Abstract
Public funders of health research have been widely criticized on the grounds that their allocations of funding for disease-specific research do not reflect the relative burdens imposed by different diseases. For example, the US National Institutes of Health spends a much greater fraction of its budget on HIV/AIDS research and a much smaller fraction on migraine research than their relative contribution to the US burden of disease would suggest. Implicit in this criticism is a normative claim: Insofar as the scientific opportunities are equal, each patient merits research into their condition proportional to the burden of disease for which that condition is responsible. This claim—the proportional view—is widely accepted but has never been fully specified or defended. In this paper, I explain what is required to specify the view, attempt to do so in the most charitable way, and then critically evaluate its normative underpinnings. I conclude that a severity-weighted proportional view is defensible. I close by drawing out five key lessons of my analysis for health research priority-setting.
Keywords
Introduction: The critique
Funding for health research is limited. Not every valuable research question can be investigated. Health research funders (henceforth “funders”) must therefore make decisions about how to allocate the resources they have. The choices they make affect which populations will benefit from improved health in the future. They are therefore choices that depend on value judgments.
On what basis should public funders allocate their limited resources among different patient groups with different disease profiles? One possible answer to that question can be found in a common criticism of how governmental and intergovernmental funders allocate money across diseases. For example, the US National Institutes of Health (NIH)—the world's largest funder of biomedical research—has been repeatedly criticized for the apparent mismatch between its allocation of funding among different conditions and the burden of disease attributable to those conditions. 1 This criticism has prompted multiple analyses of the relationship between NIH funding and measures of disease burden in the US—and occasionally global—population (Gillum et al., 2011; Gross et al., 1999; Sampat et al., 2013; Vargas et al., 2019). Likewise, public and non-profit funded health research in other countries and regions—including Australia, Canada, China, Latin America, Norway, and the United Kingdom—has been assessed against the benchmark of whether it is proportional to the local burden of disease (Kinge et al., 2014; Kmietowicz, 2002; Lamarre-Cliche et al., 2001; Luengo-Fernandez et al., 2012; Mitchell et al., 2009; Perel et al., 2008; Xu et al., 2014). Even the World Health Organization (WHO) has not escaped scrutiny. David Stuckler et al. (2008) compared WHO's budget allocation by disease area with burden of disease data for the world and did the same comparison for two WHO regions with starkly different distributions of disease (the Western Pacific and Africa). They found that 87% of WHO's 2006-7 research budget was allocated to infectious diseases, 12% to non-communicable diseases, and less than 1% to injuries and violence. This, they argue, shows a “misalignment of global health priorities and disease burden for health research worldwide” (Stuckler et al., 2008: 1563).
Very few of the authors who compare allocations of research resources to the burden of disease claim that the two should be exactly proportional. They accept, for example, that the state of science may be such that the opportunities for a breakthrough are much greater in one area of disease research than another, or that research into one—perhaps rare—disease may offer insights into the treatment or prevention of other—more common—diseases. 2 Nevertheless, such technical complications aside, there is implicit agreement on the underlying value judgment. Insofar as the scientific opportunities are equal, each patient merits research into their condition proportional to the burden of disease for which that condition is responsible.
Despite the apparent agreement on values, no one has yet provided a complete specification of this proportional view, nor offered a defense of the value judgment that underlies it. In this paper, I explain what is required to specify—that is, unambiguously state—the view, attempt to do so in the most charitable way, and then critically evaluate its normative underpinnings. My analysis reveals that making good on this common criticism of public research funders is much more complex than its exponents acknowledge. Nevertheless, given some normative and empirical assumptions, I conclude that a modified version of the view is defensible. According to the severity-weighted proportional view, diseases that are globally under-funded are those that receive a smaller fraction of total funding, conditional on scientific opportunity, than their severity-weighted contribution to the global burden of disease.
The paper proceeds as follows. In section 2, I consider which questions need to be answered in order to specify the proportional view sufficiently to assess whether research funding is consistent with it. In section 3, I describe the moderate prioritarianism that I will use to evaluate the proportional view. In section 4, I develop a simple model with which to assess the extent to which the proportional view is consistent with this moderate prioritarianism. Section 5 argues on the basis of this model that insofar as expected health gains are proportional to research funds invested into a disease, research investments should be proportional to disease prevalence but disproportionately skewed towards diseases that are worse for individual patients. Sections 6 and 7 explore what happens if we relax two of the key assumptions of the model in ways that make it more realistic. In section 8, I summarize the implications of my analysis for research priority-setting and suggest some directions for future research.
Specifying the proportional view
Proponents of the proportional view hold that when a funder is allocating money across a population, each individual in the population has a claim on research resources proportional to the burden of their disease. This normative claim seems at least prima facie plausible. However, as stated, the view is underspecified and so hard to evaluate. First, we need to know how its proponents propose to measure the burden of disease. Second, we need to know which population has standing—that is, whose disease burden counts. Third, we need to know how to count the money that has and will be allocated.
The metric of disease burden
How should we measure the burden of disease to which funding is supposed to be proportional? Various metrics are used in the analyses of funding allocations I mentioned above. These include disease incidence or prevalence, years of life lost, death rates, disability-adjusted life-years (DALYs), hospitalizations, and economic burdens. Which of these we use can make a substantial difference to the relative burden contributed by each disease. For example, osteoarthritis is a major problem for aging populations around the globe. However, most individuals who suffer from osteoarthritis will not die from it—the main impact of the disease is from pain and impaired function (Palazzo et al., 2016). Calculating disease burden in terms of death rates will clearly give osteoarthritis a different ranking than calculating it using a measure that captures morbidity. To give another example, in low-income countries, one predictor of households experiencing catastrophic health expenditures appears to be chronic illness (Brinda et al., 2014; Su et al., 2006). If economic burdens count as part of the burden of disease, that might support greater research investment into chronic illnesses.
