Abstract
Sex differences in spatial cognition are well documented; males typically outperform females on tasks dealing with mental rotation and spatial navigation, while females tend to outperform males on tasks dealing with object location, relational object location memory, or spatial working memory. Here we investigated both behavioral and neural sex differences in sex-specific spatial abilities. In Experiment 1, sixty-six (30 males, 36 females) participants completed computerized mental rotation (MR) and spatial working memory (SWM) tasks. In Experiment 2, twelve (6 males, 6 females) participants were given slightly modified versions of the same tasks during functional magnetic resonance imaging (fMRI). In both experiments, males outperformed females on the MR task, but no behavioral sex difference was observed on the SWM task. Males showed more activation in left parahippocampal gyrus, right medial frontal gyrus, inferior parietal lobe, inferior frontal gyrus in the MR task. Females showed activation in the left parahippocampal gyrus only. For the study condition of the spatial working memory task, females showed activation in left inferior frontal gyrus, while males activated left inferior parietal and medial frontal areas. In the test conditions, females showed activation in the right inferior frontal gyrus, left middle temporal gyrus, and left parahippocampal gyrus. Males activated right medial frontal gyrus and inferior parietal lobe. Interestingly, similar regions – parahippocampal gyrus, inferior parietal lobe, and middle temporal gyrus - were found to be active when males solved mental rotation tasks and females solved spatial working memory tasks. Further, performance was modulated by activation in the parahippocampal gyrus and middle temporal gyrus for males and the middle temporal gyrus and inferior frontal gyrus for females. These data extend previous claims for sex differences in sex specific spatial cognitive abilities by demonstrating both behavioral and neural sex differences consistent with an evolutionary model, which suggests sexual selection may have favored sex-differences in such abilities and the neural substrates that sub-serve those processes.
Keywords
Introduction
Sex differences in spatial cognition are well documented (see Gaulin, 1995 for review) and appear to have evolved as a consequence of sexual selection for such neurocognitive capacities (Geary, 1995). Current behavioral (Geary, Gilger, and Elliott-Miller, 1992; McBurney, Gaulin, Devineni, and Adams, 1997; Moffat, Hampson, and Hatzipantelis, 1998; O'Laughlin and Brubaker, 1998; Silverman, Kastuk, Choi, and Phillips, 1999), comparative (Daly and Wilson, 1983; Gaulin, 1992; Gaulin, FitzGerald, and Wartell, 1990; Lacreuse, Herndon, Killiany, Rosene, and Moss, 1999; Roof and Stein, 1999; Stavnezer et al., 2000), and neuroscientific (Gur et al., 2000; Jordan, Wustenberg, Heinze, Peters, and Jancke, 2002; Thomsen et al., 2000) literature has provided convergent support for the development of the sex difference in spatial abilities (Silverman and Eals, 1992). The evolution of sex differences in spatial abilities related to navigation may have resulted from one or more of the following: intra-sexual competition for resources (e.g., access to members of the opposite sex), direct intra-male competitive mechanisms such as hunting and / or warfare (Alexander, 1979; Hill, 1982; Symons, 1979), or as a byproduct of increased home range (Ecuyer-Dab and Robert, 2003; Gaulin, 1992; Gaulin and FitzGerald, 1989). Local object, or spatial relational working memory (i.e. indicating the location of a previously viewed object in an array), which is a typically female dominated task, may have evolved in females for locating food sources and indicating the location of offspring (Ecuyer-Dab and Robert, 2003; Geary, 1995; Silverman and Eals, 1992). This conception of the dimorphism of sex-specific spatial cognition forms the basis for a hunter-gatherer based model for sex differences in spatial abilities. Here we focus on this hunter-gatherer hypothesis as a theoretical guidance for our investigation.
Behavioral investigations of sex differences have shown a male advantage (e.g., quicker reaction time and increased accuracy) on mental rotation (MR) tasks (see also Geary et al., 1992; Linn and Petersen, 1985; Masters and Sanders, 1993; McBurney et al., 1997; Silverman, Choi, Mackewn, Fisher, Moro, and Olshansky, 2000; Vandenberg and Kuse, 1978; Voyer, Voyer, and Bryden, 1995). Vandenberg and Kuse (1978) provided an ecologically valid analysis of spatial abilities by investigating mental rotation of 3-dimensional objects, which has been replicated, and is now considered the standard task for measuring mental rotation. Jordan et al., showed that task difficulty (i.e. 3-D objects vs. 2-D Letters) correlated with increased cortical activation in the parietal cortex. Similarly, Shepard (1994) indicated a larger effect for tasks requiring higher cognitive demand due to their analogous relationship to real-world 3-dimensional space, or foraging tasks. Therefore, increased parietal cortex activation in 3D MR tasks may be due to the higher cognitive demand the task requires (Barnes et al., 2000).
Neuroscientific support for sex differences is provided through neuropsychological and neuroimaging studies on, typically, male dominated spatial tasks. Gur et al., (2000) measured hemodynamic response to spatial tasks that varied in difficulty. They showed a proportional increase in hemodynamic response in the right parieto-occipital sulcus and left motor cortex as task difficulty increased. Males showed right lateralized activation in the more difficult spatial tasks and bilateral activation across all spatial tasks, whereas females showed less activation in both levels of the task. Due to their findings, Gur et al., (2000) suggested that increased cerebral blood flow in the right hemisphere of males is correlated with increased performance on spatial tasks. The male advantage in mental rotation may be a result of a strategy that entails creating a mental image of the object and turning it in virtual (egocentric) motor space. Theoretically, this would have been naturally selected for due to the advantage these abilities would have conferred for males in a hunting environment enabling them to rotate their personal, or egocentric, space and weapon in relation to what they were hunting.
