Abstract
Background
There is evidence of the beneficial effects of swimming training (ST) after myocardial infarction (MI) on heart function. However, the actions of ST preconditioning (STP) in MI on vascular function require further research.
Objective
This work focused on evaluating whether STP has beneficial nitric oxide-mediated effects on cardiovascular function in chronic MI.
Methods
Four months female Wistar rats were randomized as follows: (1) sham; (2) six weeks left anterior descending coronary artery ligation (LADL); (3) LADL + valsartan; (4) ST + LADL. Eight weeks of STP were performed in the ST + LADL group, then LADL was induced on the estrus stage and evolved for six weeks. The LADL + valsartan group was treated a day after surgery, for six weeks. Subsequently, vascular reactivity to Ang II and phenylephrine in aortic rings, quantification of nitric oxide (NO) on serum, heart, and aorta, and echocardiographic parameters were measured.
Results
ST + LADL reduced both vasoconstrictors reactivity beyond sham values. Ejection fraction (EF) and shortening fraction (SF) decreased in the LADL group vs sham, while ST + LADL and LADL + valsartan remained unchanged. NO levels decreased in LADL + valsartan and ST + LADL groups compared to sham and LADL groups.
Conclusion
STP in chronic MI improves cardiac function and reduces vascular reactivity through a non-related NO mechanism.
Introduction
Cardiovascular disease (CVD) remains the leading cause of death globally and has increased, particularly in post-menopausal women. 1 Since 2014 the incidence of myocardial infarction (MI) in women has been higher than in men, which has increased the interest in understanding the importance of sex on cardiovascular function. 2 Several studies have shown that CVD in women has different risk factors, pathophysiological evolution, and treatment response.3,4
Sedentary behavior (SB) is recognized as a major risk factor for cardiovascular health in women. The Objective Physical Activity and Cardiovascular Health (OPACH) study shows that girls and women tend to be more sedentary than men. 5 According to the Lancet Women and Cardiovascular Disease Commission, boys are more likely to be encouraged to be physically active at an early age, and this encouragement continues into adulthood, contrary to women, where declines with age.5–7 Thus, SB alters hemodynamic, inflammatory, and metabolic processes, leading to impaired arterial health, which directly and indirectly promotes the development of CVD. 8 Therefore, regular physical activity has been proposed as a highly effective therapeutic strategy for the prevention and treatment of CVD.9,10 For those reasons, over the years, exercise has become increasingly important in patients with acute myocardial infarction (AMI). 11
Exercise preconditioning (EP) induces cardioprotective effects, including anti-atherosclerotic and anti-thrombotic benefits, and triggers protective cellular responses that may prevent acute coronary events. 12 The beneficial effects of EP have been studied in male models, finding that high-intensity interval training for 4 weeks significantly improves mitochondrial biogenesis and angiogenesis in cardiac tissue, reducing cardiac injury and necrosis after MI. 13 In addition, EP can reduce the damage to vascular function, and ventricular remodeling and improve the quality of life of patients as well as post-infarction mortality. 14 These effects are important to preserve cardiac function during AMI (first 72 h). 15 However, in chronic myocardial infarction (CMI), the preservation of vascular reactivity as a compensatory mechanism for cardiac viability is more relevant. 16
During chronic adaptation in the myocardium, there is a humoral activation of the renin-angiotensin and the sympathetic nervous systems. The release of angiotensin II, norepinephrine, and endothelin-1 directly contributes to the hypertrophy and reduced distensibility of myocardial and vascular smooth muscle fibers, which increases peripheral resistance and thus pressure overload in the heart. 17 The treatment of myocardial infarction and heart failure includes angiotensin II receptor (AT1R) antagonists, these drugs prevent the increase in vascular reactivity, ventricular remodeling, and cardiac hypertrophy, improve ventricular function in the heart, and stimulate growth in vessels.18,19
Although pharmacological treatment helps to mitigate the long-term adverse effects of MI, physical activity has been proposed as a preventive and complementary therapy. In this sense, the combination of angiotensin receptor blockers and moderate exercise has shown good results in improving arterial compliance and cardiac function. 20 Some have demonstrated that short-term swim training (ST) can improve mitochondrial function by reducing reactive oxygen species (ROS) production and increasing antioxidant superoxide dismutase 2 (SOD2) activity in aged male mice. 21 Dayan et al., also reported that four weeks of swim training preconditioning (STP) for MI improved cardiac function in male rats. 22 Some others have shown that exercise training (ET) reduces remodeling, and hypertropia, and prevents MI. 23 Regarding the effects of ET on vascular function, de Moraes et al. showed in males that running training protects vessels by increasing NO and SOD2 activity in mesenteric aortic rings. 24 ET has also been shown to normalize vascular reactivity in aortic rings from males fed a high-fat diet by decreasing iNOS expression.25,26 However, no one has demonstrated the effects of STP on vascular reactivity on CMI in female rats. Therefore, this work aimed to evaluate whether STP has beneficial nitric oxide-mediated effects on cardiovascular function 6 weeks after MI in female Wistar rats.
