Abstract
Structural and social determinants of health are environmental conditions that affect individuals’ health and functioning across the life course. These determinants are salient factors at the apex of individual psychology, community variables, and larger sociological factors, exerting prominent effects on individuals’ wellbeing. The National Institute on Aging seeks to understand their influence on aging and Alzheimer’s disease. We propose that the benefits of doing so may reach beyond knowledge of disease mechanisms – it may also improve clinical outcomes. We describe efforts introducing structural and social determinants of health data into the national network of Alzheimer’s disease research centers. We report results of pilot testing 225 structural and social determinants of health items at the University of Pennsylvania’s center among 135 cognitively unimpaired research participants. We summarize item and instrument completion rates, participant feedback, and survey adjustments. We describe the initiative, potential for its impact, pilot testing results, and future directions.
Keywords
Structural and social determinants of health are environmental conditions in daily life that affect an individual’s health and wellbeing (U.S. Department of Health and Human Services, 2023; World Health Organization, 2010). The relevance of structural and social determinants of health is recognized across the globe, including in England (Watson et al., 2021), Sri Lanka (Jayakody & Arambepola, 2022), Hong Kong, Japan, South Korea, Taiwan, and Thailand (Kang et al., 2023), South Africa (Trani et al., 2022), Cuba, Dominican Republic, Mexico, and Peru (Llibre-Guerra et al., 2024), and the United States (US) (Idorenyin Imoh & Charity, 2023). Structural and social determinants of health were codified into US policy in 1980, when they were used to ground Healthy People, the United States’ plan for addressing preventable threats to population health and wellbeing (Centers for Disease Control, 2022). During the decades that followed, evidence and interest has grown in considering the function of structural and social determinants of health in models of population health and disease (Andermann, 2016; Braveman & Gottlieb, 2014).
Structural and social determinants of health are often anchored to individual and group identities defined by age, sex, race, sexual orientation, gender identity, disability status, learning differences, and other groupings. Structural and social determinants of health reflect social pathways consisting of complex interplays between individuals and society, which vary with time and place. Structural and social determinants of health and their effects in dementia may be best considered and addressed at the individual, family, and societal levels (Stites et al., 2021).
Social determinants include aspects of the social environment (e.g., education years) and physical environment (e.g., built neighborhood). Structural determinants refer to social processes (e.g., employment), systems (e.g., healthcare), and policy (e.g., financial practices). Social determinants comprise an individual’s immediate surroundings, such as family and healthcare team, while structural factors include society level factors, such as healthcare access and insurance coverage. Ecological Systems Theory poses that these are interconnected environmental systems that shape human development, whereby individuals’ immediate surroundings form the microsystem and mesosystem (Bronfenbrenner, 1992). Broad societal contexts form the macrosystem, and the influence of time and historical events are called the chronosystem. While Ecological Systems Theory focuses on early development life stages, the systems of interaction that it postulates and premise that these systems shape experience are relevant to diagnosis, treatment, care, and wellbeing in dementia.
Emerging evidence suggests that structural and social determinants of health explain heterogeneity in aging trajectories and health-related patient outcomes (Adkins-Jackson et al., 2023; U.S. Department of Health and Human Services, 2023; Wang et al., 2019). Previous research has demonstrated the influence of seven structural and social determinants of health domains on aging and Alzheimer’s disease outcomes (Stites et al., 2021) including: (1) education –years (Hill et al., 2015), quality (Chin et al., 2012), and attainment (e.g., undergraduate degree) (James et al., 2012); (2) occupation –type (Qiu et al., 2003; Stern et al., 1994) and conditions (Marquez et al., 2010); (3) socioeconomic positioning (Greenfield & Moorman, 2019; Hill et al., 2015); (4) social stressors and perceived stress (Shields et al., 2017; Zuelsdorff et al., 2020); (5) social support and social positioning (Amir et al., 2019; Hill et al., 2015; Rote et al., 2021); (6) built environment\neighborhood (Besser et al., 2019); and (7) social identity, such as age, race, ethnicity, language, disability, sexual orientation (Flatt et al., 2018); sex and gender (Alzheimer’s Association, 2021; Stites, 2020) –with each serving as both a group identity and structural determinant (See Stites & Velocci, 2024 for further discussion).
The discoveries from investigating structural and social determinants of health have the potential to move structural and social determinants of health from a sociological concept into a recognized part of the Alzheimer’s disease medical model. In clinical practice, providers could routinely consider the relevance of structural and social determinants of health in diagnosis, treatment, and care of individuals with Alzheimer’s disease and their care network. In addition to disease-modifying medications, clinicians could also recommend behavioral and cognitive therapies to lessen the negative effects of stress related to Alzheimer’s disease and the cognitive and functional impairments it causes. Furthermore, they could partner with patients to complete genograms to identify and organize social networks to distribute care responsibilities and develop targets for system- and practice-level interventions to address barriers to clinical presentation, diagnosis, and care.
Moreover, medicine is influenced by systemic and structural factors that shape the access and delivery of care for individuals from different groups. For example, racial discrimination from health professionals has been shown to negatively impact the perceived quality of care given to African-American and Hispanic individuals (Sorkin et al., 2010). Sexism persists in the form of underrepresentation of females in biomedical research and poor reporting and investigation of sex and gender-related effects in health and disease, feeding into patterns of misdiagnoses and treatment delays for female patients (Merone et al., 2022). People from sexual and gender minoritized communities have reported being discriminated against in healthcare settings, leading to mistrust of medical providers, avoidance of seeking care, and diminished mental and physical health outcomes (Ayhan et al., 2020). The many forms of bias permeating the medical system must be addressed as part of addressing structural and social determinants of health with the individuals who are accessing care and treatment for dementia. Collecting data on structural and social determinants of health can be an early stepping stone towards prioritizing the needs of distinct social groups to ensure healthcare is accessible and effective for all. Uncovering structural and social determinants of health that may impact individuals from given populations can be foundational to determining how healthcare can be improved to be less biased and more equitable. Stated more starkly, collecting structural and social determinants of health without a steadfast goal to use those data to correct systemic bias risks compounding existing harms.
To achieve these benefits, scientists and clinicians alike must understand structural and social determinants of health mechanisms and their modifiers to translate them into clinical practice. The National Institute on Aging Alzheimer’s Disease Research Centers are the ideal setting to do this, as they comprise a discovery network with multidisciplinary scientific expertise positioned to make breakthroughs in empirical understandings of Alzheimer’s disease mechanisms (National Institute on Aging, 2023).
The addition of structural and social determinants of health to the large repertoire of data collected and analyzed in the Alzheimer’s Disease Research Center network is expected to advance scientific discoveries and assure the place of structural and social determinants of health in the translation of those advances into the diagnosis, treatment, and care of older adults. However, we must first understand needed data regarding the process of implementing measures of structural and social determinants of health into Alzheimer’s Disease Research Center study cohorts. The purpose of the current study was to pilot test structural and social determinants of health measures included in one Alzheimer’s Disease Research Center, where study cohort participants undergo comprehensive clinical, genetic, and biological testing (National Institute on Aging, 2023). If successful, the rollout of the measures across the Alzheimer’s Disease Research Center network will garner data on sociological effects in older individuals. These data will allow a more complete understanding of Alzheimer’s disease mechanisms, which is expected to inform novel interventions that can more effectively optimize aging outcomes.
Background
National Institute on Aging Support and Infrastructure
The National Institute on Aging is prioritizing funding for the collection and study of structural and social determinants of health data (National Institute on Aging, 2021; National Institutes of Health, 2023a). To date, efforts to collect structural and social determinants of health in Alzheimer’s disease research have been supported largely by independent investigator research grants. This has generated a tapestry of data. However, with broad organizational support from the National Institute on Aging, cogent longitudinal data can be collected across larger samples in the national network of more than 30 Alzheimer’s Disease Research Centers. Soon, a module of 40 structural and social determinants of health questions will be included in the Alzheimer’s Disease Research Center’s uniform data set. The data presented herein contain pilot information on 25 of the 40 questions.
Alzheimer’s Disease Experience
Alzheimer’s disease is one of several causes of dementia, which refers to a syndrome of impairments in cognition and function. Discoveries made in Alzheimer’s Disease Research Centers have aided in identifying groups of proteins considered to cause Alzheimer’s disease dementia, including amyloid and tau (Jack et al., 2018; Pereira et al., 2021; Snitz et al., 2015). Adding structural and social determinants of health into Alzheimer’s Disease Research Centers will move this science forward via three specific benefits. First, incorporating investigation of structural and social determinants of health into Alzheimer’s Disease Research Centers may aid in understanding mechanisms by which population-level sociocultural factors affect Alzheimer’s disease outcomes. For example, research indicates that Black populations meet clinical criteria for Alzheimer’s disease dementia at higher rates, are more likely to be undiagnosed or diagnosed at later stages of disease, receive lower quality of care, and have worse mortality rates than their White counterparts (Lennon et al., 2021; Lines & Wiener, 2014).