It is hypothesized that the neurocognitive capacity needed to effectively rotate 3-diminesional objects in space is related to navigational abilities, and is more dominantly expressed in males (Geary et al., 1992; Gur et al., 2000; Jordan et al., 2002; McBurney et al., 1997; O'Laughlin and Brubaker, 1998). Mental rotation involves mentally representing and manipulating space and objects, and these mental simulations may serve to guide present and future behavior. The cognitive processes involved in mental rotation/simulation have yet to be completely elucidated, but it may be part of a larger network involved in predictive motor cognition or mental mirroring (e.g., Jackson and Decety, 2004). Motor cognition related to the so-called mirror neuron system (Gallese, Keysers, and Rizzolatti, 2004; Oztop and Arbib, 2002; Rizzolatti and Craighero, 2004) has traditionally been used to explain motor processes such as reaching and intentional grasping, but it is possible that mirror neuron systems exist across other neurocognitive modular systems such as audition, emotion, and possibly also spatial cognition (e.g., Oztop, Wolpert, and Kawato, 2004). A spatial mirroring system might allow an individual the capacity to mentally model an object, the self, and self-relational perspective to that object in space. If spatial cognition were related to these processes we would expect to find activation in pre- and supplementary motor areas, midline cortical structures such as the medial frontal lobes and precuneus, and the inferior parietal lobes (Decety and Sommerville, 2003; Jackson and Decety, 2004; Rizzolatti and Craighero, 2004). Because of the possible role of mental simulation in hunting and competition, it appears that sexual selection may have also been a driving force toward the sexual dimorphism in spatial cognition and appears to have enabled males to maintain a benefit/advantage in tasks that drew on the capacity to mentally rotate objects in space.
While males excel at navigational and rotational spatial tasks, females show an advantage in spatial abilities relating to memory for object locations and spatial working memory (Duff and Hampson, 2001; Eals and Silverman, 1994; Silverman and Eals, 1992). For example, females make fewer errors and take less time to complete tasks that tap object location memory (OBL) (James and Kimura, 1997; McBurney et al., 1997; Tottenham, Saucier, Elias, and Gutwin, 2003) and spatial working memory (SWM) (Duff and Hampson, 2001a). Alexander et al., (2002) expanded the research on this task by discovering that female performance was enhanced when objects were placed in the right hemispace, suggesting left hemisphere dominance for this type of processing. The hemispheric asymmetry when solving these spatial tasks, therefore, may be related to remembering the location of objects or the relational locations of objects (Alexander et al., 2002). Previous research has indicated a role for “serial processing” or spatial working memory for spatial task completion in women (Duff and Hampson, 2001a). Further investigation of spatial working memory indicates that the inferior frontal gyrus is implicated in working memory and short-term episodic memory. According to the Hemispheric Encoding-Retrieval Asymmetry (HE/RA) (Nyberg, Cabeza, and Tulving, 1998) model - the left frontal lobe should be implicated in encoding and right frontal lobe in retrieving spatial episodic information, as well (Jordan et al., 2002).
Further support of this female strategy was observed by Gur et al. (2000) who found that only men show a right hemispheric shift in activation for MR spatial tasks. Even though the task used was male-dominated, this suggests that females continue to use the left hemisphere to solve spatial tasks ranging in time and difficulty. Males and females, therefore, appear to implement different cognitive strategies, originating in different neural substrates when solving spatial cognitive tasks (Jordan et al., 2002).
The aim of the current study was to investigate whether the behavioral differences in sex-specific spatial cognition correlated with differences in neurocognitive activation using functional magnetic resonance imaging (fMRI). We conducted two studies – one behavioral and one functional neuroimaging. We hypothesized that 1) consistent with existing studies (Geary et al., 1992; Gur et al., 2000; Jordan et al., 2002; McBurney et al., 1997; Moffat et al., 1998; O'Laughlin and Brubaker, 1998; Silverman et al., 1999), males would outperform females on a 3-dimensional mental rotation task and 2) males would show greater neural activation when solving mental rotation tasks. We also hypothesized 3) that women would outperform males on a SWM task and that 4) a sex difference in neural activation associated with solving the SWM task would be observed, with females showing significantly more left hemisphere activation. Also because we expected females to show a behavioral advantage in SWM tasks we predicted that (5) females would show left hemisphere activation while encoding stimuli and right hemisphere activation while retrieving spatial information, consistent with the HE/RA hypothesis (Nyberg, Cabeza, and Tulving, 1998).
Experiment 1: Behavioral sex differences in mental rotation and object location memory
Subjects
Sixty-seven (35 males, 32 females; Mean age=20.67; 62 right handed, 5 left handed) Drexel University undergraduate students volunteered to participate in the study for course extra credit. All subjects provided written informed consent in compliance with the University's Institutional Review Board. Subjects participated in two tasks - mental rotation and spatial working memory.
Stimuli and Procedures
Because the goal of this study was to compare sexes on sex-specific spatial cognitive tasks, we made every effort to create stimuli that were as similar as possible in color, cognitive demand, and experimental instructions and procedures. For example, all stimuli were presented as white on a black background and subjects were instructed to respond using same – different discriminations in both experiments. All stimuli (MR and SWM) were presented using Neurobehavioral Systems Presentation version .71 experimental design software (http://www.neurobs.com).
Mental Rotation (MR)
Standard abstract 3-D block design mental rotation stimuli (Shepard and Metzler, 1971) were adapted for this study. Stimuli were scanned into a computer, enlarged, resolution-corrected, and color inverted using Adobe Photoshop Elements. Three classes of experimental stimuli were then constructed and presented in three conditions: 1) spatial: block designs were the same but rotated out of phase (blocks were rotated either 90 or 180 degrees out of phase; see Figure 1a); 2) same: blocks were the same but not rotated (see Figure 1b); and 3) different: two different blocks were presented so that rotation could not produce the same stimuli side by side (see Figure 1c). Subjects were asked to make same-different judgments using the computer mouse. Order of stimuli and button used for same/different was randomized between subjects.

