Abstract
Background
Reablement is recommended to maximise functioning in people with dementia, yet in Australia, is not routinely available. This study aimed to provide insight into the implementation and program outcomes of reablement in real-world practice for a person living with dementia.
Methods
Reablement was implemented for a client with dementia. In parallel, a formative mixed-methods pilot evaluation was performed, using single-case experimental A-B-A design (n = 1), supplemented by routinely collected pre-post program clinical measures. Implementation was evaluated qualitatively via clinical notes for fidelity, feasibility and client engagement.
Results
Single-case experimental design outcomes indicated the program positively impacted the participant’s physical functioning. Additionally, most routinely collected pre-post clinical measures demonstrated improvement. Intervention fidelity varied, with differences in length and client engagement.
Conclusion
Implementation of evidence-informed reablement has been shown to be feasible in real-world practice for a community-dwelling person living with dementia. Larger implementation trials are needed to build on preliminary outcomes to ultimately improve access to these important programs.
Keywords
Introduction
Dementia is one of the leading causes of disease burden and disability in Australia (Australian Institute of Health and Welfare, 2022) and worldwide (World Health Organisation, 2021). People with dementia experience progressive loss of functional ability necessitating increasing support to complete activities of daily living (Giebel et al., 2014; Liu-Seifert et al., 2015; Martyr & Clare, 2012). Functional ability is determined by the combination of an individual’s intrinsic capacity (the composite of their physical and mental capacities) and their environment (World Health Organisation, 2015). Accordingly, it is one of the leading determinants of quality of life (QoL) for people with dementia (Burks et al., 2021). Consequently, interventions that delay functional decline are highly valued by people with dementia (Guideline Adaptation Committee, 2016), particularly as pharmacological interventions offer limited efficacy in improving outcomes or slowing disease progression (Szeto & Lewis, 2016; Tisher & Salardini, 2019).
Healthcare systems are under pressure to provide quality dementia care services to a growing population using limited resources (World Health Organisation, 2017). Functional ability and length of admission to residential care (largely determined by functional ability) (Olsen et al., 2016) are the most important cost drivers in dementia care (Brown et al., 2017; Gustavsson et al., 2011; Standfield et al., 2019). At an individual level, transition to residential care is also predictive of significant negative changes in QoL (Olsen et al., 2016). Therefore, interventions that maintain function to prolong people with dementia living at home are desirable from both person-centred and economic view-points (Standfield et al., 2019; Tisher & Salardini, 2019).
Reablement is an emerging multidisciplinary approach that uses an individual’s intrinsic capacity and environmental modifiers (e.g. housing modifications, mobility aids and assistive technologies) to maintain function, regain lost function where possible and compensate for changes in function (Poulos et al., 2017). By maximising functional ability, reablement aims to increase QoL, reduce service utilisation (Tessier et al., 2016), decrease carer burden, and potentially delay the need for long-term residential care (Poulos et al., 2017). Reablement aligns with the broad recommendations for a wellness approach to care stated in the Australian Clinical Practice Guidelines for People with dementia (Guideline Adaptation Committee, 2016). However, a lack of clinical detail within the guidelines (for example, recommended intervention length, frequency and structure) mean challenges remain around delivering reablement in practice. A recent study of the Australian aged care sector found that reablement has thus far been inconsistently defined in the literature, leading to potentially improper implementation. As providers urgently offer interventions labelled ‘reablement’ to align with funding schemes, services delivered are not necessarily evidence-informed (O'Connor et al., 2020). To address this gap, a series of evidence-informed, freely available ‘reablement in dementia’ resources were developed (O'Connor, Gresham, et al., 2022; O'Connor, Rowlands, & Poulos, 2022) to support health professionals in designing, delivering and evaluating reablement programs for clients with dementia (Gresham et al., 2019; O'Connor et al., 2019; O’Connor & Poulos, 2021; Poulos et al., 2019).
It is widely acknowledged that successful implementation of evidence into clinical practice is challenging as research and clinical environments can be vastly different (Clemson et al., 2021; Culph et al., 2021; Proctor et al., 2009). Real-world clinical environments present a range of individual and system-wide factors that can impact the successful translation of research evidence (Azim et al., 2022). Studies to test the effectiveness of the translated research are needed to aid health professionals and potential clients in their decision making pertaining to funding, delivering or participating in these programs (O'Connor, Gresham, et al., 2022; Poulos et al., 2018). While the reablement in dementia resources have been freely available since 2018, there are yet to be any implementation studies of their use, and there is presently limited uptake of these resources within the Australian aged care sector (O'Connor et al., 2020; O'Connor, Gresham, et al., 2022). Greater understanding of the implementation of these resources within real-world practice, using existing sustainable funding sources is needed to help bridge the evidence-practice gap. The outcomes of these evaluations have the potential to inform future practice (Highfield et al., 2015), increase sustainability and drive uptake of evidence-informed reablement in the broader dementia care sector (Proctor et al., 2009).