In discussions of health care priority-setting it is widely agreed that premature mortality and morbidity caused by a disease should be taken into account. It remains an open question whether the burden of a disease should be conceptualized so as to include additional welfare effects, such as economic losses or the impacts on third-parties (like caregivers). 3 Insofar as including or excluding these effects would affect the relative burden of disease attributed to different conditions, a full specification of the proportional view must take a stand on whether they are part of the measure.
For the sake of argument, it will sometimes be necessary for me to adopt a metric of disease burden in the remainder of this paper. Except where the relevant data is not available, I use the DALY, which is perhaps the most widely used metric for assessing the burden of disease in a population. The DALY is a composite measure that incorporates both years of life lost from premature mortality and time spent at sub-optimal health as a result of disease. One DALY can be conceptualized as the equivalent of one healthy year of life lost. As noted, this choice of metric incorporates value judgments about what matters for the purposes of distributing scarce resources. The arguments that I make could equally be made with any other measure of disease burden, so using the DALY does not beg any questions here. However, which metric we choose will matter when it comes to implementing the view, since it will affect which diseases are considered to have higher burdens.
Population with standing 1: Temporal considerations
Two questions must be answered regarding standing—that is, regarding who counts as a member of the population whose health matters. The first concerns at what time point we should assess the burden of disease in a population. The funding analyses conducted of the distribution of actual research funding typically compare funding to contemporaneous patterns of disease. A small gap—usually two years—between burden and funding is often set, on the grounds that even if funding reflects burden we should expect a slight delay before changes in disease burden are reflected in funding. 4 The basic assumption of these analyses, though, is that we want to know whether funders are allocating resources proportional to disease burden at the time. 5
A natural alternative to asking about contemporaneous proportionality is to ask whether funding is proportional to the expected burden of disease at some future time. 6 For certain conditions, the time for which we estimate the burden of disease will make a substantial difference. For example, Alzheimer's disease and other dementias were responsible for an estimated 2 million deaths worldwide in 2016, which is 3.5% of all deaths. This is estimated to increase to 3.3 million (4.8%) in 2030, 5.4 million (6.3%) in 2045, and 7.3 million (7.1%) in 2060 (World Health Organization, 2018). If funding should be proportional to disease burden—here using deaths as a rough proxy for burden—then the share of funding that goes to dementia research will be substantially affected by whether we allocate based on the current or a future burden.
Moreover, there are good reasons to think that funders, insofar as they take disease burden into account, should consider some future disease burden, not the current disease burden. This is because there is usually a substantial time lag between research being conducted and that research having any effect on clinical practice or public health measures. For example, if a funder decides now that it will start to support the development of new antibiotics, there will be a substantial time lag between when it issues its first grants and when any resulting product is ready to go into clinical trials, a further lag before any successful new medication has marketing approval, and a further lag before the medication's use diffuses through health care systems so that clinicians start to prescribe it to patients who can benefit. On the plausible assumption that health research is valuable because of its beneficial effects on health, funders should be taking account of the disease burden at the time when those effects are realized. 7
Recognizing that the disease burden that matters is the disease burden that will be faced by the population at the time when health research has its effects introduces several technical complications. One is simply that predictions of the future burden of disease for a population are less reliable than estimates of the current burden of disease. The former invariably make use of the latter and must additionally extrapolate in the face of uncertainty about likely epidemiological and demographic trends, changes in health care systems, and the results of current research programs.
A second complication is that the time lag between research being funded and it having its first effect on health varies considerably. Some types of research, such as implementation science or comparative effectiveness research are likely to have a short timeline. For example, a study comparing the effectiveness of interventions that are currently used to eradicate methicillin resistant Staphylococcus aureus (MRSA) from hospitals during outbreaks will likely produce results that are immediately usable by other facilities. 8 The process of drug development, on the other hand, has a much longer timeline. It takes years to move from the identification of a promising new molecular entity through the various clinical trials that are required to obtain regulatory approval. 9 Even after regulatory approval, it takes time for a new treatment to be incorporated into regular medical practice. Time lags for basic science research to have any effect will typically be even longer and harder to predict.
Population with standing 2: Political boundaries
The second question about standing that must be answered in order to specify the proportional view concerns political boundaries. Is the (future) population whose health matters for the purposes of assessing the distribution of research funds the global population or some smaller group? The mission statements of some national funding bodies direct them to focus on the health of their country's population (Pierson and Millum, 2022). Others simply aim to improve human health without mention of political geography. Meanwhile, most studies that analyze the relationship between funding and disease burden do so for a particular funding body and a particular country or region.
Given that the largest public funders of health research are almost all high-income country (HIC) institutions, whether those institutions have a national or global purview will make a huge difference. 10 In HICs, 86% of the total DALY burden is associated with non-communicable diseases, 10% with injuries, and just 5% with communicable, maternal, neonatal, and nutritional conditions. 11 While globally the relative burden of injuries is the same, communicable, maternal, neonatal, and nutritional conditions comprise 26% of global DALYs. There are also multiple conditions that impose a substantial burden in LMICs, but almost none in HICs. These include tuberculosis, malaria, and the so-called “neglected tropical diseases.” For example, tuberculosis is responsible for just 0.08% of the DALYs in HICs. It is responsible for 3.4% of the DALYs in sub-Saharan Africa, where it is a major killer, and nearly 2% of global DALYs. Is tuberculosis research a high priority for a HIC funder on the proportional view? That clearly depends on whether the population with standing is limited by national borders.
Whether a government funding agency ought to take the disease burden of populations outside of the country into account presumably depends on whether the government itself has obligations to non-citizens outside its borders. Most political theorists believe that HIC governments do have some such obligations. They disagree on the nature and extent of those obligations. Those with more statist leanings think that obligations to people outside of the state are limited. 12 Those with more cosmopolitan leanings think that people have similar justice-based claims on state governments no matter where in the world they are. 13 Presumably, then, statists would think that the population whose burden of disease matters most is the population within the state whose research funding is being scrutinized. The health needs of others would get lower weight. 14 Cosmopolitans, on the other hand, would expect the government of an HIC to take the global disease burden into account when making its allocation decisions.