Examples of stimuli used in the MR experiments. Subjects were instructed to respond by making same – different discriminations to three conditions: (a) spatial condition, (b) same condition, and (c) different condition.
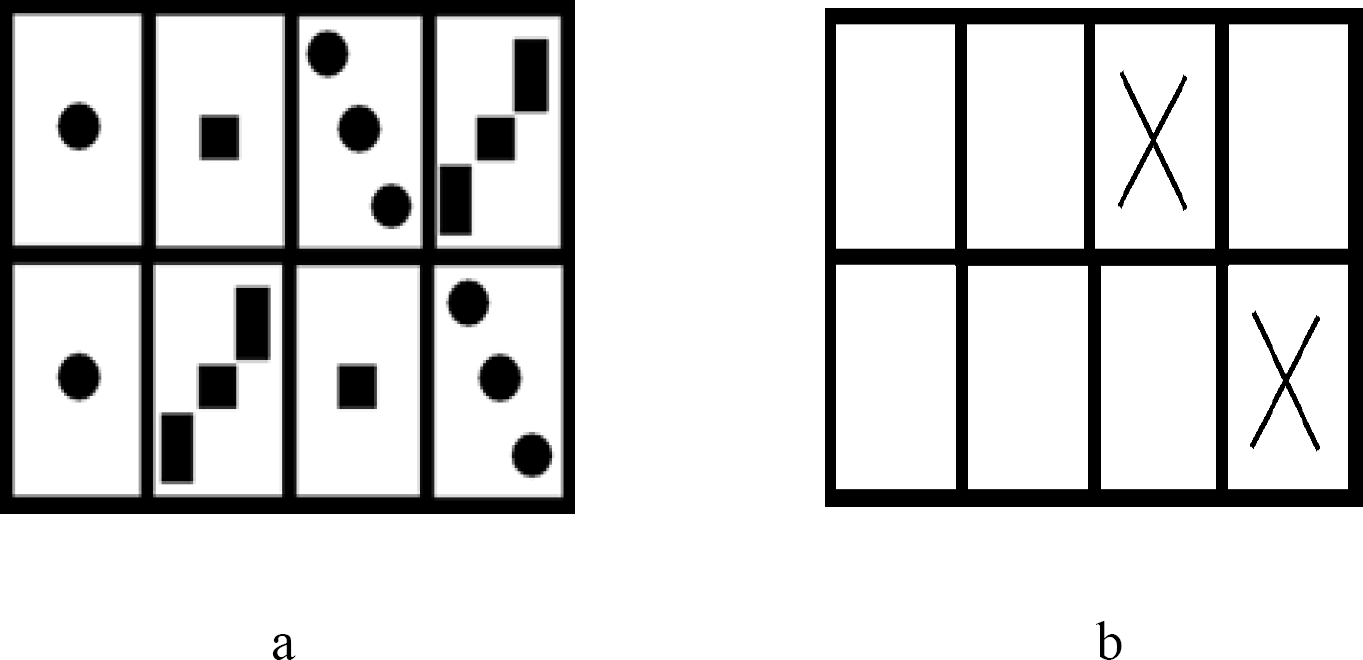
Examples of stimuli used in the SWM experiments. Subjects were instructed to study (try to remember) the location of pairs in (a) study condition. Then subjects were asked to respond making same – different discriminations to stimuli in (b) test condition. Subjects were instructed to choose same when the X's were placed on positions that previously were associated with the same object configurations and different when they did not.

Spatial parametric BOLD activation maps for MR condition contrast spatial vs. same (p<.001, spatial extent = 8 voxels; Red = males; Blue = Females).
Spatial Working Memory (SWM)
Stimuli were created from modified versions of what was described in Duff and Hampson (2001b). Participants were presented with a “study” stimulus for 20 seconds that resembled the game “concentration”. The study stimulus consisted of 8 “cards” that presented 4 pairs of object configurations (see Figure 2a). Subjects were instructed to try and remember the location of the pairs of objects. Immediately after presentation of the study stimulus subjects had their memory for pairs tested. Subjects were presented with an array of 8 white cards two of which had “X” printed on them (see Figure 2b). Subjects were asked to make same – different judgments as to whether those cards with X's on them had the same (or different) objects on the other side. Subjects were presented with 8 test stimuli four of which were correct and 4 of which were incorrect; correct and incorrect cards were presented randomly. After responding to all 8 test stimuli, a new study stimulus was presented (20sec) and followed by 8 new test stimuli. We used five study-test pairs of stimuli. Order of study-test stimuli was randomized between subjects.
Results
Mental Rotation
Twelve subjects' (7 males and 5 females) data were not included in our analyses because of technical difficulties experienced with data collection software and response collection device, which resulted in loss of the data being recorded. Additionally, 6 (3 males, 3 females) subjects who responded incorrectly to every trial of the same (control) condition were removed for probable inattention or misunderstanding of the instructions (e.g., key assignment per condition). We, therefore, present behavioral data for forty-nine (24 females, 25 males) of the 67 originally recruited subjects.
Males responded correctly (73.6%) significantly more often than females (68.4%) (Mann-Whitney U, p<.05). No significant difference was found for reaction time in any condition or collapsed across conditions (all p's >.05).
Spatial Working Memory
No differences in performance or reaction time for the spatial working memory test were found (all p's > .1)
Experiment 2: Neurocognitive sex differences in mental rotation and object location memory (fMRI)
Subjects
Twelve (6 male, 6 female; Mean age = 20.67) Drexel University students volunteered for participation in the fMRI study. Handedness was assessed using a modified version of the Edinburgh Handedness Inventory (Oldfield, 1971). One subject presented as left-handed. We analyzed his data separately and found no significant differences from the right-handed subjects therefore we included this subject in all group analyses.
