Abstract
One of the most fascinating problems in comparative psychology is how learning contributes to solving specific functional problems in animal life, and which forms of learning our species shares with non-human animals. Simulating a natural situation of territorial conflicts between predatory carabids and red wood ants in field and laboratory experiments, we have revealed a relatively simple and quite natural form of learning that has been overlooked. We call it catalog learning, the name we give to the ability of animals to establish associations between stimuli and coherent behavioral patterns (patterns consist of elementary motor acts that have a fixed order). Instead of budgeting their motor acts gradually, from chaotic to rational sequences in order to learn something new, which is characteristic for a conditioning response, animals seem to be “cataloguing” their repertoire of innate coherent behavioral patterns in order to optimize their response to a certain repetitive event. This form of learning can be described as “stimulus-pattern” learning. In our experiments four “wild” carabid species, whose cognitive abilities have never been studied before, modified their behavior in a rather natural manner in order to avoid damage from aggressive ants. Beetles learned to select the relevant coherent behavioral patterns from the set of seven patterns, which are common to all four species and apparently innate. We suggest that this form of learning differs from the known forms of associative learning, and speculate that it is quite universal and can be present in a wide variety of species, both invertebrate and vertebrate. This study suggests a new link between the concepts of cognition and innateness.
Keywords
Introduction
In his On the Origin of Species by Means of Natural Selection, Charles Darwin (1859) theorized that evolutionary theory would change the foundation of psychology. Although many researchers have appreciated Tinbergen's (1963) four fundamental questions concerning the ontogeny and casual mechanisms of cognition, and the phylogeny and functions of cognitive skills, only in recent years have all of the divided sub-disciplines of psychology started to implement evolutionary principles into their literature and research (Fitzgerald and Whitaker, 2010). Comparative psychologists have developed new techniques to probe the cognitive mechanisms underlying animal behavior, and they have become increasingly skillful at adapting methodologies to test diverse species (MacLean et al., 2012; Vonk and Shackelford, 2012). However, the schemata of ordering learning classes is still flexible (Reznikova, 2012), and it is possible that some forms of learning are not yet discovered. The most promising field here is studying how advanced intelligence interacts with inherited preparedness in a wide variety of animal minds.
It is known that individual adaptive behavior involves different kinds of learning together with innate behavioral patterns. Development of ethology, comparative psychology, and behavioral ecology enabled researchers to bundle together “innate” and “not innate” traits when considering animal cognition (Bateson and Mameli, 2007; Haldane, 1946; Marler, 2004; Reznikova, 2007). Researchers of animal cognition discovered a great deal of cognitive adaptations, tightly connected with species' modes of survival in their ecological niches. Members of many species, both vertebrates and invertebrates, demonstrate sophisticated cognitive abilities within relatively narrow domains. Examples include extraordinary classificatory capacities in pigeons (Herrnstein and Loveland, 1964; Huber, 1995; Watanabe, 2009) and honey bees (Mazokhin-Porshnyakov, 1969; Mazokhin-Porshnyakov and Kartsev, 2000; Menzel and Giurfa, 1999), abilities of group-retrieving ant species to grasp regularities, use them for flexible coding and “compression” of information to be transferred to their nest mates, and even add and subtract small numbers in order to optimize their messages (Reznikova and Ryabko, 1994, 2011; Ryabko and Reznikova, 1996, 2009), the ability to memorize and recognize many mates by facial features in paper wasps (Sheehan and Tibbetts, 2011; Tibbetts and Dale, 2007), huge data storage in food caching birds and mammals (Shettleworth, 1998), manifestations of “theory of mind” (Bugnyar and Kotrschal, 2004; Emery and Clayton, 2004), logic and counting (Smirnova, Lazareva, and Zorina, 2000) and sophisticated tool use (Kacelnik, Chappell, Weir, and Kenward, 2006) in corvids, and so on. It is a challenging problem for comparative psychologists to understand to what extent these cognitive adaptations can be attributed to flexible learning abilities versus innateness. To solve this problem, different forms of learning should be investigated in natural situations in which animals can perform their innate behavioral repertoire, together with flexible components of their behaviors.
Considering that insects, at least social hymenopterans, hold particular places of honor in studies of sophisticated cognitive abilities of animals, they can serve as good models for investigating different forms of learning within a functional and evolutionary framework. Certain forms of associative learning are considered basic elements of animal cognition (Wasserman and Miller, 1997). Associative learning is necessary when animals have to learn something new, from the simple paring of one stimulus with another (or with a motor pattern) to learning complex sequences of acts. Until the classic studies of Schneirla (1929) on maze learning in ants, it was repeatedly suggested that insects show little or no learning (Hollis and Guillette, 2011). However, long-term studies make clear that associative learning appears to be universal within insects. There are many examples in the literature on associative learning in insects, particularly in Hymenoptera (including parasitoids, solitary and social wasps and bees, as well as ants), Orthoptera (cockroaches, grasshoppers, locusts, crickets), Lepidoptera (moths, butterflies), Diptera (flies) and some others (for detailed reviews see Hollis and Guillette, 2011; Matthews and Matthews, 2009). To study “elementary” Pavlovian conditioned responses to rewarded stimuli such as odors and tastes, the standard assay was elaborated based on the proboscis extension responses of a harnessed bee which is restrained in an experimental apparatus in a way that resembles Pavlov's experiments with dogs (Bitterman, Menzel, Fietz, and Schafer, 1983; Carcaud, Roussel, Guirfa, and Sandoz, 2009; Frasnelli, Vallortigara, and Rogers, 2010; Reinhard, Sinclair, Srinivasan, and Claudianos, 2010; Takeda, 1961). Similar “Pavlovian-like” conditioning protocols were elaborated for cockroaches based on their “antenna-projection response” (Lent and Kwon, 2004) and for ants based on their “maxilla-labium extension” response (Guerrieri and d'Ettorre, 2010). All studied species quickly learned to associate visual cues through classical conditioning with rewarded odors. Freely walking bees (Chaffiol, Laloi, and Pham-Delègue, 2005; Sandoz, Laloi, Odoux, and Pham-Delègue, 2000) and ants (Dupuy, Sandoz, Giurfa, and Josens, 2006) successfully learned to associate an odor with food and approached that odor in a Y-maze. More complex examples concern the ability of honey bees to associate a rewarded smell exposed to them in a hive with their way back to the food in the field using visual clues to guide them (Reinhard, Srinivasan, Guez, and Zhang, 2004), as well as complex maze negotiations in ants (Cammaerts and Lambert, 2009; Karas and Udalova, 2001; Reznikova and Ryabko, 1994).