My own view is that some form of cosmopolitanism is correct. However, this paper is not the place to adjudicate the rich debate about the correct theory of global justice. Since disease burden varies greatly across countries, however, it is another question that needs to be answered if we are to know to which disease burden funding should be proportional. In section 7, I explain how comparisons between global disease burden and the global allocation of research funds can be of value for cosmopolitans, at least.
Past and future funding
It might seem that how to calculate how much money is spent on specific diseases poses only technical, not normative, challenges. Indeed, the technical challenges are substantial, since many research investments are expected to benefit multiple patient groups and others—basic research into cellular processes or population-wide biobanks—may appear disease-neutral. However, a further, normative question must also be answered in order to fully conceptualize the proportional view. Is the funding to which (future) patients have a proportional claim, the current year's funding, the total future funding, or the total past and future funding? The analyses that have been conducted of individual funders’ allocations usually consider the distribution of an annual budget in one or a few years. This makes sense looking forward, if we assume that without intervention the relative funding per disease will continue to be decided in a similar way. But it does not take account of historical misallocations of funding. For example, Faheem Farooq et al. looked at US federal and foundation funding for sickle cell disease research and cystic fibrosis research over the course of a decade (Farooq et al. 2020). Though comparable in their impact on patients’ quality and length of life, cystic fibrosis received about three times more federal funding per patient. On the proportional view, sickle cell disease looks to have been underfunded for some years. Similar points could be made about the global allocation of research funds: the “neglected tropical diseases” have been neglected for decades (Bosman and Mwinga, 2000).
What relevance, if any, does this prior misallocation of funding have for future allocations? At least two views have prima facie plausibility. First, we might consider the present to be year zero. The effects of prior years’ research funding are already captured in our estimates of (future) disease burden. If earlier research funds resulted in an effective treatment for a disease, then that disease will have a lower burden and so patients with the disease will have a smaller claim to current resources. If earlier disease-specific research funds did not benefit patients with that disease, then they are no better off as a result and so their claim to assistance has not been reduced. On the other hand, we might think that the relevant claim is a claim to resources. If a population's claims have been neglected in the past, then they should be compensated for that in the present.
This is an issue that merits further attention. Space precludes my adjudicating it here. For the time being, I assume that we can treat a funder's research budget as though we were at year zero—that is, without taking into account whether that funder misallocated its resources in the past.
Moderate prioritarianism for research priority-setting
In the previous section, I identified three questions that proponents of the proportional view must answer in order to specify the content of their view. Once these questions are answered we know what the view is. We can then evaluate it. In order to do so, we must next specify the ethical principles that are relevant to assessing different distributions of research resources among the members of the population with standing. In the academic literature on priority-setting, there is widespread agreement that allocators should take into account both the magnitude of benefits that they can produce and the distribution of benefits among potential beneficiaries (World Health Organization, 2014: 7–8). All else equal, a distribution of resources is better the greater the total benefits it produces. All else equal, a distribution of resources is better the more its benefits accrue to disadvantaged groups. In addition to agreement among academics, studies of public preferences reliably show that most people also care about these two considerations. While all health benefits are valued, respondents generally think that it is more important to provide a benefit of the same magnitude to someone who is badly off than someone who is better off (Shah, 2009). 15
We can conceptualize a view that takes these two considerations into account as either egalitarian (caring about inequality in addition to magnitude of benefits) or prioritarian (caring about disadvantage in addition to magnitude of benefits). For the purposes of this paper, it does not matter which we adopt. 16 In the following, I speak in prioritarian terms and adopt a moderate prioritarianism, according to which, “the strength of our reasons to benefit people depends both on the size of these possible benefits and on how well off these people are” (Parfit, 2012: 402). Everything I say could be restated in egalitarian terms instead.
One issue on which there is not agreement is the relative weight we should give to maximizing benefits versus giving higher priority to beneficiaries who are worse off. To what extent should we be willing to sacrifice efficiency in order to help the disadvantaged? In between the extremes—which I reject—of directing all our resources to the worst off and caring only about getting the most benefits possible are a wide range of possible positions. 17 Since there is no consensus on this matter, I do not adopt any particular weighting of the two considerations here. Fortunately, we can draw some interesting conclusions about the proportional view without doing so.
A model for disease-specific research investments
Assume that the DALY is an appropriate measure of disease burden and that we have specified the population that has standing. I now present a simple model for thinking about the health impact of disease-specific research investments. The goal of the model is to allow us to judge allocations of disease funding that correspond to the proportional view according to how well they maximize population health benefits and how far they prioritize more disadvantaged patient groups. The model is based on five empirical assumptions. These necessarily simplify a very complex reality for the purposes of ethical analysis. However, starting with this simple model will allow us to assess and ultimately revise the proportional view on the basis of its fit with moderate prioritarianism. While describing the assumptions below, I note how empirically accurate they must be in order for the model's verdicts on the proportional view to be true. In sections 6 and 7, I consider the implications for research priority-setting when the first and last assumptions are relaxed in ways that make them more realistic.
Assumption 1: Uniform gains from research
The first assumption is that the gains from research investments are uniform. For the model, this means that each dollar invested in research into a particular patient population leads to a unit of expected benefit of equal magnitude to each member of that patient population. A patient population for disease-specific research will typically comprise all the patients with that disease in the population with standing. So, for example, the assumption of uniform gains implies that a dollar invested into Type 2 diabetes research provides an expected benefit b to each patient with standing who has Type 2 diabetes, while a dollar invested into pancreatic cancer research provides expected benefit b to each patient with standing who has pancreatic cancer.
The assumption of uniform gains incorporates two distinct and important assumptions. One is that diseases offer equal scientific opportunity. Thus, the model assumes that research investment into a disease has not reached the point of diminishing marginal returns, that we are not at such an early stage of research that there are low-hanging fruit that offer cheap health gains, that we can ignore synergistic effects from existing research efforts, and so forth. Further, the assumption that each patient with the disease stands to benefit implies that all patients with standing have access to new health care interventions—that is, there is a seamless transition from research advances to improved clinical care for all. In section 6, I discuss the implications for priority-setting of adjusting the model to make both of these components of the uniform gains assumption more realistic.