Second, the process will bring attention to the representation of lived experiences of research participants. Some ways that Alzheimer’s disease biomarkers and other outcomes differ across populations may be attributable to the underrepresentation of social groups in research on the topic. Alzheimer’s disease research samples are disproportionately comprised of White individuals (Lim et al., 2023). Discoveries made with data from those samples may not be applicable to Black populations and other groups not represented in the samples (Barnes, 2022; Barnes & Bennett, 2014; Mehta et al., 2008). Additionally, the specific focus on structural and social determinants of health in Alzheimer’s Disease Research Centers may aid in making research settings and practices more welcoming and inclusive to broader groups of people.
Third, a major focus of Alzheimer’s disease research is to transform clinical syndromes to biologically defined and treatable diseases (U.S. Department of Health and Human Services, 2022). Because disease biomarkers can be detected at any stage of disease, even in persons not yet experiencing symptoms, the continuum of cognitive and functional impairment that can describe a person with Alzheimer’s disease is widening. On one end, it can include individuals in the earliest disease stages having Alzheimer’s disease biomarkers – such as amyloid and tau (Jack et al., 2018) – without dementia (Sperling et al., 2014). They can be people employed or volunteering in the workforce and who provide social resources for younger members of their families. On the other end of this continuum are individuals in late-stage Alzheimer’s disease, who may require intensive assistance with activities of daily living (Bonnel, 1996). In turn, there is a diverse range of social experiences that can also fall under the umbrella of “living with Alzheimer’s disease”. The widening continuum of experience underscores the importance of understanding influences of structural and social determinants of health in aging and Alzheimer’s disease and, when needed, addressing structural and social determinants of health in the relevant clinical settings.
Health Disparities Framework and Fundamental Cause Theory
A framework, with measures, was developed for gathering structural and social determinants of health data in Alzheimer’s Disease Research Centers (Stites et al., 2021). The choice of measures was informed by extant literature on known social disparities in Alzheimer’s disease patient outcomes, as well as Bronfenbrenner’s Ecological Model, which proposes individuals’ outcomes arise from a complex system of relationships affected by multiple level experiences, and Fundamental Cause Theory, which explains the consistent association between socioeconomic status and health across time, place, and outcomes (Link & Phelan, 1995). The approach is also informed by the National Institute on Aging’s diversity and disparities framework (Hill et al., 2015), which emphasizes unifying existing measures, setting research priorities across the centers, adopting a life-course perspective, and identifying opportunities for intervention.
Outcomes and Mechanisms
Advancing knowledge of structural and social determinants of health in Alzheimer’s disease will aid in elucidating disease mechanisms and discovering therapies (Hill et al., 2015; National Institute on Aging, 2020). This line of research and its embedment into the major infrastructure for research discoveries also has the potential to beneficially transform the experience of living with Alzheimer’s disease. By studying structural and social determinants of health as a component of the scientific discovery process, it may be possible to be cognizant and intentionally direct social perceptions of the disease, its diagnosis, and treatment. The approach could also aid in addressing healthcare disparities earlier in the process than is currently the case (Cabin & Stites, 2021).
The medicalization of conditions has been shown to shape stigma and discrimination associated with diseases. In fact, the greater the recognition of a condition as having a biological underpinning and more sensitive and specific the testing is in identifying it, the worse the stigma (Canadian Health Services Research Foundation, 2013; Loughman & Haslam, 2018). Studies of imaging and other biomarkers in Alzheimer’s disease have demonstrated this (Stites & Coe, 2023; Stites et al., 2018, 2022). By including structural and social determinants of health early in the scientific discovery process, it may be possible to address social and structural factors – i.e., discrimination, institutional and attitudinal barriers – that may make living with the disease an impediment to participating in social and political life.
Study Team
We, a group of self-selected Alzheimer’s Disease Research Center researchers, set out to establish collection of a core set of structural and social determinants of health data in the National Institute on Aging’s Alzheimer’s Disease Research Centers (Stites et al., 2021; Streitz et al., 2022). The study team reflects multidisciplinary scientists with expertise in diverse areas, all with interest and applicability to aging and dementia. These areas of expertise include Alzheimer’s disease etiology and neurocognitive assessment, social cognitive processes, research methodologies, clinical practice and care of dementia, family and informal caregiving, and psychosocial experience. The combined lived experience of the team includes diverse racial, sexual, gender, national and linguistic identities; micropolitan and rural geographic areas; educational pathways; and socioeconomic backgrounds.
In addition to building diverse study teams, engaging impacted groups in the collection, analysis, and reporting of structural and social determinants of health data are also crucial steps to the pursuit of appropriate use of structural and social determinants of health data. It is essential, for example, when engaging individuals who identify as American Indian, Alaskan Native, or Native Hawaiian that indigenous data sovereignty and governance principles are followed. Information on the CARE Principles for Indigenous Data Governance and published frameworks on access and use of racialized data are provided in Online Appendix A. These frameworks may serve to guide the collection, use, and interpretation of data.
Study Purpose
This article reports results of piloting the structural and social determinants of health measures in an Alzheimer’s Disease Research Center clinical research cohort, which is essential to supporting the successful ongoing and expanded collection of these measures. We report on the feasibility and acceptability of administering structural and social determinants of health questions in a cohort of adults ages 65 and older. We present basic descriptive data on the measures to inform design of future studies, which will further support the development of this line of research. We summarize participant response patterns and answers to quality assurance questions. We describe the changes that were made in response to participant feedback. Our results offer data to inform collection of structural and social determinants of health data in aging and Alzheimer’s disease research cohorts and pilot data to guide future studies.
Embedding structural and social determinants of health measures into an Alzheimer’s Disease Research Center cohort presents substantial challenges but also potential for major benefits. To realize the ambitious goal of incorporating structural and social determinants of health into Alzheimer’s disease research and care models, we needed to develop a set of measures and an administration protocol that: (1) was feasible in the Alzheimer’s Disease Research Center, which has capitations in the type, volume, and complexity of data that can be collected due to a need to minimize participant burden and the availability of personnel resources; (2) would have strategies in place for harmonizing multiple data sources, which is essential for robust study of structural and social determinants of health to generate sample sizes sufficient to examine population subgroups and discrete experiential interactions; (3) could make the data widely-available to researchers across the nation and, potentially, the globe; (4) would use an instrument that could capture current and life course experiences of older adults and do so in a culturally acceptable and scientifically useful fashion; and (5) could be accessible and understandable to a diverse range of investigators who may not have prior experience with human subjects data or sociological data.
Methods
Design
This study reports results of pilot testing routine collection of structural and social determinants of health data in a research cohort of older adults. Data were collected between December 9, 2021 and March 14, 2022. We report response and completion rates, participant feedback, and modifications based on that feedback.
Participants
Study Participants
Participants were recruited from the Alzheimer’s Disease Research Center at the University of Pennsylvania clinical core cohort, including those who are co-enrolled in the Aging Brain Cohort Dedicated to Diversity study. The Aging Brain Cohort Dedicated to Diversity study is funded by a grant from the Pennsylvania Department of Health to increase diversity in research cohorts and aims to enroll an additional 160 Black adults from the Philadelphia region.
Study Eligibility
Eligible adults were ages 65 and older, native English speakers, who were enrolled in the above longitudinal studies. All participants were required to have a study partner. All participants with unimpaired cognition were invited to participate in this pilot study. After completion of data collection in cognitively unimpaired participants, data collection began in individuals with mild cognitive impairment (MCI).
Participants’ cognitive status was determined by expert consensus conference. Participant data from their annual National Alzheimer’s Coordinating Center visit were reviewed. This included data from the uniform data set, version 3 (Besser et al., 2018), clinical dementia rating® scale, a neurological exam, and several questionnaires of emotional, behavioral, and everyday functioning. Participants were deemed to have unimpaired cognition if their performance on age and education normed neuropsychological testing was within normal limits, they were functionally intact, and they did not meet clinical criteria for either Mild Cognitive Impairment or Alzheimer’s disease dementia. Mild Cognitive Impairment and Alzheimer’s diagnoses were defined based on the National Alzheimer’s Coordinating Center and Petersen criteria (Petersen et al., 1997; Shiloh, 2006) and National Institute of Neurological and Communicative Disorders and Stroke and the AD and Related Disorders Association (McKhann et al., 1984).
Interested Groups or Parties
This work reflects the contributions of many groups. Content was informed through collaboration with the Community Advisory Boards at the University of Pennsylvania Alzheimer’s Disease Research Center and Knight Alzheimer’s Disease Research Centers as well as the workgroups on structural and social determinants of health and sex, sexual orientation and gender identity measures that were part of the efforts to revise the National Alzheimer’s Coordinating Center’s uniform data set, version 4 (National Alzheimer’s Coordinating Center, 2023).
Data Collection: Design and Rollout
The survey was designed in REDCap, a Health Insurance Portability and Accountability Act of 1996 (HIPAA) compliant application used to collect data for databases and projects using surveys that can then be transferred to statistical programs (Patridge & Bardyn, 2018). Following the annual visit and consensus conference, eligible participants were sent a survey invitation via REDCap introducing the survey. The invitation was linked to a unique participant identifier. Reminder emails were automatically sent via REDCap one week and two weeks after the first invitation. Participants who did not complete the survey using the REDCap link after the second reminder were offered options to complete the survey in-person during a routine follow-up visit. This option also included a choice of iPad or paper versions. Data from various modalities could be linked by study coordinators using QR codes generated in REDCap. In addition, the option to have a proxy answer the survey on the participant’s behalf was available in each modality of data collection.