Whilst large scale implementation trials are recognised as important, the quality of many implementation trials over more than a decade has been criticised (Lewis et al., 2018; Powell et al., 2012, 2015) and as such it has been proposed that pilot studies should play a greater role in preceding definitive studies in this area (Pearson et al., 2020). The role of a formative pilot study in an implementation context should extend to identifying potential causal mechanisms of changes to the research evidence in clinical practice, and facilitate an iterative process of refining intervention strategies and optimising their impact (Pearson et al., 2020). Pragmatic implementation-effectiveness studies have already been used in a similar Australian context to aid the translation of other evidence-based reablement programs (Clemson et al., 2021; Jeon et al., 2020).
Therefore, in advance of larger definitive implementation studies, this formative pilot study aimed to explore: (1) program outcomes for a community-dwelling person living with dementia who participated in a reablement program delivered by an existing allied health service, and (2) implementation of the ‘reablement in dementia’ resources, mainly the reablement handbook, within routine clinical practice.
Materials and methods
Design
To address the dual research foci above, this study used a hybrid type 2 design with mixed methodology (Curran et al., 2012) to undertake a formative pilot evaluation (Stetler et al., 2006) of a reablement program designed using the reablement handbook (Poulos et al., 2019), delivered within an existing community aged care service setting. A simplified overview of the study design can be seen in Figure 1 Overview of the mixed-methodology used within this study. 5×STS = Five Times Sit to Stand Test; MCID = Minimal clinically important difference; MDC = Minimal detectable change. See Table 1 for a more comprehensive list of outcome measures. Figure 1 Alt Text: A multi-level flow chart illustrating the hybrid design of the study. The first level shows that aims pertain to both program outcomes and implementation exploration. The second level shows methodologies used; specifically that exploration of program outcomes involved single case experimental design (SCED) and routine pre-post clinical measures, and exploration of implementation outcomes involved review of clinical notes. The final level shows measures and analysis processes used, involving 5xSTS for SCED, change scores and clinical differences for pre-post measures, and qualitative description of clinical notes for implementation exploration.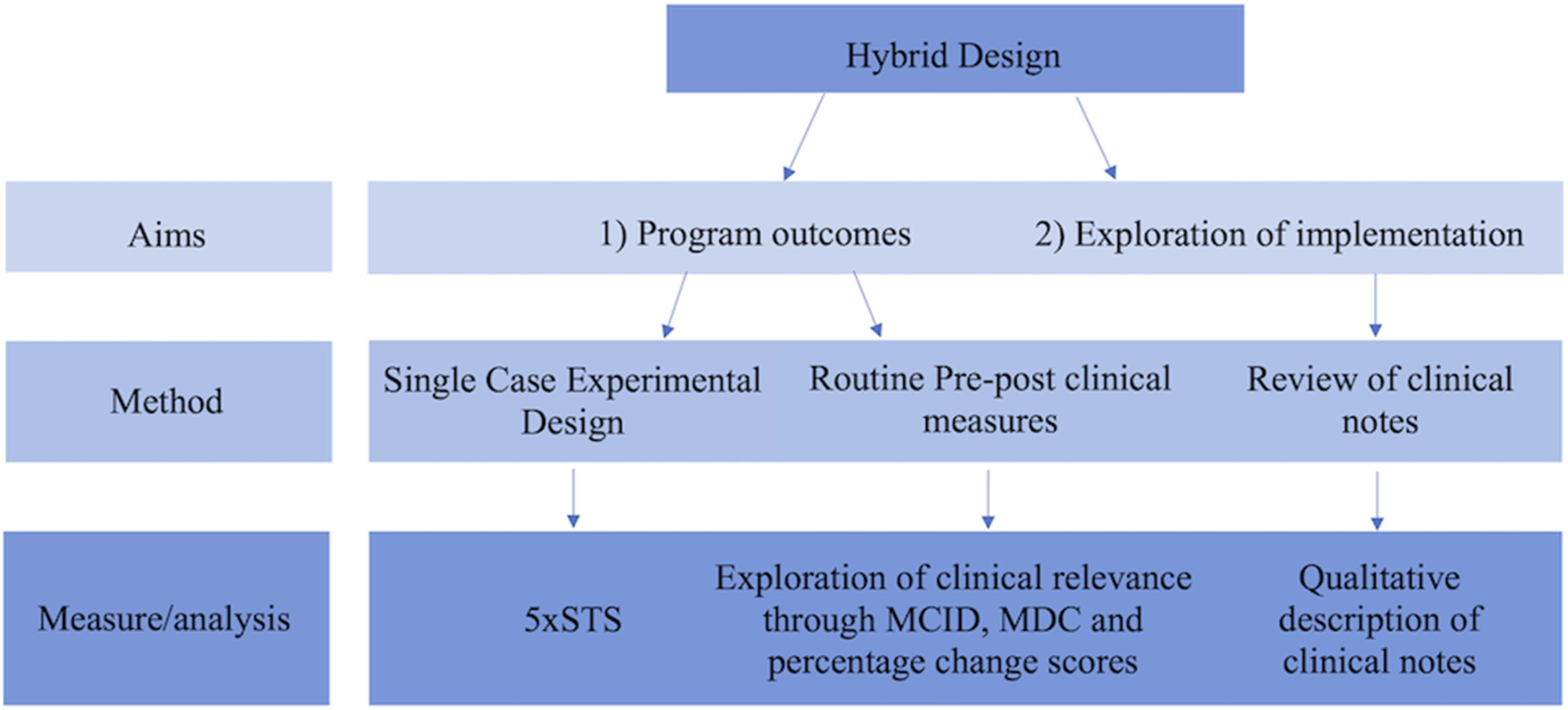
To add methodological strength to this study, a single-case experimental design (SCED) of A1BA2 withdrawal/reversal type was superimposed on the reablement program using a proxy outcome measure for functional change (Tate et al., 2016). In the baseline (A1) phase and the post-intervention (A2) phase the participant maintained their daily activities as usual. During the intervention (B) phase, the allied health team delivered a reablement program over eight weeks. The A1 and A2 phases lasted approximately two weeks, with A1 phase length determined by the time elapsed between program referral, study recruitment and reablement program start; A2 phase mirrored A1 in terms of both length and the number of datapoints collected. There was no washout period between the A and B phases.