Methods
Animals
Female Wistar rats (4 to 5 months old; 320 g average body weight) were obtained from the animal facility of the Cuautitlan Superior Studies Faculty. All animal procedures were performed in accordance with the Federal Regulation for Animal Experimentation and Care (SAGARPA, NOM-062-ZOO, 1999, Mexico), and the National Institutes of Health Guide for the Care and Use of Laboratory Animals (NIH Publications No. 8023, revised 1978, USA). This protocol was evaluated and approved by the Internal Committee for the Care and Use of Laboratory Animals of the Facultad de Estudios Superiores Cuautitlan UNAM (CICUAE-FESC) protocol number C21_04.
Experimental design
Animals were randomized to the following groups (n = 6): (1) sham; (2) six-weekleft anterior descending coronary artery ligation (LADL); (3) LADL + valsartan (1.14 mg/Kg/day, intramuscularly); (4) swim training + LADL (ST + LADL). Animals were maintained on a 12 × 12 light/dark cycle and had free access to food and water. Laboratory rodent diet 5001 was given to all experimental rats (LabDiet CA, USA). Before MI induction, the ST-LADL group was started on ST, for 30 min, five days a week for eight weeks. After this period, the ST preconditioning was stopped, and all groups except the sham group underwent LADL surgery (Figure 1). LADL was performed in the estrus phase of the sexual cycle of the female rats, because previous evidence from our group showed that the slight depletion of estrogens promotes larger infarct areas, 27 and to select rats in the estrous phase, a vaginal smear was performed. Treatment with valsartan (1.14 mg/Kg/day i.m. Sigma, St, Louis, MO, USA) was started 1 day after coronary occlusion. Previously, valsartan dose has been studied for our group to reduce vascular reactivity in male and female rats after myocardial infarction and i.m. route of administration is used on large terms to avoid drug metabolism. 28 Recovery was allowed, and after six weeks cardiac function was assessed by echocardiography, concentration-response curves to angiotensin II (Ang II) and phenylephrine were performed, and finally, serum and tissues (aorta and heart) were obtained to determine nitric oxide (NO), infarct area, and heart weight index.
Swimming training (ST)
As previously described by other authors, swimming training without any additional load on the rats is considered a low-intensity aerobic resistance exercise.29,30 During the first week of training, rats were conditioned by swimming for 10 min on the first day, increasing by five min each day until 30 min was reached in a rectangular tank (60 × 50 × 60 cm) with water leveled out at 27°C. After swimming, the animals were dried and kept in a warm place and then returned to their cages. After this period, the training consisted of having the rats swim for 30 min, five days a week for eight weeks. 31 The point of fatigue was determined when the animal could not continue to rise to the surface of the water to breathe within a period of 10 s, and when the rat could not return to a normal standing (prone) position when removed from the water and placed in the supine position. 32
Vaginal smear
A vaginal swab was taken to identify females in estrus. A cannula filled with saline solution was gently and quickly inserted into the vaginal opening to a shallow depth of approximately 0.5 cm to avoid cervical stimulation. A small amount of vaginal fluid was then aspirated. The rats were not anesthetized during this procedure. The collected epithelial cells were placed on a microscope slide, dried at 37°C, and fixed in a 1:1 ethanol-ether solution for one minute before staining with gentian violet.33,34
LAD ligation
Rats were anesthetized by intraperitoneal administration of 40 mg/kg ketamine + 5 mg/kg xylazine. During anesthesia, skin color, respiratory pattern, and consciousness were closely monitored. The rats were intubated with 14-gauge IV catheters equipped with wire stylets, and a small laryngoscope was used for direct laryngoscopy. After the catheter was properly placed in the trachea, the stylet was removed. Chest expansion during oxygen delivery confirmed the correct placement. The catheter was then connected to a three-way stopcock, with one port connnected to a 0.5 L/min oxygen tank. To maintain pulmonary pressure and prevent collapse during thoracotomy, the third port was adjusted 70–80 times per minute to achieve a tidal volume of 1.5–3 mL. 35 The left anterior coronary artery was ligated with an atraumatic needle and 5/0 silk suture, and the heart was returned to the thoracic cavity. Analgesics and topical erythromycin were administered to all animals undergoing surgery including the sham group, where, the animals were underwent to the same procedure except for the LADL. After LADL, animals were observed for two days (recovery period) and then allowed to perform LADL for six weeks.