Because the acceptability of scientific measures is often known only for a narrow field of social and clinical groups, we rolled out distribution of the survey slowly, sending it to a few participants to begin, where we could closely monitor how questions were received and follow up with participants once they did or did not complete the survey to understand their experiences. We made adjustments to the survey based on these data. This close monitoring applied for about the first 10 to 20 participants. Data collection was accelerated to routine collection once we could identify no further changes or problems. Psychologists on the study team were available to assist participants who may have unexpected distress. Other support services, such as mental health resources, were also readily available and accessible via an affiliated social work team. During this rollout, we extended the preamble instructional text for each section to give participants more advanced notice of topics. Additionally, our Alzheimer’s Disease Research Center hosted monthly Advisory Board meetings where members of our research team would present our recruitment, retention, and research ideas for feedback from our advisory board members-research participants, former employees, and community members. During one of our meetings, we consulted with the advisory board about the survey.
Data Storage and Access
Data are cleaned and transferred from REDCap to the Integrated Neurodegenerative Disease Database (Xie et al., 2011). A custom Application Programming Interface links the survey data with National Alzheimer’s Coordinating Center questionnaires and biomarker data. While initially being made available only with the Alzheimer’s Disease Research Center network, the data will be made available in the future via an Alzheimer’s Disease Research Center data request process (https://www.med.upenn.edu/adrc/datasharing.html). A public folder, located at https://upenn.box.com/s/zgo58re9netwxphbiixthjmjhsg6cae0 is updated regularly and includes the Life Experiences Survey, the REDCap codebook and a read me file. The steps in data acquisition and storage are described in Figure 1. Steps in Data Acquisition and Storage of Structural and Social Determinants of Health Data in the UPenn Alzheimer’s Disease Research Center (ADRC)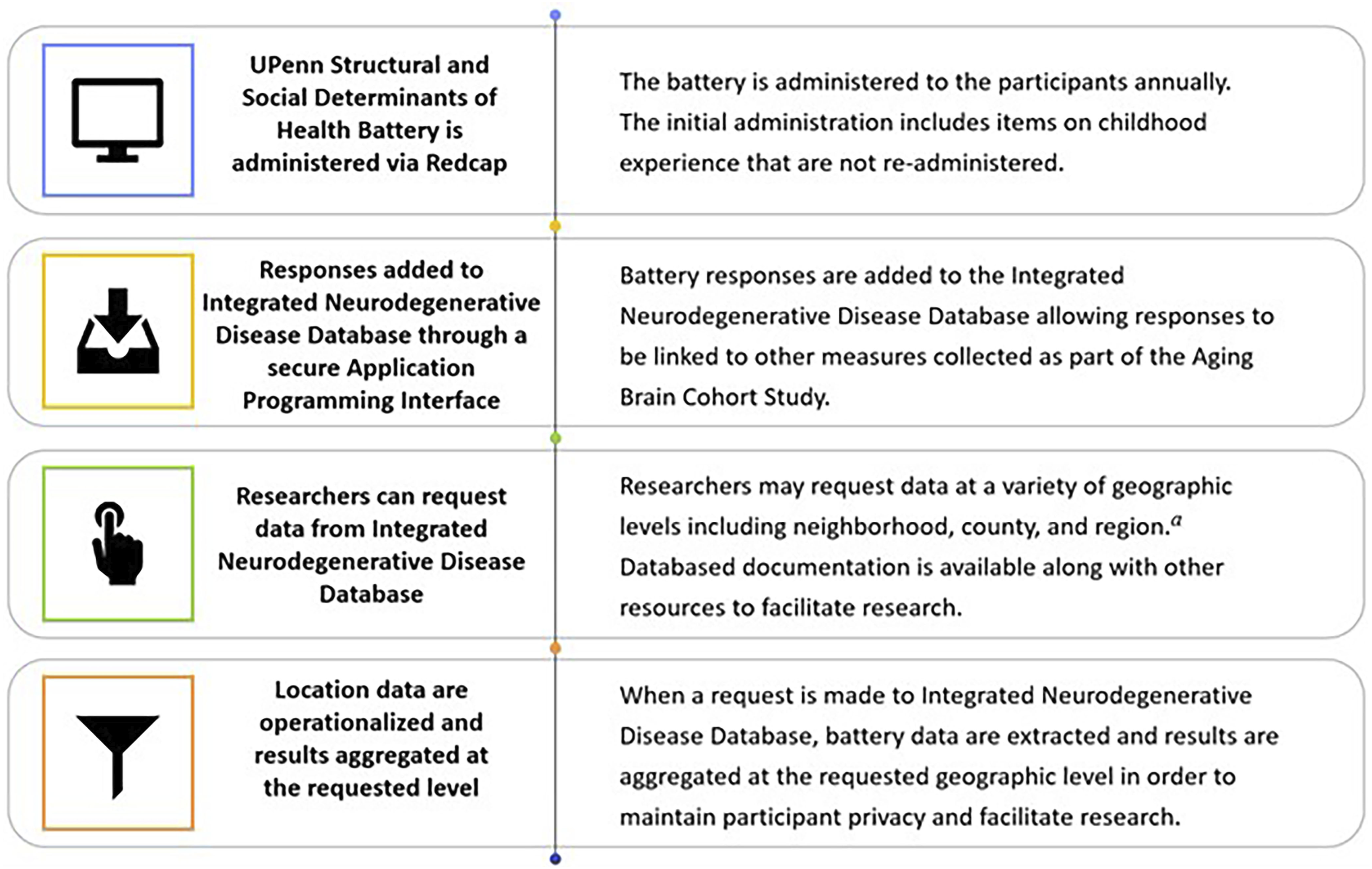
Measures
Social Identity
Basic demographic characteristics, including age, sex and race, were gathered using the National Alzheimer’s Coordinating Center Uniform Data Set A-1 Subject Demographics form (National Alzheimer’s Coordinating Center, 2015). Additional demographic characteristics were gathered using questions about marital status and living situation (Beekly et al., 2007). Sex, sexual orientation and gender identity were assessed using three questions asking sex assigned at birth, self-defined gender identity, and current sexual orientation (Flatt et al., 2022). Language was assessed using questions from the Home Language Survey Data Quality Self-Assessment. This consisted of three questions about primary language spoken, language most often spoken, and first language learned (Henry et al., 2017). The full survey is in Online Appendix B.
Disability status was assessed with six items from the Washington Group on Disability Statistics short set on functioning (Washington Group on Disability Statistics, 2022). The six questions separately asked if an individual had difficulty seeing, hearing, walking or climbing steps, remembering or concentrating, carrying out self-care, or communicating or being understood. Answer choices were no difficulty, some difficulty, a lot of difficulty, cannot do at all, refused, and don’t know. Learning disability was assessed using a single item from the National Health Interview Survey, which asked, “Has a representative from a school or a health professional ever told you that you have a learning disability?” (Centers for Disease Control, 2019). Answer choices included yes, no, refused, and don’t know.
Education
Schooling experiences were assessed using three questions from the that asked whether participants attended a racially segregated school, and if so, whether it was legally segregated and for how many years (Barnes et al., 2012). Health literacy was measured with the Calgary Charter on Health Literacy Scale, which is a 5-item Likert-type scale that asks about comprehension and other skills such as reading, writing, listening, speaking, numeracy, critical analysis, communication, and interaction skills (Pleasant et al., 2018). Four response options range from never to always. Higher scores correspond to better health literacy.
Occupation and social and socioeconomic positioning (Amir et al., 2019; Hill et al., 2015; Rote et al., 2021). Cognitive demands of work were measured using the Occupational Cognitive Response Score (Pool et al., 2016), which was determined using primary occupation as assessed on the National Alzheimer’s Coordinating Center Uniform Data Set A-1 Subject Demographics form (National Alzheimer’s Coordinating Center, 2015) and a follow-up free-text question asking for individuals’ main occupation. Socioeconomic Status and Strain was measured with a 3-item index, with a range from 2 to 14 where higher scores indicate greater financial strain: (1) How satisfied are you with your or your family’s current financial situation, (2) Currently, how difficult is it to meet monthly bill payments, (3) In the last year, which (if any) have you done as the result of economic problems to cover daily expenses (Sternthal et al., 2011). Social positioning was assessed with a modified MacArthur Scale of Subjective Social Status (Adler et al., 1994). Participants are asked to rank themselves on a 10-interval scale from worst off to best off using factors such as education, income, and jobs. A picture of a ladder with ten rungs offers a visual representation to participants when responding.