Stimuli
Stimuli from Experiment 1 were used in Experiment 2. All stimuli were presented using goggles designed for use in the fMRI environment and same – different responses were collected using a response pad designed for use in the fMRI environment (Resonance Technologies). Stimuli were presented using Neurobehavioral Systems Presentation version .71 experimental design software (http://www.neurobs.com).
fMRI Imaging Parameters
Images were collected using a Siemens Magnetom Vision 1.5 T scanner with echoplanar capability (25mT/m, rapid switching gradients). Initially, the scanning began with collection of a contiguous (no gap, whole brain) 26 slice high-resolution T1-weighted imaging sequence acquired in the axial plane to locate the positions for in-plane structural images (matrix size = 256*256; TR (repetition time) = 600 ms; TE (echo time) = 15 ms; FOV (field-of-view) = 21 cm; NEX (number of excitations) = 1; and slice thickness = 5mm). Precise localization based standard anatomic markers (AC-PC Line) were used for all subjects (Talairach and Tournoux, 1988). Next, functional images were acquired with echo planar free induction decay (EPI-FID, T2* weighted) sequence in the same plane as the structural images (128*128 matrix; FOV = 21 cm; slice thickness = 5mm; TR = 4 s; and TE = 54 ms minimum). The size of the imaging voxel was 1.72 mm × 1.72 mm × 5 mm. We collected 109 volumes per condition (MR or SWM) per subject using an event-related design. Head movement was limited by placing foam pads within the head coil and by instructing subjects to lie still throughout the entire experiment. Audio contact with participants was maintained throughout the duration of the study for safety purposes.
fMRI Experimental Design
The study was designed to measure significant changes in blood oxygenation-level dependent (BOLD) signal when solving MR and SWM tasks. We ran two separate series for each experimental condition. Additionally, we investigated whether there are sex-specific BOLD responses during these tasks. An event-related design was used (ISI jittered range: 17 seconds). Subjects were instructed to respond using same – different judgments as quickly and as accurately as possible using the dominant hand in the same fashion as Experiment 1.
Processing and Analysis of fMRI Data
The post-acquisition preprocessing and statistical analysis was performed using SPM′2 (Statistical Parametric Mapping, Wellcome Department of Cognitive Neurology, University College of London, UK), run under the Matlab® (The Mathworks, Inc., Natick, MA) environment. Images were converted from the Siemens format into the ANALYZE (AnalyzeDirect, Inc., Lenexa, KY) format adopted in the SPM package. A 3D automated image registration routine (six-parameter rigid body, sinc interpolation; second order adjustment for movement) was applied to the volumes to realign them with the first volume of the first series used as a spatial reference. All functional and anatomical volumes were then transformed into the standard anatomical space using the T2 EPI template and the SPM normalization procedure (Ashburner, Andersson, and Friston, 1999; Ashburner and Friston., 1999). This procedure uses a sinc interpolation algorithm to account for brain size and position with a 12 parameter affine transformation, followed by a series of non-linear basic function transformations seven, eight, and seven nonlinear basis functions for the x, y, and z directions, respectively, with 12 nonlinear iterations to correct for morphological differences between the template and given brain volume. Next, all volumes underwent spatial smoothing by convolution with a Gaussian kernel of 8 cubic mm full width at half maximum (FWHM), to increase the signal-to-noise ratio (SNR) and account for residual intersession differences.
Subject-level statistical analyses were performed using the general linear model in SPM2. The conditions and the baseline were modeled using a canonical hemodynamic response function with time derivative. Contrast maps were obtained through the following linear contrasts of event stimuli type – for the MR session: spatial vs. same and spatial vs. different; for SWM: study only was modeled as a boxcar function and compared to baseline MR signal, test correct + test incorrect vs. baseline and test correct vs. test incorrect were modeled as events. Group-level random effects analyses for main effects were accomplished by entering whole brain contrasts into one-sample t-tests. Sex differences were investigated by entering whole brain contrasts into two-samples t-tests. A significance threshold based on a t value of >3.17 and spatial extent of 8 voxels was applied to the effects of interest and surviving voxels were retained for further analyses.
Results
Experiment 2a: Behavioral data
One of the fMRI subject's behavioral data was lost due to complications with the interaction of the response pad and the MRI environment and therefore we present behavioral data for eleven (5 females, 6 males) of the 12 subjects.
Mental Rotation
Males responded correctly significantly more often (77.9%) than females (64.2%) (Mann-Whitney U, p<.01). There were no differences in reaction time for any condition, nor was there any effect for reaction time when collapsing across MR conditions (all p's >.05). There was also no significant difference between the results in experiment 1 and those in Experiment 2 (all p's>.05).
Spatial Working Memory
There were no differences in performance or reaction time for the spatial working memory test (all p's > .1), and no significant differences were found between the results in experiment 1 and those in Experiment 2 (all p's>.05).
Experiment 2b: fMRI data
All 12 subjects' (6 female, 6 male) data were included in the fMRI analysis. One-sample t-tests, as implemented in SPM2, were used to identify areas of activation common between sexes. Two samples t-test were computed to identify significant voxels/volumes of interest that differed between the sexes.
Mental Rotation
There were three conditions in the MR study: spatial (blocks with a solution), different (blocks without a solution), and a control condition that consisted of presenting a pair of the same block configurations. When collapsing across sex, the contrast spatial vs. same revealed activation in left inferior occipital gyrus, cuneus, and fusiform gyrus, and right middle occipital gyrus (Table 1).
Local maxima of BOLD changes during mental rotation contrast spatial vs. same when collapsing across sex. Cluster detection corrected p<0.001; spatial extent = 8 voxels.

When investigating sex differences for this contrast, males vs. females revealed activation in right paracentral frontal lobe, medial frontal gyrus, anterior cingulate gyrus, middle temporal gyrus, inferior parietal lobe, and inferior frontal gyrus, and left parahippocampal gyrus and medial frontal gyrus. Females vs. males revealed activation only in the left parahippocampal gyrus (see Figure 3 and Table 2).
