days to confirm the previous positive reporting. The participants then continued to perform the test daily until negative results were observed for three consecutive days. All participants in this study observed a luteinizing hormone surge in their cycles. The participants received monetary compensation for the time they spent participating in the present study. Ethical approval for the present study was obtained from the institutional review board of the author's university.
Results and Discussion
The average cycle duration of the participants was 29.45 days (SD = 3.15). On average, their luteinizing hormone surge occurred 15.64 days (SD = 1.91) before their next menses. This finding is consistent with the general consensus that ovulation typically occurs 14 days prior to the next menses. On the basis of the participants’ sexual desire data (daily data) and conception risk data (actuarial data) for each day of their cycle, we conducted hierarchical linear modeling or multilevel analysis (Raudenbush & Bryk, 2002) using M-plus (Muthén & Muthén, 2012) to examine the interaction between life history and conception risk in relation to sexual desire. For these analyses, conception risk and sexual desire were within-subject variables and life history was a between-subject variable. The intraclass correlation coefficient (ICC) was 0.56, indicating that between-subject differences accounted for 56% of the variance of sexual desire. In general, when ICC is 0.25 or above, a multilevel analysis should be considered (Guo, 2005; Heinrich & Lynn Jr, 2001). In the initial regression without the between-subject predictor, conception risk was positively predictive of sexual desire (β = .70, standard error [s.e.] = .047, p <.001). The regression of sexual desire on conception risk differed significantly across individual women (τ = .085, s.e. = .017, p <.001), warranting multilevel analysis to account for the between-subject variation. We included life history as a between-subject predictor of sexual desire in a multilevel regression. The results revealed a significant predictive effect of life history (β = −.41, s.e. = .172, p = .02). The between-subject predictor explained 9.41% of the variance of the within-subject slope for regressing sexual desire on conception risk. Specifically, in the participants with fast life history, the overall within-subject regression of sexual desire on conception risk was positive and significant (β = .51, s.e. = .238, p = .03), whereas for participants with slow life history, conception risk was not predictive of sexual desire (β = −.12, s.e. = .343, p = .72). Figure 2 illustrates daily sexual desire movement as a function of conception risk for fast and slow life-history women identified by a median split on the life-history scores.

Sexual desire as a function of the day of the cycle in Study 2. Points are 3-day moving averages.
Patterns of the sexual desire distribution derived from Study 2 were, in general, highly similar to those detected in Study 1. Specifically, fast life-history women exhibited a single peak of sexual desire midcycle, the peri-ovulatory peak, whereas slow life-history women exhibited the peri-ovulatory peak as well as a second heightening of sexual desire around (i.e., immediately before and immediately after) menstruation. In Study 2, the second heightening around menses of slow life-history women was lower than their peri-ovulatory peak midcycle. By contrast, the peri-menses heightening and the peri-ovulation peak, both associated with slow life history, were of similar magnitude in Study 1. For both Study 1 and 2, the overall levels of sexual desire across the full cycle were lower for slow compared to fast life-history women. Regardless of the overall levels of sexual desire displayed by fast and slow life-history women, the heightening of sexual desire associated with slow life history was consistently observed in both studies to appear twice, once around ovulation and a second time around menstruation.
General Discussion
In Studies 1 and 2, we examined how the pattern of the cyclical change of female sexual desire was associated with women's life history. Each participating woman reported her level of sexual desire on the survey day and the first day of their current and subsequent cycle, respectively (Study 1), or recorded her sexual desire throughout an entire cycle by submitting daily reports (Study 2). Both studies revealed that the participants with a fast life history experienced a high level of sexual desire midcycle, whereas those with a slow life history experienced a high level of sexual desire both midcycle and around menstruation. The results support our hypothesis that, following the parenting-oriented reproductive strategy, slow life-history women experience genuine sexual desire that drives their sexual behaviors in the nonfertile phase more closely related to the function of parenting rather than mating. By contrast, the mating-oriented fast life-history women experience a single peak of sexual desire of high magnitude midcycle when their conception probability is high.
The aforementioned results reveal that sexual desire, which indicates psychological engagement in sex and genital sexual arousal (Spector et al., 1996), changes across the ovulatory cycle as a function of life history strategies. The essence of life history is a reproductive strategy varying by the fast–slow speed of reproduction (Ellis et al., 2009). Fast reproduction involves sexually maturing and debuting early and mating frequently, immediately, and presently. RS is achieved by prioritizing mating and offspring quantity over parenting and offspring quality. The mating-oriented fast reproductive strategy attends to offspring quality primarily by prezygotic effort in selecting genetically well-endowed partners and mating with extrapair partners to ensure genetic quality. The prezygotic efforts of the fast life history strategy yield immediate benefits commencing at insemination. Therefore, fast life-history women, who pursue immediate reproductive benefits, should experience heightened sexual desire and motivation to mate only during the period when the chance of insemination is high. By contrast, the parenting-oriented slow reproductive strategy shifts from more instantaneous prezygotic effort, including heightened motivation and spontaneity, to postzygotic longer-term effort and investment aimed at raising the competitiveness of a small number of young. Therefore, slow life-history women, who pursue reproductive benefits through long-term investment, should have higher motivation for partner retention by experiencing sexual interest during both fertile and nonfertile phase of the menstrual cycle to secure continuous parental and spousal investment. As an integral part of reproductive or life history strategies, sexual desire and its timing that proceed reproductive activities should coordinate with the fast-slow strategic prioritizations and schedules to maximize RS of different life histories.