Although recent publications have demonstrated that insects can serve as good models for investigating universal regularities of learning, there are several limitations in the literature on insect learning. First, despite the fact that learning studies cover a wide range of insect species, some families, though promising, have not attracted much attention. For example, learning abilities of the species belonging to the order Coleoptera (beetles) are still under-estimated, and very few experimental investigations have been conducted on these insects. In particular, it has been revealed recently that mealworm beetles possess the ability to estimate numerosity ratios within four (Carazo, Font, Forteza-Behrendt, and Desfilis, 2009). From carabids (ground beetles), although they live in almost every terrestrial habitat on earth and possess long life spans and complex and rather flexible behavior (for a review, see Kotze et al., 2011), as far as we know, only Pterostichus melanarius has been studied for its learning abilities (Plotkin, 1979). Members of this species performed poorly in the maze which, possibly, caused aversion in insect cognitivists for many years. Second, in many learning studies of insects the training environment is very different from the animals' natural environments. However, we think that simulating insects' day-to-day problems, as it was done in the experiments with honey bees (e.g., Collett, Harland, and Collett, 2002; Giurfa, 2003) and ants (Reznikova and Ryabko, 1994, 2011), offers greater opportunities for discovering new forms of flexible behavior. Third, recent studies of insect learning are focusing mainly on the panoply of traditional associative learning phenomena, including extinction and spontaneous recovery, compound and context conditioning, as well as blocking, overshadowing, and various inhibitory phenomena (for a review, see Hollis and Guillette, 2011). A different approach has expanded our knowledge of insect cognition to the highest forms of learning, such as rule extraction (Avargués-Weber, Deisig, and Giurfa, 2011; Menzel, 2008; Reznikova and Ryabko, 2011) and social learning (Leadbeater and Chittka, 2007; Reznikova, 1982, 2007). Although insect behavior is based on innate templates to a great extent, surprisingly not enough attention has been paid to those forms of learning that concern innate predisposition to build up one set of associations more readily than another (see Reznikova, 2012), such as guided learning (Gould and Marler, 1987), or “biased learning” (Westerman, Hodgins-Davis, Dinwiddie, and Monteiro, 2012).
In the experiments described here, we study common and mass “wild” species of ground beetles whose learning abilities have never been examined before in order to investigate basic, relatively simple elements of animal cognition. Here the point is to demonstrate that there is a certain form of flexible behavior in between learning something new and applying the gained experience to a novel situation (cognition), and using available behavioral patterns that are apparently innate. Thus, the present study provides a rare case of finding a new link between the concepts of cognition and innateness.
As an experimental setting we chose a natural situation of interference competition between predatory ground beetles and red wood ants Formica s.str. Both red wood ants and carabids are generalist predators of comparable size and are very abundant in forest habitats. Within their large feeding territories the ants create “black holes” in the habitat, i.e., areas that are highly dangerous for other species, where intruders can be killed or at least injured. Earlier we revealed that ants actively force carabids out of their feeding territory, and beetles are able to change their trajectories of movement and to use different behaviors in order to avoid collisions with ants (Reznikova and Dorosheva, 2004). In order to imagine vital situations of encounters between carabid beetles and red wood ants on their common territory, it is important to note that the danger presented to a beetle by an ant could be of varying degree: A beetle can meet a single ant, or a group, or it can find itself in the vicinity of an ant foraging route overcrowded with ants. Besides, within a colony of red wood ants individuals belonging to different “professions” differ essentially by their behavior and aggressiveness, from peaceful honeydew collectors and transporting ants to aggressive guards and hunters (Dorosheva and Reznikova, 2006; Reznikova, 2008, 2011). It would be adaptive for beetles to distinguish between these situations and change their behavior flexibly and adequately.
Here we suggest that in order to gain experience from their encounters with ants and to avoid conflicts, beetles apply a form of learning that has been overlooked. We call this “catalog learning,” the name we give to the ability of animals to establish associations between stimuli and coherent behavioral patterns. The idea is that in some situations animals do not learn to do something truly novel in order to gain advantage from their environment; instead, they learn to draw a relevant innate and coherent behavioral pattern that consists of constantly ordered and non-changeable sequences of behavioral acts (or “elementary motor patterns”) out of several available ones. This differs significantly from associative learning that has been studied in many vertebrates and invertebrates (for a detailed review, see Reznikova, 2007). Instead of arranging elementary behavioral acts gradually from chaotic to rational sequences in order to learn something new, animals learn to choose the most relevant one from a set of fixed inherited behavioral patterns. It is worth noting that a coherent behavioral pattern here is not the same as a single motor pattern, or a reaction (for definitions, see Reznikova, 2012). We consider “catalog learning” to occur when an animal forms a “stimulus-pattern” association instead of a “stimulus-reaction” one.