Echocardiography
Six weeks after LADL, echocardiography was performed once for each rat for 10 min under anesthesia with ketamine (90 mg/Kg, i.p.) on the M-mode echocardiograph (Sonoscape X5V; Guangdong, China). The anterior chest wall was shaved with cream, and the rats were placed in a lateral decubitus position. Body temperature was maintained at 37°C on a pad during the study. Intraventricular septal thickness (IVSd), left ventricular internal dimension at the end of diastole (LVIDd), left ventricular posterior wall thickness at the end of diastole (LVPWd), interventricular septal thickness at the end of systole (IVSs), left ventricular internal dimension at the end of the systole (LVIDs), left ventricular posterior wall thickening fraction (LVPWs), LV ejection fraction (EF), stroke volume (SV), shortening fraction (FS) and total left ventricular mass (LVM) were measured. 36
Determination of cardiac hypertrophy
Heart weight index (ratio of heart weight to body weight) was an important parameter to evaluate hypertrophy. This parameter was determined by the following formula: WIx = Wx / Wc, where WIx is the wet weight index of the specific chamber, Wx is the wet weight of the heart, and Wc is the body weight of the animal.27,37
Vascular reactivity
The thoracic aorta was removed and placed in Krebs-Henseleit solution containing 118 mM NaCl, 4.7 mM KCl, 1.2 mM KH2PO4, 1.2 mM MgSO4–7H2O, 2.5 mM CaCl2–2H2O, 25 mM NaHCO3, 11.7 mM dextrose, and 0.026 mM calcium disodium ethylenediaminetetraacetic acid (EDTA) bubbled with 95% oxygen (O2)/5% carbon dioxide (CO2). The aorta was cleaned of connective tissue and cut into 4 to 5 mm long rings, then mounted in 10 mL isolated tissue baths filled with Krebs-Henseleit solution at 37°C (pH 7.4) and were continuously aerated with 95% O2/5% CO2. To record the development of semi-isometric force, a transducer (BIOPAC TSD105; BIO- PAC systems Inc, Santa Barbara, California) was connected to a BIOPAC MP100WSW system (from BIOPAC Systems Inc) and Acq Knowledge 8.1 (software) was used. The aortic rings were adjusted to a tension of 3 g. Concentration–response curves of Ang II or phenylephrine (10−10 to 10−6 M) were performed. 38
Infarcted size
Animals were euthanized by cervical dislocation. Hearts were frozen for an hour at −20°C and then cut into 1 mm slices, slices were incubated in nitro blue tetrazolium (NBT) 0.1% pH = 7.4 at 37°C for 10 min (Sigma Aldrich St, Louis, MO, USA) to differentiate the non-infarcted area (red tissue) from the infarcted area (white tissue). ImageJ
NO quantification
NO was quantified with Griess reaction, an indirect method based on measurement of nitrite (NO2−) concentration, since NO2−/ nitrate (NO3−) are the stable products of NO metabolism. To reduce NO3 to NO2 in the samples VaCl3 (0.8% diluted in hydrochloric acid (HCl) 10%) was used. Proteins were precipitated using ethanol (EtOH)/H2O (7:1); we used 200 ul of the solution for each 100 mg of tissue, then we homogenate, followed by centrifugation at 14,500 rpm per 20 min. We add in a microtiter plate 100 ml of supernatant sample, 100 ul of VaCl3, 50 ul of Sulfanilamide (2% diluted in HCl 10%) and 50 uL of N-(1-Naphthyl)ethylendiamine (0.2% diluted in HCl 10%). After 20 min incubation at room temperature, absorbance was read at 450 nm. 39
Statistical analysis
All data are expressed as means ± (standard error of the mean) SEM. Data were analyzed with Sigma Plot 12.0 using a one-way analysis of variance and the Student-Newman-Keuls post-hoc analysis for all experiments except the vascular reactivity test. Two-way ANOVA and Sidak's post hoc were used to assess differences between the groups in the concentration-response curves experiments. *P < 0.05 indicated statistical significance versus SHAM; #P < 0.05 indicated statistical significance versus LADL.
Sample size calculation
Several factors were considered to calculate the sample size: 1) confidence level (95%, corresponding to an alpha of 0.05, the critical value of Z for α = 0.05 is 1.96), 2) statistical power (86%), 3) effect size (0.8) and variability. The calculation was done in the G-Power program. 40 The family of tests used was F tests, with the ANOVA test: Fixed Effects, Omnibus, One-Way. Type of power analysis: “A priori: Calculate required sample size - given α, power and effect size”. We get N = 28 with a statistical power of 92%. After this, looking forward to reducing the number of animals, we test if N = 24 could be enough, finding in the same program that the statistical power is 86%, which is within the reliable range for this type of study (<80%). 41
Results
Heart weight index and infarcted size
First, we analyzed the heart weight index (heart weight/body weight ratio) to identify hypertrophy between groups. Still, there were no differences, although the ST group tended to be greater (Figure 2(a)). Also, there were no differences between infarcted size, although the group treated with valsartan and the group with training showed a trend of a smaller infarction size (27% and 28% respectively) (Figure 2(b)).
