Abstract
Keywords
Introduction
Music-based interventions (MBIs) for people with dementia, that is uses of music without continued direction by a qualified therapist, have recently gained attention (Garrido et al., 2017). Due to their malleable nature, MBIs are of particular interest to the approximately 70% of people with dementia who reside in the community (Brijoux & Zank, 2022). In a recent systematic review, it was found that MBIs –singing or music listening – benefit cognition, anxiety, and pain in community-dwelling people with dementia (Hofbauer et al., 2022).
Music listening MBIs might be particularly suitable for community-dwelling people with dementia, as they are less effortful to implement than singing interventions, as indicated by better intervention adherence (Särkämö et al., 2014). Yet, rigorous research on the effects of music listening is lacking (Garrido et al., 2017; Gaviola et al., 2019; Tsoi et al., 2018). A 2017 review found that only 10 of 28 (36 %) of music listening studies reviewed were randomised in some form and had a control condition or group (Garrido et al., 2017). Only five studies were set in the home (Garrido et al., 2017), of which only one (Särkämö et al., 2014) was a randomised-controlled trial (RCT). The latter observed an improvement in cognition, quality of life, and depression in the music listening group compared to the control group (Särkämö et al., 2014, 2016). Feasibility investigations have also indicated the capacity of at-home music listening to improve behavioural and psychological symptoms (BPSDs; Buller et al., 2019; Garrido et al., 2021; Park & Specht, 2009; Shelton, 2018), cognitive function (Li et al., 2015), and quality of life (Kulibert et al., 2019).
In previous research, MBIs have typically relied on a preference-based music selection (e.g., Buller et al., 2019; Leggieri et al., 2019). However, guidelines released by the US National Institute of Health (NIH) in 2023 underscore that the choice of music needs to be made according to intervention goals (Edwards et al., 2023). This may be particularly difficult for informal caregivers. Indeed when Garrido and colleagues trialled a guide (Garrido et al., 2022) with recommendations on symptom-based music selection (e.g. slow, positively valenced music to target agitation) caregivers often dismissed symptom-specific recommendations, reporting to find these overwhelming and unnecessary (Garrido et al., 2021). To reduce the burden for caregivers, it may be useful to supply them with pre-validated playlists targeting specific outcomes. Ultimately, large pre-validated playlists could serve as a foundation from which caregivers could further personalise choices, ensuring that the music used in caregiver-directed MBIs is not only personally meaningful for the person with dementia but also optimised for intervention goals. A combined approach would require a solid evidence base on which types of supports which intervention goal, which is currently not established.
Evidence directly comparing the effects of different music is thus needed. Slow, positively valenced music is thought to enhance parasympathetic nervous system activity (Kulinski et al., 2022), indicating that this may ease neuropsychiatric symptoms. However, music listening studies achieving reductions in BPSDs (e.g., depression, anxiety, agitation) have insufficiently characterised the music selection to support this line of reasoning (Chang et al., 2010; Guétin et al., 2009; Heim et al., 2003; Whear et al., 2014). Similarly, it is unclear which type of music may benefit cognition in people with dementia. Experiments reporting effects of music on cognitive domains in people with dementia (Baird et al., 2018, 2020; El Haj et al., 2012; Irish et al., 2006; Meilán García et al., 2012) do not provide sufficient detail. In a recent experiment, a positive effect of fast (130 bpm) positively valenced music on cognitive performance in healthy individuals was observed (Hofbauer et al., 2024). The ‘stimulating’ music may have resulted in the appropriate state of arousal to support cognitive performance on the selected tasks (Collins et al., 2012; Thompson et al., 2001). Yet, whether this will apply to people of dementia in the context of an MBI remains to be seen. Previous research with fast (>120 bpm) background music has reported that people with dementia experiencing a particularly pronounced raise in arousal were also particularly low in enjoyment of this music (Garrido et al., 2019). The arousal response of people with dementia to fast music may thus not necessarily represent positive stimulation and thus may not be conducive to cognitive performance.
Aim
In this randomised, controlled crossover pilot trial, we therefore compared two music listening MBIs: (1) a MBI with fast, positively valenced music, and (2) a MBI of slow, positively valenced music with care-as-usual. We hypothesised that the fast-positive MBI benefits cognitive performance compared to care-as-usual and the slow-positive MBI. Further, we expected that the slow-positive MBI will benefit BPSDs in comparison to both care-as-usual and the fast-positive MBI. Additionally, we expect that both MBIs will benefit well-being and quality of life compared to care-as-usual.
