Abstract
Objectives
Sleep disturbances are highly prevalent and have adverse health consequences for both people living with dementia and their carepartners. Despite this, they are under-addressed caregiving settings. This study aimed to explore these sleep disturbances and co-design a multimodal sleep intervention for people living with dementia and their carepartners.
Methods
We conducted two focus groups and five semi-structured interviews (n = 4 people living with dementia, n = 6 carepartners). Active involvement of community advisors was sought throughout the design, development, and facilitation phases. Reflexive thematic analysis was used to explore sleep-related experiences and receive feedback to shape intervention development.
Findings
People living with dementia reported disruptions to sleep and circadian rhythms, including sleep disturbances and confusion between day and night. Multiple sleep challenges were encountered by carepartners including insomnia, hypervigilance, and daytime impairment. The proposed sleep intervention was received positively, with significant insights emphasising the need for a multimodal toolkit approach, adaptation of the intervention across different dementia stages, and a focus on tailoring the program to carepartners.
Conclusion
Sleep interventions for caregivers and care-recipients should target both sleep and daytime functioning to ensure holistic support. Participants were receptive towards time-friendly, online, multimodal sleep interventions that combine cognitive behaviour therapies, light therapy, mindfulness, and exercise elements.
Introduction
Sleep disturbances are prevalent and challenging for both people living with dementia and their carepartners. Up to 90% of people with dementia experience sleep disturbances, often accompanied by disrupted circadian rhythms (Hjetland et al., 2020; Rongve et al., 2010). Sleep and circadian disruption are associated with negative outcomes for people living with dementia, including confused awakenings, hallucinations and recurring nightmares (Hjetland et al., 2020; Volicer et al., 2001), increased severity of dementia, and a greater likelihood of transitioning to an aged care facility due to safety concerns and excessive night-time activity (Gaugler et al., 2009). Comorbid medical conditions, such as chronic pain, cardiovascular diseases, diabetes, and respiratory disorders (e.g., obstructive sleep apnoea), often exacerbate sleep difficulties experienced by people living with dementia (Cordone et al., 2021; Deschenes & McCurry, 2009; Foley et al., 2004).
Family carepartners often provide round the clock care and frequently experience sleep difficulties themselves. Up to 75% of carepartners report poor sleep (Reynolds et al., 2020), and over 91% report regular sleep problems (Peng et al., 2019). Difficulties in initiating and maintaining sleep stem from sleep fragmentation and increased vigilance to support the night-time needs of care recipient (Brewster et al., 2023; Mahoney, 2003; Smyth et al., 2020). Even though good sleep is recognised as a key factor in helping carepartners cope better, it appears to be vastly under-addressed in caregiving settings (Gibson et al., 2023). Compared to the general population, carepartners of people living with dementia are at a higher risk of reporting elevated levels of stress, anxiety, depression (Ferrara et al., 2008; Wiegelmann et al., 2021), and insomnia (Brewster et al., 2023). This is often due to caregiving needs, where care responsibilities coupled with sleep loss exacerbate sleep disturbances and distress in carepartners (McCurry et al., 2015). Addressing sleep issues in people living with dementia and carepartners is crucial, given sleep’s protective role in health, quality of life, and cognitive functioning (Gao et al., 2019; Scott et al., 2021).
Existing sleep treatments primarily focus on managing insomnia in general populations (Gu & Lee, 2023), but are under-tested for people living with dementia and carepartners. Pharmacological interventions, such as benzodiazepines, antipsychotics, antidepressants, and hypnotics are widely prescribed but carry substantial risks, including increased risks of falls, early mortality, drug dependency, and impaired cognition (Lader, 2011; Maher et al., 2014; Zisapel, 2012). Melatonin, which has fewer side effects, is increasingly used in this population to improve sleep onset latency, though findings on its efficacy are mixed (Bedward et al., 2024; Cruz-Aguilar et al., 2018). Orexin antagonists have shown improvements in total sleep time, wake-after-sleep onset, sleep efficiency (Herring et al., 2020), and overall insomnia symptoms (Hamuro et al., 2018), however have been associated with next-day somnolence, which is a reported side effect (Bedward et al., 2024; Blackman et al., 2021). For carepartners, pharmacological sleep treatments may be used in conjunction with treating depression and anxiety symptoms, however, significant side effects, as mentioned above may limit their practicality (McCurry et al., 2007). Consequently, pharmacological treatments are recommended for short-term treatment or as a final option for managing and treating sleep-related difficulties for people living with dementia (Javed et al., 2023; Ooms & Ju, 2016), and may not be a suitable choice for carepartners, prompting a growing interest in the non-pharmacological interventions.
Cognitive Behavioural Therapy for Insomnia (CBT-I) is a non-pharmacological sleep intervention that addresses sleep-related thoughts and behaviours (Pignatiello et al., 2022). Recent research has found CBT-I effective at improving sleep and daytime impairment in individuals living with dementia and mild cognitive impairment (Naismith et al., 2019; O’Caoimh et al., 2019), carepartners of people living with dementia (Gao et al., 2019; Pignatiello et al., 2022) and treating insomnia in the general population (Mitchell et al., 2012; Trauer et al., 2015). However, CBT-I is not intrinsically tailored to address the unique challenges across caregiving contexts, where sleep disturbances may occur as a result of daily responsibilities and stress related to caregiving (Baron & Hooker, 2017). Mindfulness-based therapies have also been shown to reduce insomnia symptoms in carepartners (Jain et al., 2014) and improve cognitive functioning in people living with dementia (Hoffman et al., 2020), but require additional behavioural support.