Local maxima of BOLD changes during mental rotation contrast spatial vs. same when comparing sexes. Cluster detection corrected p<0.001; spatial extent = 8 voxels.

When collapsing across sex, the contrast spatial vs. different (the tightest contrast) revealed activation in right and left precuneus only (see Figure 4 and Table 3). When investigating sex differences for this contrast, males vs. females revealed activation only in the right hemisphere: middle frontal gyrus, inferior parietal lobe, and inferior occipital gyrus. Females vs. males revealed activation only the right caudate head (Table 4).
Local maxima of BOLD changes during mental rotation contrast spatial vs. different collapsed across sex. Cluster detection corrected p<0.001; spatial extent = 8 voxels.

Local maxima of BOLD changes during mental rotation contrast spatial vs. different when comparing sexes. Cluster detection corrected p<0.001; spatial extent = 8 voxels.


Spatial parametric BOLD activation maps for MR condition contrast spatial vs. different (p<.001, spatial extent = 8 voxels; Red = males; Blue = Females).
Spatial Working Memory
The spatial working memory experiment consisted of two parts: study, which required the subjects to remember the spatial relational positions of objects and test, which tested subjects' ability to recall the location of paired objects. Collapsing across sex, the study condition revealed activation only in the right temporal lobe (Table 5). When contrasting activation of males vs. females, activation in right precuneus, middle frontal gyrus, and middle occipital gyrus, and left precuneus, inferior parietal lobe, medial frontal gyrus, middle frontal gyrus, and superior parietal lobe was observed (see Figure 5a and Table 5). Females vs. males revealed significant activation in right superior temporal gyrus, and left inferior frontal gyrus, superior frontal gyrus, and caudate (see Figure 5a and Table 5).
Local maxima of BOLD changes during spatial working memory study condition collapsed across sex and between sex. Cluster detection corrected p<0.001; spatial extent = 8 voxels.


Spatial parametric BOLD activation maps for SWM experiment. (a) Activation maps for the “study” condition; (b) activation maps for the test correct + incorrect vs. baseline condition, and (c) activation maps for test correct vs. incorrect condition (p<.001, spatial extent = 8 voxels; Red = males; Blue = Females).
In order to examine the activation associated with the test phase of the experiment we conducted two sets of contrasts: 1) test correct condition + test incorrect condition vs. baseline (MR null signal) and 2) test correct vs. test incorrect. Collapsing across sex for the test correct + incorrect vs. baseline revealed no suprathreshold voxels. However, when comparing between the sexes, male vs. female revealed activation in right middle frontal gyrus and parietal lobe. Females vs. males revealed activation in right precuneus, inferior frontal gyrus, cuneus, lingual gyrus, and insula, and left middle frontal gyrus, middle temporal gyrus, posterior cingulate, middle occipital gyrus, parahippocampal gyrus, and middle temporal gyrus (see Figure 5b and Table 6).
Local maxima of BOLD changes during spatial working memory contrast test correct+incorrect-baseline between sex. Cluster detection corrected p<0.001; spatial extent = 8 voxels.

Relationship between activation and performance: Volume of Interest Analysis
When collapsing across sex, test correct vs. incorrect revealed activation in right supramarginal gyrus, superior temporal gyrus, parahippocampal gyrus, middle frontal gyrus, superior frontal gyrus, inferior frontal gyrus, middle occipital gyrus, precuneus, and middle temporal gyrus, and left anterior cingulate gyrus, superior temporal gyrus, middle frontal gyrus, superior parietal lobe, superior frontal gyrus, thalamus, and inferior temporal gyrus (see Table 7). When investigating sex differences, males vs. females revealed activation in right postcentral gyrus, middle occipital gyrus, fusiform gyrus, lingual gyrus, medial frontal gyrus, cingulate gyrus, inferior parietal lobe, and inferior temporal gyrus, and left inferior parietal lobe, inferior occipital gyrus, middle temporal gyrus, middle frontal gyrus, and middle temporal gyrus (see Figure 5c and Table 7). Females vs. males revealed activation in right parahippocampal gyrus and left insula, parahippocampal gyrus and inferior temporal gyrus (see Figure 5c and Table 7).
Local maxima of BOLD changes during spatial working memory contrast test correct vs. test incorrect between sex. Cluster detection corrected p<0.001; spatial extent = 8 voxels.

In order to further investigate the role of certain substrates in spatial cognition we built 4mm spherical volumes of interest (VOI) around several regions using SPM2's VOI tool. We only did this for areas that revealed significant activation in both sexes to sex-specific spatial tasks (e.g., medial frontal gyrus, inferior parietal lobe, middle temporal gyrus, parahippocampal gyrus, and inferior frontal gyrus) and correlated the degree of activation with behavioral performance (number correct). Most of the correlations reported in this section are not statistically significant, which is probably a product of low sample size. Therefore, we only report those correlations that showed a trend toward significance; however, these data should be interpreted with caution.
Mental Rotation
For males, performance (number correct) correlated with BOLD activation in the left parahippocampal gyrus (r=.764, p=.07: see Figure 6) and right middle temporal gyrus (r=.478, p=.3). For females, only the right middle frontal gyrus was correlated with performance (r=.766, p=.13). Figure 6 shows the correlation of performance with activation in sub-regions of the parahippocampal gyrus. Interestingly, regardless of overall activation in these sub-regions of the parahippocampal gyrus, males show a positive relationship with performance.

Degree of activation as a function of performance (number correct responses) in the parahippocampal gyrus during MR for males and females. (Blue = females; Red = males; p<.001, spatial extent = 10 voxels).