Social Support
Participants’ social support from family and friends was assessed using the Lubben Social Network Scale, which asks 3 questions about family connections and 3 questions about friendships (Lubben et al., 2006). The questions are: (1) How many [relatives or friends] do you see or hear from at least once a month, including relatives with whom you live; (2) How many [relatives or friends] do you feel at ease with that you can talk about private matters; (3) How many [relatives or friends] do you feel close to such that you could call on them for help. Total scores range from 0 to 30 with higher scores indicating better social support. Religiousness and spirituality were assessed using four questions from the Brief Multidimensional Measure of Religiousness/Spirituality, which asks 12 questions about religious and spiritual practices and communities (The Fetzer Institute & National Institute on Aging Working Group, 1999): (1) To what extent do you consider yourself a religious person? (2) To what extent do you consider yourself a spiritual person? (3) My religious beliefs help me find a purpose in even the most painful and confusing events in my life. (4) My spiritual beliefs give my life a sense of significance and purpose. Responses were given on 4-point scales. The scale for the first two items ranged from very religious to not religious at all. The scale for the second two items ranged from strongly disagree to strongly agree. Hobbies were assessed using 4 researcher designed items: (1) Do you regularly engage in hobbies and activities? (Y/N) (2) Would you like to share more information about hobbies and leisure? (Y/N) (3) If yes, which of these activities do you participate in?: physical activities (walking, bike riding, going to the gym), community activities (involvement in religious community, volunteering), mental activities (reading, puzzles and games, playing cards, painting, musical instruments), other (free text), (4) Other (describe more activities below).
Social Stressors and Perceived Stress
Perceived stress was assessed using the Perceived Stress Scale, which is a 10-item scale that asks how often a participant felt stress over the past 30 days (Teresi et al., 2020). Answer options range from 0 to 4, with higher scores indicating more often experiencing stress. Daily encounters with discrimination were assessed with the Everyday Discrimination Scale, which is a 9-item measure with total score that ranges from 1, meaning never, to 4, meaning often (Williams, 2016). A 10th item asks the reason(s) to which the respondent attributes the biased treatment. Exposure to stressors was measured with the Social Readjustment Rating Scale. This is a 43-item list of events, including death of a spouse, divorce, being fired at work, major changes in living conditions, and death of a close friend or relative (Holmes & Rahe, 1967). Participants indicated whether each event occurred in the last year.
Adverse experiences in childhood were assessed using the Adverse Childhood Experiences scale, which is a 20-item questionnaire that includes multiple choice and yes/no responses. Scores range from 0 to 7, with higher scores indicating more exposure to abuse (Felitti et al., 1998). Experiences with childhood caregivers were assessed with 13 items from the Health and Retirement study (University of Michigan Health and Retirement Study, 2020). The questions asked about caregiver relationships, level of education, occupational status, and general health. Assessment of early-life socioeconomic status is described in Social Support.
Built Environment/Neighborhood
Neighborhood quality, feelings about one’s community and sense of belonging, and early-life socioeconomic status were assessed using the Modified Life-course Sociodemographic Neighborhood Exposures Questionnaire, which includes 19 items that are multiple-choice, fill-in-the-blank, and a ten-point rating scale (Mendes de Leon et al., 2009). Geography was assessed using three items that asked about upbringing in the United States, childhood 5-digit zip code, and current 5-digit zip code. For childhood zip code, participants could list up to three zip codes and the commensurate ages until the age of 18 years. A crosswalk of geographic units of analysis are listed in Online Appendix C.
Participant Acceptability Questionnaire
We asked participants for their feedback via 9-questions at the end of the Life Experiences Survey. The questions included several aspects of the participant experience, including: approximate survey completion time (in minutes), the participant’s opinion on the length of the survey (too long, too short, or just the right length), identifying confusing questions (with a free text option), identifying inappropriate or offensive questions (with a free text option), identifying questions that the participant wanted to expand on (with a free text option), and additional questions the participant would like to see included in the survey.
Brain amyloid
Brain amyloid data were collected for a subset of participants via positron emission tomography (PET) imaging. Deposition of Aβ plaques are measured using PET radiotracers, wherein an increase of A
Statistical Analysis
Descriptive analyses were used to characterize the sample and participant survey responses. We report measures of central tendency and variation. Data were reviewed for outliers and erroneous responding. All statistical tests were 2-sided. All analyses were conducted in Stata v. 16.0 and R v 1.4.
We present sample size determination to support the planning of future studies with these measures. The form of the sample size calculation depends on the scientific question and type of statistical test. For example, to compare group difference of demographic variables, the two-sample t-test (2-sided) can be implemented. The key components in a sample size calculation for this test include: 1. standardized effect size/Cohen’s d (d): The ratio of absolute difference in group means and the pooled standard deviation. Cohen classified effect sizes as small (d = 0.2), medium (d = 0.5), and large (d ≥ 0.8); 2. desired level of significance (α): The probability of claiming a significant effect when the truth is the opposite (Type I error), commonly set at 0.05; 3. power (1 − β): The probability of claiming a significant effect when that is truth. The National Institutes of Health recommended value of power is 0.8 (National Institutes of Health, 2023b). Here β is the chance of Type II error, which is failed to claim difference when the truth is the opposite. The formula of determining sample size is:
Results
Participant Sample
Sample Characteristics
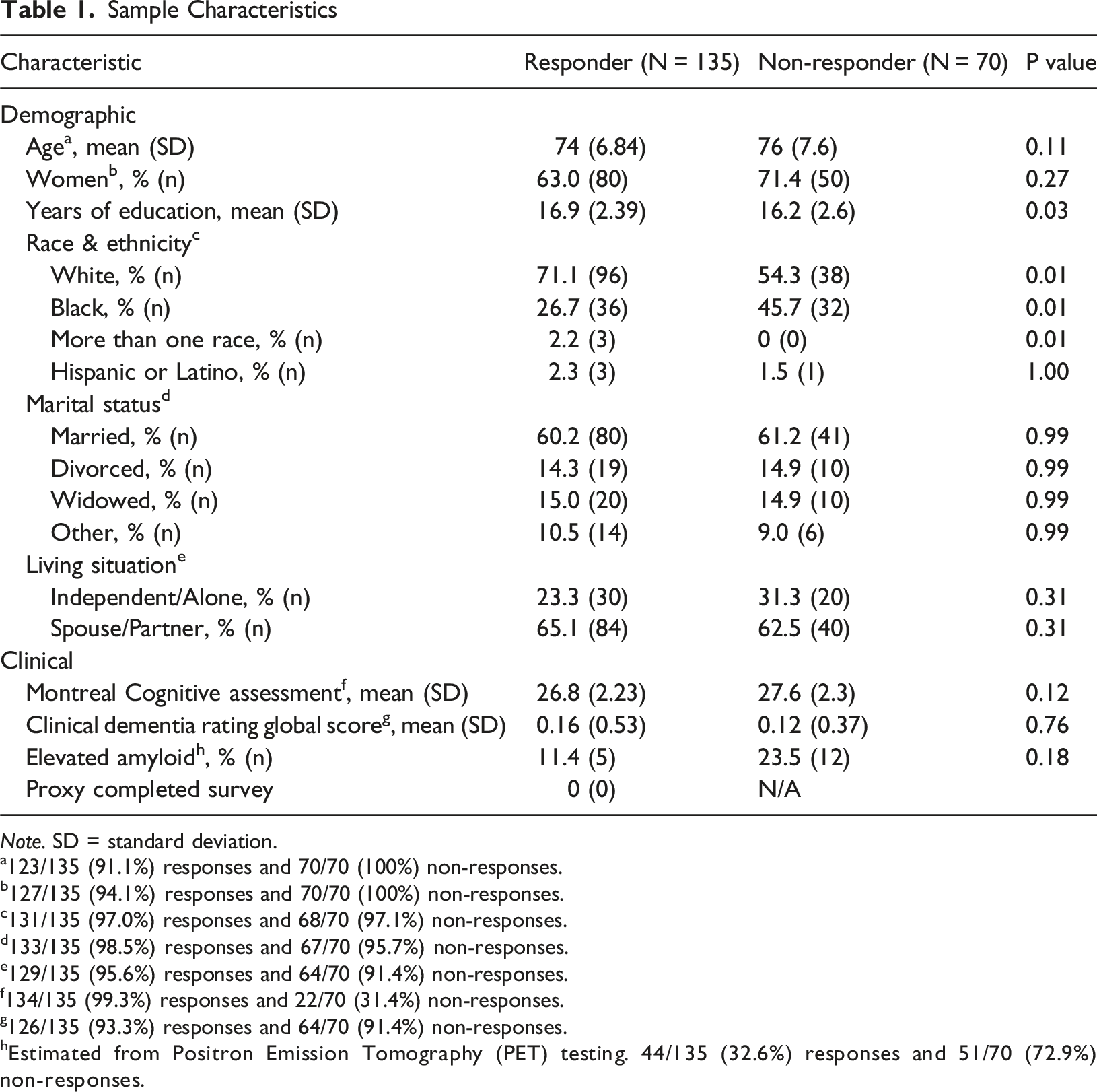
Note. SD = standard deviation.
a123/135 (91.1%) responses and 70/70 (100%) non-responses.
b127/135 (94.1%) responses and 70/70 (100%) non-responses.
c131/135 (97.0%) responses and 68/70 (97.1%) non-responses.
d133/135 (98.5%) responses and 67/70 (95.7%) non-responses.
e129/135 (95.6%) responses and 64/70 (91.4%) non-responses.
f134/135 (99.3%) responses and 22/70 (31.4%) non-responses.
g126/135 (93.3%) responses and 64/70 (91.4%) non-responses.
hEstimated from Positron Emission Tomography (PET) testing. 44/135 (32.6%) responses and 51/70 (72.9%) non-responses.