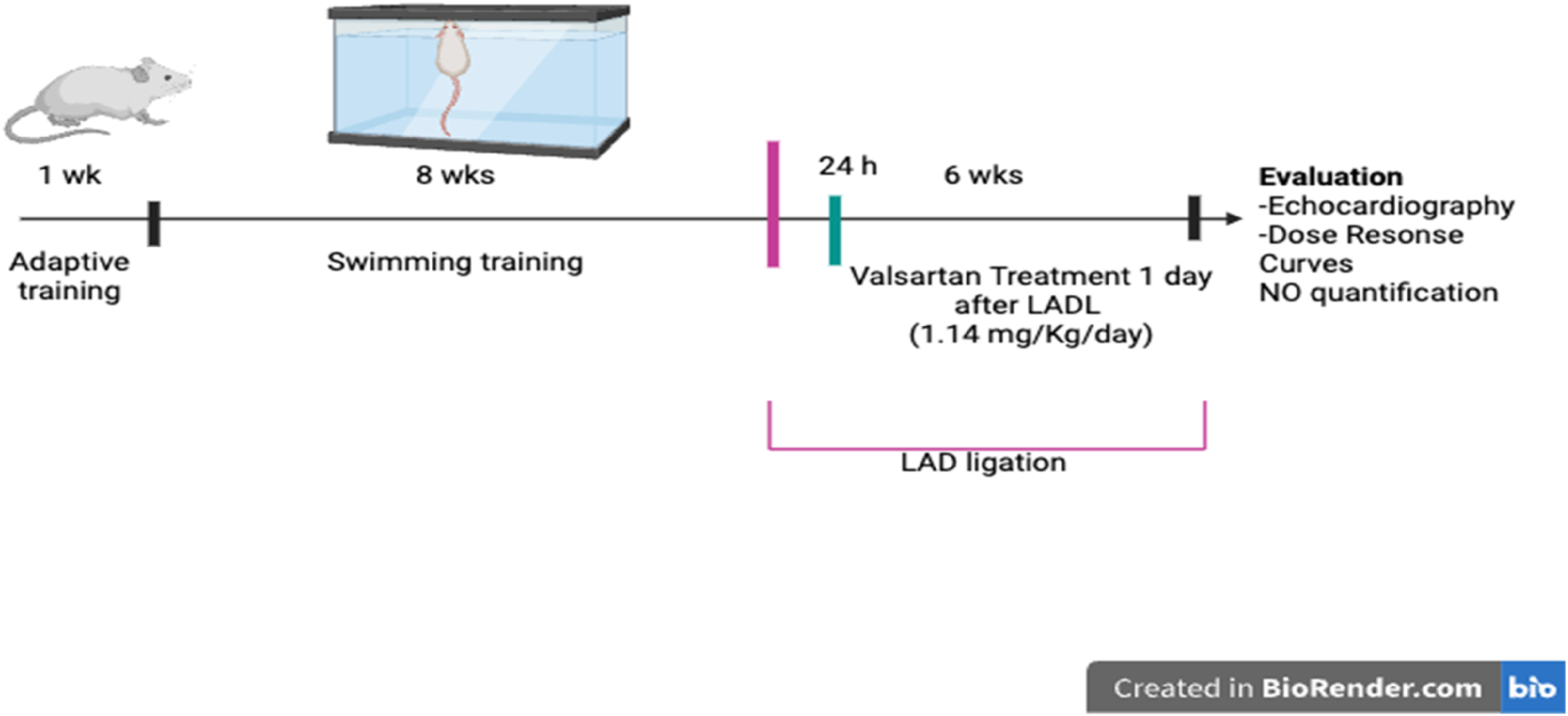
Experimental design.

Heart weight index and % infarct size. (a) Heart weight index, no significant changes were found. (b) Infarct size, no significant changes were observed. Results are shown as the mean ± SEM (n = 6). One-Way Analysis of Variance (ANOVA).
Echocardiographic parameters were necessary to evaluate cardiac function. Accordingly, Table 1 shows an increase in the IVSd, LVPWd, IVSs, LVM, LVIDs, and IVS/LVPW ratio in the LADL group vs the sham group, which indicates that the left ventricle has lost its contractile capacity and increased wall thickness. We also found a decrease in the ejection fraction (EF) and the SF in the LADL group vs the sham group but not in the trained and treated groups (Table 1). In the ST + LADL group, we found an increase in LVPWd, IVSs, LVPWs, and LVM, while end-diastolic volume (EDV), end-systolic volume (ESV), and stroke volume (SV) were decreased vs the sham group. Regarding valsartan treatment, we only found a decrease in ESV and SV in comparison to the sham group. In other measures of cardiac structures, we did not find differences between groups.