Materials & methods
Participants
We recruited via public advertisement in doctor’s offices, relevant non-governmental organisations, support groups for informal carers of people with dementia, and online. Inclusion criteria were that (1) one person in the dyad was a person with a self-reported dementia diagnosis living in the community, and (2) the other was an informal caregiver on an at least weekly basis. Dyads were excluded if: (1) the person with dementia was institutionalised, (2) visual/hearing/language abilities were insufficient for participation, or (3) the researcher deemed there to be a lack of physical or psychological capacity required for safe and effective participation (e.g. severe mental illness). No interested dyads had to be excluded. The convenience sampling procedure over a pre-determined time period (January to December 2023) resulted in a total of n = 17 dyads being included. One dyad was lost to follow-up in the first MBI period (see Figure S1) and excluded from analysis. Thus, data of n = 16 dyads was analysed. Post-hoc power analysis estimated 80% power to detect a within-participant difference of 1 unit between conditions given an alpha level of .05 (Schoenfeld, 2010). The small sample size is therefore considered appropriate for an initial assessment of the piloted intervention’s feasibility and effectiveness before larger-scale testing.
In order of enrolment, dyads were assigned to one of four possible condition sequences (see Figure 1). Half of participants first underwent a control condition (care-as-usual). The assessments for this trial were conducted between the 25th of February and the 12th of December 2023. For a detailed sample description, see Table 1. Based on Montreal Cognitive Assessment (MoCa) scores (mild: 25-18, moderate: 11-17, severe: ≤10; MoCa Test Inc., 2023), the sample included six people with mild (37.5%), three with moderate (18.75%), and seven with severe (43.75%) dementia. Visualisation of intervention sequences used. In total, four sequences were used: (1) MBI A first, then MBI B, (2) MBI B first, then MBI A, (3) control (care-as-usual), MBI A, and MBI B, (4) and control (care-as-usual), MBI B, and MBI A. Participant demographics. Abbreviations: MoCa: Montreal Cognitive Assessment, NPIQ-D: Total Caregiver Distress Score on the on the Neuropsychiatric Inventory–Questionnaire, NPIQ-S: Total Severity Score on the Neuropsychiatric Inventory–Questionnaire, QoL-AD: Quality of Life–Alzheimer’s Disease score, SD: Standard Deviation, WHO-5: 5-Item Wellbeing Index of the WHO.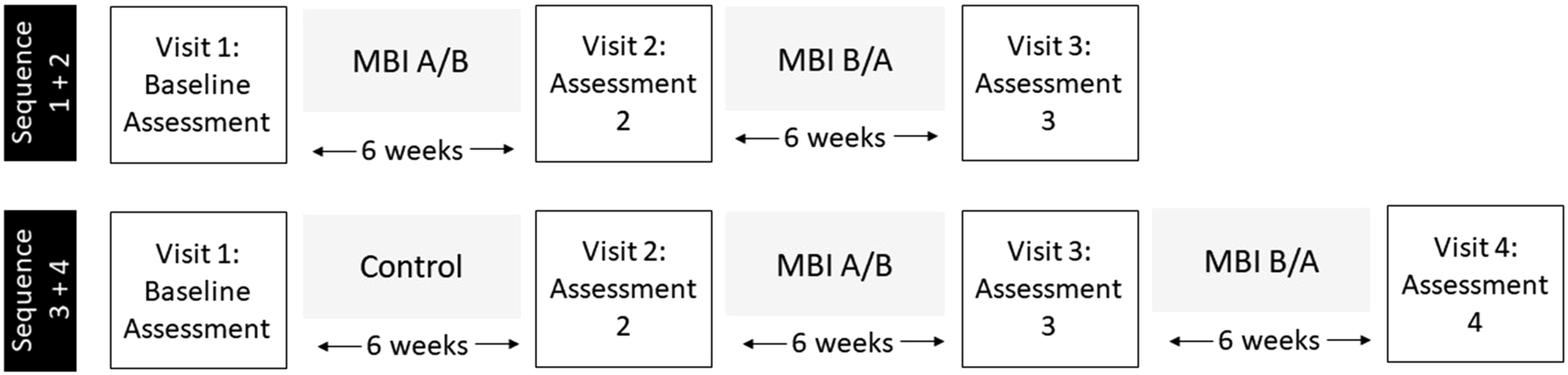

Procedure
In a crossover design, all dyads underwent both MBIs – the fast-positive MBI (henceforth ‘MBI A’) and the slow-positive MBI (‘MBI B’). Half also underwent a control period. The target duration for each period (control, MBI A, MBI B) was 6 weeks (M: 6.5, SD: 1.5, range: 4.3–11.9). Each period was preceded and followed by a home visit by a researcher (see Figure S1). When a visit preceded a MBI period, dyads received a tablet (Samsung Galaxy S6) with the respective playlist on it, headphones, and an hourglass indicating 10 minutes. They were asked to aim for at least three weekly sessions of music listening à 10 minutes. The researcher demonstrated how to use the tablet’s music player. If caregivers preferred, they received a CD (n = 2). Caregivers were asked to protocol MBI session date and duration, as well as any comments, in a logbook. Quantitative data (see the following sections) were collected at each visit. After each MBI, the researcher also collected dyads’ verbal descriptions of the dyads experience with the MBI. During MBI periods, support phone calls were made to the informal caregiver. These calls were made at an interval of their preference but at least once per MBI period (M: 1.9, SD: 0.8, range: 1–3).