Multi-Item Scales and Domains
Completion Rates and Summary Coefficients for Scales and Multiple Item Domains
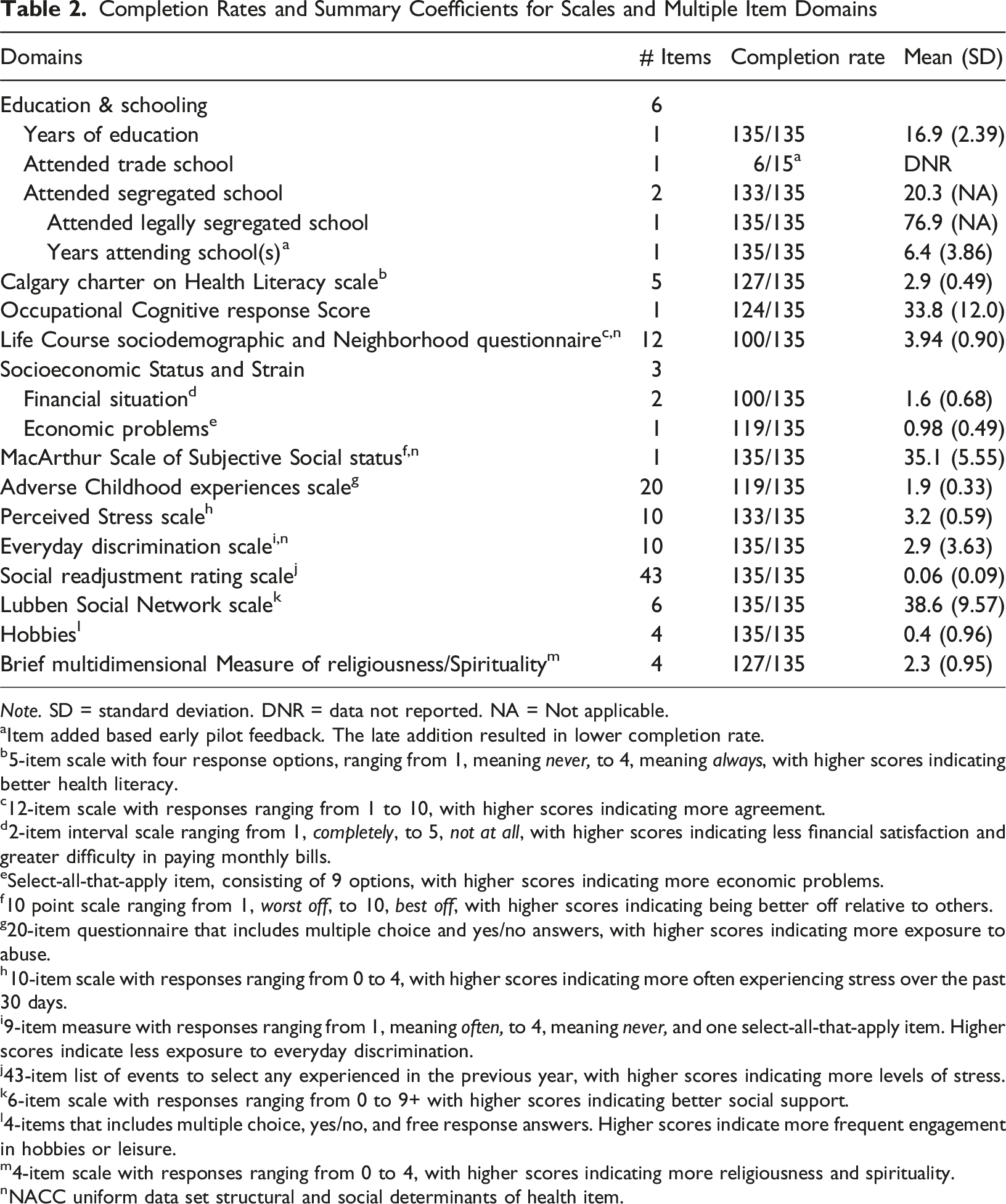
Note. SD = standard deviation. DNR = data not reported. NA = Not applicable.
aItem added based early pilot feedback. The late addition resulted in lower completion rate.
b5-item scale with four response options, ranging from 1, meaning never, to 4, meaning always, with higher scores indicating better health literacy.
c12-item scale with responses ranging from 1 to 10, with higher scores indicating more agreement.
d2-item interval scale ranging from 1, completely, to 5, not at all, with higher scores indicating less financial satisfaction and greater difficulty in paying monthly bills.
eSelect-all-that-apply item, consisting of 9 options, with higher scores indicating more economic problems.
f10 point scale ranging from 1, worst off, to 10, best off, with higher scores indicating being better off relative to others.
g20-item questionnaire that includes multiple choice and yes/no answers, with higher scores indicating more exposure to abuse.
h10-item scale with responses ranging from 0 to 4, with higher scores indicating more often experiencing stress over the past 30 days.
i9-item measure with responses ranging from 1, meaning often, to 4, meaning never, and one select-all-that-apply item. Higher scores indicate less exposure to everyday discrimination.
j43-item list of events to select any experienced in the previous year, with higher scores indicating more levels of stress.
k6-item scale with responses ranging from 0 to 9+ with higher scores indicating better social support.
l4-items that includes multiple choice, yes/no, and free response answers. Higher scores indicate more frequent engagement in hobbies or leisure.
m4-item scale with responses ranging from 0 to 4, with higher scores indicating more religiousness and spirituality.
nNACC uniform data set structural and social determinants of health item.
Extended Demographic Variables
Completion Rates of Added Demographic Characteristics (N = 135)
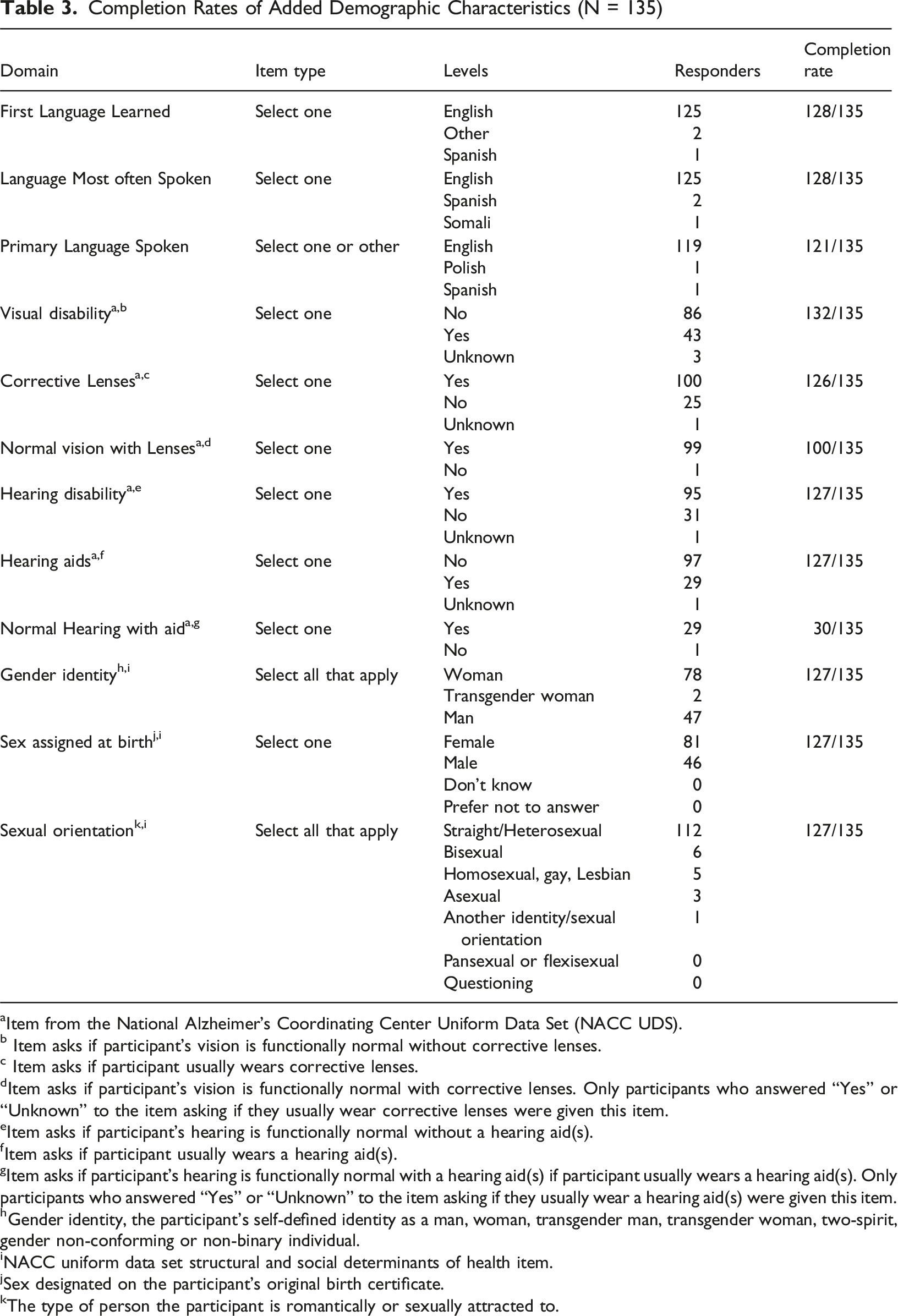
aItem from the National Alzheimer’s Coordinating Center Uniform Data Set (NACC UDS).