The SCED methodology was designed according to the Risk of Bias in N-of-1 Trials Scale (Tate et al., 2013). In addition, routinely collected pre-post program clinical data were collated and qualitative clinical notes pertaining to program delivery and implementation were extracted (Kim et al., 2017).
Setting
This study was conducted via an established community aged care service in Sydney, Australia. The research team worked collaboratively with the allied health team within the service to coordinate recruitment and data collection in parallel with routine service provision. The reablement intervention was delivered as part of a short-term restorative care program (STRC) (Australian Government, 2024). STRC is a federal government funded program for community-dwelling people over 65-years-old, which aims to improve functional ability to promote independence (Australian Government, 2024). Intervention delivery and outcome measurement collection were undertaken at times of mutual convenience within the participant’s home.
Participant
Purposive sampling was undertaken to recruit the participant. Inclusion criteria were that the participant must: (a) have a diagnosis of dementia or identified memory impairment, (b) be living in the community and enrolled to receive a reablement program, (c) have capacity to provide informed consent or, if not, have a responsible guardian willing to provide consent on their behalf and support their participation, and (d) be 18-years-old or above.
Ethics
Study approval was obtained from the University of New South Wales Human Research Ethics Committee (HC210759), and written informed consent was obtained from the participant.
Intervention
During the B phase, the participant engaged in a reablement program based on the ‘reablement in dementia’ handbook (Poulos et al., 2019), tailored specifically to address their identified therapeutic goals. To promote fidelity to the resources in delivery of the reablement program, a virtual training session was provided to the allied health team by one of the researchers who was involved in development of the resources (last author). The reablement program was designed by the clinical allied health team (an occupational therapist and physiotherapist) in reference to the reablement handbook, but accounting for any restrictions of their clinical environment. For example, program length (8 weeks) was determined by the STRC funding structure. At the beginning of their program, the participant identified two primary goals for their program in collaboration with the treating occupational therapist and physiotherapist. The therapist-adaptation of these goals (O’Connor & Poulos, 2021) were:
Mobilise safely outdoors with a mobility aid for approximately 60 metres. (physiotherapist) Increase safety and independence within the home using small aids for showering provided through the STRC program. (occupational therapist)
Based on these goals, program 4.1 (“supporting mobility and physical function through a falls prevention program” (p.38; Poulos et al., 2019) from the reablement handbook was used to inform the program that was delivered.
The occupational therapist assessed the participant’s functional ability, prescribed equipment to increase home safety (including a personal response system, a stair rail for home access and bathroom aids), and coordinated the participant’s care throughout the program. In parallel with the occupational therapist, the physiotherapist also performed an independent assessment of physical functional ability, delivered a series of face-to-face exercise sessions tailored to the participant’s physical and cognitive abilities, and prescribed a home exercise program to be completed outside these visits using physiotherapist-provided equipment (e.g. resistance bands and a pedal exerciser). A more in-depth exploration of the fidelity of the delivered program to the reablement handbook is included in the results section.
As part of the broader STRC program, through which the reablement intervention was being delivered, the participant received two in-person consults from a dietitian and podiatrist and one in-person consult from an audiologist.
Outcome measurements
The SCED outcome measure was the Five Times Sit to Stand (5xSTS) (Bohannon, 2011). The 5xSTS has excellent reliability in demonstrating physical functional ability and falls risk in community dwelling older people (coupled with ease of administration and low cost), therefore the clinical allied health team deemed it an adequate proxy for the participant’s reablement goals (Buatois et al., 2008; Melo et al., 2019). The 5xSTS involves measuring the time taken for the participant to transfer five times from a seated position to a standing position as quickly as possible using a standard height chair (43–45 cm) with straight back (Melo et al., 2019). The less time taken to complete the test, the better the outcome. Performance times greater than 13.6 seconds are associated with increased disability and morbidity in community dwelling older adults (Ardali et al., 2020). As per SCED guidelines (Tate et al., 2016), a minimum of five 5xSTS data points were collected per phase. The proxy outcome measure was collected by members of the research team (A-phases; AC and CMCOC) and the clinical allied health team (B-phase).