In our study, we simulated routine territorial interferences between ground beetles and red wood ants and investigated how members of four carabid species modified their behavior (i.e., how they learn) in order to avoid damage from ants. Further detailed studies were conducted on one carabid species displaying the most multi-faceted behavioral inter-relations with red wood ants. As far as we know, this is the first study of different learning abilities in ground beetles, and the first experimental paradigm in which simulation of natural inter-specific interference serves as a basis for studying learning processes in insects.
In order to examine whether carabid beetles are able to change their behavior in such a way as to avoid collisions with ants, we simulated situations of territorial conflicts in field and laboratory experiments.
Experiment 1: The Ability of Beetles to Avoid Collisions with Ants in a Y-Shaped Maze
Materials and Methods
As a first step of investigating whether beetles can adjust their behavior in response to clashes with their natural enemies, we conducted experiments in which repeated collisions with aggressive ants were simulated in Y-shape mazes. Experiments were performed in 1997 and 1998.
Animals and housing
Beetles and red wood ants were collected in their shared feeding territory in the mixed pine-birch forest, in the University campus in Academgorodok near Novosibirsk. Beetles were collected by pitfall traps and by manual sampling and then were housed in the laboratory during three days before the experiment. The beetles of each species were housed in groups in containers with moist litter, and they were fed by minced meat once a day. We used 31 specimens of Carabus regalis, 52 Pterostichus magus, 20 P. niger, and 42 P. oblongopunctatus. During the main part of the experiment, the beetles were housed solely in individual containers with moist litter. Ants were collected from the top of an ant-hill; the most aggressive individuals were chosen among “guards”: those ants that demonstrated a “dead grip” as a reaction to a needle approaching the top of the ant-hill (Dorosheva and Reznikova, 2006). Ants were housed in a group of about 40 individuals in a small artificial nest on a separate arena where they received water, carbohydrate and protein food. After finishing the experiments, ants were returned to their ant-hill in the forest and the beetles were released in a remote area of the forest.
In the first laboratory experiment a simple Y-shaped maze was used. One section was empty, while the other contained an aggressive ant tied to a wall of the maze with a thin thread, so that it could move freely within the length (4 cm) of the thread (see Fig. 1). As in our earlier experiments (Reznikova and Dorosheva, 2004), in these conditions aggressive tied ants attack and bite approaching beetles; encounters usually lasted 1–3 seconds because a tied ant was not able to run down a beetle, and neither the ants' aggressiveness nor the beetles' fearfulness increased during repeated trials. In all trials a beetle was placed at the entrance of the maze, and subsequent events were recorded manually with the use of a stop-watch. Typically, a beetle spent less than 4 s motionlessly with its antennae stretched and then began to move. Each beetle was tested 30 times with the interval of 5 minutes between runs. Each run took about 1 minute. To avoid the possible influence of smell tracking by beetles themselves, the paper on the floor of the maze was changed after each test. To exclude possible influence of preference for the left or the right section, previous subsidiary tests for all individuals were conducted a day before the main experiment in empty mazes lacking any stimuli (30 runs per individual). For 145 out of 147 specimens, no preference for the left or the right section was observed, and these beetles were used in the main experiment.
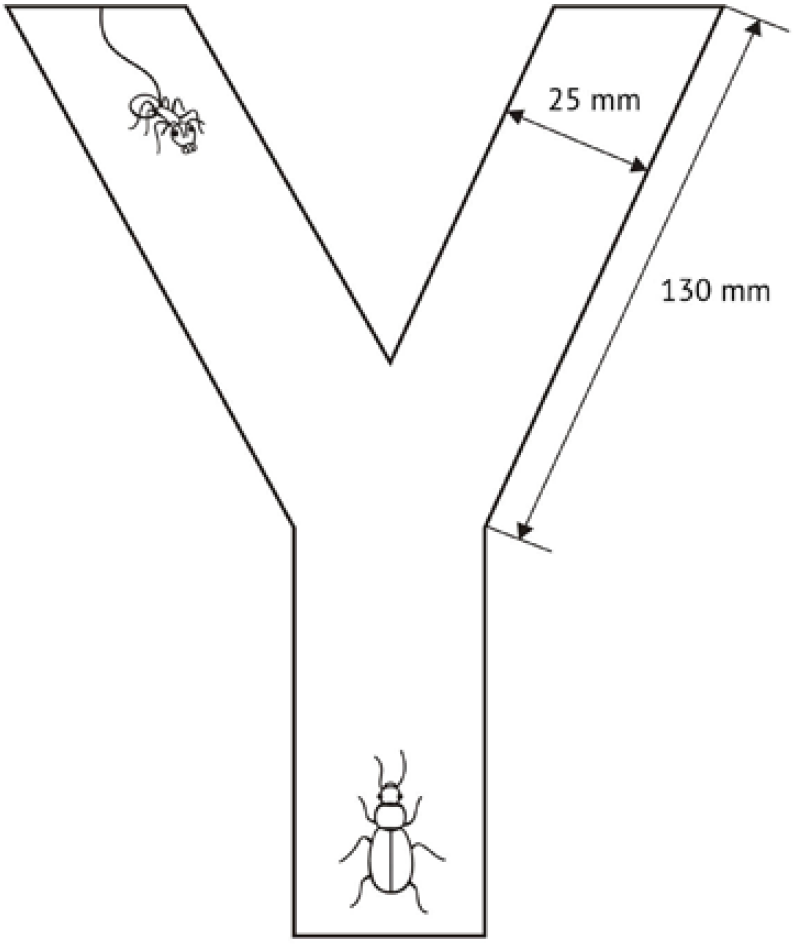
A scheme of a maze with an ant attached in one section with a thread
We considered insects' encounters as “collisions” (or “clashes” or “conflicts” – used here interchangeably) when they resulted in bites, on one side or on both. The ability of the beetles to avoid collisions with ants was estimated as the ratio between “erroneous” actions (“mistakes”) and “correct actions.” We define here the “correct action” as a modification of behavior that allowed the beetle to avoid a clash successfully. Such modifications included both avoidance of the dangerous section of the labyrinth and the use of specific behavioral patterns (see below). We considered “erroneous” those reactions when a beetle did not take into account the presence of ants and did not modify its behavior. As noted earlier, the beetles were collected in the forest; therefore, some specimens may have had previous experience of contacts with ants, whereas others may have had none. In order to minimize the influence of preceding experience, we took into account only those (both correct and incorrect) actions that were taken after the beetle managed to avoid conflict with the ant for the first time.