Spatial Working Memory
For males, performance correlated with BOLD activation in the parahippocampal gyrus (r=.639, p=.17) (see Figure 7). For females, performance was correlated with activity in the left middle temporal gyrus (r<.05) (see Figure 8).
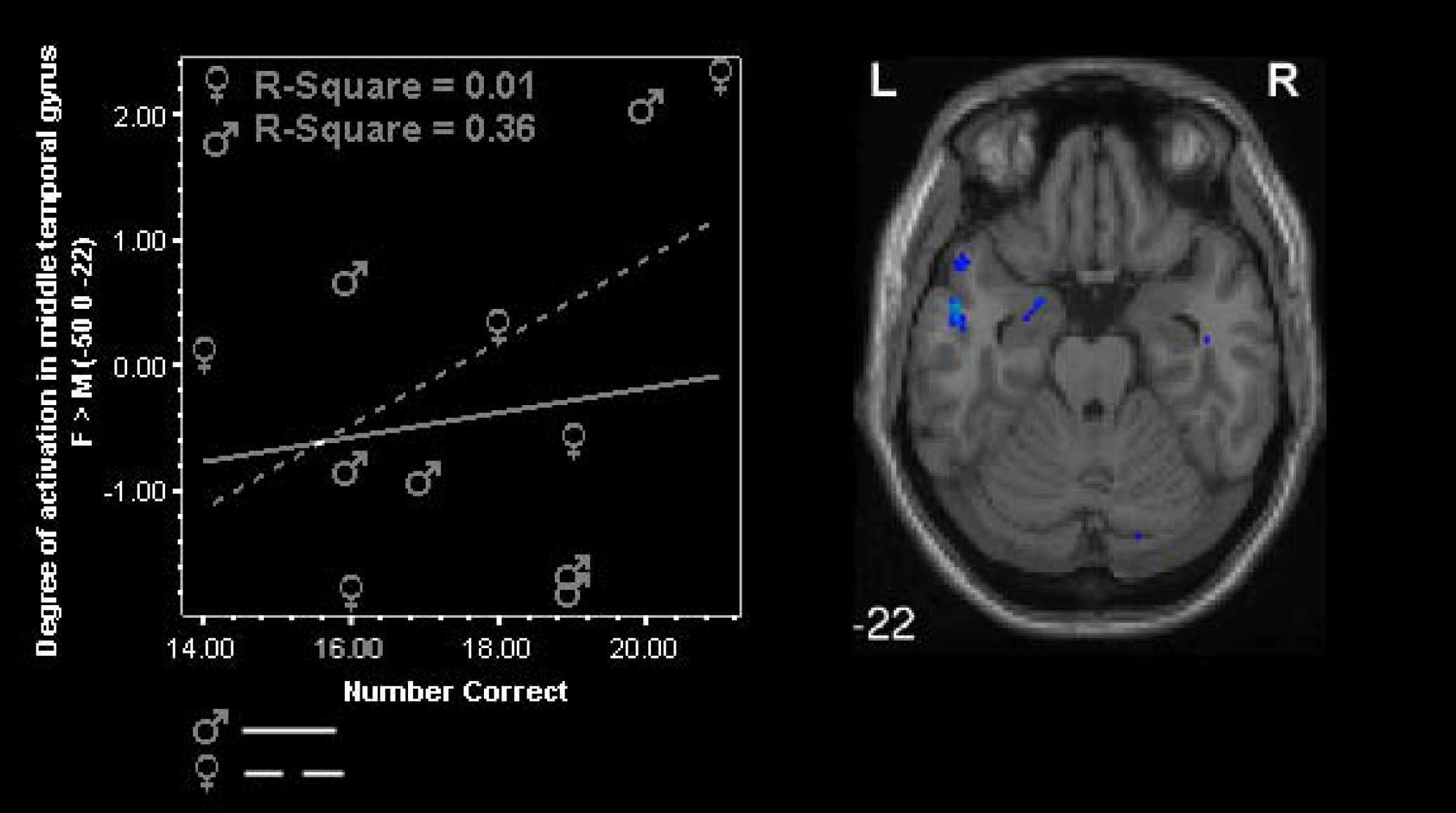
Degree of activation as a function of performance (number correct responses) in the middle temporal gyrus during SWM test correct + incorrect vs. baseline for males and females. (Blue = females; p<.001, spatial extent = 10 voxels).

Degree of activation as a function of performance (number correct responses) in the inferior frontal gyri during SWM test correct + incorrect vs. baseline for males and females. (Blue = females; p<.001, spatial extent = 10 voxels).
Discussion
This study, designed to measure behavioral and neural correlates of sex differences in spatial cognition, supports previous research suggesting a male dominance for mental rotation tasks (Geary et al., 1992; Gur et al., 2000; Jordan et al., 2002; McBurney et al., 1997; O'Laughlin and Brubaker, 1998). Although we were not able to replicate a performance difference in spatial working memory, as has been demonstrated in the literature, these data do extend previous claims for sexually differentiated performance in spatial cognitive abilities by demonstrating neurocognitive sex differences. The discovery that males and females appear to employ left parahippocampal gyrus to solve tasks that are associated with behavioral advantages (i.e. MR for males and SWM for females) is novel. Interestingly, left parahippocampal gyrus correlated with increased performance in the mental rotation task for males, but was inversely correlated for the spatial working memory task in females. Both males and females also showed significant activation in the right inferior frontal gyrus when responding to sex-specific spatial tasks. This finding suggests that each sex may capitalize on retrieval mechanisms for previously encoded spatial episodic memories, which supports the notion that retrieval of spatial information is in part related to hemispheric specificity (Jordan et al., 2002). The exact role each of these regions plays in sex-specific spatial cognition is unknown, and an obvious avenue for further research.