Outcome measures assessed in this study.

PROMIS = Patient-Reported Outcomes Measurement Information System.
SMART = Specific, Measurable, Attainable, Relevant and Time-bound.
Data analysis
The SCED outcome data was graphed for each stage and analysed visually as per recommendations for SCED studies (Kratochwill et al., 2010; Lane & Gast, 2014; Tate et al., 2016). Changes in mean, trend and variability of data across phases was used to evaluate the impact of the reablement program on the 5xSTS using systematised guidelines by Kratochwill et al. (Kratochwill et al., 2010) to increase the accuracy of interpretation (Krasny-Pacini & Evans, 2018; Kratochwill et al., 2010). In addition, percentage of non-overlapping data between phases was used to provide an indication of effect size (Chen et al., 2016).
For the routinely collected pre-post clinical measures, percentage change scores (baseline to post-program) were analysed to explore program outcomes. Percentage change scores were calculated for each outcome by dividing the change score (baseline score subtracted from the post-intervention score) by the absolute value of the baseline score, multiplied by 100 (Zhang & Han, 2009). Percentage change scores present results in clinically relevant terms to patients and clinicians and have been used previously in small sample studies (O'Connor et al., 2021; Savage et al., 2014). Where available, the authors report the meaningful clinical significance of change scores.
For the implementation data, clinical notes were content analysed to extract information around program feasibility, funding, fidelity, participant engagement, and whether or not there were adverse events associated with participating in the program. These details were compared to the reablement handbook (Poulos et al., 2019). No interviews were conducted with the participant, family or delivering service providers.
Results
Demographic information
Despite aiming for 3-6 participants to provide inter-subject replication, only one eligible participant was successfully recruited within the study timeframe. This participant completed their reablement program between June and August 2022.
Demographic and clinical characteristics of the participant at baseline.

RUDAS = The Rowland Universal Dementia Assessment Scale.
aInitials changed for anonymity.
NL lived in the community with her son’s family, was home alone a minimum of three days per week, and attended a day respite centre two days per week. The STRC referral reported that NL received minimal support from those she lives with apart from shopping and the preparation of evening meals. Whilst NL reportedly “feels she can manage herself” (dietitian notes), her healthcare was coordinated by her nephew who does not live with her but who “expressed concerns regarding her memory and her ability to manage” (Aged Care Assessment Team support plan). NL’s nephew was acting as her primary informal carer who “phones daily and provides regular monitoring and prompts with medication and meals, transport to appointments and other tasks as required” (Aged Care Assessment Team support plan). The physiotherapist identified that “since the knee operation [year unknown] and moving houses [NL’s] mobility has declined” (physiotherapist discharge summary).
SCED outcomes
Baseline A1 phase had a mean 5xSTS score of 20.4 seconds (standard deviation [SD] = 4.9 seconds). Visual inspection of the baseline phase suggested an increasing (i.e. worsening) trend across the phase with a clear pattern of all data points being above the clinically significant cut-off, indicating increased risk for disability and morbidity. During the intervention B phase, the mean 5xSTS score improved to 16.3 seconds (SD = 5.8 seconds), representing a 20% mean baseline reduction. This phase had a decreasing (improving) trend, with the latter three data points below the performance cut-off, indicating lower risk for disability and morbidity. When the intervention was removed in the A2 phase, the mean 5xSTS score further improved, decreasing to 14.9 seconds, with the narrowest variability of all the phases (SD = 1.8) and a decreasing (improving) trend (Figure 2). Reviewing the percentage of non-overlapping data, 50% (three data points) of the B phase were below the lowest A1 data points. When comparing B and A2 phases, there were no overlapping data. This indicates that while there was latency of effect between A1 and B phase initially, there was a sustained effect of intervention into A2 phase. In addition, there was a mean difference of 5.5 seconds between A1 and A2 phases, which is more than double the minimum detectable change identified for 5xSTS performance in community-dwelling females aged 60-years-old and above (Goldberg et al., 2012). 5×STS SCED outcomes across all three phases (A1BA2). A lower time indicates better performance. The dashed horizontal line at 13.6 seconds indicates the clinically significant cut-off over which there is an association with increased disability and morbidity in community dwelling older adults (Ardali et al., 2020). An extended celeration line has been added from A1 to B phase, and B to A2 Phase. A linear trend line has been added to A2 Phase for visual interpretation. Figure 2 Alt Text: three panel graph showing STS time points from each study phase. Panel one shows A1 phase times, panel 2 shows B phase times, and panel 3 shows A2 phase times.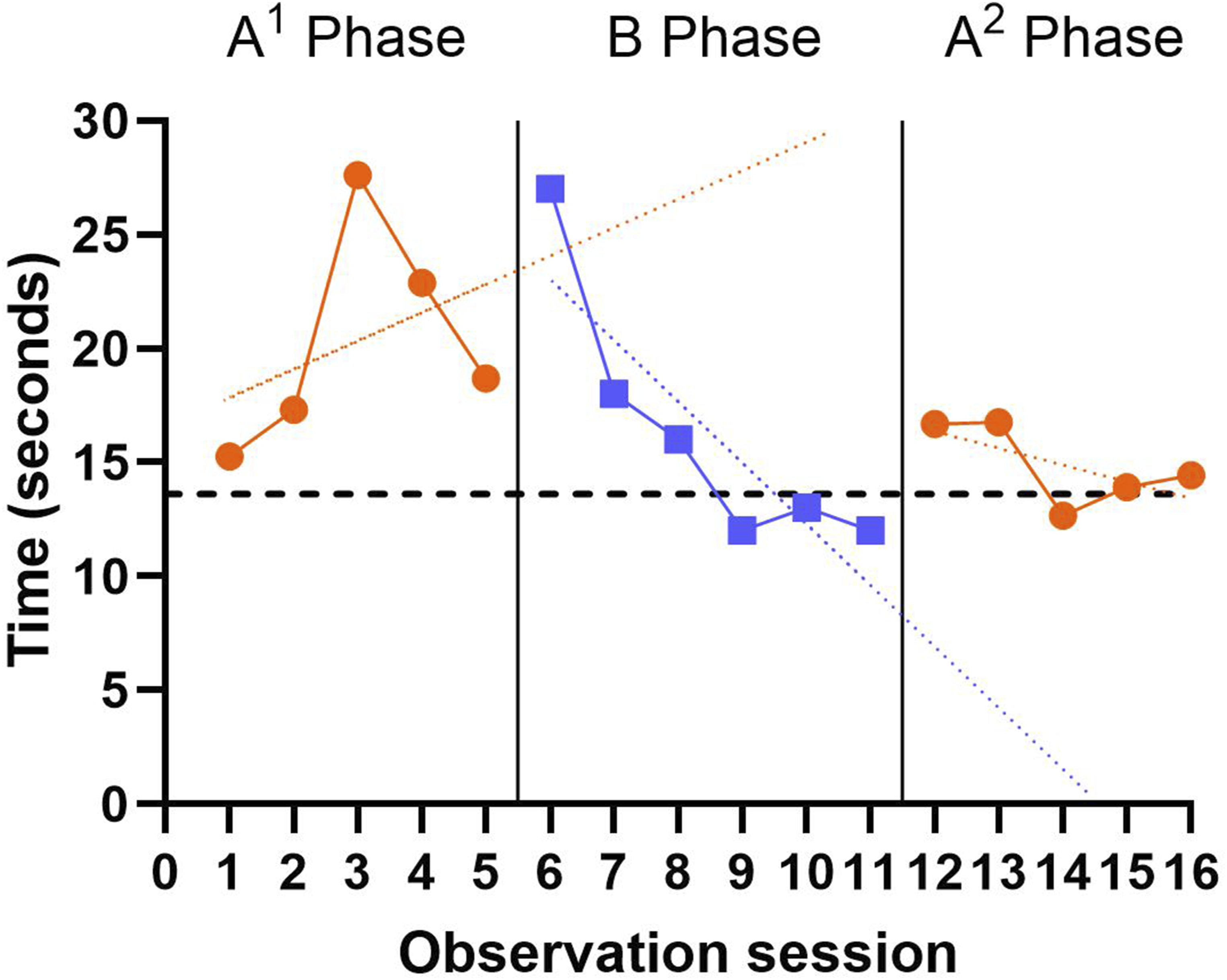
Routinely collected clinical outcomes
Outcomes of the routinely collected pre-post clinical measures.
