Abstract
Introduction:
The effect of renal sympathetic denervation (RDN) on neurohormonal responses is largely unknown. We aimed to assess the effect of RDN on the renin–angiotensin–aldosterone system (RAAS) and endogenous catecholamines.
Methods:
A total of 60 patients with hypertension underwent RDN and remained on a stable antihypertensive drug regimen. Samples for plasma aldosterone, plasma renin and urine (nor)metanephrine were collected at baseline and at 6 months post procedure. Ambulatory blood pressure (BP) recordings were obtained at baseline and at 6 months post procedure.
Results:
Mean age was 64±9 years, and 30/60 patients were male. At 6 months, average daytime systolic and diastolic ambulatory BP decreased by 10 and 6 mmHg, respectively (p<0.001). No significant change was observed in plasma aldosterone (median=248.0 pmol/L (interquartile range (IQR) 113.3–369.5 pmol/L) vs. median=233.0 pmol/L (IQR 110.3–360.8 pmol/L); p=0.66); renin (median=19.5 µIU/mL (IQR 6.8–119.5 µIU/mL) vs. median=14.3 µIU/mL (IQR 7.2–58.0 µIU/mL); p=0.32), urine metanephrine (median=0.46 µmol/L (IQR 0.24–0.77 µmol/L) vs. median=0.46 µmol/L (IQR 0.22–0.88 µmol/L); p=0.75) and normetanephrine (median=1.41 µmol/L (IQR 0.93–2.00 µmol/L vs. median =1.56 (IQR 0.74–2.50 µmol/L); p=0.58) between baseline and 6 months, respectively. No correlation was found between the decrease in mean systolic daytime BP and changes in RAAS hormones or endogenous catecholamines.
Conclusion:
Despite significant reductions in ambulatory BP, RDN did not result in a significant change in endogenous catecholamines or in RAAS hormones at 6 months.
Keywords
Introduction
Recent randomised, sham-controlled studies have proven the blood pressure (BP)-lowering effect of renal sympathetic denervation (RDN) in patients with mild and uncontrolled hypertension.1–4 Although the procedure has been shown to be efficacious and safe, many questions remain about the exact pathophysiological mechanisms by which RDN lowers BP. 4 The antihypertensive effect of reducing sympathetic tone by RDN is hypothesised to be driven by the interaction between renal sympathetic nerves and the renin–angiotensin–aldosterone system (RAAS). 5 The renal afferent nerves are the physiological conduit of central sympathetic drive and are involved in the control of peripheral vascular resistance. 6 The efferent nerves increase renin secretion by juxtaglomerular cells using norepinephrine (NE) as a neurotransmitter, stimulate tubular reabsorption of sodium and directly increase renal vascular resistance.6,7
Previous studies have shown that there is a causal relationship between renal norepinephrine spillover, renin release and arterial plasma renin activity. 8 Moreover, the first proof-of-concept trial for RDN showed a mean reduction of 47% in renal noradrenaline spillover accompanied with a significant BP drop in patients with hypertension. 9 Conversely, the effects of RDN on the RAAS has only been studied in a limited number of preclinical studies, failing to demonstrate a correlation between BP reduction and change in sodium excretion or in RAAS activity.10,11 To date, dedicated studies focusing on the link between RDN and neurohormones in vivo are lacking. Therefore, in the present study, we aimed to assess the effect of RDN on RAAS hormones and endogenous catecholamines as an attempt to understand the BP-lowering effect.
Methods
Study population
This was a single-centre (Erasmus Medical Centre) prospective study including 63 hypertensive patients undergoing RDN between November 2012 and February 2016. Patients had to be on a stable antihypertensive drug regimen for at least 4 weeks prior to inclusion. Patients were excluded when their antihypertensive drug regimen was changed during the course of the study (n=3). The study was approved by the Erasmus Medical Centre Ethics Committee, and written informed consent was obtained from all participants.
RAAS hormones and endogenous catecholamines were prospectively measured at baseline, after a 4-week stabilisation protocol and at 6-month follow-up. Enrolment occurred when the following criteria were met: a mean office BP of ⩾140 and/or 90 mmHg (based on three consecutive measures), use of two or more antihypertensive drugs, age 18–75 years and an estimated glomerular filtration rate (eGFR) of >45 mL/min/1.73 m2. Secondary causes for hypertension were ruled out in all patients after extensive laboratory analysis and computed tomography angiography or magnetic resonance imaging assessment.
Study measurements and end points
Laboratory data were drawn pre-procedurally (baseline) and at 6 months post procedure (RDN) during morning hours. Laboratory analyses based on blood samples were obtained in a sitting position. Blood sampling was done at four to 8 weeks prior to the procedure. Urine samples were collected to measure the levels of (nor)metanephrine. The blood and urine sampling protocol was similar for all time points. Creatinine-corrected urinary sodium and potassium levels were obtained at baseline and at 6 months in order to correct for possible confounding by changes in diet and sodium intake. Antihypertensive drugs that could interfere with the plasma renin and/or aldosterone remained unchanged during sampling at both baseline and at the 6-month follow-up.
The primary end point was the change in plasma renin, plasma aldosterone and endogenous catecholamine (urine (nor)metanephrine) concentrations at 6 months post procedure compared to baseline. Secondary analyses included the change in renal function as measured using both eGFR (mL/min) and cystatin C (mg/L). eGFR was estimated based on the following the following: GFR in mL/min per 1.73 m2=175×serum creatinine–1.154×age–0.203×1.212 (if patient is black)×0.742 (if patient is female).
Plasma aldosterone, urinary metanephrine and normetanephrine concentrations were measured by liquid chromatography-mass spectrometry (Waters Xevo TQS, Milford, MA). Plasma renin concentration was measured using the IDS-iSYS assay system (Immunodiagnostic Systems, Boldon, UK). Automated seated office BP measurement was performed with the Omron M10-IT (Omron Healthcare Co. Ltd, Kyoto, Japan). An average value of three consecutive BP measurements was used (measured on the upper arm with the highest BP value). The 24-hour ambulatory BP measurements (24h ABPM) were performed with an oscillometric device (model 90217A; Spacelabs Healthcare, Snoqualmie, WA), and the mean (systolic and diastolic) 24h ABPM and mean daytime (systolic/diastolic) ABPM were included in the analysis. A patient with a decrease of ⩾5 mmHg in mean daytime systolic ABPM at 6 months was considered as a responder to RDN. 12 Patients in which samples were only available either at baseline or at follow-up were excluded.
RDN
All patients not using aspirin were preloaded with 300 mg aspirin and were advised to continue with aspirin for at least 1 month. Pre-procedurally, 100 IU heparin/kg were administered to achieve an active clotting time >250 seconds. All procedures were performed under conscious sedation. After administration of local anaesthesia, RDN was performed according to standard instructions for use using either the Paradise™ (n=13; Recor Medical, Palo Alto, CA), Vessix V2™ (n=5; Boston Scientific, Natick, MA), Symplicity™ (n=19; Medtronic, Minneapolis, MN), OneShot™ (n=3; Covidien, Campbell, CA) or EnligHTN system (n=20; St Jude Medical, St Paul, MN).13,14
Statistical analysis
Continuous data are expressed as the mean±standard deviation (SD) when normally distributed, or as median (interquartile range (IQR)) when non-normally distributed. Categorical data are presented as percentages. Changes in BP between baseline and 6 months were assessed using the paired t-test. Changes in RAAS hormones and endogenous catecholamines between baseline and 6 months were assessed by using the Wilcoxon signed-rank test. The Spearman or Pearson correlation coefficient was used when appropriate to evaluate the relationship between the change in RAAS hormone concentration and the change in BP. The change in RAAS hormones and endogenous catecholamines in responders versus non-responders was tested using the Mann–Whitney U-test. All statistical tests were two-tailed. A p-value of <0.05 was considered statistically significant. Statistical analysis was performed using IBM SPSS Statistics for Windows v24.0 (IBM Corp., Armonk, NY).
Results
Mean age was 64±9 years, and 30/60 patients were male. The mean number of antihypertensive drugs at baseline was 3±1. The antihypertensive drug regimen did not change between baseline and 6-month follow-up in any of the patients. Mean office BP was 172±23/94±14 mmHg and mean 24h ABPM was 144±17/81±12 mmHg. Baseline characteristics are presented in Table 1. No differences were found in baseline characteristics between responders and non-responders.
Baseline characteristics.