Echocardiographic parameters.
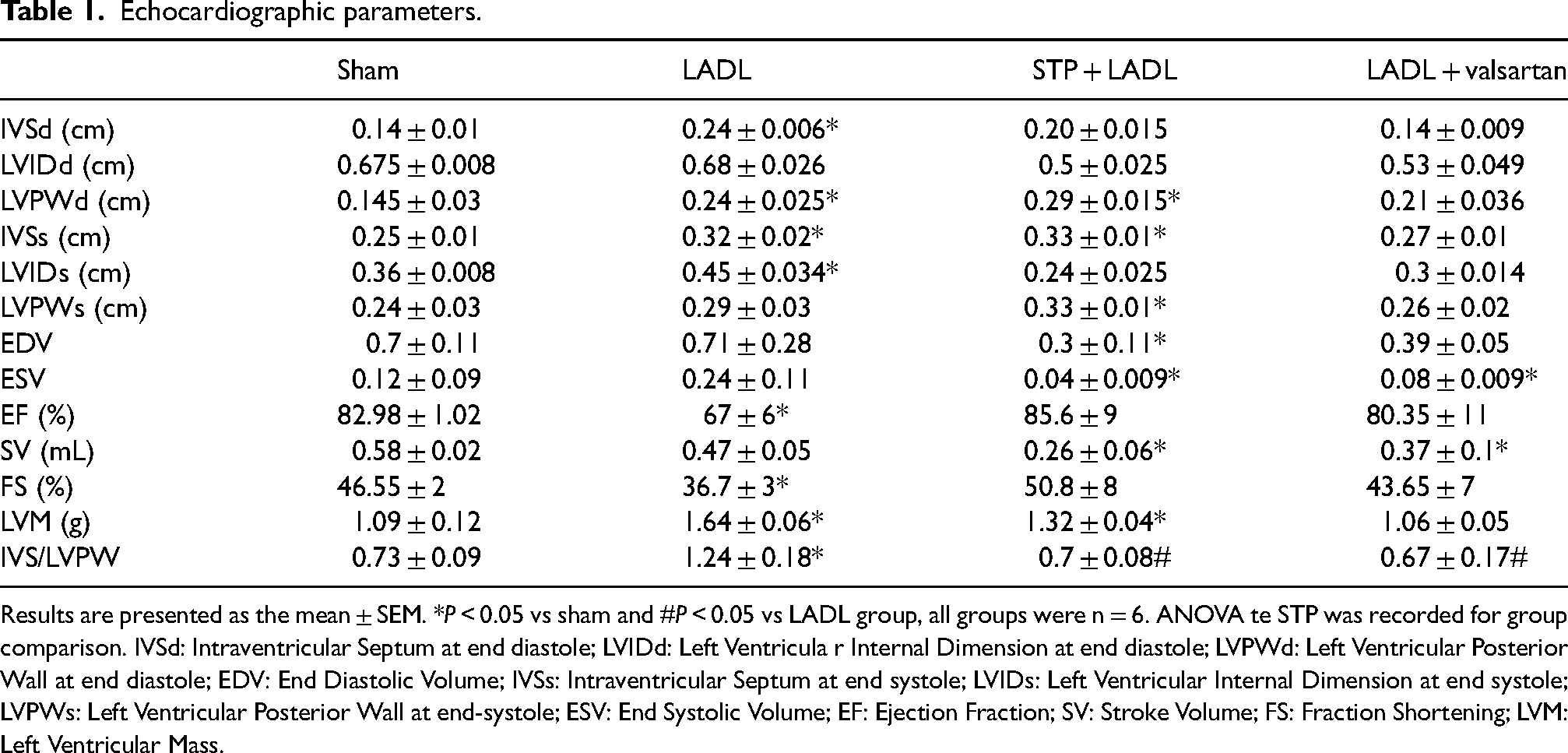
Results are presented as the mean ± SEM. *P < 0.05 vs sham and #P < 0.05 vs LADL group, all groups were n = 6. ANOVA te STP was recorded for group comparison. IVSd: Intraventricular Septum at end diastole; LVIDd: Left Ventricula r Internal Dimension at end diastole; LVPWd: Left Ventricular Posterior Wall at end diastole; EDV: End Diastolic Volume; IVSs: Intraventricular Septum at end systole; LVIDs: Left Ventricular Internal Dimension at end systole; LVPWs: Left Ventricular Posterior Wall at end-systole; ESV: End Systolic Volume; EF: Ejection Fraction; SV: Stroke Volume; FS: Fraction Shortening; LVM: Left Ventricular Mass.