Other approaches, such as bright light therapy, tailored sleep education programs, and exercise have also been trialled separately in people living with dementia and carepartners, with promising effects on sleep outcomes (Figueiro et al., 2019; Fontana Gasio et al., 2003; Forbes et al., 2014; Gibson et al., 2017; McCurry et al., 2007; Tewary et al., 2018). However, the effectiveness of bright light therapy for people living with dementia varies greatly, possibly due to heterogeneities in study designs and the need for a behavioural component (Figueiro et al., 2019; Fontana Gasio et al., 2003; Hjetland et al., 2020; McCurry et al., 2011; Wilfling et al., 2023). Exercise interventions have been found to improve total sleep time, sleep efficiency and wake-after-sleep onset, but must be carefully individualised to the recipient with carepartner assistance (McCurry et al., 2011; Richards et al., 2011). Additionally, carer-focused interventions, including education about dementia, sleep hygiene, and relaxation techniques, have been shown to improve an array of sleep outcomes, enabling carepartners to return to sleep faster after awakenings (McCurry et al., 2007). Other multi-modal carer-focused approaches such as the CAregiver Sleep Intervention (CASI) and the NITE-AD intervention have been well-received and show improvements in sleep quality for carepartners (Simpson & Carter, 2010) and people living with dementia (McCurry et al., 2005; Tewary et al., 2018).
Combining optimal strategies from all evidence-based therapies holds the potential to address practical challenges with caregiving, including the complexities in care, time constraints and financial limitations, while also improving sleep and well-being among families living with dementia. Recent systematic reviews (O’Caoimh et al., 2019; Pignatiello et al., 2022) advocate tailored multi-modal interventions, integrating therapeutic components such as cognitive and behavioural techniques, physical activity therapy, light therapy, and mindfulness. These recommendations aim to comprehensively address the factors contributing to sleep disturbances among carepartners and people living with dementia, while also acknowledging the association between sleep, cognitive changes, and functional decline. While there are a growing number of studies employing a multimodal approach to treat sleep disturbances for people living with dementia (McCurry et al., 2005, 2011; Richards et al., 2011), and dyads including carepartners (Gibson et al., 2017; Rapaport et al., 2024; Song & Kim, 2021), few have been co-designed with input from those with lived experience. Recognising the importance of lived experience and tailored approaches, co-design has an important role in developing interventions aimed at addressing the unique sleep disturbances experienced within care-recipient and caregiver dyads (Blomkamp, 2018; Trischler et al., 2018). This collaborative approach emphasises the integration of perspectives and experiences of those living with dementia and their carepartners, shaping interventions that address their specific needs (Schleider, 2023; Steen et al., 2011).
Based on this understanding, our research team proposed an evidence-based multi-modal sleep intervention incorporating CBT-I, mindfulness-based and fatigue management strategies. We adopted a participant-centred approach to understand sleep disturbances and practical challenges experienced by people living with dementia and carepartners. We aimed to identify perceived barriers to implementing sleep strategies in their day-to-day lives and to gain feedback and perspectives on our proposed multimodal sleep program for dyads of carepartners and people living with dementia. Specifically, we focused on the intervention design (including the frequency and duration of the sessions), the content (such as potential strategies to include in the intervention and topics covered) and the delivery (for example, in-person vs. online sessions, individual vs. group sessions).
Methods
Participants
We engaged with Dementia Australia’s Consumer Engagement Officer and their network of community advocates to distribute information about our project on our behalf. Participants were recruited through convenience sampling based on their willingness to engage in open discussions about their sleep experiences and offer feedback on the proposed sleep intervention. Individual carepartners and people living with dementia were recruited for the study, with separate focus groups/interviews conducted for carepartners and people living with dementia. While dyads could participate, we used broader recruitment to allow people at various stages of caring to participate (e.g., people who were carepartners in the past). This approach allowed participants to reflect on their experiences, and engage in discussions with others in similar situations, while also being asked to reflect on the dyadic nature of the proposed sleep intervention.
Inclusion criteria for carepartners included those with past or current caregiving role(s) for a person living with dementia. Former carepartners were also included in the study based on the finding that, as caregiving roles evolve, some individuals develop a more nuanced understanding of the effects of sleep deprivation (Gibson et al., 2023). For people living with dementia, inclusion was based on self-reported or carepartner-reported diagnoses, confirmed through a phone interview. During the call, information was gathered regarding the date of diagnosis and current symptoms, however this information was not recorded to ensure the confidentiality and privacy of participants. Exclusion criteria involved individuals who were unable to provide informed consent on their own behalf due to significant cognitive impairment. This was due to the potential challenges that individuals living with significant cognitive impairment might encounter in responding to questions during the focus groups and interviews, and their potential inability to provide consent for study participation (Clarke & Keady, 2002; McKeown et al., 2010).
Community engagement
Two community advisors (author AP, who has lived experience with dementia, and author TV, who has personal experience with caregiving) were invited to assist with designing and reviewing the focus group/interview discussion guide, facilitating the focus groups and interviews (with participant’s consent), and reviewing participant related documentation, including explanatory statements, consent forms and advertisement materials. Their attendance throughout the focus group and interview sessions, in particular, aimed to create a sense of comfort for participants and to ensure that the researchers facilitated the sessions with a respectful and considerate approach. They actively engaged during the discussions by asking questions and follow-up inquiries and shared their personal experiences. The input of the community advisors was also sought during the redesign phase of the Dementia, Sleep and Wellbeing intervention (Verma et al., 2023).