Uniform gains was picked for its simplicity, not its realism. It makes modeling the effects of different disease-specific investments straightforward and therefore easy to compare for their fit with moderate prioritarianism. However, the lessons that I draw from the model regarding the proportional view could equally be drawn using a different function relating investment to patient benefits, provided the function ensures that: (1) average expected benefits increase as funding increases and the number of patients with the targeted disease increases; and (2) differences in scientific opportunity do not substantially covary with disease prevalence, severity, or prior funding. As far as I can tell, those two conditions are met in practice.
Assumption 2: Non-arbitrary disease classifications
The second assumption is that there is a non-arbitrary way to classify diseases for the purposes of applying the assumption of uniform gains. By non-arbitrary I mean that the classification reflects, to some extent, what is known about the etiology of the diseases, possible ways to treat or prevent them, or similarities and differences in symptoms. If, for example, Type 1 diabetes and Type 2 diabetes are classified as two distinct diseases then there should be similarities between patients with Type 1 diabetes that are not shared with patients who have Type 2 diabetes and vice versa. It then makes sense to distinguish money spent on Type 1 diabetes research from money spent on Type 2 diabetes research. We can model money spent on diabetes research simpliciter as though it is split between Type 1 and Type 2 diabetes. 18 This assumption prevents “gaming” of the model by simply redescribing diseases to encompass more patients and—given the assumption of uniform gains—thereby providing greater benefits per dollar.
This assumption is also a simplification and so, strictly speaking, false. The level of granularity at which we distinguish diseases is to some extent driven by our interests. 19
However, the simplification will not affect the model's verdicts regarding the proportional view provided that the classification to some extent maps onto real differences between diseases. The model would have the same implications provided it is true that for any two diseases, A and B, investing money into research on disease A is more likely to benefit patients with disease A than patients with disease B.
Assumption 3: Independence of diseases
The third assumption is that whether a patient has one disease is independent of whether they have another. On this assumption, the fact that someone is bitten by a poisonous snake, for example, does not make it more or less likely that they contract malaria or develop colon cancer. In fact, of course, many conditions are risk factors for other conditions. For example, heart disease, diabetes, and stroke are risk factors for Alzheimer's disease (Alzheimer's Association, 2022). The fact that disease risks are not actually independent presents a further challenge to those who are crunching the numbers in order to assess whether disease burden is proportional to funding. It makes the accounting considerably harder. In principle, we could create a model of disease-specific research investment that included patients with multiple conditions in order to take account of this. However, it would not affect the results of the comparison of the proportional view and the alternative principled ways to allocate funding I describe below; it would just make their calculation more complicated.
Assumption 4: No cure
The fourth assumption is that no patient population will reach full health before this funding cycle is complete. This means that no matter how much of the available funding is invested in a particular disease, none of it will be wasted—it will all generate the uniform returns described in Assumption 1. For most serious diseases, this assumption is realistic.
It is worth noting that this assumption is likely to hold even when research programs aim at curing a disease, rather than aiming at making incremental progress in its treatment. This is because when thinking about the benefits generated by research funding we are considering expected benefits. The expected benefits of an investment are given by summing the products of the probability of each outcome of the investment and the magnitude of the benefits of each outcome. In the vast majority of cases, the probability that a particular research program will generate a cure within a funding cycle is very low.
Assumption 5: Sole funder
The fifth assumption is that all the research funding for the population is distributed by a single public funder. This assumption allows us to ignore, for now, the question of how individual funders should coordinate with one another (or, equivalently, it means we are modeling what would happen if all the funders did coordinate). It also allows us to set aside the complications that arise when different types of funder have different obligations to different sub-populations. For example, we ignore, for the moment, the possibility that a charity whose mission is mental health research has a special obligation to fund mental health research. I return to this assumption in section 7.
Given these assumptions, we can model how different distributions of research funding would affect expected disease burden. To illustrate, consider a stylized population containing 90 patients with disease A and 10 patients with disease B. The sole funder makes its decisions about how to allocate research resources at time t0. The effects of the funding occur at time t1. The funder has $12 million to allocate between the two diseases. Following the assumption of uniform gains, each $1 million allocated to a disease reduces the disease burden per patient with that disease by one DALY. Following the assumption of no cure, the per patient DALY burden for each disease is greater than 12 (so that even if all the money is allocated to one disease it will not eliminate that disease). If the funder spends all $12 million on research on disease A at t0, then it will reduce the per patient DALY burden of disease A at t1 by 12 and thereby reduce population DALYs by 1080. If the funder spends all $12 million on research on disease B at t0, then it will reduce the per patient DALY burden of disease B at t1 by 12 and thereby reduce population DALYs by 120. And if the funder divides the funding equally between diseases A and B, then it will reduce the per patient DALY burden of each disease at t1 by 6, which will translate into a total reduction in population DALYs of 600.
Evaluating the proportional view
Using similar stylized populations, I now use the model to evaluate the proportional view against the standard of moderate prioritarianism. I consider three principled schemes for allocating the funding: allocating to each disease proportional to the disease's contribution to total DALYs in the population (the proportional view), allocating to reduce total population DALYs as much as possible (maximize benefits), and allocating to benefit the worst off as much as possible (equalize outcomes).
As before, there are 100 patients, two diseases, and $12 million of funding. In order to see the implications of different ways to allocate funding by disease, I vary two factors: the prevalence and the severity of each disease.
First, consider Scenario 1: Same prevalence, different severities. 50 patients have each disease. Disease A causes a shortfall of 40 DALYs in each patient with the disease. Disease B causes a shortfall of 20 DALYs in each patient with the disease. Table 1 shows the burden associated with each disease at t0 and then at t1 after funding has been distributed between the two diseases according to each of the principled schemes.
Scenario 1: Same prevalence, different severities.