In order to assess comparability of results obtained in nature and in the laboratory, we conducted an additional experiment in which new beetles were tested in Y-shaped mazes just after they had been tested in the field where they were placed in the vicinity of an ant foraging route. In sum, 32 individuals were examined: 5 C. regalis, 17 P. oblongopunctatus, and 10 P. magus. For each specimen, sets of behavioral patterns were compared in field and laboratory experiments, in addition to recording the efficiency of avoidance of collisions with ants.
Results
Tests in the maze demonstrated that members of all species surprisingly did not learn “simply” to avoid a “dangerous” section of the maze; instead, they learned to successively avoid or to end conflicts with ants applying one of the following set of coherent behavioral patterns:
Behavioral patterns of avoidance:
turning away from the ant
turning away after touching the ant with the antennae (“touch-turn”)
turning away without a contact; (see Fig. 2) (“turning away”)
going around the ant; (see Fig. 3) (“going around”)
stopping near the ant, often with legs and antennae hidden (freezing before the contact) (see Fig. 4) (“stopping”)
Defensive behavioral patterns applied by beetles during contacts with ants:

A beetle is turning away from an ant (photo by Nail Bikbaev)

A beetle is attempting to go around the ant (photo by Nail Bikbaev)

A beetle is freezing before the contact with an ant (photo by Nail Bikbaev)

A beetle is attempting to change the direction of movement after encountering an ant in the maze (photo by Nail Bikbaev)

A beetle is freezing during the contact with the ant (photo by Nail Bikbaev)

Fighting in the maze: A beetle flips over on its back to gnaw at the ant (photo by Elena Dorosheva)

Fighting in the maze: An ant just killed the beetle (photo by Elena Dorosheva)
The use of these coherent behavioral patterns as a reaction to bites from the aggressive ant can be considered a result of evoking an innate response that consists of ordered motor acts integrated into a rather appropriate stereotyped sequence.
Four species tested in the maze seemed to use specific preference for definite sets of stereotyped patterns, although the simplest pattern 1.2 (“turning away”) was practically universal for all species (see Fig. 9, Table 1). P. oblongopunctatus showed preference for the patterns 1 (“turning away”) and 2 (“going around”), i.e., during first tests all specimens tried to round the ant. When facing the threat they began to use the pattern 1.2 (“turning away”) after 3–6 runs. P. magus showed preference for the patterns 1 (“turning away”) and 5 (“freezing”). It is worth noting that we observed several cases of direct beetle-ant antennal contacts between the tied ants and the members of species that are comparable with red wood ants in size: P. magus and P. oblongopunctatus. In insects, such contacts can be considered attempts of closer identification, similar to sniffing under the tail in mammals (for details, see Reznikova, 2007). Members of two species that are much bigger than an ant, P. niger and C. regalis, did not try to go around the ant in the maze. They often turned away at some distance from the ant (the pattern 1.2) and avoided the section with the tied ant (the pattern 7). These beetles more frequently attacked and bit the tied ant.
Pairwise comparison of the number of cases where beetles of different species used different behavioral patterns (1–7) in order to avoid collisions with ants in the mazes

Notes: n = number of beetles
p < 0.5
p < 0.01
p < 0.001

Numbers of members of different species that used different behavioral patterns to avoid conflict with a tied ant in the maze
Computing pairwise differences between four beetle species by chi-square test, we found that in all cases, with the exception of one pair (P. niger and C.regalis), the obtained values of chi-square exceed the tabulated critical number (13.3 at the significance level 0.01) and thus frequencies of use of different patterns differ in all species. P. niger and C. regalis do not differ significantly by the set of patterns of avoidance of conflicts with ants (χ 2 = 7.48) (see Table 1). Other species, although they use similar sets of behavioral patterns in their conflicts with ants, display different preferences for certain patterns.
Laboratory findings were confirmed by field experiments. It turned out that those specimens of P. magus and P. oblongopunctatus that successfully applied different patterns to avoid clashes with ants in the vicinity of their foraging routes also successfully avoided the danger in the maze. From 10 specimens of P. magus tested in this series of trials, four managed to avoid conflicts with the tied ant in the maze, and the same individuals successfully maneuvered among ants on their routes: The average number of bites by ants was 0.25 (SD = 0.5, M = 0.25) per beetle. Those four individuals that learned poorly in the maze got 3.25 (SD = 1.26, M = 0.63) bites per beetle on ants' routes. Two individuals from those 10 belong to the intermediate group (see above). In P. oblongopunctatus, four individuals successfully avoided conflicts with ants both in the maze and in the field (0.8 bites per beetle on ants' routes, SD = 0.96, M = 0.48), and 3 beetles did not learn to avoid ants in either of the situations (2.7 bites per beetle, SD = 1.15, M = 0.67).