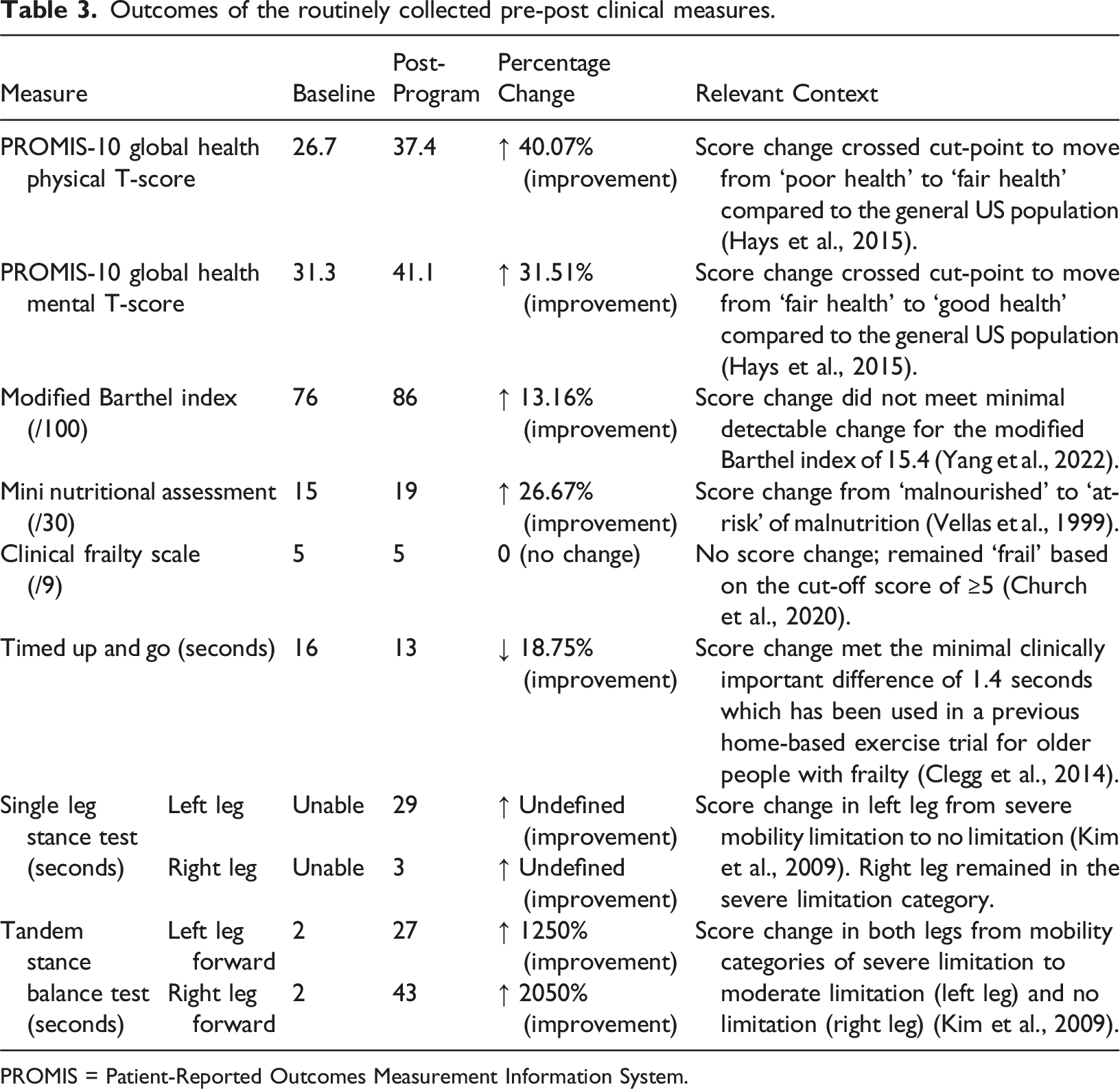
PROMIS = Patient-Reported Outcomes Measurement Information System.
Goal Attainment Scaling-Light scores.

Implementation outcomes
Feasibility and funding
The STRC package was a viable funding avenue for delivering an evidence-informed reablement program to a community-dwelling person with dementia. Feasibility of implementation was demonstrated by the collaborative approach exhibited by the occupational therapist and physiotherapist to supply equipment, assistive technology, education and tailored exercise sessions as per the reablement handbook.
Fidelity
Comparison of the reablement program delivered versus program 4.1 outlined in the ‘reablement in dementia’ handbook (Poulos et al., 2019).

Importantly, handbook consistent practice was also noted, including session lengths of 30–60 minutes and the tailored nature of the exercises and home safety modifications that were delivered and/or prescribed. As per handbook recommendation, considerable attempts at increasing the sustainability of the reablement program through involving an allied health assistant and family were also made; these are discussed below.
Participant engagement
During face-to-face sessions, NL was able to “engage in exercises with proper support and prompts” (physiotherapist). However, it was surmised that outside of the face-to-face sessions NL was “unlikely to complete her home exercise program independently due to apathy and memory issues” (physiotherapist, discharge summary). This was potentially exacerbated by the above-mentioned lack of engagement from NL’s family. To support the participant in completing the exercises between visits when home alone and thus increase handbook fidelity of the program, the physiotherapist trialled visual aids, placed in clearly visible spaces in the home. These “didn’t work well with NL as she doesn’t like things hanging on the wall/fridge” (physiotherapist). Similarly, equipment provided by both the occupational therapist and physiotherapist was “stored…in a cupboard to keep them neat and out of the way and [NL] would therefore forget to use them” (occupational therapist).
Consequently, as it appeared NL needed social support to perform the exercises, an allied health assistant was engaged to assist with extra exercise sessions. However, they were only able to complete one session before scheduling conflicts meant that no further allied health assistant-led sessions were provided.
Another finding relevant to participant engagement was that NL “continued to complain of isolation [and was] excited to have someone to talk to” (physiotherapist) throughout the program. On multiple occasions therapist notes indicated the perception that NL received significant social benefits from the reablement program which were an additional benefit to the functional improvement aims of the reablement handbook.
Adverse events
No adverse events were reported for the duration of the program.