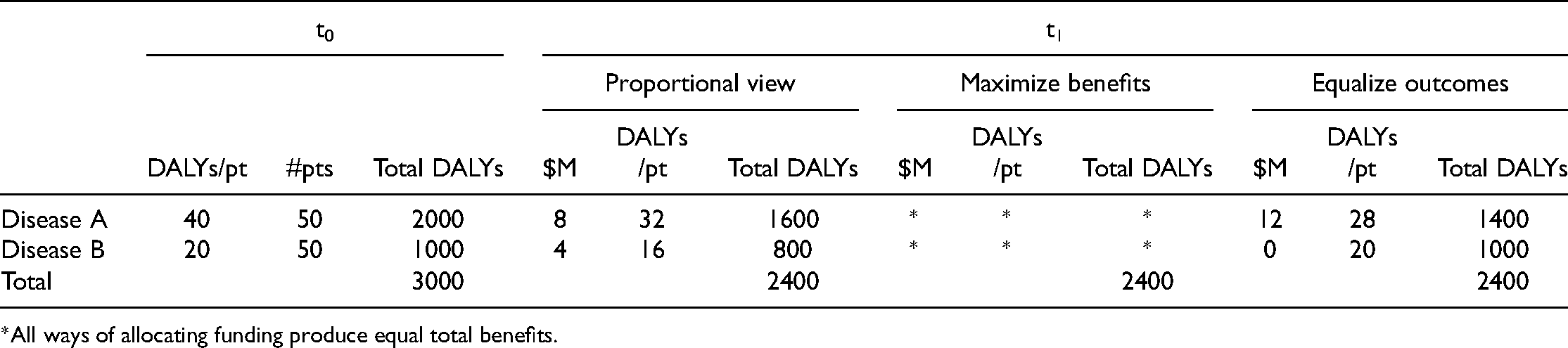
Scenario 1: Same prevalence, different severities.

*All ways of allocating funding produce equal total benefits.
When prevalence is the same, the proportional view tells us to allocate twice the funding to the disease that is twice as severe. This results in a reduction in burden by eight DALYs per patient for the more severe disease A and a reduction by four DALYs per patient for the less severe disease B. As a result of this allocation, at t1, the disease burden in the population associated with disease A is reduced from 2000 to 1600 and the disease burden associated with disease B from 1000 to 800. When prevalence is the same, all allocations will provide the same total DALY reduction and so the principle of maximizing benefits does not provide a verdict. If we aim to equalize patient outcomes—that is, to optimize the situation of the worst off—we can best do so by putting all our funding towards the more severe disease A, which will reduce its per patient burden to 28 DALYs and its population burden to 1400 DALYs.
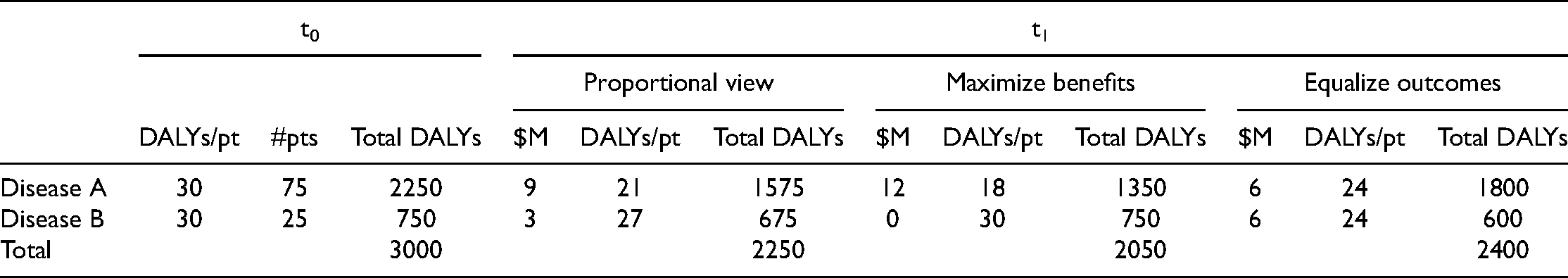
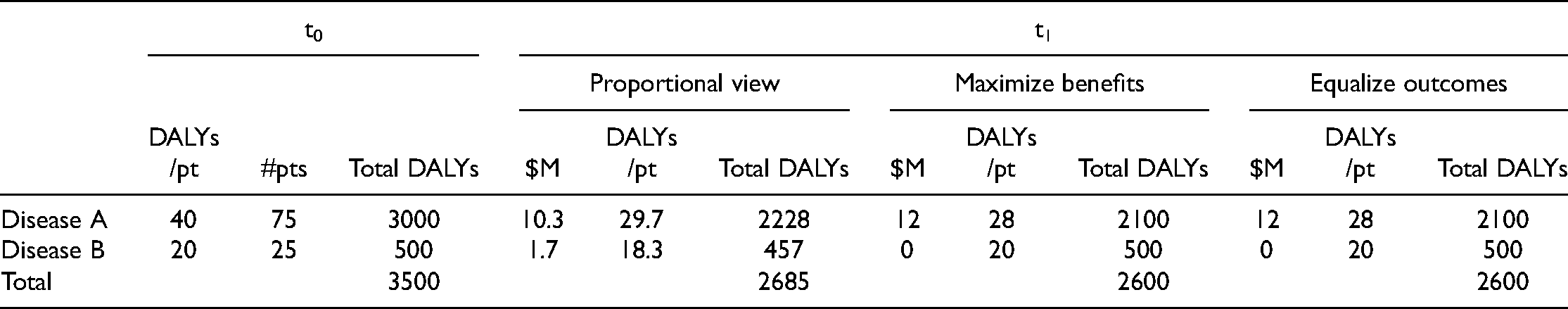
Tables 2–4 provide the same results for three other populations: Scenario 2: Different prevalence, same severity. 75 patients have disease A. 25 patients have disease B. Both diseases cause a shortfall of 30 DALYs in each patient with the disease. Scenario 3: Different prevalence, different severities, more badly off. 75 patients have disease A. 25 patients have disease B. Disease A causes a shortfall of 40 DALYs in each patient with the disease. Disease B causes a shortfall of 20 DALYs in each patient with the disease. Scenario 4: Different prevalence, different severities, more better off. 25 patients have disease A. 75 patients have disease B. Disease A causes a shortfall of 40 DALYs in each patient with the disease. Disease B causes a shortfall of 20 DALYs in each patient with the disease.