b Item asks if participant’s vision is functionally normal without corrective lenses.
c Item asks if participant usually wears corrective lenses.
dItem asks if participant’s vision is functionally normal with corrective lenses. Only participants who answered “Yes” or “Unknown” to the item asking if they usually wear corrective lenses were given this item.
eItem asks if participant’s hearing is functionally normal without a hearing aid(s).
fItem asks if participant usually wears a hearing aid(s).
gItem asks if participant’s hearing is functionally normal with a hearing aid(s) if participant usually wears a hearing aid(s). Only participants who answered “Yes” or “Unknown” to the item asking if they usually wear a hearing aid(s) were given this item.
hGender identity, the participant’s self-defined identity as a man, woman, transgender man, transgender woman, two-spirit, gender non-conforming or non-binary individual.
iNACC uniform data set structural and social determinants of health item.
jSex designated on the participant’s original birth certificate.
kThe type of person the participant is romantically or sexually attracted to.
Completion Rates of Added Demographic Characteristics (N = 135)
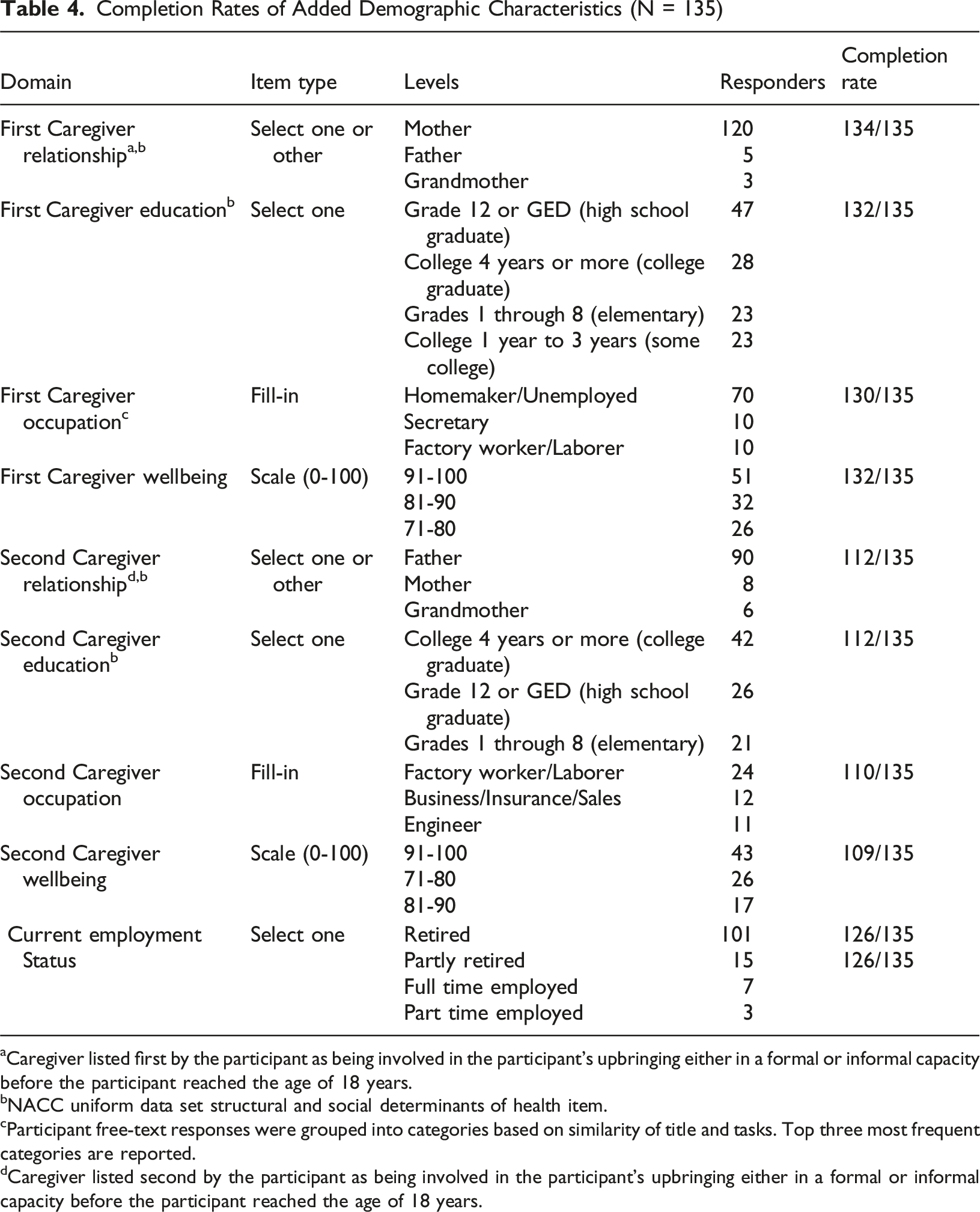
aCaregiver listed first by the participant as being involved in the participant’s upbringing either in a formal or informal capacity before the participant reached the age of 18 years.
bNACC uniform data set structural and social determinants of health item.
cParticipant free-text responses were grouped into categories based on similarity of title and tasks. Top three most frequent categories are reported.
dCaregiver listed second by the participant as being involved in the participant’s upbringing either in a formal or informal capacity before the participant reached the age of 18 years.
Participant Geographic Data
Completion Rates of Participant Reported Location Data (N = 135)
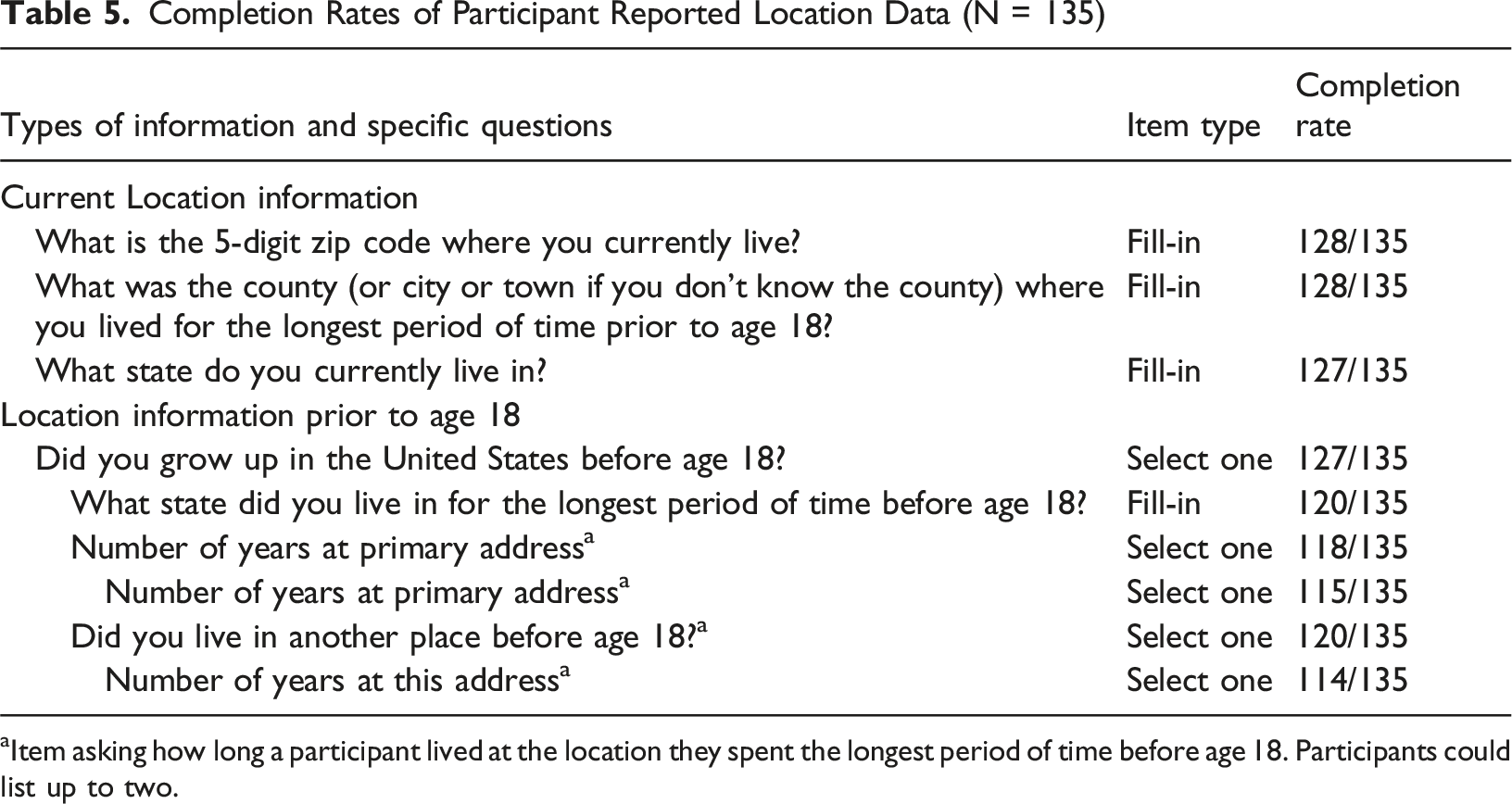
aItem asking how long a participant lived at the location they spent the longest period of time before age 18. Participants could list up to two.