Vascular reactivity
The vascular contractile response to Ang II and phenylephrine (Figure 3), showed that in both cases, the LADL group increased vascular reactivity compared to the sham group. The LADL + valsartan group returned both vasoconstrictors reactivity to baseline values. Interestingly, the ST + LADL group also decreased the reactivity of Ang II and phenylephrine beyond the sham group. Table 2 shows the analysis of the curves, where the LADL group had an increase in maximum effect (Emax) to both vasoconstrictors. The ST preconditioning group showed a lower Emax for both Ang II and phenylephrine. The effective concentration 50 (EC50) for Ang II remained unchanged between groups, but for phenylephrine, it was decreased in both treated groups. In addition, following Brooks et al., 42 we used a mathematical method to determine the vessel diameter, which indicated that the LADL group had the shortest diameter.
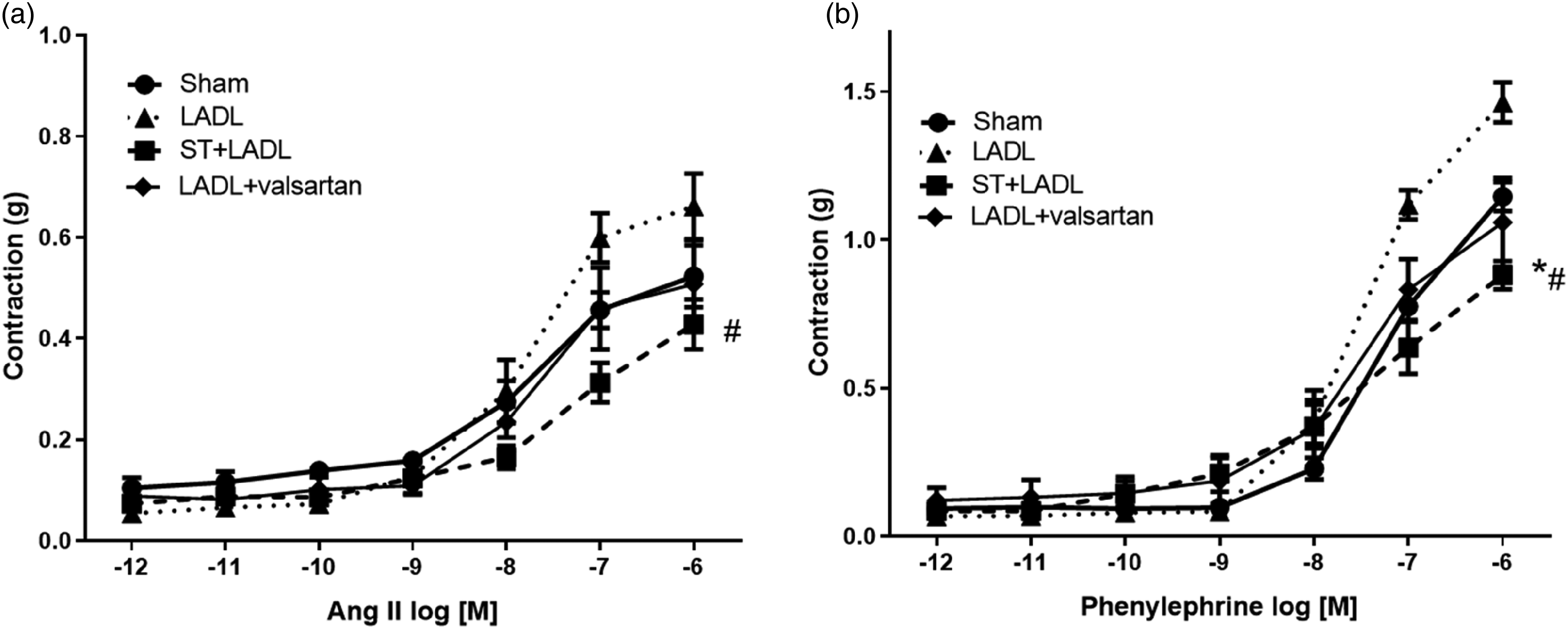
Vascular contractile response to angiotensin II and phenylephrine.
Pharmacodynamic analysis of the concentration-response curves to Ang II and phenylephrine.

Results are presented as the mean ± SEM. *P < 0.05 vs sham and #P < 0.05 vs LADL group, all groups were n = 6. ANOVA te STP was recorded for group comparison.
Finally, the production of NO in the heart is similar to blood levels (Figure 4). In both groups, we found that NO tended to increase in the LADL group and returned to normal values with valsartan treatment and exercise. However, in the aorta, NO regulation is different. In the sham group, we found values of 600 uM, which significantly decreased in the treated groups (ST + LADL and LADL + valsartan).