Focus group and interview discussion guide
The focus groups and interviews followed the same discussion guide and included open-ended questions with two primary objectives. Firstly, to identify sleep difficulties and subsequent daytime impairments experienced by both carepartners and people living with dementia and included questions about common sleep changes, when the changes typically occurred, and common barriers to seeking help for sleep-related challenges. Secondly, to collect feedback and recommendations related to the content, structure, and delivery of the proposed sleep intervention. Specifically, participants were asked to provide their input on the likelihood of using the proposed sleep strategies (e.g., mind-body techniques, sleep hygiene and light strategies etc) and the potential barriers to adopting them. Participants were asked to provide their feedback on the appropriateness of the individual components of the sleep intervention. To facilitate this discussion, descriptions of the proposed components were screen-shared with the participants over Zoom. Participants were also asked questions regarding the delivery (in-person or virtual), duration and length of the intervention, and their perspectives on having a manual as a reference throughout the intervention.
The discussion guide was developed by the research team and underwent iterative reviews, including input from the Dementia Australia Consumer Engagement Officer and consultation with our two community advisors. The guide was modified following each review phase. The discussion guide can be found in Table 1 of the Supplemental materials.
Proposed sleep intervention: Dementia, sleep and wellbeing
The development of the proposed sleep intervention, tentatively named the Dementia, Sleep and Wellbeing project, was led by author SV in collaboration with authors TV and AP (lived experience community advisors) and the research team, which included experts in sleep and dementia. Prior to developing the intervention, author SV engaged in dementia awareness training, including sleep and related psychosocial experiences of carepartners. The 6-week intervention was tailored toward dyads of carepartners and people living with dementia. It incorporated the principles of CBT-I with elements of light therapy, mindfulness techniques and strategies to manage fatigue. The intervention comprised of six structured modules: (1) introduction to the program and psychoeducation on sleep and insomnia; (2) light and sleep interventions for optimising circadian rhythms; (3) relaxation and mindfulness; (4) fatigue management; (5) recap of topics covered; and (6) sustaining sleep/mood strategies for the future. Each module included a review and reflection on the previous session, psychoeducation on key concepts and concept-specific strategies, as well as goal setting for the upcoming week. Each module was accompanied with a handout outlining the key concepts and strategies. An outline of the intervention is available in Table 2 of the Supplemental materials.
Procedure
This manuscript presents a pilot feasibility study of the Dementia, Sleep and Wellbeing intervention, as well as a framework for co-designing a multi-modal intervention with lived experience. The study was approved by Monash University Human Research Ethics Committee (Approval no. 29545). Participants were recruited via online advertisement on the Dementia Australia research website during September 2021. Those who expressed interest were contacted via telephone for screening, and had the study procedures explained, including recording of sessions and presence of community advisors. Consent was obtained both verbally and electronically during the phone call. One week before their scheduled focus group or interview, participants were offered the opportunity to review the discussion guide and the proposed Dementia, Sleep and Wellbeing intervention. They were encouraged to share their written comments/feedback via email if they found it more convenient and were also invited to contact the research team after the focus groups for individual discussions if desired.
Authors PV (a postdoctoral research fellow with experience in co-design and sleep interventions) and SV co-facilitated the focus groups; SV facilitated the five semi-structured interviews. Both PV and SV completed further learning in consumer and community involvement and received guidance from community advisors in attempts to enhance safety and sensitivity of facilitation for participants. Community advisors TV and AP attended both focus groups and a majority of the interviews. The duration of the focus groups and interviews ranged between 60 and 90 minutes. They were conducted virtually via the videoconferencing platform Zoom (Zoom Video Communications, Inc, 2021) and recorded using the Zoom local recording feature and transcribed verbatim using a transcription service.
Data analysis
The data was analysed using reflexive thematic analysis (TA) as proposed by (Braun & Clarke, 2013, 2021a), within an interpretivist framework. An interpretivist approach recognises that reality and knowledge are shaped through the interpretation of diverse experiences, contexts, and perspectives, acknowledging the existence of multiple and varied realities (Hudson & Ozanne, 1988). A key aspect of reflexive TA is acknowledging positionality and maintaining reflexivity throughout both data collection and analysis. Authors PV, SV, and MJ bring backgrounds in psychology and sleep intervention development, offering insights into common sleep difficulties and treatments. Author AB has a background in working with people living with dementia and carepartners and author JD has a background in psychology. During the focus groups and interviews, authors PV and SV recognised the inherent dynamics in research and adopted a flexible and sensitive approach. This included being adaptable in the structure of the focus groups and interviews, such as allowing for breaks, following participant’s lead on discussion topics and allowing flexibility in attendance. Additionally, they built rapport and established trust with participants before and during the sessions and created a supportive environment by asking the groups to maintain respectful and safe group guidelines.