Scenario 2: Different prevalence, same severity.
Scenario 3: Different prevalence, different severities, more badly off.
Scenario 4: Different prevalence, different severities, more better off.
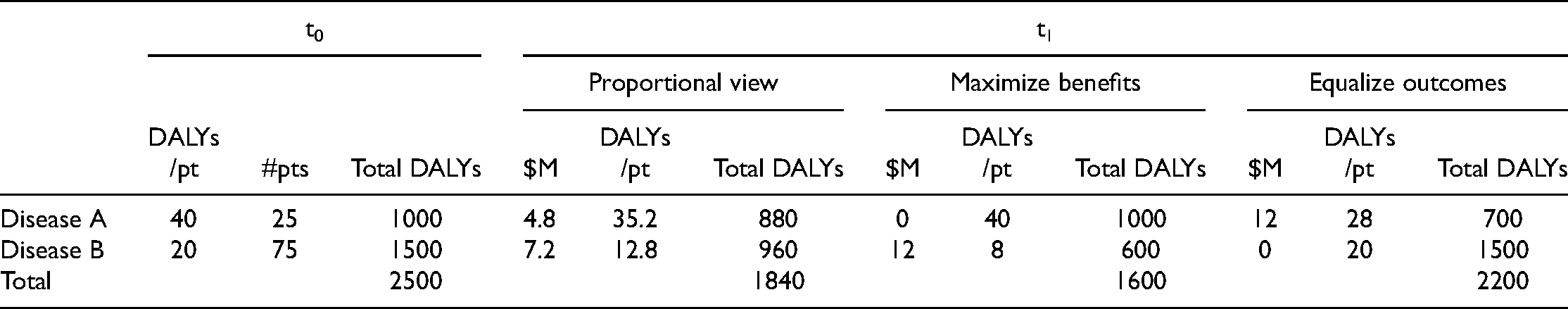
Modeling these four patient populations suggests some verdicts on the proportional view. For populations where the principles of giving priority to the worst off and maximizing total benefits pull in different directions—Scenarios 2 and 4—the proportional view strikes a balance between these two considerations. For example, in the case of Scenario 2, the proportional view leads to a difference in DALYs per patient at t1 (27 to 21) that is smaller than if we maximize benefits but greater than if we equalize outcomes. It leads to fewer total DALYs associated with the two diseases (2250) than if we equalize outcomes (2400), but more than if we maximize benefits (2050). Whether this balance between the two considerations is the right balance depends, as noted earlier, on the relative weight that should be given to each.
We can gain some insight into whether the proportional view is striking the right balance by seeing how it fares in the other two scenarios. For populations where there is no conflict between the allocation that maximizes the expected benefits of research and the allocation that most improves the outcomes for the worst off, the proportional view is clearly non-optimal. In Scenario 1, it produces the same total benefits but does not improve the situation of the worse off as much as equalizing outcomes. Worse, in Scenario 3, it sacrifices those who are worse off and produces less total population benefit. In both cases, this is because it allocates too much funding to the better off disease group.
The simple model suggests, then, that the proportional view misallocates resources. To bring the disease-specific allocation of research funding more into line with a moderate prioritarian principle, the view should be amended to give greater weight to investigating diseases that are worse for individual patients than proportionality alone would suggest.
To give a concrete example, according to the Institute for Health Metrics and Evaluation, the total DALY burden attributable to lung cancer in some HICs is similar to that attributable to low back pain (Institute for Health Metrics and Evaluation, 2021b). Both are the cause of very substantial disability and—in the case of lung cancer—many early deaths. But the prevalence of low back pain is several orders of magnitude greater than the prevalence of lung cancer. Low back pain is just not as bad for an individual patient as lung cancer (this is like Scenario 4). If the arguments I have given so far are correct, this suggests that, conditional on scientific opportunity, more funding should be allocated to lung cancer research than to low back pain research.
Note that we can restate the proportional view in a way that captures this prioritarian adjustment. On the priority-weighed proportional view, each person has a claim to health research resources proportional to their priority-weighted burden of disease. If patient A's disease is twice as bad as patient B's disease, then A will have a claim to research resources more than twice B's claim. How much more is a matter for further investigation. It will depend on how much we value improving the lot of the disadvantaged versus increasing total benefits (or, for the egalitarian, the relative weight given to equality versus total advantage).
I now turn to some adjustments to the model that make it more complicated but more realistic.
The simple model is much too simple to accurately model how research funding translates into health benefits. In particular, we should not expect a linear relationship between the two. As the Institute of Medicine report on NIH funding put it: “the relationship between such data and allocations of research funding will not be simple because health problems are not equally ripe for research advances.” (Institute of Medicine, 1998: 5). 20 One disease area may have been recently transformed by a technological advance, a new understanding of the disease's etiology, or a novel class of therapeutics, such that there are many promising avenues of research. Another might have few obvious research questions that can be answered with existing tools, few experts proposing novel projects, or the disease may persist because existing preventive or treatment options have not been implemented rather than because of a lack of scientific knowledge. 21 As a result of these differences, the same amount of research funding may have greater or lesser expected benefits.
Three key points are worth noting about this imperfect correlation between research investments and expected benefits. First, no matter how individuals’ claims to assistance relate to their disease burden, these differences in scientific opportunities will introduce noise such that the ethically optimal allocation of funding will not be a proportional match between strength of claim and funding. Studies that look at the relationship between burden of disease and disease-specific research funding therefore face the challenge of distinguishing a justified deviation from a problematic mismatch.