Materials
Stimuli were selected from a pre-validated pool of orchestral musical pieces. In the pre-validation process, 102 healthy adults rated the perceived emotional valence of musical excerpts on a visual analogue scale ranging from ‘very negative’ to ‘very positive’. From their ratings, the perceived valence was estimated using mixed-effect models (Hofbauer & Rodriguez, 2023). We selected pieces with perceived positive valence. For each MBI, six musical pieces (total duration: approx. 22 minutes) were included. For MBI A, stimuli had a tempo of ≤110 beats per minute (bpm; M: 129, SD: 26, range: 116–184). For MBI B, the tempo was ≤90 bpm (M: 78, SD: 12, range: 59–87). For a detailed list of stimuli see Table S1.
Outcomes
Demographics
We recorded the gender, age, marital status and level of education for the person with dementia and the caregivers. We further asked caregivers’ relation to the person with dementia.
Adherence
For dyads who exclusively used the tablet (n = 14, 88%), it was possible to reliably assess the total duration of music listening using the meta-data of the music player. Adherence was determined for each MBI separately. If the total listening time exceeded the recommended minimal duration of 180 minutes (10 minutes × 3 × 6 weeks), the dyad was determined to have adhered to the respective MBI. For dyads using a CD, we used logbook entries to estimate listening duration: the listening durations reported were added up. For one dyad, adherence could not be determined, as they used a CD but did not keep a log of their sessions. For one other dyad, adherence could only be determined for MBI B but not A, as they used a personal device for the latter and kept no log.
Cognition
We used standardised tests to assess a range of cognitive domains affected in dementia. These tests exist in parallel versions so that the person with dementia was presented with a new version each time. To assess semantic memory, we used word list tests (Alenius et al., 2019). 10 words were read out-loud and the person with dementia was asked to recall these thereafter (‘immediate recall’). This process was repeated three times in total, the word list being presented in a different order each time. The number of correctly recalled words was summed (range: 0–30). To get an indication of processing speed, we used the Zahlenverbindungstest (ZVT; Oswald & Roth, 1987) which is the German version of the non-alternating trail making test (Rammsayer & Stahl, 2007). The ZVT was administered in a pencil-paper-format and requires the person with dementia to draw a line between numbers from 1-90 in ascending order. Mistakes had to be corrected. Time to completion was recorded. To measure word fluency and executive control (Dekhtyar et al., 2022), we used a phonemic fluency task (letters: F/A/S/L). After these testing procedures, the person with dementia was asked to recall the word list again (‘delayed recall’, theoretical range 0–10).
Quality of life and well-being
We used the Quality of Life–Alzheimer’s Disease (QoL-AD; Logsdon et al., 2002) scale in an interview format. On the QoL-AD, the person with dementia rated 13 aspects of their life as poor, fair, good, or excellent. These ratings translate to scores from 1-4, which were summed across items to give the final score (range: 13–52). As a brief measure of well-being, we used the 5-Item Wellbeing Index of the WHO (WHO-5; World Health Organization, 1998). On the WHO-5, the person with dementia indicated whether five statements applied to them on a scale from all of the time to never (scores: 0–5). Answers were summed to give an overall well-being score (range: 0–25).
Behavioural and psychological symptoms
On the Neuropsychiatric Inventory–Questionnaire (NPIQ; Cummings et al., 1994; Kaufer et al., 2000), caregivers reported on 12 core symptoms (e.g., agitation/aggression) with regards to the past four weeks. A score of zero indicates that the symptom was not observed. If the symptom was present, its severity was judged (mild, moderate, and severe; score: 1–3) and caregivers rated their own distress in response on a scale from 0-5 (no distress to very severe distress). Scores for severity (range: 0–36) and distress (range: 0–60) were summed.
Descriptive data
During the support phone calls, we asked caregivers whether there had been any personal/technical barriers to implementing the MBI since the last call. We also asked about any initial impressions (questions in Text S1). Further, at the end of each MBI period, dyads were asked if there had been any problems, any observable effects, and whether there were suggestions for improvement of the MBI (Text S2). Answers to these structured questions were recorded in bullet point form.
Statistical analysis
All analyses were conducted using R (Version 4.2.2.) in RStudio. We set the significance level α < 0.05.
Adherence analysis
We calculated percentage of adherent dyads, median listening time, and interquartile range (IQR) per MBI. Listening times were compared using non-parametric paired Wilcoxon test, given violations of normality.