Survey Feedback and Modifications
Survey Domains, Participant Feedback, and Corresponding Modification
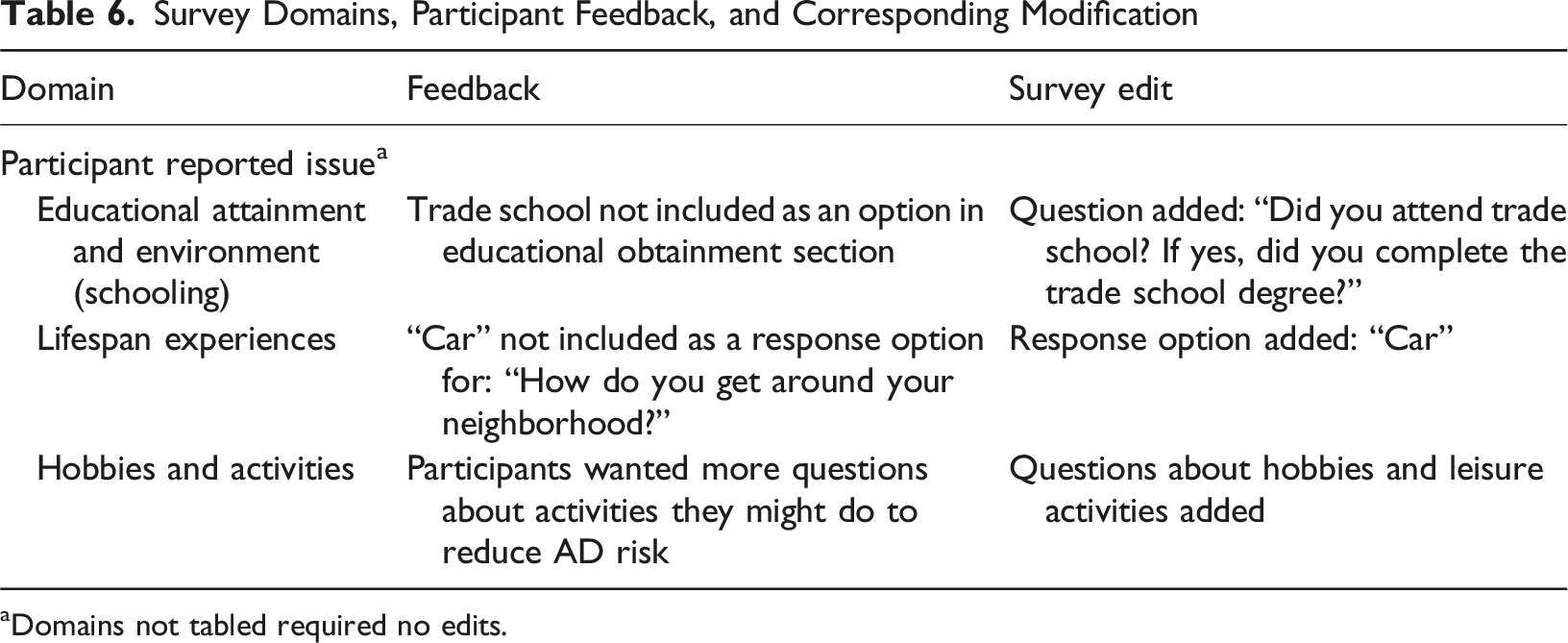
aDomains not tabled required no edits.
Early data on Completion of the Survey in Persons with MCI
Completion Rates and Summary Coefficients for Scales and Multiple Item Domains for the Subsample With Mild Cognitive Impairment (MCI)
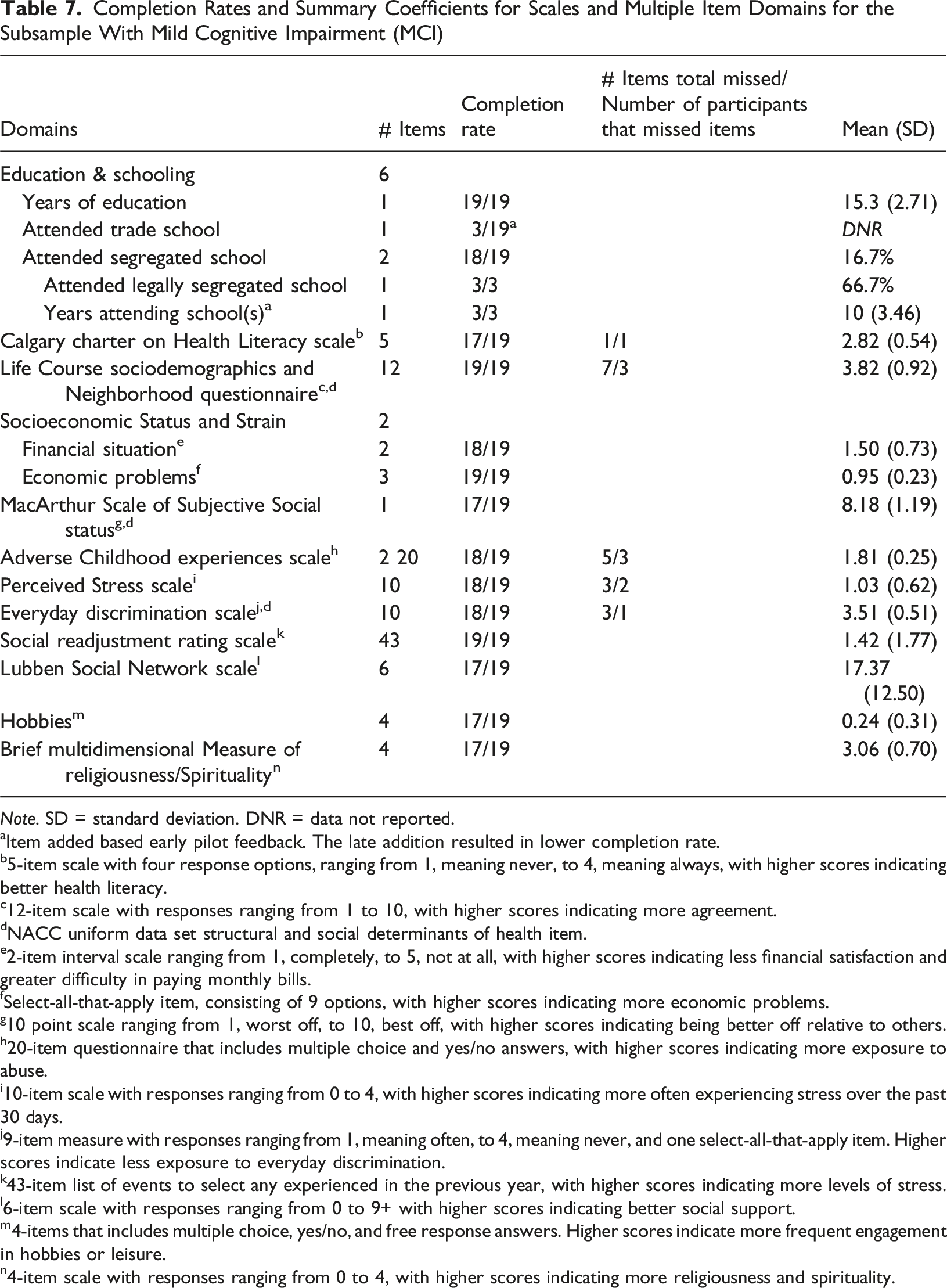
Note. SD = standard deviation. DNR = data not reported.
aItem added based early pilot feedback. The late addition resulted in lower completion rate.
b5-item scale with four response options, ranging from 1, meaning never, to 4, meaning always, with higher scores indicating better health literacy.
c12-item scale with responses ranging from 1 to 10, with higher scores indicating more agreement.
dNACC uniform data set structural and social determinants of health item.
e2-item interval scale ranging from 1, completely, to 5, not at all, with higher scores indicating less financial satisfaction and greater difficulty in paying monthly bills.
fSelect-all-that-apply item, consisting of 9 options, with higher scores indicating more economic problems.
g10 point scale ranging from 1, worst off, to 10, best off, with higher scores indicating being better off relative to others.
h20-item questionnaire that includes multiple choice and yes/no answers, with higher scores indicating more exposure to abuse.
i10-item scale with responses ranging from 0 to 4, with higher scores indicating more often experiencing stress over the past 30 days.
j9-item measure with responses ranging from 1, meaning often, to 4, meaning never, and one select-all-that-apply item. Higher scores indicate less exposure to everyday discrimination.
k43-item list of events to select any experienced in the previous year, with higher scores indicating more levels of stress.
l6-item scale with responses ranging from 0 to 9+ with higher scores indicating better social support.
m4-items that includes multiple choice, yes/no, and free response answers. Higher scores indicate more frequent engagement in hobbies or leisure.
n4-item scale with responses ranging from 0 to 4, with higher scores indicating more religiousness and spirituality.