For the male dominated spatial task, we found sex differences in behavioural performance and BOLD response activation associated with mental rotation (see Figure 9 for summary of activation). When comparing for sex differences, males showed significantly more cortical activation over the whole brain (p<.001; 8 voxel cluster; see Figure 1 and Table 1) than females when solving mental rotation (MR) tasks. Specifically, males revealed more activation in the medial frontal lobes as well as regions of the right inferior parietal lobe, right inferior frontal gyrus, and left parahippocampal gyrus. Activation of right inferior parietal lobe and inferior frontal gyrus, is consistent with previous literature investigating MR (Gur et al., 2000; Jordan et al., 2002; Thomsen, Hugdahl, Ersland, Barndon, Lundervold, Smievoll, Roscher, and Sundberg, 2000).

Summary of activation for the Mental Rotation Task
Activation in the inferior parietal lobes is the most common finding and suggests that this area holds specific importance for male spatial cognition. It is likely that the inferior parietal lobe which allows for mental representation of self (Jackson and Decety, 2004; Platek, Keenan, Gallup, and Mohamed, 2004) is also responsible for self-relation (egocentricity) to objects in space that can then be mentally manipulated (rotated). This process likely works in concert with regions in the lateral (inferior) and medial frontal lobes (i.e. mirror neuron system and social cognitive systems) and may be related to the hypothesis of spatial cognition via egocentric processing (Chan, Peelen, and Downing, 2004; Committeri et al., 2004; Platek, Loughead et al., in press; Seurinck, Vingerhoets, de Lange, and Achten, 2004). Cortical midline structures, such as the medial frontal gyrus have been implicated in self-representation in space and time (e.g., Northoff and Bermpohl, 2004; Platek, Thomson, and Gallup, 2004; Platek, Mohamed, and Keenan, in press; Vogeley et al., 2001). This would suggest that solving spatial cognitive tasks that deal with mental rotation or navigational skills is related to some type of mental motor cognition – mentally simulating actions.
The relationship between spatial processing, motor cognition and mental state prediction/representation may represent a specific functional network of neural substrates that would have conferred an advantage to males in learning and/or developing navigational skills, thus increasing cognitive efficiency while engaging in these tasks. In other words, integration of systems that allow for efficient spatial and motor processing could benefit learning spatial navigational routes, while integration of mental state predictions and spatial cognition might have aided combat techniques.
Interestingly, we also found activation in the parahippocampal gyrus, a region that has received little attention in the spatial cognition literature. The parahippocampal gyrus is, however, tightly linked in a neuroanatomical loop with the hippocampus and the entorhinal cortex, both of which have been implicated in spatial cognition in animals (Committeri et al., 2004; Deacon and Rawlins, 2005; Geary, 1995; Heilman, Jeong, and Finney, 2004; Jones, Braithwaite, and Healy, 2003; Lacreuse et al., 1999; Poti, 2004) as well as the inferior parietal lobe, which has been implicated in spatial cognition in humans (e.g., Jordan et al., 2002). Furthermore, we found that performance (number correct) in males was correlated greatest with activity in this region (see Figure 6), even in regions of the parahippocampal gyrus that females showed more overall activation during MR tasks (see Figure 6). This suggests that this region may be an integral substrate that allows males to easily maximize and implement their navigational abilities and may have been subjected to selective pressures as a result. Investigation of patients with specific lesions to this area may help identify the extent to which this region is involved in MR.
BOLD activation associated with spatial working memory (SWM) was also found (see Figure 10 and 11 for summary of activation). When comparing for sex differences, females showed significantly more cortical activation (p<.001; 8 voxel cluster) over the whole brain when solving SWM tasks. Females revealed significantly more activation in the areas of the frontal gyrus as well as regions of the temporal lobe and parahippocampal gyrus. Specifically, females employ left inferior frontal gyrus while encoding an array in the study condition and show activation in the right inferior frontal gyrus while retrieving information in the test condition. This is consistent with literature on short-term episodic memory encoding and retrieval (Nyberg et al., 1998). However, we also showed left parahippocampal activation when contrasting females with males on SWM, which is similar to the activation we found when contrasting males with females on the MR task. One could speculate that the left lateralized activation could be due to an internal verbalization strategy, however the current study does not provide evidence for or against this claim.

Summary of activation for the Spatial Working Memory Task: Study Phase
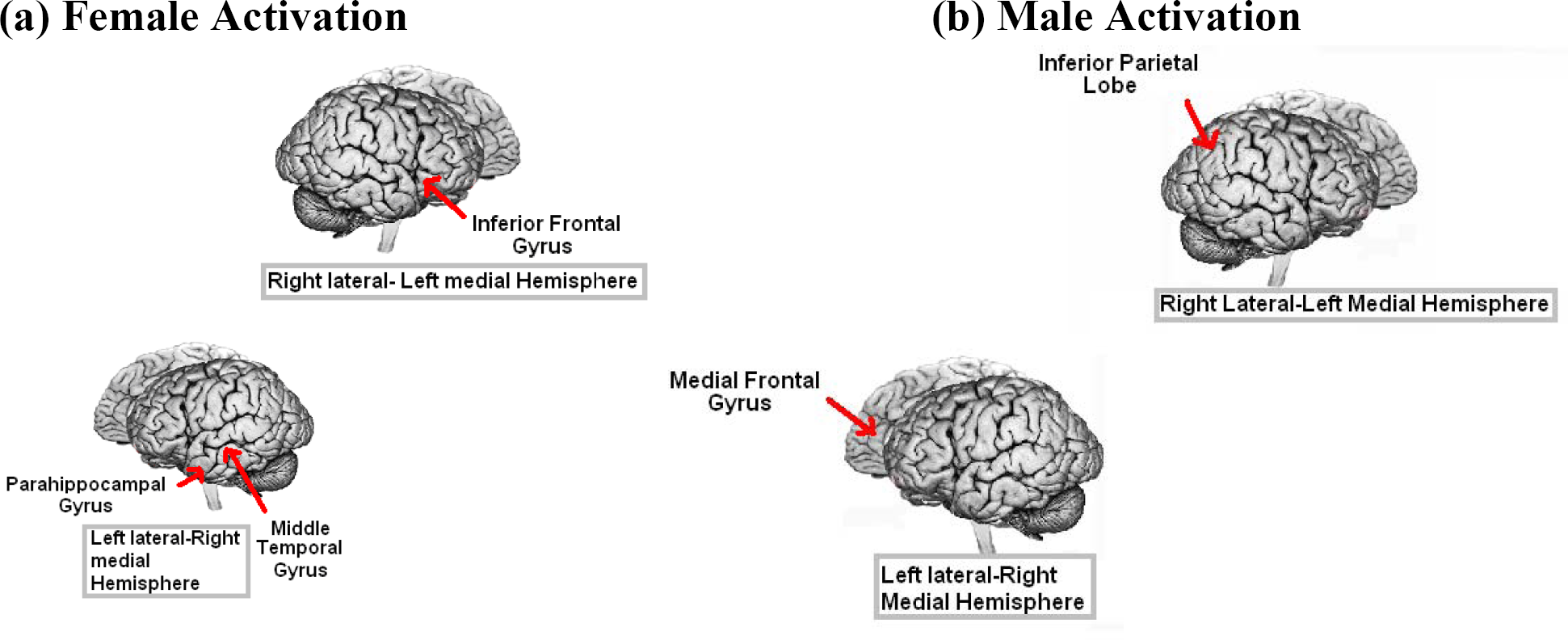
Summary of activation for the Spatial Working Memory Task: Test Phase