Main analyses
Data were analysed using mixed-effect models. Before modelling, we inspected data for extreme outliers (i.e., observations more than 3 IQR above or below, respectively, the third and first quartile of observations in a condition; R package ‘rstatix’). If removing such an extreme outlier affected results, the observations was excluded. Separate models were used for each outcome. All models included a random subject effect. In the first, unadjusted set of models, we included only a fixed effect of intervention (control, MBI A, MBI B). In the second, adjusted set of models, we added the fixed effect for period (after period 1–3), the baseline value for the respective outcome as a covariate (Metcalfe, 2010), and adherence in the respective intervention (yes/no). In line with recommendations, we did not include an interaction term, assuming carryover to be negligible (Senn, 1992; Senn & Lambrou, 1998), and we did not include a fixed effect of sequence (Kenward & Roger, 2010). We report mixed model output and corresponding marginal mean tables (R packages ‘lmer’, ‘lmerTest’). Mean and standard deviation (SD) of outcomes as measured post interventions and the mean (SD) of differences between these can be found in Table S2. Descriptive data were analysed by identifying content categories, defined as statements that shared similar meanings. Each category was quantified based on its frequency of occurrence, enabling insights into the predominant topics and sentiments expressed by participants. While this approach aligns closely with the principles of content analysis (Hsieh & Shannon, 2005), the bullet point style data collected in this exploratory pilot study lacked richness required for the iterative rigor typical of comprehensive qualitative analysis.
Exploratory analysis
It is conceivable that using an MBI with positively valenced music may result in outcome improvements compared to care-as-usual regardless of whether tempo was fast or slow. In this exploratory analysis, we thus treat the MBIs A and B as a single MBI and compare outcomes after 6 weeks and 12 weeks with outcomes after the control condition, adjusting for baseline values.
Results
Adherence
The adherence rates for MBI A and B were 54% and 53%, respectively. The median listening duration for MBI A was 180 minutes (IQR: 172, range: 39–555), while MBI B was listened to for a median of 237 minutes (IQR: 290, range: 22–1631). Listening times did not differ significantly between MBIs (Z (11) = -1.90, p = .057).
Behavioural and psychological symptoms
Marginal means and difference in marginal means (with 95% confidence intervals) by intervention (Control, MBI A, MBI B) for unadjusted and adjusted (for baseline values, period, adherence) outcome specific models.
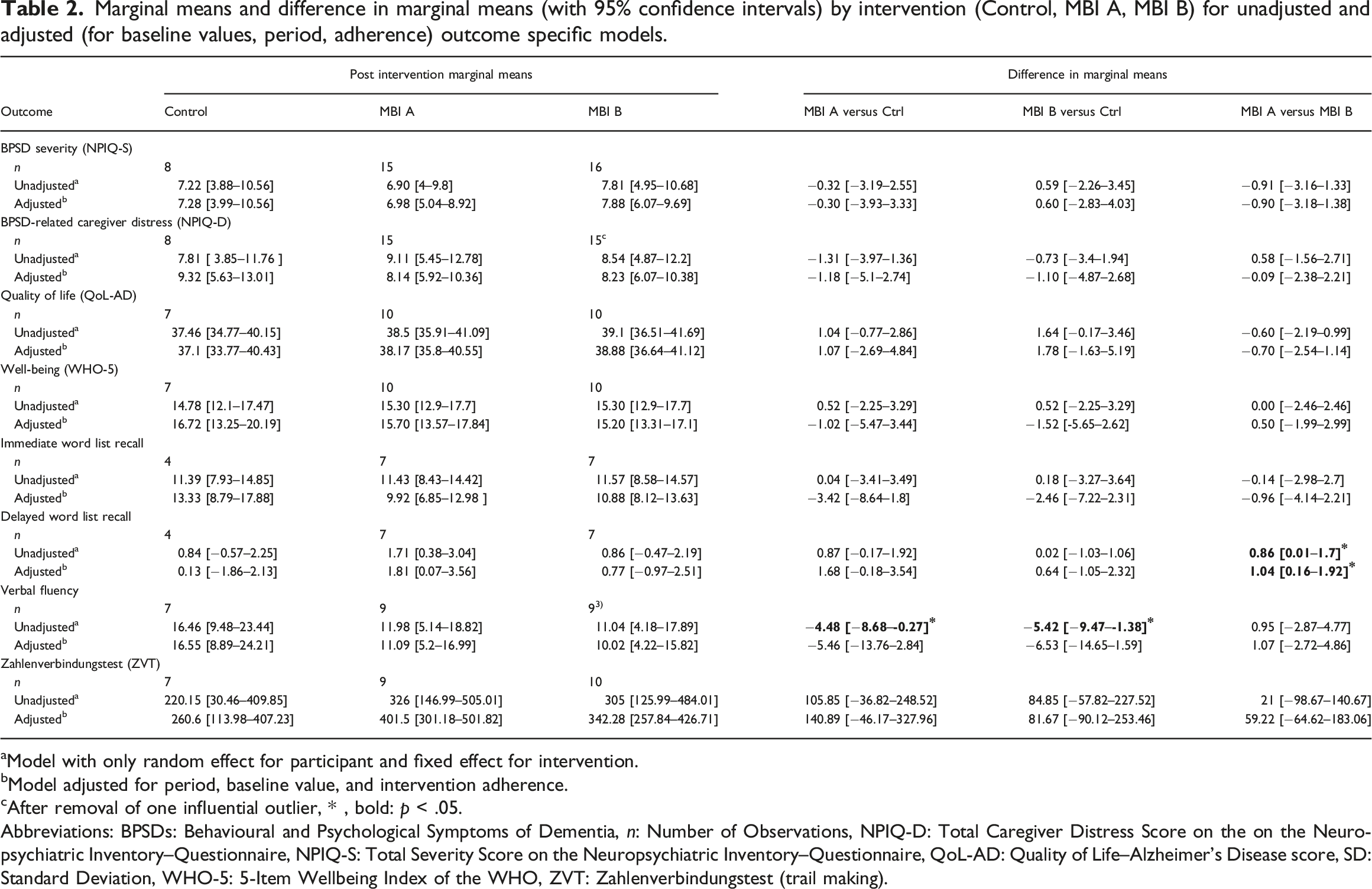
aModel with only random effect for participant and fixed effect for intervention.