Discussion
This study provides preliminary evidence regarding the feasibility of implementing evidence-informed reablement for community-dwelling people with dementia and the potential for positive program outcomes within a real-world clinical setting. There is a growing body of evidence for home-based reablement programs to enhance function and independence in community dwelling people with dementia (Cezar et al., 2021; Dawson et al., 2019; LeDoux et al., 2020; Teri et al., 2020; Ávila et al., 2018). However, most of these studies were performed in a research context. A reablement intervention has undergone implementation research in Australia, however this program requires specialised training of practitioners with associated financial investment (Clemson et al., 2021). To the best of the author’s knowledge, the current study is the first in Australia to specifically explore the implementation of, and program outcomes from, freely available, evidence-informed reablement resources in a real-world setting (O'Connor, Gresham, et al., 2022). Therefore, this study provides an important link between preliminary works that have been conducted within a research setting, and the translation of research evidence into practice, by continuing to bridge the reablement evidence-practice gap (Clemson et al., 2021; Corvol et al., 2018).
Positive outcomes were observed around both aims of this mixed-methods study, with reablement program benefits identified via the SCED and routinely collected clinical data, and feasibility of implementation of the reablement handbook demonstrated within an established community aged care service. Variable program fidelity to the reablement handbook was identified, often due to clinical constraints (Kourouche et al., 2021; McGee et al., 2018). This mirrors previous research, which has found that complex healthcare interventions are often not delivered as planned, as health professionals modify interventions to fit within their service context (Cross et al., 2022; French et al., 2021; Lorencatto et al., 2014; Toomey et al., 2015; Wenborn et al., 2021). Importantly, the reablement handbook itself acknowledges that the research evidence collated to form its recommendations are “often generated using strict protocols and specifically trained staff, and may be conducted within a research environment [meaning] that not all research protocols may be suitable to, or sustainable outside of, this environment” (p.9; Poulos et al., 2019). Therefore, despite the differences in program delivery that were reported, the positive quantitative outcomes observed for this participant suggest that pragmatic modifications to the ‘reablement in dementia’ resources to facilitate implementation within real-world practice may still generate positive outcomes for people with dementia, but further research is needed to determine the limits of this flexibility.
An important clinical challenge identified by this study was the difficulty of engaging the participant with dementia in their reablement program outside of face-to-face sessions with treating therapists. NL was identified by the physiotherapist as potentially being impacted by apathy which affects 50-70% of people with dementia (Selbæk et al., 2013). Apathy is defined as a loss of motivation that is accompanied by diminished self-initiated behaviour and reduced goal-directed cognitive activity (Tampi & Jeste, 2022). Consequently, it is unsurprising that NL did not participate in her between-session home exercise program, and it is likely that many others with dementia would face similar challenges as supported by previous work that proposes apathy creates barriers to goal pursuit in reablement (Golenko et al., 2022).
Considering these challenges with program engagement, informal carers such as family have long been established as integral to motivate the person with dementia and support them in activity initiation and engagement (Baber et al., 2021; Chang et al., 2021). Indeed, family carers are a vital determinant of positive health outcomes for people with dementia, providing a protective effect against the risk of transition to residential care, and a better quality of life (Farina et al., 2017; Olsen et al., 2016). As encouraged by the sustainability recommendations of the reablement handbook, the physiotherapist and occupational therapist endeavoured to engage the family as active members in the reablement program, though this was ultimately unsuccessful. Prior studies have noted that opinions and expectations of health professionals and patient families can often be mismatched in reablement programs (Hjelle et al., 2016; Jakobsen et al., 2019). This has previously been theorised to be due to insufficient clarity regarding the limits of involvement of next of kin, how the distribution of responsibility should be assigned between next of kin and health professionals, and what each party hopes the program will achieve at the start (Jakobsen et al., 2019). There is potential that such a mismatch between client, family and health professional expectations about the reablement program contributed to the communication challenges and consequent departures from fidelity (e.g. lack of occupational therapist in-person follow up) found in this study.
Clinicians should aim for a more family-centred approach that encourages greater dialogue between people with dementia, their next of kin and health professionals (Doh et al., 2020; Hjelle et al., 2016; Jakobsen et al., 2019). However, challenges exist around successfully engaging family in reablement programs. A recent study (Jakobsen et al., 2019) outlined the paradox that even when health professionals acknowledge next of kin as important partners in reablement, they are simultaneously unsure how to effectively collaborate, particularly in Western cultures where there is significant value placed on the autonomy of the client with dementia. Healthcare professionals must navigate maximising family involvement without exerting pressure on next of kin to fulfill a role beyond their capacity as this has been shown to have negative health consequences for this group (Jakobsen et al., 2019; Poulos et al., 2019). Clear front-end communication of program extent, objectives, and review appointments both with participants and their family may help to align program aims for each person, thus improving compliance with the planned intervention and avoiding some of the challenges encountered in the present study.