No levels in serum, heart, and aorta. (a) Serum NO levels, no significant differences were found. (b) Heart NO levels, no significant differences were found. (c) Aorta NO levels, a decrease was found in STP + LADL and LADL + valsartan groups (*P < 0.05) vs the sham group, STP + LADL group also decrease NO levels vs the LADL group (#P < 0.05).
Discussion
Our work was focused on studying the effect of ST preconditioning on the development of CMI on vascular reactivity and cardiac function in female rats, where we found that ST reduced vascular reactivity to both the major vasoconstrictors of the renin-angiotensin system and the sympathetic system. However, the modulation of vascular reactivity was not due to NO production as we expected. Furthermore, ST preserved cardiac function as well as valsartan treatment. ST has been proven due to the beneficial effects reported in patients after a coronary event which reduces mortality rate, cardiac remodeling, and improves left ventricular function.43–45 The cardioprotective mechanism of aerobic exercise was first reported by McElroy et al., 46 later some found that it could increase cardiac antioxidant capacity in rats, improve the systolic function of the aged myocardium, and also improve recovery of blood pumping capacity after myocardial ischemia.47,48 However, most STP studies were performed in males, with little detraining time, and in acute myocardial infarction models, focusing on cardiac function. 49 Our results suggest that long-term ST performed before LADL surgery improves vascular and cardiac function in chronic myocardial infarction in female rats by a NO-independent mechanism.
Despite the beneficial effects on cardiovascular function, we did not find differences in infarct size between the groups, in agreement with other authors,23,50 although the valsartan and STP groups showed a smaller infarct area. Previously, Yang et al. 51 reported that valsartan reduces the infarct size since it normalizes the balance of matrix metalloproteinases (MMPs)/ Tissue inhibitors of metalloproteinases 1 (TIMP-1) and fibronectin in rodents. Also, valsartan treatment reduces cardiac remodeling through Ang II receptor blockade and its hypertrophic effects on cardiomyocytes, fibroblast hyperplasia, and an increase in the amount of collagen deposited in the interstitial space. 52 On the other hand, it has been reported that swimming training, reduces inflammation, fibrosis, and scar thinning 53 due to the increase in peroxisome proliferator-activated receptor alpha (PPAR-α) levels 54 in rats, a finding that could be related to our results. Besides, the exercise induces an increase in oxygen delivery and molecular mechanisms to avoid ventricular remodeling through gene expression, paracrine factors, and signaling pathways that promote cardiac regeneration. 55
Regarding cardiac remodeling, we found an increase in the LVIDs of the LADL group, which is related to a decrease in ventricular contractile capacity, and it is reflected in the reduction of EF. 56 This decrease in contractile capacity is determined by changes in cardiac structures during myocardial infarction known as cardiac remodeling. 57 In our results the reduction of LVIDs is due to the increase in the thickness of the intraventricular septum and the LVPW in the LADL group, increasing the LVM. The hypertrophic process after MI has been widely reported. In this sense, in the LADL group, the increase in the IVS/LVPW ratio could indicate concentric hypertrophy. 58 On the other hand, STP prior MI, caused an increase in LVM, and LVPW, suggesting a hypertrophic process. These results could be related to the overexpression of Myosin heavy chain-ß (MHC- ß) and MMP-2. 59 Notwithstanding the increase in LVM, the IVS/LVPW ratio of the ST + LADL, did not show significant increases, suggesting that ST preconditioning before MI attenuated cardiac hypertrophic remodeling. Our results could be related to the study of Lin et al., where they found that physiological myocardial hypertrophy has a regression after a detraining period, which leads to antihypertrophic effects through myosin Heavy Chain Associated RNA Transcript (Mhrt779)/ brahma-related gene 1 (Brg1)/ histone deacetylase 2 (Hdac2)/ phosphorylated Akt (p-Akt)/ phosphorylated glycogen synthase kinase 3 beta (p-GSK3β) signal pathway, enhancing myocardial resistance to subsequent pathological hypertrophic stress, such as that caused by coronary occlusion. 60 Concerning cardiac function, ST avoided EF decrease after MI, this has been reported previously by Maessen et al., where athletes with MI showed a better ejection fraction in comparison to the sedentary MI group. 61 Furthermore, valsartan treatment prevented changes in cardiac morphology and function, as have been described by other authors. 19
We evaluated vascular reactivity to Ang II and phenylephrine because it has been reported that athletes have higher vascular compliance and better vascular wall structure compared with sedentary subjects. 62 However, the STP effect prior to MI on the vasculature is not fully understood. About this, an increase in peripheral resistance due to renin-angiotensin system (RAS) and sympathetic nervous system (SNS) hyperreactivity is associated with an augmented afterload of an infarcted heart, promoting cardiac hypertrophy. 63