These findings support a hypothesis that males and females use similar cortical structures when solving sex-specific spatial tasks. Males showed a lateral shift in cortical activation, similar to that found in MR tasks, for the study condition of the SWM task: left inferior parietal lobe and medial frontal gyrus. Interestingly, there is no significant left inferior frontal gyrus activation in males, which females appear to use during the encoding portion of this task. This suggests that sex differences in this task might be related to poor encoding ability by males of spatial object locations, but this hypothesis demands further investigation to clarify this issue.
The correlation found between performance on SWM tasks and middle temporal gyrus and inferior frontal gyrus in females suggests that increased activation in middle temporal gyrus may improve female performance, while activation in the inferior frontal gyrus may be a measure of cognitive demand. In other words, since females showed a negative relationship between performance and activation in the inferior frontal gyrus, perhaps females employ the inferior frontal gyrus only when they have to work harder to produce a response. This notion is speculative and demands further research.
When analyzing the data between sexes at a macro level, it appears that females employ left hemisphere substrates, primarily frontal gyrus activation when solving both types of spatial tasks – MR and SWM. While males, conversely, showed more activation in right hemisphere substrates, primarily parietal regions (see also Weiss et al., 2003).
Unique is the discovery that males and females appear to employ a number of similar neural substrates, and perhaps cognitive strategies, across mental rotation and spatial working memory tasks. It is possible that males' disadvantage in solving spatial working memory tasks (Duff and Hampson, 2001a; Eals and Silverman, 1994; James and Kimura, 1997; McBurney et al., 1997; Silverman and Eals, 1992; Tottenham et al., 2003) is due to their strategy and recruitment of specific neural substrates. They may be “stuck in the parietal lobe,” or unable to encode the same mental object memory template that females may be creating, which appears to be related to frontal activation. The decreased performance on mental rotation tasks for females may be due to the lack of lateral shift as indicated by Gur et al. (2000), but also because of an inability to activate extended areas of the parahippocampal gyrus (i.e. no significant activation in the inferior parietal lobe or medial frontal gyrus).
This study had several limitations. First, these data should be interpreted with caution due to our small sample size. Second, the use of more ecologically valid stimuli in the SWM task (e.g. faces, everyday objects) may have produced the behavioural sex difference seen in other studies (e.g., Silverman and Eals, 1992). Our use of a small object matrix size (8 “cards”) may have limited our ability to detect sex differences. The use of larger object matrices, or a number of differing object matrix sizes may be a more sensitive test of sex differences in spatial working memory and may allow for systematic psychophysical investigation of spatial working memory. Future studies would benefit from systematically controlling for object matrix size and stimuli type. This approach would allow for a better understanding of the neurocognitive strategies involved in, and the sex differences associated with spatial working memory. The lack of a behavioural performance difference in the SWM portion of this study may be explained by this lack of sensitivity. The MR task could be modified to resemble the SWM task by presenting pairs of 3-D block designs in a memory task that entails a level of encoding beyond the current implementation of the task. We were also limited in our power to detect significant activation by performance effects; an increased sample size in future studies could rectify this issue. Finally, controlling for egocentric processing/self-referent mental simulation may help elucidate the role of self-processing in spatial cognition.
In conclusion, we found common activation in sex-specific spatial abilities in the areas of the parahippocampal gyrus, middle temporal gyrus, inferior frontal gyrus, and medial frontal gyrus. This suggests that males and females may use similar cortical regions when solving spatial tasks at which they tend to excel. These data, and the cross-species findings of these areas being involved in spatial cognition, suggests that these regions may have been a substrate of sexual selection for sex differences in spatial cognition. In humans, it appears that a neural network that includes the parahippocampal gyrus, middle temporal lobes, parietal lobes, and the medial frontal lobes has been selected to allow each sex to adequately engage in spatial cognitive tasks that provided adaptive survival advantages for that sex. Additionally, males employ a spatial network that may recruit the so-called mirror neuron system that may create a mental simulation of motor-based spatial/navigational maps.
Footnotes
Acknowledgements
The authors thank Danielle Raines, Thomas Myers, Jaime Thomson, and Scott Faro for their assistance with data collection. The authors also thank Steve Gaulin for his assistance with stimulus design. Portions of this work were funded by a Neuroscience Research Fund donation from Hans and Dolores Levy.