The findings of the present study can help explain the inconsistencies in the cyclical sexuality literature (for a review, see Crowley 2020; Motta-Mena & Puts, 2017; Regan, 1996). First, for the luteal phase between ovulation and the next menses, several studies have reported decreased sexual desire due to an increase in progesterone levels (Jones et al., 2018; Roney & Simmons, 2013), whereas others have reported similar levels of sexual desire across all time periods with no correlation with progesterone level (Grebe et al., 2016). Our finding of individual differences in life history may explain the inconsistencies in the aforementioned literature. Specifically, fast life-history women experienced a low level of sexual desire, whereas slow life-history women experienced a high level of sexual desire in the luteal phase. Second, during the fertile window of the menstrual cycle, although increased sexual desire is widely observed (Bullivant et al., 2004; Pillsworth et al., 2004), decreased sexual behavior is also reported and explained as general attempts by women to avoid conception (Burley, 1979). The ovulatory shift interpretation of increased sexual desire and sexual activities also cause controversies (Gildersleeve et al., 2014; Havlíček et al., 2015; Jones et al., 2019; Welling & Burriss, 2019; Wood et al., 2014). Supportive of the ovulatory shift hypothesis, most of the findings are associated with female preference for good-genes mate values and extra pair partners (e.g., Gangestad et al., 2019; Little & Jones, 2012). A large number of studies refute the ovulatory shift of mate preference (e.g., Arslan et al., 2021; Jünger et al., 2018; Marcinkowska et al., 2016; Shimoda et al., 2018; Stern et al., 2019, 2021). The counter explanation refers to an undifferentiated surge of sexual interest upregulated by additive hormonal fluctuations (e.g., Arslan et al., 2021; Roney & Simmons, 2013). The present study lends support to both explanations. The finding of differential heightening of sexual desire associated with fast (single peri-ovulatory surge) and slow life history (a second heightening around menses) matches with the ovulatory shift of mate preference from good genes for ovulatory sexuality during the fertile phase to paternal and spousal investment during the nonfertile extended sexuality (Gangestad et al., in press; Gangestad & Dinh, 2022). The finding of a peri-ovulatory surge of sexual desire being experienced by both life history strategists, yet with fast strategists reporting higher intensity than slow strategists, supports the undifferentiated sexuality theory (Roney & Simmons, 2013) in explaining additional variance beyond the ovulatory shift explanation.
This study has several limitations. First, the luteinizing hormone test alone might not accurately identify the fertile window because false positive or negative may occur (Marcinkowska, 2020) and the fertile window may have been passed if participants came to the laboratory to complete tasks after detection of a positive result (Lobmaier & Bachofner, 2018). However, in Study 2, we adopted a continuous measure throughout the ovulatory cycle and did not apply discrete laboratory session. Moreover, we verified the luteinizing hormone test results with the backward-counting method basing on a known onset of the next cycle. Consistent with the general understanding of using the test of luteinizing hormone surge that ovulation occurs within 24–48 h after the start of the surge, the reported surge day in Study 2 was average 15.64 days prior to the next cycle or 1.64 days prior to a typical ovulation day which is 14 days prior to the next cycle. Therefore, the luteinizing hormone test in the present study was assumed to be reliable. Future studies can apply multiple methods including observation of cervical mucus quality to determine ovulation (Lobmaier & Bachofner, 2018). Second, sexual desire is associated with other life-history-related traits, such as a sociosexual orientation (Timmers & Chivers, 2012) and satisfaction with a pair-bonding relationship (Hurlbert & Apt, 1994). Sociosexual orientation was also examined in the studies of ovulatory cycles, although inconsistent results were reported, with its cyclical change being confirmed (Marcinkowska et al., 2021; Shirazi et al., 2019) or not confirmed (van Stein et al. 2019). Future studies can include these variables to examine their moderating effects on the relationship between sexual desire and the ovulatory cycle and examine sexual desire and sociosexual orientation together as functions of ovulatory cycle and life history. Finally, among different criticisms of the psychometric approach to study human life history (e.g., Zietsch & Sidari, 2020), the mini-K, which we used to measure slow life history, has been criticized for potentially conflating causes with outcomes concerning some of its items (Gruijters & Fleuren, 2018). Whereas our study bears the same blames, we made an effort to supplement the mini-K with another life history measurement, FSS, which has been designed to be reflective indicators of life history as the underlying construct and is thus better fitted for the psychometric and statistical analyses we applied on the aggregates of the two measures. Despite these and other limitations, the present study is among the first to employ life-history theory as a framework within which to investigate women's cyclical sexuality and provide additional insights to help integrate some of the existing explanations of women's sexual desire throughout the ovulatory cycle.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Grants Council of Hong Kong (grant number Project Number: 15602518).