Reflexive TA comprises six-phases, including familiarisation, coding, generating initial themes, developing and reviewing themes, defining and naming themes, and writing (Braun & Clarke, 2021a). Throughout the data analysis, the research team acknowledged their own potential biases and varied experiences and perspectives. They focused on understanding participants’ experiences and continuously referred back to the aim of the research. The authors moved between the different phases of the analysis to develop a comprehensive and nuanced understanding of the findings (Terry et al., 2012). This approach facilitated the emergence of deeper insights and interpretations as the authors continually engaged and reflected upon the data (Terry et al., 2012). During the familiarisation phase, authors PV, JD and AB re-read the transcripts and recorded their initial impressions. Using an inductive approach, PV and JD assigned code labels to meaningful data sections, which were repeatedly reviewed and revised to ensure rigour throughout the analysis. Authors PV and JD created initial themes by grouping codes that shared meaning within the data set using QSR International NVivo 12 Plus (QSR International Pty Ltd, 2023). These themes were evaluated by PV, JD, MJ, and AB to ensure that the codes best reflected the themes and the broader research objective. A visual thematic map was developed to depict theme relationships and names were assigned to each theme. Extracts that best represented the themes were included in the final report. Braun and Clarke’s checklist was referred to for methodological rigour throughout the data analysis process (Braun & Clarke, 2021a). Following Braun and Clarke’s reflexive thematic analysis, data saturation was not aimed for (Braun & Clarke, 2021b). Instead, the focus centred on the depth and quality of the collected data, ensuring that the data was relevant to the aim of our research (Braun & Clarke, 2021b; Malterud et al., 2016).
Findings
Participant characteristics
The study included a total of 10 participants, including six carepartners and four people living with dementia. Among the carepartners, two identified as current carers, while four were former carers. The mean age of carepartners was 63.68 years (SD = 15.50, range = 34–75), and 69.0 (SD = 4.7, range = 62–72). The majority of the sample were female (n = 8).
The focus groups comprised three carepartners in Group 1 and two people living with dementia in Group 2. In addition, three one-on-one interviews were conducted with carepartners and two interviews with people with dementia to gather further detailed information after the focus groups. These additional interviews were conducted (i) to gather further detailed information from carepartners who were not spouse/partners, (ii) due to increased interest in the study and (iii) for participants who were unable to attend the focus groups.
Challenges in sleep for people living with dementia and carepartners
Three themes were constructed relating to sleep challenges experienced by people living with dementia: (1) Insomnia, (2) The impact of medical conditions on sleep, and (3) Collaborative sleep strategies for supporting people with dementia. Four themes were also constructed concerning sleep difficulties among carepartners: (1) Insomnia, (2) Hypervigilance and stress, (3) Daytime impairment, and (4) Effective strategies used by carepartners. Figure 1 presents the thematic map that illustrates the relationship between sleep-related difficulties as reported by people living with dementia and carepartners, along with their feedback regarding the proposed Dementia, Sleep and Wellbeing intervention. Thematic map of the themes related to sleep difficulties and feedback on the Dementia, Sleep and Wellbeing intervention as constructed by people living with dementia and carepartners.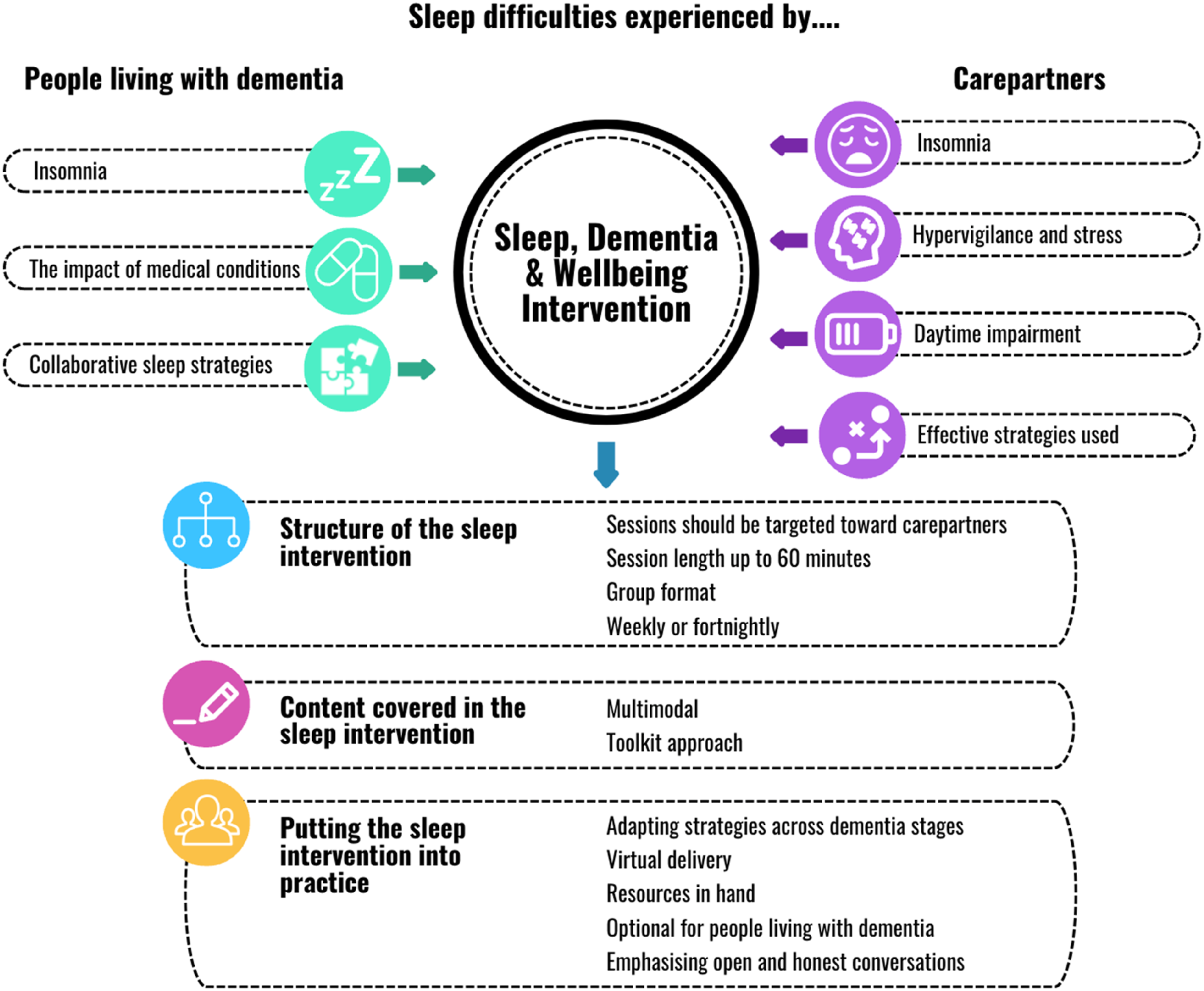
Sleep-related difficulties experienced by people living with dementia
People living with dementia discussed a number of sleep-related difficulties that they frequently experience.