bModel adjusted for period, baseline value, and intervention adherence.
cAfter removal of one influential outlier, * , bold: p < .05.
Abbreviations: BPSDs: Behavioural and Psychological Symptoms of Dementia, n: Number of Observations, NPIQ-D: Total Caregiver Distress Score on the on the Neuropsychiatric Inventory–Questionnaire, NPIQ-S: Total Severity Score on the Neuropsychiatric Inventory–Questionnaire, QoL-AD: Quality of Life–Alzheimer’s Disease score, SD: Standard Deviation, WHO-5: 5-Item Wellbeing Index of the WHO, ZVT: Zahlenverbindungstest (trail making).
Mixed model effect estimates (with 95% confidence intervals) for adjusted outcome specific models.
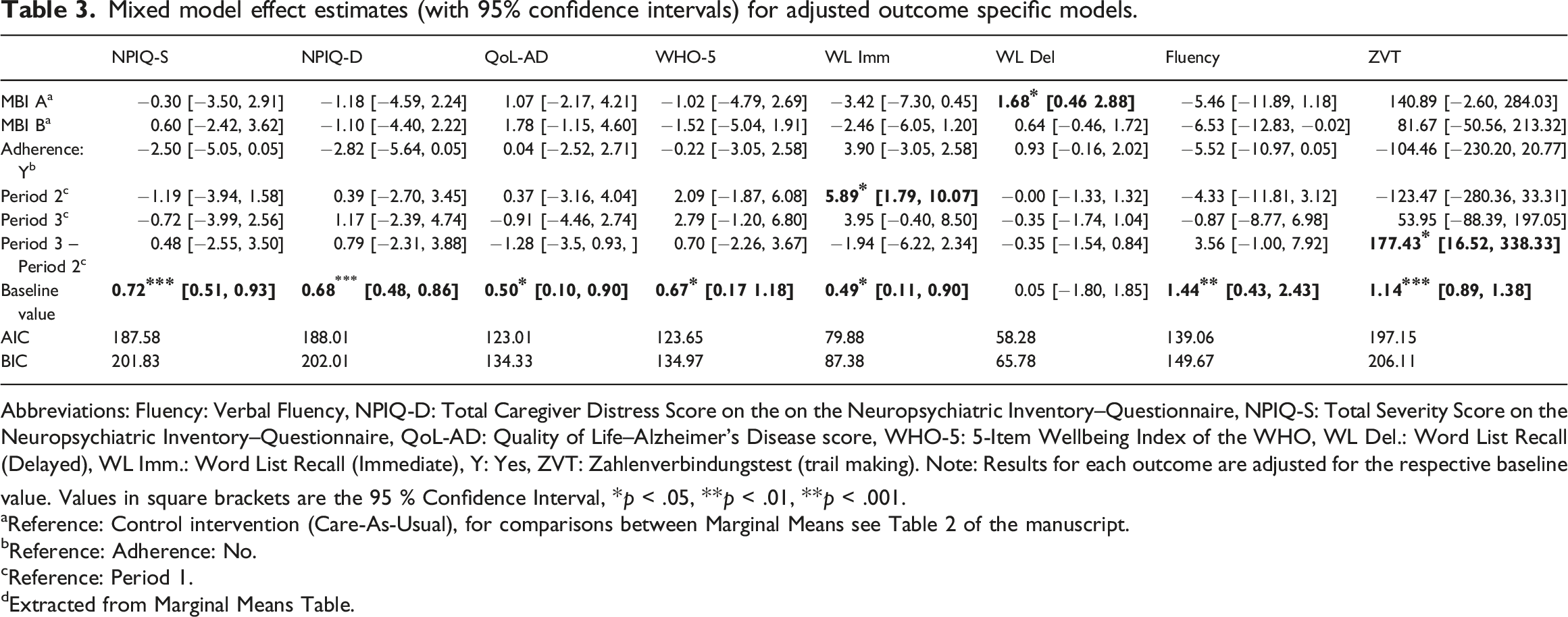
Abbreviations: Fluency: Verbal Fluency, NPIQ-D: Total Caregiver Distress Score on the on the Neuropsychiatric Inventory–Questionnaire, NPIQ-S: Total Severity Score on the Neuropsychiatric Inventory–Questionnaire, QoL-AD: Quality of Life–Alzheimer’s Disease score, WHO-5: 5-Item Wellbeing Index of the WHO, WL Del.: Word List Recall (Delayed), WL Imm.: Word List Recall (Immediate), Y: Yes, ZVT: Zahlenverbindungstest (trail making). Note: Results for each outcome are adjusted for the respective baseline value. Values in square brackets are the 95 % Confidence Interval, *p < .05, **p < .01, **p < .001.