Flexibility of Beetles' Behavior When Interacting with Ants in Different Situations
This series of experiments simulated vital situations of beetles' encounters with a “peaceful” ant and with an aggressive one. We examined whether beetles changed their behavior in repeated and changed situations and whether they were able to gain experience from their encounters with their enemies. These experiments were conducted in small separate laboratory arenas with freely moving ants and P. magus beetles. This is the most abundant species of carabids on ant territories, as it has the same seasonal peak of activity with red wood ants; furthermore, this species is closer to ants by its size and characteristics of movements than other species studied (Reznikova and Dorosheva, 2004).
Experiment 2: Beetles' Reactions towards “Peaceful” and Aggressive Ants
Materials and Methods
This experiment was aimed at examining beetles' abilities to discern characteristic features of ants' behaviors in order to avoid clashes with aggressive individuals and waste no energy to run away from peaceful ants. The experiment was conducted in 2003.
Animals and housing
Beetles were collected and housed as described in Experiment 1. Forty specimens of P. magus were used in the main experiment and 40 ones in auxiliary tests. Ants were collected in the vicinity of an ant-hill. In order to select surely aggressive and peaceful ants for the main part of the experiment, we conducted auxiliary tests in which the ants were taken from nature and housed in the laboratory. Ants interacted with carabid beetles, one by one, on arenas (15×15 cm) for 10 minutes each. Each ant was tested once, and the number of its chance encounters with a beetle on the arena was recorded (about 8 times, in average). We considered an ant aggressive if it displayed a high degree of aggression during all encounters, such as the “dead grip” and “long bites” (for a detailed description of aggressiveness in ants, see Reznikova and Yakovlev, 2008). We considered an ant “peaceful” if it demonstrated only a low level of aggressiveness: no more than a “short bite” (Reznikova and Yakovlev, 2008). Finally, 28 aggressive and 12 peaceful ants were selected and then used in the main part of the experiment.
Procedure
Beetles and ants interacted in pairs, on arenas 15×15 cm, during 10 min per test; that is, 28 pairs of beetles and aggressive ants, and 12 pairs of beetles and peaceful ants. Each beetle, as well as each ant, took part in one trial only. Insects could freely move on arenas, and we recorded all their behaviors as a manual protocol using a stop-watch. The frequencies of all behaviors recorded were compared in situations of interactions of beetles with aggressive and peaceful ants. Comparisons were between subjects that took part in interactions with aggressive versus peaceful ants.
Results
Before examining whether beetles can learn lessons from their collisions with ants, we tested whether these insects in principle react differently to aggressive and peaceful ants. In order to examine whether beetles apply behavioral patterns of avoidance more frequently when interacting with aggressive ants than with peaceful ones, we tested P. magus on laboratory arenas with the ants of different behavioral characteristics.
First, it turned out that when interacting with ants on arenas, beetles displayed the same stereotyped behavioral patterns as in mazes, except for pattern 7 (that was possible to use in the maze only). We then compared relative frequencies of patterns of avoidance (patterns 1–3: “turning away,” “going around,” and “stopping”) in beetles interacting with aggressive ants (n = 28) and in beetles interacting with peaceful ants (n = 12) by Fisher's angular transformation, and differences appeared to be significant: (φ = 5.735, p < 0.01). This shows that beetles avoided aggressive ants more frequently than peaceful ones (see Fig. 10).

Comparison of relative frequencies (proportion of the total number of encounters with ants) of patterns of avoidance (patterns 1–3) in beetles interacting with aggressive ants (n = 28) and in beetles interacting with peaceful ants (n = 12)
The difference in the beetles' behavior can be considered a consequence of overt differences in the behavior of peaceful and aggressive ants. The situations that we simulated in our experiments correspond to the situations in nature when beetles encounter members of “aggressive professions,” such as guards and hunters, or “peaceful professions” such as aphid milkers and transporters of honey dew (see Reznikova, 2007, 2011). In our earlier publications devoted to the behavior of ants being exposed to beetles (Dorosheva and Reznikova, 2006; Reznikova and Yakovlev, 2008) we described typical behaviors of ants during their encounters with beetles. In descending order of aggression they were: “death grip” (the ant grasps the beetle and does not let go); “long attacks” (attacks lasting more than 5 s, after which the ant releases the beetle and gets away); “short attacks” (attacks lasting less than 5 s). It is worth noting that “long attacks” usually last about a minute and even more, and an ant is quickly moving along the beetle's body biting it here and there. As opposed to aggressive ants, peaceful ones displayed indifference (the ant shows no response at all, or explores the beetle with its antennae and moves on) or evasion (the ant changes direction of its movement to avoid the encounter).
In the experiments described here, all reactions of ants towards beetles were the same. This is of no surprise that beetles discern behavioral features of aggressive ants and apply behavioral patterns of avoidance after 1–4 encounters with them; that is, after a short interval of interaction, usually much shorter than an experimental trial.
Experiment 3: Beetles' Ability to Gain Experience from Interrelations with Peaceful and Aggressive Ants
Materials and Methods
This experiment was conducted in 2009 to test whether beetles are able to retain for at least three days the memory about interactions with ants.
Animals and housing
Beetles and ants were collected and housed as described in Experiment 1. All insects had no prior experience of being involved in experiments.
Procedure
Beetles and ants interacted on arenas, as described in Experiment 1. We tested 20 specimens of P. magus, one by one, with a sole aggressive ant and 20 with a sole peaceful ant, 10 min each, each time a new one. Three days later we again tested 10 beetles with unfamiliar aggressive ants, and 10 with unfamiliar peaceful ants. From the remaining 20 beetles, those 10 individuals that earlier had contacted aggressive ants now had to interact with peaceful ants, and vice versa. Insects could freely move on arenas, and we recorded all their behaviors as a manual protocol using a stop-watch. The frequencies of all behaviors were compared in situations when beetles encountered aggressive and peaceful ants in repeated and changed situations.