Second, though the assumption of uniform gains is surely mistaken, the reasons why it is likely to be mistaken do not map in any obvious way onto disease prevalence, severity, or prior funding. That is, we cannot generalize to say that research into more common diseases will usually be more or less fruitful than research into more rare diseases. Likewise for diseases that are more severe and for diseases that have been heavily researched in the past. Depending on the case, research into a more common disease may be more fecund because the disease is better understood or it is easier to find sufficient patients to study. On the other hand, sometimes a rare disease will have a clearer etiology (as, for example, where a rare disease is caused by a single recessive gene). A more severe disease may have more upside potential but it might be more intractable. Prior funding may mean that the low-hanging fruit for researchers have already been plucked or it may mean that there is an existing base of knowledge that puts researchers closer to breakthrough treatments. In sum, the basic lesson of the simple model still holds: Conditional on scientific opportunity, funding by disease area should be proportional to disease burden, but skewed towards more severe conditions. Judgments about scientific opportunity will have to be made on a case-by-case basis. 22
Third, when I applied the simple model to the stylized patient populations in Section 5, it suggested that the optimal allocation of funds for a population where a more severe disease is more prevalent is one that devotes all those funds to the more severe disease. This allocation maximizes total benefits and prioritizes the worst off. The fact that the assumption of uniform gains does not hold explains why, even in these circumstances, funders should spread research money across different disease areas, not focus solely on the diseases that are worst for patients. Research investments in one area sometimes have payoffs in other areas (Institute of Medicine, 1998: 22). Research into rare diseases sometimes leads to breakthroughs that generalize to more common diseases (Lauer et al., 2015: 1302). The unpredictable nature of scientific investigation means that it is sensible to hedge our bets. 23 Thus, funding should be skewed towards more severe conditions but should not exclusively focus on the worst.
There is another reason why the assumption of uniform gains will not hold that is unrelated to scientific opportunity. As I noted above, the assumption implies that any knowledge about a condition that is generated by the research will equally benefit each person with the condition. But, in reality, people do not have equal access to the fruits of scientific knowledge. For example, a great deal of medical research is aimed at developing patented pharmaceuticals. These are typically priced in order to maximize profit, not maximize patient access, and so many patients—especially in LMICs—will predictably not get them or will not get them for many years after they are available to more wealthy patient groups. Other research products rely on an existing medical infrastructure that is not universally available. For example, research that results in improvements in equipment or techniques for intensive care will be of little benefit in countries with few intensive care units. 24
At first glance, one might think that this point supports giving higher priority to research into conditions that affect people who have (or will have) access to good quality health care. If poorer patient populations are not going to be able to afford the new drugs anyway, then the benefits of developing drugs for their conditions will be limited. Better then to focus on conditions that cause a high burden in HICs. But the implications of the global variability in access are more complicated than that. First, since choices can be made about not just what diseases to study but what sorts of interventions to study for those diseases, it militates in favor of research into types of interventions that more people are expected to be able to access. For example, this is a reason to support a research program that aims to develop a heat-stable drug over one that assumes its product will be kept under constant refrigeration. Second, according to the moderate prioritarianism that I have assumed, both the amount of benefit and the disadvantage of the beneficiaries are relevant to who should receive resources. Populations with worse access to health care are typically worse off and so will merit priority on those grounds.
This brief discussion has only scratched the surface of the complex and important issue of how research priority setting should relate to health care access. To address it fully would require at least another paper. For now, it is enough to note that disparities in access to health care do not support giving lower priority to research on diseases that affect the global poor.
Relaxing the sole funder assumption
It is not true that each population is served by one funder. Not only are there multiple entities supporting research within most countries, health research that happens outside a country will frequently be relevant to health care within it. To complicate matters further, national governments are not the only actors who support health research. Globally, as of 2009, government funders accounted for over US$64 billion of health research spending each year (Røttingen et al., 2013). 25 About US$1 billion more was spent by multilateral agencies, such as the World Health Organization and the World Bank. But more than US$130 billion was spent by for-profit companies and over US$20 billion by not-for-profit organizations, like the Wellcome Trust, the Bill and Melinda Gates Foundation, and Cancer Research UK (Pierson and Millum, 2018). What is the relevance of this fact to evaluating any individual funder's allocation of research funding?
First, it tells us that critiques of national funding bodies which point to a mismatch between their allocations of funding and national burden of disease are making a very consequential normative assumption. They assume that the proportional view holds for government funding bodies independent of what other research is being conducted. To see why this assumption is so significant, consider cancer research funding in the UK. In 2018, UK government funders spent approximately £130 million on cancer research (UK Clinical Research Collaboration, 2020: 133). This represents 13.2% of the £984 million in UK government health research funding that can be attributed to specific disease areas. 26 But 19.7% of 2016 UK DALYs were attributed to cancer and neoplasms (UK Clinical Research Collaboration, 2020: 127). If we should evaluate government funding bodies independent of the actions of other funders, cancer looks underfunded relative to its disease burden in the UK. Matters change if we expand the scope of our assessment. The UK charity and not-for-profit sector spent £353 million on cancer research in 2018. Total government and non-profit spending on cancer research was therefore £483 million, or approximately 26% of the total attributable funding. 27 Given that cancer research is a high priority for industry, it is likely that including for-profit funding would raise the fraction of total research spending devoted to cancer even higher. The judgment of whether the UK government spends too little or too much on cancer research is clearly affected by whether it should take other sources of funding into account, even if we only consider what happens within the borders of the country. 28
There is not space in this paper to adjudicate the issue of how far funders should take account of the actions of other funders. I note here simply that it seems implausible that a patient population's claim on one funder's limited resources should be wholly independent of how far other funders are responding to the needs that underlie that claim. If this is correct, then the analyses of funding that compare a single funder's allocation of resources to a population's burden of disease may tell us very little, by themselves, about whether any funder is misallocating their resources.