Discussion
Embedding structural and social determinants of health data collection in the research cohorts at major research hubs that inform Alzheimer’s disease science may advance equitable and generalizable discoveries (Centers for Disease Control, 2021) and improve conceptual understanding of disease mechanisms (Stites et al., 2021). Moreover, the effort supports developing a conceptual model to better understand life course experiences on brain aging and structural and social determinants of health as actionable targets for effective clinical interventions.
In order to successfully position the structural and social determinants of health construct in a multidisciplinary biomedical model of collaborative science, we identified the need for a set of measures and administration protocol that would: (1) be feasible in terms of participant burden and organizational resources, (2) harmonize multiple data sources to study small subgroups and discrete experiences, (3) make the data widely-available to Alzheimer’s disease researchers across the nation and, potentially, the globe, (4) capture current and life course experiences with culturally acceptable and scientifically useful methods, and (5) be accessible and understandable to a diverse range of investigators. We address each of these goals below. Our results offer data to inform the collection of structural and social determinants of health data from older adults and guide future studies using these measures.
We pilot tested structural and social determinants of health questions using an online platform among cognitively unimpaired older adults, ages 65 and older, participating in an Alzheimer’s Disease Research Center cohort. We found the response rate to be acceptable (66%, n = 135/205) (Fincham, 2008), and the completion rate to be excellent (95%, n = 128/135). Item skip rates were very low, which included items that have been shown to yield lower responses, such as questions asking about socioeconomic status. We also found this was the case for questions about sexual orientation (94.1% completion) and gender identity (94.1% completion). While our survey includes many questions (a minimum of 281 items), we designed the survey to be streamlined and convenient, taking participants an average of 25 to 30 minutes to complete.
Participants' willingness to answer a wide range of questions is notable. There has been some concern expressed among scientists about asking participants to report sexual orientation and gender identity as discomfort with those questions could lead individuals to drop out or not enroll in research. We found no evidence to support these concerns. Somewhat unexpectedly, we found that questions about childhood appeared most difficult for participants to complete; these items had relatively lower completion rates (lowest was 88.9%) and, on the acceptability survey, three participants acknowledged some discomfort with the childhood questions, but still chose to complete them.
Another important goal we had was to capture experiences across the life course. The seemingly most complex of which were questions asking participants to recall zip codes and the ages that they resided at those zip codes. The original design of those questions used free text boxes to collect zip code and respective age. Pilot testing demonstrated that most participants were willing and able to provide current resident information and where they lived prior to the age of 18.
Given wide variation in responses to childhood places of residence, particularly around units of time, we revised the items for greater structure. In the revised version, participants are asked about two time periods from their childhood: before and after age 12. This approach will garner some of the first life-course data routinely collected in Alzheimer’s Disease Research Centers.
Our pilot study contributes to the research on structural and social determinants of health in dementia that shows structural and social determinants of health questions can be completed in relatively little time with low non-response rates (Giuse et al., 2017; Mattos et al., 2019; Walters et al., 2017). Our survey, which includes structural and social determinants of health associated with both risk and resilience, addresses a tension in prior studies; researchers tend to focus on structural and social determinants of health associated with health risks (Bartley et al., 2023; Mattos et al., 2019; Rhee et al., 2020), while research participants emphasize adoption of healthy lifestyles and relationship advantages (Mattos et al., 2019). Additionally, our approach deliberately addresses both sociologically-grounded structural and social determinants of health centered on advancing social equity (i.e., anti-racism) and psychologically-oriented structural and social determinants of health that advocates for individual betterment (i.e., subjective stress).
We have worked to include the perspectives and interests of participants in the development of this instrument along with what is known in the evidence-based literature. Where possible, we have constructed questions to transcend multiple social contexts. This instrument may need to continue to evolve as science progresses as well as perspectives of new communities are incorporated. As part of this ongoing work, it is essential individuals contributing to this work are both kept aware of its progress and engaged in the discussion.
This line of research, and its translation into routine care, has the potential to improve patient outcomes – including identification and selection of biological targets for intervention, identifying patients at high risk for poor outcomes, addressing modifiable social problems, and, effectiveness of disease-modifying therapies. There is a notable risk in partially achieving the success of this undertaking; mechanisms attributed to structural and social determinants of health could be dismissed or ignored in the clinical setting (Andermann, 2016). For example, caregiver burnout is a well-recognized clinical problem in the care of patients with Alzheimer’s disease. Reducing the unhealthy behaviors associated with burnout requires creating more supportive environments that make healthy choices easy. Thus, in addition to individual counselling and education, broader interventions and programs are needed to make respite care and in-home services readily available.
Our pilot study has limitations. White research volunteers were more likely than Black research volunteers to respond to the survey, and most participants completing the survey identified as White, female with an average age of 74 years (SD = 6.8). The generalizability of our findings may be limited to groups with similar characteristics. Future studies are needed to pilot these questionnaires in more sociocultural groups. Additionally, while our response rate of 66% is typically considered acceptable in the peer reviewed literature, response rates in Alzheimer’s Disease Research Center cohorts are often higher. Thus, we plan to explore options for increasing the response rate such as using email reminders and follow-up phone calls.
For next steps, we plan to collect these structural and social determinants of health data longitudinally in this group of cognitively unimpaired older adults and continue data collection in research participants with cognitive impairment. Data collection in persons with cognitive impairment will be essential for evaluating the feasibility of collecting these data with the assistance of caregivers and other proxy reports. This step is essential for ensuring the data can be readily available for requests made through the Alzheimer’s Disease Research Centers data sharing mechanism.
Moving forward it will be essential to expand collection and study of structural and social determinants of health to research samples that are representative of the populations with Alzheimer’s disease. In particular, structural and social determinants of health exert disproportionate consequences on members of minoritized communities, including notably Black and Hispanic populations which are underrepresented in research and experience the greatest burden of Alzheimer’s disease. Gathering these data in groups from low resource settings and backgrounds will also be core to advancing this line of work.
Conclusion
Structural and social determinants of health are salient sociological factors, exhibiting prominent effects on individuals’ health and wellbeing. Yet, they are often not addressed in Alzheimer’s disease. The introduction of structural and social determinants of health data into the national network of Alzheimer’s disease research centers provides opportunities to accelerate scientific discovery and improve clinical outcomes for older adults. Findings from pilot testing showed most structural and social determinants of health items had high completion rates, low and inconsistent skip patterns, and were received well by participants. The findings lend support to the feasibility of routinely collecting structural and social determinants of health as part of the national architecture for Alzheimer’s disease research.
Supplemental Material
Supplemental Material - Recognizing Structural and Social Determinants of Health in the Diagnosis and Care of Dementia
Supplemental Material for Recognizing Structural and Social Determinants of Health in the Diagnosis and Care of Dementia by Shana D. Stites, Sharnita Midgett, Nisha Patel, Kimberly Halberstadter, Rosalie Schumann, Marissa L. Streitz, Yidan Shi, John C. Morris, Jason Flatt, Crystal M. Glover, Dawn Mechanic-Hamilton in Dementia
Footnotes
Acknowledgements
Nina Miller for technical assistance with images.
ORCID iDs
Ethical Considerations
The Institutional Review Board of the University of Pennsylvania approved all procedures involving human subjects [068200 for ABC and 844047 for ABCD2].
Consent to Participate
All participants provided written informed consent prior to participating.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Institute on Aging [P30AG012836-25-S2, P30AG010124, P30AG10161, U24AG058556, K23AG065442, P01AG03991, P01AF02676, P30AG066444, R01AG083177, R24AG066599, K01AG056669, K23AG065499]; and by the Alzheimer’s Association [AARF-17-528934, AARGD-22-929144]. The Aging Brain Cohort Dedicated to Diversity (ABCD2) Study is supported by a grant from the Pennsylvania Department of Health (2019NF4100087335). The funding sources had no involvement in the study design, collection, analysis or interpretation of data, writing of the report, or the decision to submit the article for publication.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
While initially being made available only with the ADRC network, the data will be made available in the future via an ADRC data request process (https://www.med.upenn.edu/adrc/datasharing.html). A public folder, located at ![]() , is updated regularly and includes the SSDOH survey, the REDCap codebook and a read me file.
, is updated regularly and includes the SSDOH survey, the REDCap codebook and a read me file.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