Results
This study included two experiments. In the first experiment we investigated how experience gained by beetles from their previous contacts with either aggressive or peaceful ants influences their choice of behavioral patterns during subsequent contacts with ants. We tested whether those beetles that encountered an aggressive ant during the first stage of the experiment displayed patterns of avoidance during the following meetings with ants more frequently that those beetles that interacted with a peaceful ant. The procedure was as follows: Each beetle had a single 10 minute training trial with one ant that was either aggressive or peaceful. Twenty beetles were exposed to an aggressive ant and 20 to a peaceful ant. All 40 beetles were tested 3 days later. Ten beetles of each group were exposed to an aggressive ant and 10 to a peaceful ant. It is worth noting here that members of these two ant groups reacted to beetles rather differently. Although the number of encounters of beetles with members of both groups during 10 minutes trials were the same (on average 7.8 and 7.6 times per trial with aggressive and peaceful ants, respectively), aggressive ants attacked beetles in average 5.5 times per trial, and peaceful ants attacked 2.6 times. Levels of aggression (see the previous paragraph) were different in members of these ant groups: Peaceful ants never applied “long attacks,” not to mention “death grip,” and they usually restricted their attacks by lunges with open mandibles (for details, see Reznikova and Yakovlev, 2008).
We compared relative frequencies of patterns of avoidance (patterns 1–3: “turning away,” “going around,” and “stopping”) in beetles interacting firstly with an aggressive ant and then with a peaceful ant (n = 10) and in beetles interacting with a peaceful ant twice (n = 10) by Fisher's angular transformation. The differences appeared to be significant: (φ = 1.684; p < 0.01) (see Fig. 11).

Comparison of relative frequencies (proportion of the total number of encounters with ants) of patterns of avoidance (patterns 1–3) in beetles that had previously encountered either an aggressive or a peaceful ant
Beetles exposed to aggressive ants during training responded with more avoidance behaviors on tests with peaceful ants than did beetles that had experienced peaceful ants during training and test. It is interesting that beetles retain experience during at least three days. We can suggest that the preliminary contact with an aggressive ant changes the behavior of a beetle so that it becomes careful not to encounter any kind of ants.
It is very likely that it is an ant's movement pattern that triggers the beetle's avoidance response. Ants' aggressive reactions towards beetles (see Dorosheva and Reznikova, 2006) are preceded by aggressive postures and sharp movements, so beetles are able to discern these signs of aggression and choose a relevant pattern of avoidance from their behavioral repertoire.
In the second experiment we tested whether those beetles that encountered a peaceful ant first, and thus did not gain a painful experience, display patterns of avoidance during the next encounters less frequently that those beetles that first interacted with an aggressive ant. We compared relative frequencies of patterns of avoidance (patterns 1–3: “turning away,” “going around,” and “stopping”) in beetles interacting firstly with a peaceful ant and then with an aggressive one (n = 10) and in beetles interacting with a peaceful ant twice (n = 10) by Fisher's angular transformation, and differences appeared to be not significant: (φ = 0. 154; p > 0.05) (see Fig. 11). So, we can conclude that the preliminary contact with a peaceful ant does not impact choices of behavioral patterns in beetles during their contact with the next ant.
Experiment 4: Beetles' Ability to Establish Associations between Stimuli (Bites from Ants) and Relevant Behavioral Patterns
Materials and Methods
This experiment was conducted in 2011, with a new group of beetles in order to examine their ability to learn how to avoid collisions with ants in repeated situations.
Animals and housing
The experiment was conducted in the laboratory with P. magus. Beetles were collected and housed as described in Experiment 1; aggressive ants were collected and housed as described in Experiment 2.
Procedure
The procedure was mainly the same as described in Experiment 3; 29 specimens of P. magus were used. During the experiment, one individual was badly damaged by ants and was excluded; we also did not consider three beetles that demonstrated chaotic reactions; as a result, the data on 25 specimens were obtained. We tested the hypothesis that beetles change their behavior after the first clash with an aggressive ant in such a way as to choose the relevant behavioral pattern faster than before. Each from 25 beetles interacted one by one with a sole ant in a separated arena 25×25 cm. A beetle was placed on the arena, and after 3 minutes an ant was placed there as well. All behaviors of insects were recorded manually by a stop-watch during 10 minutes of interaction. The arena was cleaned by alcohol and dried after each trial conducted with each pair of insects. After 24 hours the procedure was repeated with each beetle, one by one, confronted with another single aggressive ant.
We compared numbers of certain behavioral patterns applied by a beetle in relation to the total sum of all of its reactions towards ants. The pairwise Wilcoxon criterion was used.
Results
The experiment described here is similar in some respects to Experiment 3, in which beetles gained painful experience from their contacts with aggressive ants. Here we proceeded with investigating whether the beetles change the frequency of choice of different behavioral patterns as a reaction to clashes with their enemies. In this experiment we tested a new group of beetles in order to avoid multiple comparisons; besides, we used a different interval between trials, that is, 24 hours instead of 3 days.
Frequencies of patterns in behavioral repertoires of beetles during the first and the second encounters with sole aggressive ants are given in Figure 12.