Insomnia
People living with dementia shared their challenges in both initiating and maintaining sleep throughout the night. Multiple awakenings during the night were also a common occurrence, often attributed to concerns about frequent urination, vivid dreams or nightmares. “The nights when my brain just won’t stop and I’ve got to get up. I just can’t, it’s pointless me even trying to sleep.” (Person living with dementia). “So I find wakefulness being awake all night sometimes is really, really difficult.” (Person living with dementia). “I wake up very early and very vivid dreams that sometimes [cause] disorientation from day to night.” (Person living with dementia).
Consequently, both people living with dementia and their carepartners reported a noticeable increase in mental fog and fatigue during the daytime. “I’d be 12 hours in bed, and I’d be sleeping and I wouldn’t wake up rested. And my brain fog was so thick that I have about an hour and a half in the morning to do things with, but I could fake it till I make it.” (Person living with dementia).
The impact of medical conditions on sleep
The co-occurrence of various medical conditions alongside dementia and how these conditions affect sleep patterns were discussed. Notably, chronic pain emerged as a prominent factor leading to disruptions in sleep during the night for people living with dementia. “It’s pain…I can’t sleep and I have to get up and it’s just, it’s a nightmare.” (Person living with dementia).
Another medical condition contributing to sleep disturbances for people living with dementia was obstructive sleep apnea (OSA). Following an OSA diagnosis, some individuals accepted continuous positive airway pressure (CPAP) machine treatment if recommended, which notably enhanced their sleep quality. However, others described difficulties with the CPAP masks, leading to night-time awakenings and posing challenges to achieving restful sleep. “The biggest problem is the mask, because it is such high pressure.” (Person living with dementia).
Collaborative sleep strategies for supporting people with dementia
This theme centred on the collaborative efforts of carepartners and people living with dementia to implement strategies aimed at improving the sleep quality of individuals with dementia. During the day, intellectual and physical stimulation was emphasised, such as engaging in music, art, and exercise. Several night-time strategies such as getting into bed together and waiting until the person with dementia falls asleep, adhering to a consistent and early bedtime, going to the bathroom before bedtime, and encouraging a winding down before sleep were being used to support sleep. “My family have learnt not to wind me up before it’s bedtime because I can’t. My filters are damaged…so if there’s bad news that can wait till tomorrow.” (Person living with dementia).
Carepartner’s sleep experiences
Carepartners described insomnia and hypervigilance, associated with stress and caregiving responsibilities as some of their primary challenges with sleep.
Insomnia
Carepartners discussed their experienced symptoms of insomnia, such as difficulties falling asleep, staying asleep and waking up early. They suggested that their sleep disturbances, particularly difficulties falling asleep, largely began after their care recipient was diagnosed. “I don’t fall asleep [in]front of the telly typically I don’t go to bed till quite late. Staying asleep and getting to sleep is probably the worst for me. I get my best sleep from about five and if I didn’t have an alarm, I wouldn’t get up till about nine, but I do get up.” (Carepartner). “Early on, I had really bad sleep ‘cause I was grieving and that was the only time I could do it is because you’re facing the person all the time and there’s a bit of not wanting to believe its true and all those kind of things. I think the first year or two, that was probably the thing that stopped me sleeping the most.” (Carepartner).
Sleep fragmentation and subsequent difficulty going back to sleep were common issues. For example, night-time awakenings of the person living with dementia, night-time wandering, showering, and dressing were described as behaviours that frequently disrupted carepartners’ sleep at night. “She has frontotemporal dementia, and she has real disorientation between day and night…to the point that before she was diagnosed, she was up in the middle of the night, rearranging the shed and making lots of noise.” (Carepartner).
Lastly, some carepartners described how they still experience sleep difficulties, which have persisted following changes to their caregiving responsibilities. “[I’m] now say 14 months on [and] still have issues, which I think are slowly resolving. It obviously has a big impact on what used to be quite not a problem for me at all. That’s my experience that I’m bringing here today.” (Carepartner).