This formative pilot study faced considerable challenges recruiting participants as a result of the constraints associated with working with a singular service over a limited time-frame in a low incidence population, as previously recognised (Olive & Smith, 2005). Considerable literature highlights the difficulty research studies have meeting recruitment targets (Beckers et al., 2019; Duncan et al., 2018; Gorton et al., 2022; Realpe et al., 2021). Simultaneously, it is well documented that people with dementia are a hard-to-reach population and recruiting the numbers needed for research relevant to them is challenging (Bartlett et al., 2019; Black et al., 2014; Cooper et al., 2014). This is important as future definitive implementation studies will likely face similar challenges which should be considered in project design. A recent exploration by Field et al. (Field et al., 2019) of organisational and individual difficulties with research recruitment of people with dementia provided recommendations for future recruitment planning. These recommendations should be considered when designing future SCED based service evaluations to address the barriers faced in this project.
This study has some limitations that must be considered. As this study involved evaluation of implementation within routine clinical practice, it was not possible to provide extensive intra-case replication (e.g. ABAB design). We therefore aimed to provide inter-case replication by recruiting 3-6 participants to allow for the establishment of a multiple baseline design that would have increased replication and external validity (Krasny-Pacini & Evans, 2018; Kratochwill et al., 2010; Tate et al., 2016). Ultimately, challenges around recruiting within a clinical context led to a small sample size. Despite this, outcomes provide important preliminary evidence on the feasibility and potential positive impact of delivering reablement for people with dementia living in the community.
Despite the limitations of SCEDs, using recommended visual analysis procedures have been recognised as valuable in bridging the evidence-practice gap (Tate & Perdices, 2020). Whilst visual analysis has notable shortcomings, it is the most appropriate approach as applying inferential statistics to SCEDs, particularly single participant studies, is inappropriate as the data are inherently autocorrelated, thus limiting the choice of appropriate statistical analyses (Beeson & Robey, 2006; Olive & Smith, 2005). When compared to a pre-post design, the A1BA2 withdrawal/reversal design itself strengthens internal validity (Tate et al., 2016). While the A1BA2 design is not the highest standard of SCED (Krasny-Pacini & Evans, 2018), it was fit for purpose within this service context where only one intervention period (B) was attainable due to the financial limitations imposed by STRC funding, and therefore, provides greater applied clinical potential (Australian Government, 2024). Indeed, it has been argued, that in the age of personalised medicine, the restrictive inclusion criteria of gold-standard randomised control trials makes these difficult to generalise and translate into real-world practice (Krasny-Pacini & Evans, 2018).
As a formative pilot study, the findings presented here are only preliminary and should be interpreted with caution, however, through evaluation of handbook-recommended versus actual reablement implementation, study outcomes have potential to inform the design of more definitive implementation studies in the future and provide guidance to health professionals currently implementing the resources in practice to optimise their impact.
Recommendations for future research include further work into the flexibility of reablement resource implementation within a variety of service contexts and funding models. Replication via SCEDs can be achieved across studies as long as the 5-3-20 rule is satisfied (i.e. “a minimum of five SCED studies examining the intervention, conducted by at least three different research teams in three different geographical locations with a combined number of 20 single-cases across the papers”) (pg 21; Kratochwill et al., 2010). The present study used only one of 27 distinct ‘programs’ of the reablement handbook as this was relevant to the participant and their goals; it is likely future studies in other contexts will use other handbook subsections. Future studies therefore could have the twofold effect of extending the research to other ‘programs’ within the handbook and replicating evidence to strengthen generalisability of results regarding implementation of the ‘reablement in dementia’ resources. Finally, the authors would like to encourage research into the implementation of reablement in rural areas, particularly via telehealth to determine its unique challenges and outcomes for this underserviced population (Bauer et al., 2019; Sekhon et al., 2021).
Conclusion
This study provides insight into how evidence-informed reablement can be implemented within clinical practice to support community dwelling people with dementia. Despite variable fidelity to the reablement handbook, improvements to the participant’s physical functional abilities were observed. Larger implementation trials are the vital next step to build on these preliminary positive findings. In addition, whilst this study reported outcomes from one specific ‘reablement in dementia’ program, future work should explore the feasibility of other sections of the ‘reablement in dementia’ resources in different contexts. It is hoped that healthcare professionals will consider the challenges outlined above when designing reablement programs, and also the potential for modifying programs to fit within real-world contexts. The benefits of engaging in evidence-informed reablement programs for people living with dementia should be a key focus in future service development. By demonstrating a financially feasible pathway the authors hope that future service providers will be empowered to identify similar funding avenues, and inspired to seek new ones to improve access to reablement for people with dementia living in the community.
Footnotes
Acknowledgements
The authors would like to thank the participant, their family, and the clinical allied health team for participating in and collaborating on this study. We would also like to acknowledge and thank Angela Collins for supporting the conduct of this research within the community allied health team.
Author’s contributions
Study concept and design: AC, CJP, CMCOC, KL. Acquisition of data: AC, CT, JA, CMCOC. Analysis and interpretation of data: AC, CMCOC. Preparation of manuscript: AC, CMCOC. Critical revision of manuscript for important intellectual content: All authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: COC is funded by a Dementia Centre for Research Collaboration (DCRC) Post-doctoral Fellowship.