aReference: Control intervention (Care-As-Usual), for comparisons between Marginal Means see Table 2 of the manuscript.
bReference: Adherence: No.
cReference: Period 1.
dExtracted from Marginal Means Table.
Quality of life and well-being
No significant effects of intervention were seen for quality of life (QoL-AD) and Well-Being (WHO-5) (Table 2). There was no significant effect of period or adherence (Table 3).
Cognitive testing
In the unadjusted model of delayed word list recall, MBI A was associated with significantly more recalled words than MBI B. This effects survived the adjustment; MBI A was associated with 1.04 ([95% CI: 0.16, 1.92], p = .028, Hedge’s gav = 0.70) more recalled words (see Figure 2). Other intervention comparisons were not significant (Table 2). One extreme observation was excluded from the fluency task data. In the unadjusted model, the control condition was associated with superior fluency performance compared to MBI A and B (Table 2), however, these effects did not persist after adjustment. There were no effects of period or adherence for delayed recall or fluency performance (Table 3). There were no effects of intervention or adherence on immediate recall and ZVT (Table 2). Yet, there were period effects for both (Table 3). Post-Intervention Delayed Recall Performance. Squares with error bars represent the marginal means (adjusted for period, adherence, and baseline performance) with 95% confidence interval. Coloured circles/triangles/diamond) represent the raw performance scores. Points were jittered for visibility.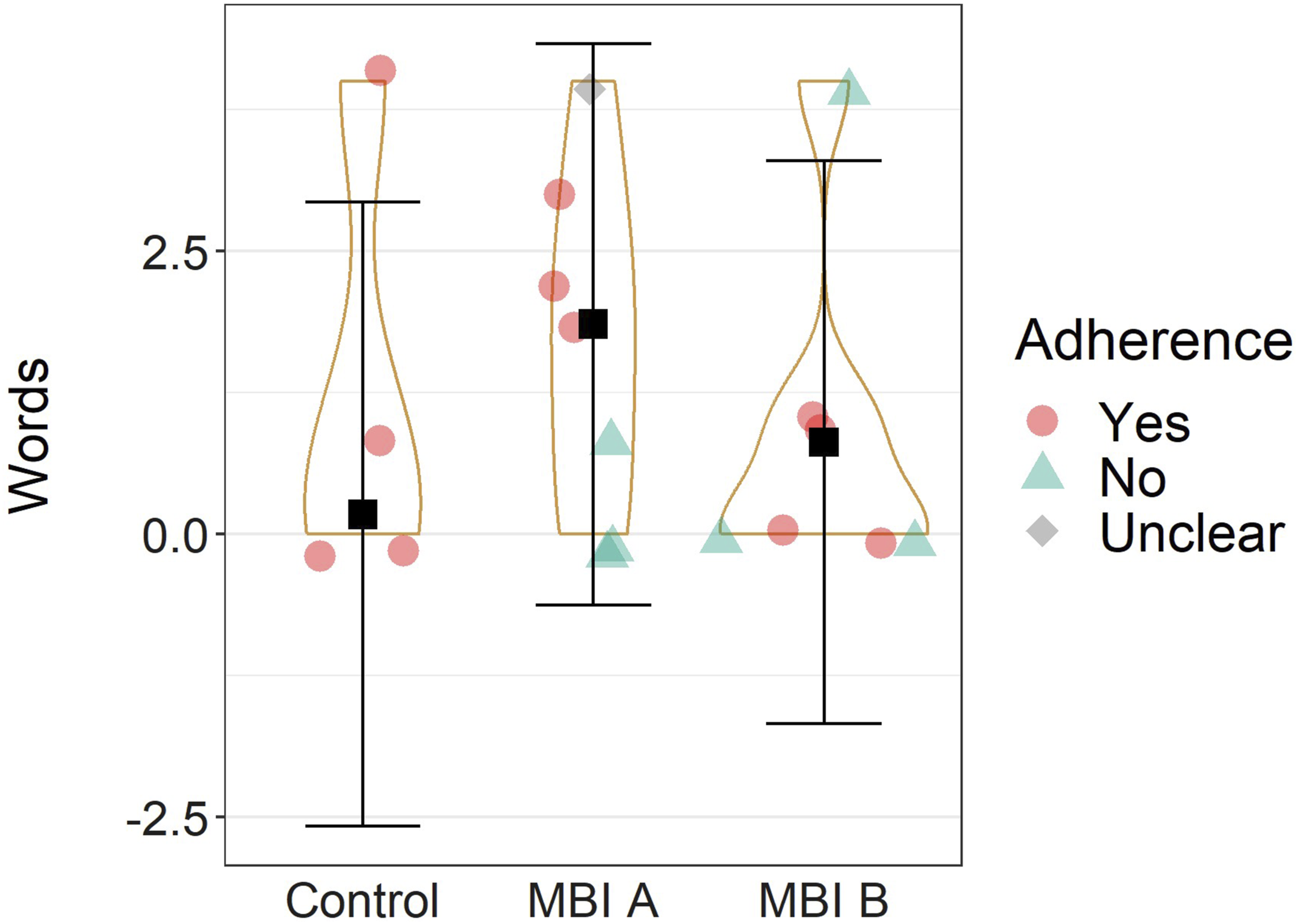
Exploratory analysis
Combining MBI data and analysing the effect at 6 and 12 weeks compared to control, we found a significant, positive effect of the MBI on quality of life after 6 weeks (6 weeks MBI- Control: 1.79 [95% CI: 0.06, 3.52], p = .04, Hedge’s g av = 0.51). No other effects of measurement time point were observed (see Table S3).
Analysis of descriptive data
Nine (56 %) dyads reported focused music listening, while five (31 %) described a mix of focused listening and using the music as a background sound, and two (13 %) reported the music being used exclusively as a background sound. In line with quantitative results, none of the dyads reported notable symptom changes. However, most reported in-the-moment effects of the MBIs on the person with dementia (MBI A: 73%, MBI B: 81%). During MBI A, six (40 %) showed physical engagement, such as clapping, whistling, swaying or humming. Five (33 %) dyads highlighted that MBI A elevated the person’s mood or reinforced a pre-existing good mood. Conversely, five (33 %) noted that the person with dementia seemed agitated by MBI A but indicated no longer lasting negative effect. For MBI B, nine (56 %) dyads reported a calming effect on the person with dementia. Notably, one of the caregivers said playing MBI B at bedtime helped the person with dementia sleep through the night, which had not been possible previously. Another highlighted that the relaxation eased a Parkinson-related upper body rigor. Three dyads (19 %) said that MBI B induced some transient emotional upset/sadness in the person with dementia.