Hypervigilance and stress
Because of night-time awakenings, carepartners shared that they experienced pre-bedtime stress and were hyper-aware about being woken by the person living with dementia. They referred to “shallow” sleep, characterised by the feeling of not entering deep sleep during the night due to anticipation of being woken up or to check for any safety concerns. The additional stress experienced by carepartners due to separation anxiety exhibited by the person living with dementia was also discussed. “After the morning that I found him upstairs with the gas heater on, I realised that I was now at a stage where I was needing to care for someone who had the cognitive ability of a younger child, and he could get himself into any sort of danger.” (Carepartner). “I realised that if I got into bed with him and sat up on my laptop he was happy, as long as I was in bed because he had separation anxiety and he wanted to be where I was.” (Carepartner).
Daytime impairment
Many carepartners described feeling exhausted throughout the day as a consequence of inadequate sleep, which affected their daily functioning, and resulted in increased irritability and mood changes: “I guess what I call survival mode in that there are certain things that I need to get done today…washing and cleaning that sort of thing. It would take me more effort to do those things because I was so tired.” (Carepartner).
Effective strategies used by carepartners
To address their insomnia symptoms and daytime impairments, carepartners described a number of strategies that they had employed to help manage these changes. These included establishing bedtime routines or engaging in sleep hygiene practices, such as taking a shower before bed to relax, turning the lights low or using soft lighting before bed, going to bed only when tired, and eliminating or reducing caffeine intake. Physical activity was also utilised to promote better sleep and overall health. “I just can’t stress enough the importance of exercise and there is just more and more evidence every day coming out from researchers around Australia about the importance of exercise for managing all sorts of other issues as we age in terms of diseases.” (Carepartner).
In relation to sleep medications, carepartners shared varied approaches, some refrained from using any, while others mentioned utilising melatonin or antihistamines to aid in sleep initiation. Additionally, some carepartners had tried or been prescribed benzodiazepines and mirtazapine. However, most carepartners, regardless of whether they used medications or not, expressed concerns about their addictive nature and either used them cautiously or avoided them altogether.
Feedback on the sleep intervention design and delivery
The next set of themes centres around the input and recommendations provided by both people living with dementia and carepartners regarding the proposed sleep intervention. Participants shared their views on the structure of the intervention, the content, and how the intervention should be delivered.
The structure of the sleep intervention
Sessions should be targeted towards carepartners
Both people living with dementia and carepartners advised that given each person with dementia has a different journey, it may be more beneficial to ‘upskill’ carepartners with sleep strategies that they can implement for themselves and for the person living with dementia rather than having sessions as a dyad. Ways in which this could be achieved were discussed, including potentially having carepartner-only sessions, which would allow carepartners to discuss their own experiences freely and gain insights from others in similar situations. “It might be easier to have the carer speak away from the person they’re caring for to be able to provide some frank and honest sort of answers or be able to participate freely.” (Carepartner).
Session length of up to 60 minutes
The consensus was that an hour-long session would be sufficient to maintain people’s attention and would also be more manageable for carepartners to incorporate into their schedules. I think about an hour as you don’t want too much because I can’t concentrate more than that. (Person living with dementia).
Group format
Both carepartners and people living with dementia agreed that conducting the sleep intervention sessions in group formats would be effective and the benefits of this approach were discussed. The group-based format was seen as an opportunity for carepartners and people living with dementia to form meaningful connections with others and reduce feelings of isolation. Smaller groups (up to six) were considered ideal, and privacy and confidentiality were flagged as key concerns that needed to be managed. Participants appreciated the opportunity to listen and learn from others with similar experiences, enabling them to connect and gain insights. “The benefits around a group format [are] that people are hearing other people’s stories as both carers and people living with dementia, and you can actually connect with those stories and gain information from insights, but also solutions that they may have” (Carepartner). “Carers are quite isolated and looking for that connection and sharing those stories are really important, but also for someone living with dementia, having a connection with other people who have dementia, because I think it normalises dementia then.” (Carepartner).
Weekly or fortnightly
Both carepartners and people living with dementia suggested either weekly or fortnightly sessions (i.e., every two weeks) as their preferred frequency for the sleep intervention. They also discussed the importance of maintaining flexibility in scheduling the intervention throughout the day and highlighted the importance of considering various factors that can influence levels of engagement. “The other thing is the time of the day, you’ve really got to allow the person living with dementia to name what time of the day that their brain works best.” (Person living with dementia).
The content covered in the sleep intervention
Multimodal
A program with multiple aspects of sleep improvement, such as CBT-I, mindfulness, light therapy and sleep education was positively received. Carepartners also emphasised the need to incorporate other strategies that may have indirect effects on sleep, such as physical activity, daytime stimulation, unwinding and pain management techniques. These additional elements were considered valuable in promoting better sleep patterns and supporting overall well-being. “I think that part of it is just acknowledging that added level of complexity, that whatever intervention you are providing, that it is not going to be an isolated bubble in amongst all the complexity of the other stuff going on.” (Carepartner).
Toolkit approach
Carepartners emphasised the need for regular check-ins and the use of real-time examples to ensure ongoing support and enable practical implementation of the program. To tailor the program, carepartners and people living with dementia recommended a toolkit approach, to ensure greater flexibility to choose from different strategies that suit their individual needs and circumstances. Within the toolkit approach, it was advised that the first session should be individual/dyad to establish initial circumstances and set up goals. “For me, that sounds really good because you’d be working with a carer around their specific areas then also have that more general approach around the toolkit in terms of the strategies that you might be able to utilise.” (Carepartner).