Our results showed that Ang II induced greater vascular reactivity in groups with LADL as we previously reported. 28 In this sense, Ku et al. demonstrated an enhancement of vasoconstriction and a loss of vasodilating response to thrombin occurring in ischemic coronary arteries. 64 On the contrary, STP and valsartan treatment prevented the increase of Ang II vascular reactivity. After having obtained the concentration-response curves to Ang II and phenylephrine, we assumed that the result was because STP generates an increase in NO production. Previously in our group, we reported that females treated with valsartan showed a decrease in vascular reactivity to Ang II after two weeks of MI, by that time we proposed that the mechanism was due to the production of NO and cardioprotective estrogen effects. 28 However, in the present work, we did not find an increase in NO levels on ST + LADL and LADL + valsartan, on the contrary, a decrease was shown in the aorta. Interestingly, we found that NO regulation is very different in the vasculature than at the systemic level. According to our results, Agnaldo et al. reported that repeated STP reduces vascular reactivity to phenylephrine in mesenteric arteries, through a non-endothelial NO-related mechanism. 65 In addition to this, we performed vascular reactivity experiments in the aorta, which is a conductance artery. However, we do not exclude that NO production is also regulated by STP in smaller arteries. Another consideration to be made is that we quantified NO to relate it to the vasodilatory effect, however, there could be pathways not related to cGMP such as the activation of non-endothelium-dependent calcium channels that could generate the vasorelaxing effect, as reported by Ling et al., when analyzing the vasodilatory effect of the guanxinning tablet. 66 Therefore, our results suggest that changes in vascular reactivity due to MI and STP could be related to the modulation of the angiotensin and adrenaline receptors.
For Ang II, we only found differences in Emax, but not in EC50, and this could indicate that both STP and valsartan treatment influence receptor density but do not modify Ang II affinity to the AT1 receptor as other groups have reported.67,68 In addition, valsartan treatment modifies Emax and EC50 to phenylephrine despite valsartan being a drug that acts directly on AT1 receptors but not on adrenergic receptors. This could be explained by the influence of valsartan on adrenergic receptor density and signaling pathway, where some researchers have reported that Ang II through its AT1 receptor can induce the transcription of α1b and α1d adrenergic receptors.69–71
On the other hand, the STP group prior to myocardial infarction showed the lowest Emax to phenylephrine, as reported by Bianchi et al., 72 which is related to and Acetylcholine dependent vasodilation. 73 In addition, we found that STP is as effective as valsartan treatment in reducing phenylephrine EC50 and the diameter of the vessel compared to the LADL group. 74 The relationship between adrenergic receptors and exercise has been studied for some time, and it has been suggested that the adrenergic system is mainly responsible for maintaining vasoconstriction during exercise training and metabolism change through alpha-adrenergic receptors 75 and triggering sympathetic nerve activity. 76 Changes in adrenergic receptors at the central nervous level are due to exercise 77 and vagus nerve activity. 78 This work proves that STP had impact on the adrenergic and RAS receptors-signaling pathway in vascular smooth muscle. Furthermore, through a mathematical model, we found changes in the diameter of the vessel due to each therapy. This also indicates that each treatment not only influences at the molecular level or contractile function but also the tissue morphology, since, as reported, the vascular smooth muscle phenotype depends on calcium-dependent transcription factors79,80 and exercise training exerts a reestablishment in calcium channels function.81,82
Limitations of this study
This study has several limitations that should be acknowledged. First, the use of only female Wistar rats limits the generalizability of the findings to other sexes and species. Additionally, the study focused on the effects of STP in CMI but did not explore the impact of different types or intensities of exercise, which may produce varying effects on vascular and cardiac function. The six-week period between LADL induction and assessment may not fully capture the long-term effects of STP or the chronic progression of MI. Lastly, while NO levels were measured, other potential mechanisms underlying the observed improvements in cardiac function and vascular reactivity were not investigated, limiting our understanding of the full spectrum of ST's benefits before MI.
Conclusion
Long-term STP prior to coronary occlusion improves cardiac function and reduces vascular reactivity to Ang II and phenylephrine in chronic myocardial infarction, through an independent NO mechanism in female rats.
Footnotes
Acknowledgments
To Crisoforo Mercado-Marquez for his support at the Cuautitlan Superior Studies Faculty animal facility.
Author contributions
Flores-Monroy Jazmin Ramirez-Hernandez Diana, and Lezama-Martinez contributed to the design, implementation of the research, and supervised the project. Velazco-Bejarano Benjamin, Valencia-Hernandez Ignacio, Lopez-Sanchez Pedro, and Fonseca-Coronado Salvador contributed to the analysis of the results and to the writing of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Research carried out thanks to the program of the Dirección General de Asuntos del Personal Académico, Universidad Nacional Autónoma de México, Catedra. FES Cuautitlan, (grant number IN204925, IA204924, IN202022, CI2429, CI2211 and PIAPIME 2.11.21.24).