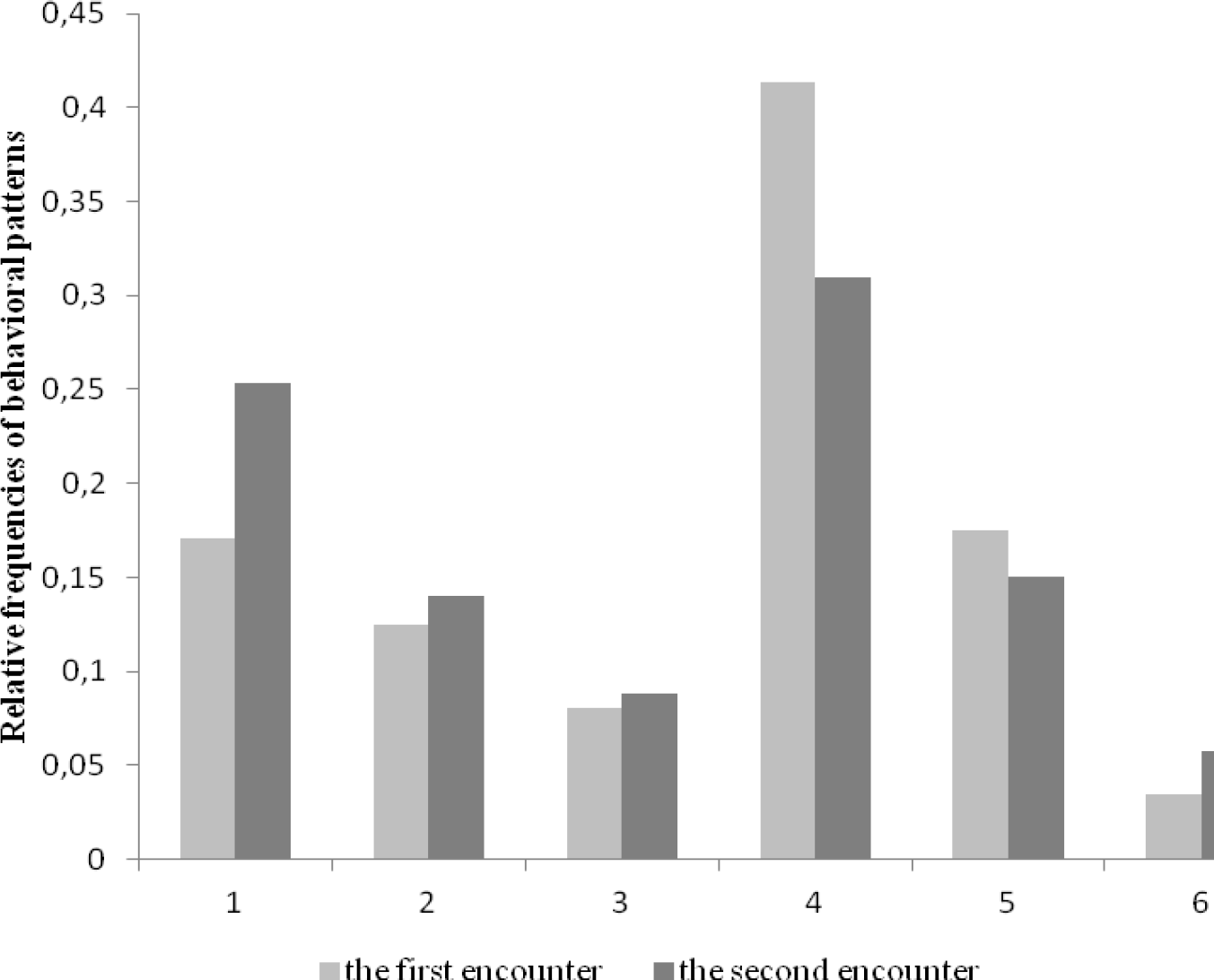
Relative frequencies (proportion of the total number of encounters with ants) of behavioral patterns applied by P. magus to avoid or end conflicts during the first the second encounters with sole aggressive ants in repeated situations
In Table 2 the results of pairwise comparisons by Wilcoxon criterion are given. It turned out that the frequency of pattern “turning away” (1) increased significantly (t = 18.0; p = 0.010), whereas the frequency of pattern “speeding” (4) decreased (t = 8.0; p = 0.005). Both changes mean that when meeting for the second time with an aggressive ant, beetles more frequently turn away before encountering with it, and as they do not approach the ant, they do not apply the pattern 4 “speeding” (4). So, we can conclude that having an experience of interactions with an aggressive ant changes beetles' behavior in such a way as to avoid any contacts with ants.
Pairwise comparisons of relative frequencies of patterns in beetles interacting twice with an aggressive ant

Notes: t values of Wilcoxon criterion
p < 0.05;
p < 0.01
Discussion
Predatory carabid beetles appeared to flexibly change their behavior in order to avoid dangerous encounters with red wood ants, their competitors for space. This corresponds to the accepted definitions of learning (Matthews and Matthews, 2009; Reznikova, 2007). In our previous study we revealed that interference competition is rather strong between red wood ants and carabid beetles (Reznikova and Dorosheva, 2004). In the present study, investigating cognitive aspects of interspecies interference, we revealed a form of animal learning that apparently had not been described before. When starting our maze studies, we expected that after several encounters with an aggressive ant tied within one of two sections of the Y-shaped maze, beetles should form a conditional reflex and thus begin to avoid a dangerous place. It was also expected that an innate template may help them to form this association rather quickly. The concept of innate template is rooted in early psychological experiments on fear conditioning in a human infant (Watson and Rayner, 1920). Furthermore, the presence of an innate template has been demonstrated in a great variety of species, from monkeys (Mineka, Keir, and Price, 1980) and humans (Rakison and Derringer, 2008; Soares, 2012) to ants (Dorosheva and Reznikova, 2006; Dorosheva, Yakovlev, and Reznikova, 2011; Dussutour, Deneubourg, Beshers, and Fourcassié, 2009). However, in our situation, avoidance of the dangerous section of the maze was only one (and relatively rare) out of a set of coherent behavioral patterns used by beetles to avoid the unpleasant event. We revealed a common set of coherent behavioral patterns that allowed beetles to avoid injuries from ants: (1) to turn away; (2) to go around the ant; (3) to freeze before the contact with the ant; (4) to speed up movement and change the direction of movement; (5) to freeze during the contact with the ant; (6) to fight with the ant; (7) only in the maze: steady avoidance of the dangerous section. Members of different species quickly learned to manipulate with several patterns from this set of 7 patterns, and frequencies of the use of different patterns depended on species-specific traits in the investigated species of beetles.