Putting the sleep intervention into practice
Adapting sleep strategies across dementia stages
People living with dementia and carepartners recommended that the sleep intervention should be designed in a way that is beneficial for all stages of dementia. It was recognised that certain strategies could be suitable depending on the stage of dementia of those engaging with the intervention. It was therefore recommended that the intervention strategies were adaptable to accommodate for this variability. “As things decline…the strategies that might work at the start may need to evolve over time. So, understanding where the person is in their dementia experience and being able to say, if you are experiencing X, Y and Z, here are some ideas that you might want to try. And as things change, then different strategies might be helpful.” (Carepartner).
It was emphasised that the person delivering the intervention should be experienced in responding appropriately to people living with dementia if they demonstrate confusion or if they experience distress during the sessions (e.g., providing breaks).
Virtual delivery
Carepartners and people living with dementia agreed that the virtual delivery of the intervention was an effective method. They noted that the increased familiarity with technology due to the impact of the coronavirus pandemic made it easier for people to engage in virtual communication.
Resources in hand
Given the toolkit approach to the intervention, it was recognised that a substantial amount of information would likely be presented during the sessions. As such, written materials to accompany the intervention were suggested by both people living with dementia and carepartners. Having written materials to refer to throughout the sessions was deemed valuable and was also described as a helpful resource to refer back to once the intervention had concluded. They emphasised the importance of keeping written materials concise and avoiding using excessive text, suggesting the use of short summaries presented in dot points.
Optional for people living with dementia
In certain circumstances, it was proposed to offer the sessions as optional for people living with dementia to attend. This consideration was based on factors such as level of insight and engagement. For example, a carepartner described how her father’s limited recognition of his diagnosis and sleep difficulties would have made active participation for him a challenge. “In our personal experience, my dad never acknowledged he had dementia so he probably wouldn’t have gone to a group like this…so being involved in an intervention like this [it] would have had to be presented in a different way for him to participate.” (Carepartner).
Nonetheless, it was strongly recommended that the choice for people living with dementia should be made available and that adjustments be made to facilitate this, including the option for people living with dementia to step out of the session if needed. “I think it would be quite good to have a carepartner…then sometimes the person with dementia can have a bit of a break when you talk to the carepartner.” (Person living with dementia).
Emphasising open and honest conversations
Finally, the importance of making the intervention and supporting materials easily accessible was emphasised by all participants. They recommended making the sessions as informal as possible, resembling more of a ‘chat,’ and using simple language while avoiding complex scientific terms. “Ask questions that are appropriate and don’t confuse with doublespeak. Keep things simple so that they don’t have to think a lot about what is being asked…use layman terms and be respectful and be clear.” (Person living with dementia).
The participants underlined the need for direct, clear communication, and avoiding token gestures. They preferred straightforwardness over ambiguity and emphasised the importance of respectful interaction. “A lot of us don’t remember a lot of what we’re being told. We certainly remember how it made us feel.” (Person living with dementia).
Discussion
This study examined the nature of sleep disturbances experienced by people living with dementia and carepartners. It actively sought feedback and recommendations about how to optimise a sleep intervention specifically designed to support sleep-related difficulties encountered within dyads of people living with dementia and carepartners. Overall, findings from the study demonstrate that sleep disturbances are common and persistent in informal caregiving circumstances. This is in line with other research illustrating disproportionate prevalence and effects of sleep challenges in caregiving communities (Czeisler et al., 2021; Gibson et al., 2023). The themes in this study provide a richer context into the diverse factors affecting people living with dementia and their impact on both the individual as well as the carepartner. For instance, misperceptions of time of day, confused awakenings, and night-time safety concerns linked to the person living with dementia have profound impacts on sleep quantity and quality in carepartner, leading to night-time hypervigilance and daytime dysfunction. Stress and worry about the diagnosis, and uncertainty related to future may be associated with sleep disturbances in the dyad. These findings highlight the crucial need to develop and offer multimodal interventions that not only address, but also adapt to address the dynamic sleep and daytime needs of people living with dementia and their carepartners, particularly those that are dyadic.
Sleep disturbances, while prevalent, have profound implications on the overall functioning and wellbeing of carepartners and individuals living with dementia. Hence, participants in the study were receptive and strongly supportive of a multimodal sleep intervention. They emphasised the importance of addressing multiple facets of sleep and daytime functioning. This approach caters to differences in sleep issues between different dyads, and progressions of dementia. A combination of CBT-I, light therapy, exercise, and mindfulness as suggested in the proposed program has the potential to provide an acceptable, ecologically valid, time-friendly, accessible intervention for the community, as it packages evidence-based strategies. In addition, participants also recommended having a “toolkit” approach, where they can pick and choose strategies based on their needs. Indeed, toolkit style, non-pharmacological approaches have been found to improve mental health (Morton et al., 2017), and are more flexible and tailored relative to top-down, didactic intervention approaches (Barac et al., 2014). The inclusion of both current and former carepartners provided valuable guidance on ensuring the intervention was adaptable to all stages of dementia. Based on our findings and the overall feedback from community advisors, it was important that the proposed intervention took a personalised, adaptable approach aimed at sharing skills and knowledge during the group sessions so that participants could use the strategies based on their unique circumstances, both at present and into the future.