Second, there are good reasons to think that funders differ in their obligations. According to Leah Pierson and Joseph Millum's analysis of the duties of individual funding organizations, all funders have some obligation to conduct socially valuable research, but they differ in how this obligation is constrained by other duties and in terms of the populations to whom they have special obligations (Pierson and Millum, 2018). For example, non-profit organizations have obligations to conduct socially valuable research consistent with their mission statements and commitments to patient groups. For-profit organizations have obligations to conduct socially valuable research consistent with reasonable returns on investment, as well as to avoid taking advantage of market failures. A complete picture of who should be funding what would need to take account of these different obligations, as well as a population's disease burden.
The complications introduced by acknowledging the existence of multiple funders might make one think that there is no value in comparing disease funding to burden of disease. But such pessimism is unwarranted. We just need to pick the comparators carefully and be careful about how we interpret their meaning. In particular, I now argue, there is value in comparing the global distribution of research funding to the global burden of disease, especially for those—like myself—with cosmopolitan views about global justice.
For a cosmopolitan, it is presumptively unjust if individuals are disadvantaged as a result of the region of the world in which they are born. It would therefore be presumptively unjust if disease burden were treated as more or less important depending on which side of a political border the patients lay. A government body should give priority to individuals within its jurisdiction only if the individuals within other jurisdictions are equally well served by their governments. In the current, highly unjust, global order, this suggests that governmental and intergovernmental bodies should not give normative weight to political borders when they are deciding how to allocate research funds. Following Pierson and Millum, all else being equal, they should therefore aim to maximize the global social value of the research they support, where social value is a function of both the magnitude of the benefits produced and the disadvantage of the beneficiaries. 29 Further, if we accept Pierson and Millum's arguments, the rules that govern markets are also amenable to assessment in terms of social justice. The regulations that govern for-profit research funders will be fair insofar as they tend to lead to the efficient production of socially valuable outcomes. And non-profits also have obligations to aim for socially just outcomes because their existence is subsidized by states—for example, through taxation policies—who are themselves constrained by considerations of justice.
All this suggests that the ideal overall distribution of research funding when all funders’ contributions are aggregated will be the same as the ideal overall distribution would be if there were just one neutral funder. There is therefore value in comparing the global distribution of research funding to the global burden of disease. Doing so can tell us whether specific diseases are over- or under-funded relative to their burden. It may not always tell us who has the primary responsibility to remedy the maldistribution, but it is one step towards that goal.
Implications
According to the proportional view, health research funders should allocate their resources so that, conditional on scientific opportunity, disease-specific research is funded proportional to the burden of disease caused by each health condition. Though it is not always explicitly stated, the proportional view underlies many critiques of how research funding is actually allocated. In articulating and critically assessing the proportional view, this paper has drawn a number of conclusions with relevance to research priority-setting.
First, any assessment of whether a disease is under- or over-funded relative to others must specify the metric of disease burden, the population that has standing, and the time period over which funding is measured. Specifying each entails value judgments.
In this paper I assumed that the DALY—which captures both morbidity and mortality—is an appropriate measure of disease burden. However, in the literature on health care priority setting, strong arguments have been put forward to support the use of measures that incorporate more than just the direct effects of ill-health, including effects on third parties like caregivers and economic impacts on households. 30 If these arguments are sound then the measures of disease burden used for research priority-setting should presumably be similarly widened.
The question of which population has standing has a geographic and a temporal aspect. Regarding geography, how the needs of different groups are considered will depend on one's views about global justice. In parts of this paper I have assumed, but not argued for, a cosmopolitan view according to which everyone's needs count equally no matter where in the world they are. 31 It is worth re-emphasizing, though, that even those political theorists who defend statist views of global justice do not deny that HIC governments have some obligations to benefit the people of LMICs. 32 Regarding the temporal aspect, I argued that the burden of disease that is relevant to research priority-setting is the burden of disease at and beyond the time at which the research is expected to have an effect. We should not be comparing current funding to the current burden of disease in a population.
The amount of funding allocated to a disease can be measured at a point in time (e.g., current annual budget) or over a period. The key normative question in this regard is whether and how to take account of diseases or patient populations that have historically been over- or under-funded.
Second, assessed against the benchmark of moderate prioritarianism, the proportional view is mistaken. The correct way to take burden of disease into account would be to give greater weight to diseases that are worse for individual patients. This supports a severity-weighted proportional view for disease-specific research investments. Since it is an open question how much weight should be given to degree of disadvantage versus magnitude of benefit, it is also an open question exactly how much greater weight should be given to more severe diseases.
Third, even if research funding were correctly aligned with the relative needs of patients, we would not expect a perfect match between the amount of funding and the (severity-weighted) burden of each disease. This is because funding decisions should take account of the scientific opportunities that are available to make progress on a disease. The challenge for studies that look at the relationship between burden of disease and disease-specific research funding is to distinguish a justified deviation from a problematic mismatch.
Fourth, the analyses of research funding that have prompted criticisms based on the proportional view have mostly examined just one funder. Comparing the allocation of research funds by one funder to a population's burden of disease without considering what other funders are doing makes sense only if each funder's obligations are independent of one another. This seems implausible and so critiques of individual funders may not be very informative on their own, particularly for smaller funders. However, the question of whether and how funders should take each other's actions into account is a matter for further investigation.
Finally, despite all these complications and critical remarks, there remains value in making certain comparisons between funding allocations and disease burden. In particular, given some normative and empirical assumptions, we can still say what it means for a disease to be globally under-funded. Diseases that are globally under-funded are those that receive a smaller fraction of total funding, conditional on scientific opportunity, than their severity-weighted contribution to the global burden of disease. Efforts to map global research funding are therefore valuable for funders who want to improve their disease-specific allocations of resources. 33
Footnotes
Acknowledgements
For valuable comments, my thanks to Peter Kilmarx, Leah Pierson, David Wasserman, two anonymous reviewers for the journal, and audiences at the Clinical Center Department of Bioethics and the Fogarty International Center at the National Institutes of Health, the New Scholarship in Bioethics Annual Symposium 2020, the Wharton School at University of Pennsylvania, and the World Congress of Bioethics 2018.
Declaration of conflicting interests
The author declares no potential conflicts of interest with respect to the research, authorship, and publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