Personal hurdles for using the MBIs were (1) busy personal schedules (n = 5, 31 %), (2) illness and hospitalisation (n = 2, 13 %), and (3) the person with dementia showing disinterest or resistance (MBI A: n = 3 (20 %), MBI B: n = 2 (13 %)). None reported technical issues hindering the use of the MBI. The most common suggestion for improvement, made by half of dyads (n = 8), was to extend the playlists. Further suggestions were to individualise the playlists according to person with dementia’s music taste and biography (n = 5), using more simplified technology (n = 2), and using a personal device for engaging with music (n = 1).
Discussion
The central goal of this pilot trail was to evaluate and compare the effects of two MBIs on cognition, well-being, quality of life, and behavioural and psychiatric symptoms. While our research was only sufficiently powered to detect differences of around one unit, we report the findings here as the presence of absence of such larger differences is informative to a research field that overall lacks quantitative comparison of MBIs. Limited drop-out and dyads’ largely positive descriptions indicated that the intervention was feasible. Yet, findings also highlight the importance of supporting dyads in carrying out the MBIs, as, for each MBI, only about half of the sample was adherent. This is in line with recent findings by Baker and colleagues, who, in spite of telephone support by qualified music therapists, found only 61 % adherence in their caregiver-delivered MBI (Baker et al., 2023). In the descriptive data, dyads cited busy schedules as a hurdle. Dyads suggested that ensuring that people with dementia could use the MBI on their own could increase adherence. Independent intervention use by people with dementia is a very valuable next step, particularly given that between one third and half of community-dwelling people with dementia are living alone (Eichler et al., 2016; Miranda-Castillo et al., 2010).
With respect to BPSDs, we had expected slow, positively valenced music to reduce overall severity. However, contrasting previous findings (Hofbauer et al., 2022; van der Steen et al., 2018), neither MBI showed a significant effect on BPSDs. Given our small sample size, this might not reflect a true null effect. Rather, as our research was only sufficiently powered to detect substantial differences, it may suggest the absence of such substantial differences, without excluding the presence of more subtle changes. In fact, dyad’s descriptions reveal in-the-moment effects of the MBI on behaviour and affect. People with dementia’s reaction to fast, positively valenced music was described as characterised by movement and induced/maintained positive mood. Conversely, slow, positively valenced music was described as having a calming effect on people with dementia. These reports of notable, if transient, effects might suggests that outcome measurements were either not sensitive enough or that effects are highly individual. Alternatively, effects of MBIs may be dose-dependent and null findings in caregiver-delivered MBIs could thus be the result of low adherence (Baker et al., 2023). The absence of large, long lasting benefits need not necessarily deter further research in this area. Community-dwelling people with dementia and caregivers report valuing non-pharmacologic interventions even if their benefit is restricted to the moment (MacPherson et al., 2009; Tuckett et al., 2015).