Both carepartners and people living with dementia offered specific suggestions to enhance adaptability, such as allowing flexibility in who attended the session (e.g., sessions optional for people living with dementia), targeting the intervention towards upskilling carepartners, and delivering the sessions virtually to groups. A group-based approach is pragmatic due to the therapeutic benefits of group settings and the proven efficacy of online interventions for older adults (Brewster et al., 2023; Xiang et al., 2024), including virtually-delivered CBT-I (Granberg et al., 2022). Moreover, group sessions, whether involving dyads or carepartners only, facilitate shared learning and mutual support among individuals with similar experiences (Orfanos et al., 2021). Group sessions, in particular, were recommended to ensure knowledge sharing and support, beyond one-to-one interactions with a sleep coach. Delivering and targeting the intervention toward carepartners addresses the pressing issue of sleep disturbance in this population, which often persists after changes in caregiving status (Gibson et al., 2023). Given the long-term health implications of caregiving (Corey et al., 2020), disseminating the intervention to carepartners and equipping them with strategies is a feasible way to support their own health, while also enhancing their ability to support the person within their care effectively. Additionally, optional attendance for people living with dementia ensures that they feel included and part of their treatment journey, while also having the flexibility to join the program as needed and if they are able, which might change session to session. Participants also highlighted the importance of open, informal communication and the use of clear language throughout the delivery of the intervention and included materials. This focus aimed to guide the therapist delivering the intervention to ensure they respect the individual experiences of people living with dementia and carepartners, while also fostering genuine engagement and building a sense of trust during the sessions.
The study is subject to the following limitations. First, the study included a sample of 10 community members. While the constructed themes reflect earlier qualitative studies exploring sleep-related challenges in people living with dementia and carepartners (Gibson et al., 2014). Second, dyads of people living with dementia and their carepartners were not specifically recruited for this study. This was done to encourage participants to openly discuss their perspectives, including the intensity of caregiving. Similarly, we wanted people living with dementia to independently share their personal experience. While this ensured that participants could reflect on their experiences without feeling constrained by the presence of their partner, we also acknowledge that this may have limited collaborative feedback and shared perspectives that could have emerged from their joint participation. Future research on coproduction of interventions should consider both dyadic and individual level participation to observe whether the themes change. Third, it is possible that only individuals with perceived sleep disturbances participated. This means that they may have greater awareness about sleep and more receptiveness towards participating in sleep interventions. Fourth, diagnoses or the severity of dementia for people living with dementia was not recorded as part of the study. Discussions about the type of dementia occurred when participants were initially recruited for the focus groups/interviews, but it was not formally recorded. This decision was made to safeguard the confidentiality and privacy of the participants recruited from the Dementia Australia community. Furthermore, identifying the type and severity of dementia was not essential for the current study, given that sleep issues are well documented across various types and severities of dementia (Hjetland et al., 2020). However, we acknowledge the importance of capturing this information for future clinical trials aimed at evaluating the applicability and efficacy of sleep interventions. While all study individuals provided consent and were able to participate in the study, it remains unclear whether the intervention design will require further modifications for individuals with more advanced dementia. The research team tried addressing this by inviting former carepartners to the study, who could reflect upon their experiences of caregiving and advise us on the utility of the program across different stages of dementia. Lastly, the study included participant(s) who identified as Indigenous Australians to reflect upon their experiences, however, the focus groups lacked sufficient cultural and linguistic diversity. This is important to address given the disparities in sleep health and prevalence of dementia among Indigenous Australians (Blunden et al., 2022; Lavrencic et al., 2022; Smith et al., 2008). As such, further research is required to examine the applicability and feasibility of such interventions in diverse communities.
Conclusion
The findings from this study offer valuable insights into the sleep difficulties experienced by people living with dementia and carepartners. The proposed sleep intervention was positively received by carepartners and people living with dementia. Adopting a toolkit approach, using a semi-group format for intervention delivery, and disseminating the strategies to carepartners was recommended to make the intervention more applicable across diverse diagnoses and disease severities. Overall, these findings suggest a need for evidence-based, structured multimodal sleep and lifestyle sleep interventions. It is anticipated that tailored interventions could lead to significant improvements in sleep and daytime functioning for both carepartners and people living with dementia.
Supplemental Material
Supplemental Material - Integrating lived experience to develop a tailored sleep intervention for people living with dementia and carepartners
Supplemental Material for Integrating lived experience to develop a tailored sleep intervention for people living with dementia and carepartners by Aimee Brown, Jasmine Dowling, Sumedha Verma, Rosemary Gibson, Tom Valenta, Ann Piestch, Marina G. Cavuoto, Susan M McCurry, Bei Bei, Michael Woodward, Melinda L. Jackson, Prerna Varma in Dementia
Footnotes
Acknowledgements
We would like to thank all of the participants who dedicated their time and insights to our study. We would also like to acknowledge our community advisors for their insightful guidance and continued support, and Kate Harding for her support with recruitment.
Author contributions
Project conceptualisation: PV, MJ, SV
Study design- PV, MJ, SV, BB, RG, AP, TV, MC, MW, SM, Data collection: SV, PV
Data analysis and interpretation: AB, JD, PV, MJ
Drafting of the manuscript: AB, JD, PV, MJ, Critical feedback and revision: AB, JD, SV, BB, RG, AP, TV, MC, MW, SM, MJ, PV
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Monash University’s School of Psychological Sciences and Dementia Collaborative Research Centres, Australia.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
Author biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