Our further experiments in the field plots, Y-shaped mazes and on laboratory arenas where beetles met with one or several ants, aggressive or peaceful, revealed a great deal of flexibility in behaviors applied by beetles to avoid collisions in different situations. For example, in the maze, where a tied ant was not able to pursue a beetle, carabids rarely used freezing during the contact with the ant, whereas on arenas this was a frequently used and the most effective behavioral pattern, particularly in the situation of interaction with a group of aggressive ants. It is worth noting that members of several carabid species share the same set of patterns and differ from each other only by relative frequencies of the use of some of them, thus displaying species-specificity in the preference for different patterns.
Our detailed experiments with P. magus, the closest species from other carabids to red wood ants by its behavioral and ecological characteristics, demonstrated that members of this species react flexibly to ants, differently to aggressive and peaceful ones. They appear to be able to learn lessons from their encounters with aggressive ants, and retain experience for at least three days.
Based on our study of beetles' ability to flexibly adapt their behavior in response to challenges coming from their competitors for space, we suggest that these insects establish associations between stimuli and relevant coherent behavioral patterns in their innate behavioral repertoire. We call this catalog learning and claim that this form of learning differs from those described before, in particular, from classical conditioning, operant conditioning, trial-and-error learning, and, of course, from the most complex forms of learning, such as rule extraction, learning set formation, etc. (see Reznikova, 2012). It is possible that this form of learning shares with “innate guided learning” (Gould and Marler, 1987) and “avoidance learning” (Bolles, 1970) such important features as fast conditioning and the use of species-specific defense reactions. However, the response of beetles to repeated encounters with their enemies is not simply a reaction of fear as this was described in situations of predator recognition in other species explained by means of “guided learning” (see Griffin, Evans, and Blumstein, 2002). What beetles seemed to learn in our experiments is to select the most relevant among several innate coherent behavioral patterns in order to make their defensive behavior towards their natural enemies most effective and parsimonious. We neither pretend that “catalog learning” is a complex type of learning, nor do we claim that this is the first described situation in the insect learning literature when learning is intertwined with innateness. In particular, good examples of the interaction between learning and innate behavior in insects is the way that honeybees learn the course of the sun's passage across the sky (see Dyer and Dickinson, 1994), as well as the interaction between innate “learning rules” and flexible learning to distinguish complex visual stimuli in bees and wasps (Mazokhin-Porshnyakov and Kartsev, 2000). However, we think that catalog learning, with its distinct features, fills a lacuna in our knowledge about possible innate elements of cognition in insects and, perhaps, this form of learning can be found not only in insects but in vertebrate species as well.
For the development of the comparative approach to cognition, it is important to find basic elements of animal cognition and to find a link between the concepts of cognition, intelligence, learning, and innateness. We can define learning in animals in its wide sense as the ability to establish stable changes in their behavior that allow them to solve a certain problem (Reznikova, 2007, 2012). Different classes of learning work differently in natural situations. Until recently, associative learning has been considered the main basic class accepted by ethologists. Catalog learning described here is based on innateness to a great extent and thus differs essentially from associative learning. Associative learning can be said to have taken place when there is a change in an animal's behavior as a result of one event being paired with another (Pearce, 2000). There are two main forms of associative learning: classical (Pavlovian) conditioning based on pairing an unconditional stimulus (such as food or pain) with a conditioned (previously neutral, such as bell ring) stimulus, and operant (Skinnerian) conditioning based on pairing the behavior and its consequences. Unlike the classical conditioning, where a new response is formed basing on the association with a previously neutral stimulus, in operant conditioning there is no creation of a new response; instead, there is an increase or decrease in the response that is already being exhibited (for details, see Reznikova, 2007). We claim here that, instead of an association between a stimulus and a reaction (S-R), like in classical conditioning, and R-S (or S-R-S) learning like in operant conditioning, “catalog learning” establishes an association between a stimulus and a relevant innate behavioral pattern consisting of elementary motor acts that have a fixed order. It can be described as “stimulus - pattern” learning (S-P). We suggest that, in order to optimize their reactions to the evolutionary essential stimuli, animals learn to draw relevant patterns from the “catalog” of their species-specific repertoire of innate behaviors. As with other forms of learning, catalog learning is based on stable changes in animals' behaviors; however, here these changes are based on the selection of definite innate behavioral patterns as a response to the persistent force of a natural stimulus. Catalog learning can thus be considered a natural way to learn, and an exciting area for future research in comparative psychology, because it puts the study of animal learning into an explicitly evolutionary and functional framework.
Footnotes
Acknowledgements
Research was supported by RFBR (11-04-00536), by the Integrated Project of Siberian Branch RAS (grant N 21), by the Program “Living Nature” of the Presidium of Russian Academy of Science, and by the Program of cooperative investigations of SB RAS and third parties (grant No. 63). We thank Daniil Ryabko for his fruitful comments.