Regarding cognitive performance, we hypothesised that fast, positive music would be associated with benefits in the cognitive domain. This was seen in delayed recall, for which fast, positively valenced music was associated with superior performance compared to slow, positively valenced music. While the advantage amounted to only one more word remembered, this is a notable difference of nearly one standard deviation. Fast, positively valenced music may allow for improved cognition by ways of heightening mood and arousal, an effect that has been observed for background music and music immediately preceding cognitive testing (Calabria et al., 2023; Husain et al., 2002; Mitterschiffthaler et al., 2007) but not yet for music listening over a longer period. It remains to be investigated why such an effect might differentially affect delayed recall in particular. However, low power – due to some people with dementia being unable to complete cognitive testing – showing in large confidence intervals surrounding estimates, may have led to underestimation of some effects. These findings in the context of a pilot trial are valuable, as they highlight that future research efforts interested specifically in cognitive or self-report domains will have to adjust recruitment procedures to ensure the inclusion of people with dementia capable of such reports. Alternatively, further research in this area will have to rely more heavily on proxy-report measures.
Similarly, not all people with dementia were able to provide self-reports of quality of life and well-being. Based on previous findings (Hofbauer et al., 2022; van der Steen et al., 2018), we had anticipated both MBIs to benefit these domains. Combining data of both MBIs, we see an initial increase in quality of life after 6 weeks of music listening. Yet, this effect seems to diminish over time. Comparing the effect of each MBI on quality of life to care as usual, we see non-significant increases in each MBI for which the effect sizes fall in the range identified in meta-analysis of music-based interventions (van der Steen et al., 2018). This suggests that null-findings may be the result of low power. At the same time, QoL-AD and WHO-5 are broad measures, which include domains unlikely to be affected by an MBI, such as the financial situation. As a recent review has pointed out, measures of well-being and quality of life in people with dementia are still evolving, and a greater focus on in-the-moment reports would likely provide more useful insights into intervention effects (Clarke et al., 2020).
There are limitations to this research. Overall sample size was small. Thus, the study only was sufficiently powered to observe large differences between interventions. Specifically, it can be estimated that for measures that the whole sample completed a 1 unit difference between interventions could be detected at an 80 % power level, while for outcomes with for example half the sample a difference would have to reach 1.6 units to be detected at that power (Schoenfeld, 2010). Further, we cannot make inferences about potentially varying responses to the MBIs between different dementia aetiologies, as we did not record type of diagnosis. While in the present investigation the sample size would not have sustained detailed sub-group analysis, future investigations may make such distinctions, as experimental evidence points to dementia-subtype specific responses to music (Baird et al., 2018, 2020; Garrido et al., 2018).
In conclusion, while we expected to see diverging effects of a MBIs with fast or slow positively valenced music, findings from our pilot study found little quantitative evidence of this. Fast, positive music was associated with improved delayed recall; all other comparisons were non-significant. Based on dyads’ descriptions, we conclude that additional differences likely exist but are mostly observable in-the-moment, requiring refined assessments. Moreover, given that cognitive testing and self-report outcome measures could only be completed by a subsection of participants, future research will have to either adapt the recruitment strategy to ensure that a sample with less severe impairments is recruited or rely on proxy measures. Overall, the MBI created value for the person with dementia, as music listening was associated with positive, if transient, effects. Thus, music listening may be recommended to people with dementia to gain moments of well-being. Importantly, even though further research is needed, these findings underline the necessity of carefully considering the nature of music in conjunction with the individual needs of the person with dementia. While the MBI design under investigation can be described as feasible, results also show that adapting it for independent use by people with dementia may be a valuable next step.
Supplemental Material
Supplemental Material - Comparing two caregiver-delivered music listening interventions for community-dwelling people with dementia: A randomised controlled crossover pilot trial
Supplemental Material for Comparing two caregiver-delivered music listening interventions for community-dwelling people with dementia: A randomised controlled crossover pilot trial by Lena M Hofbauer, and PD Francisca S Rodriguez in Dementia
Footnotes
Acknowledgements
We would like to thank all participants for their collaboration and contribution. The first author, LMH, would like to thank Nadja Ziegert, BSc, for her support in the data collection process.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Clinical trial registration
The protocol of this study was pre-registered with the German Clinical Trials Register (ID: DRKS00030688).
Data availability statement
The data that support the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