Values are presented as M±SD or n (%).
ARB: angiotensin receptor blocker; ACEi: angiotensin converting enzyme inhibitor; 24h ABPM: 24h ambulatory blood pressure measurement; BP: blood pressure; CCB: calcium channel blocker; eGFR: estimated glomerular filtration rate; SD: standard deviation.
Change in RAAS hormones and endogenous catecholamines
Venous blood sampling and urine collection was performed at a median of 78 days (IQR 34–150 days) prior to procedure (considered as ‘baseline’) and at 6-month follow-up at a median of 192 days (IQR 183–199 days) after RDN. Median plasma aldosterone was 248.0 pmol/L (IQR 113.3–369.5 pmol/L) at baseline compared to 233.0 pmol/L (IQR 110.3–360.8 pmol/L) at 6 months (p=0.66), and median plasma renin was 19.5 µIU/mL (IQR 6.8–119.5 µIU/mL) at baseline compared to 14.3 µIU/mL (IQR 7.2–58.0 µIU/mL) at 6 months (p=0.32). Median urine metanephrine was 0.46 µmol/L (IQR 0.24–0.77 µmol/L) at baseline compared to 0.46 µmol/L (IQR 0.22–0.88 µmol/L) at 6 months (p=0.75), and median urine normetanephrine was 1.41 µmol/L (IQR 0.93–2.00 µmol/L) at baseline compared to 1.56 µmol/L (IQR 0.74–2.50 µmol/L) at 6 months (p=0.58; Table 2).
Renin, aldosterone and (nor)metanephrine at baseline and six months post procedure.

Values are presented as median (IQR).
IQR: interquartile range.
Safety end point
Renal function remained unchanged between baseline (70.8±15.9 mL/min) and 6-month follow-up (69.4±17.6 mL/min; p=0.33). Cystatin C also remained unchanged between baseline (1.10±0.24 mg/L) and 6-month follow-up (1.11±0.26 mg/L; p=0.54).
Change in BP and correlation with neurohormones/catecholamines
Mean office BP decreased significantly between baseline and 6 months (–12±23/–8±12 mmHg; p<0.01). Similar results were observed in mean (–8±14/–5±9 mmHg) and mean daytime ABP (–10±16/–6±9 mmHg; both p<0.01; Table 3).
Change in BP at follow-up measured in office and ambulant.

Values are presented as M±SD or n (%).
No correlation was found between the change in mean daytime systolic ABP and change in plasma renin (r2=0.26; p=0.28; Figure 1). A similar lack of correlation was found for plasma aldosterone and change in mean daytime systolic ABP (r2=0.34; p=0.14), and similar findings were observed in urine normetanephrine (r2=–0.28; p=0.72) and urine metanephrine (r2=0.20; p=0.80). When comparing responders (n=36/57) and non-responders (n=24/57), no significant difference in changes between 6 months and baseline were observed in plasma renin and aldosterone levels and urine catecholamines (Table 4).
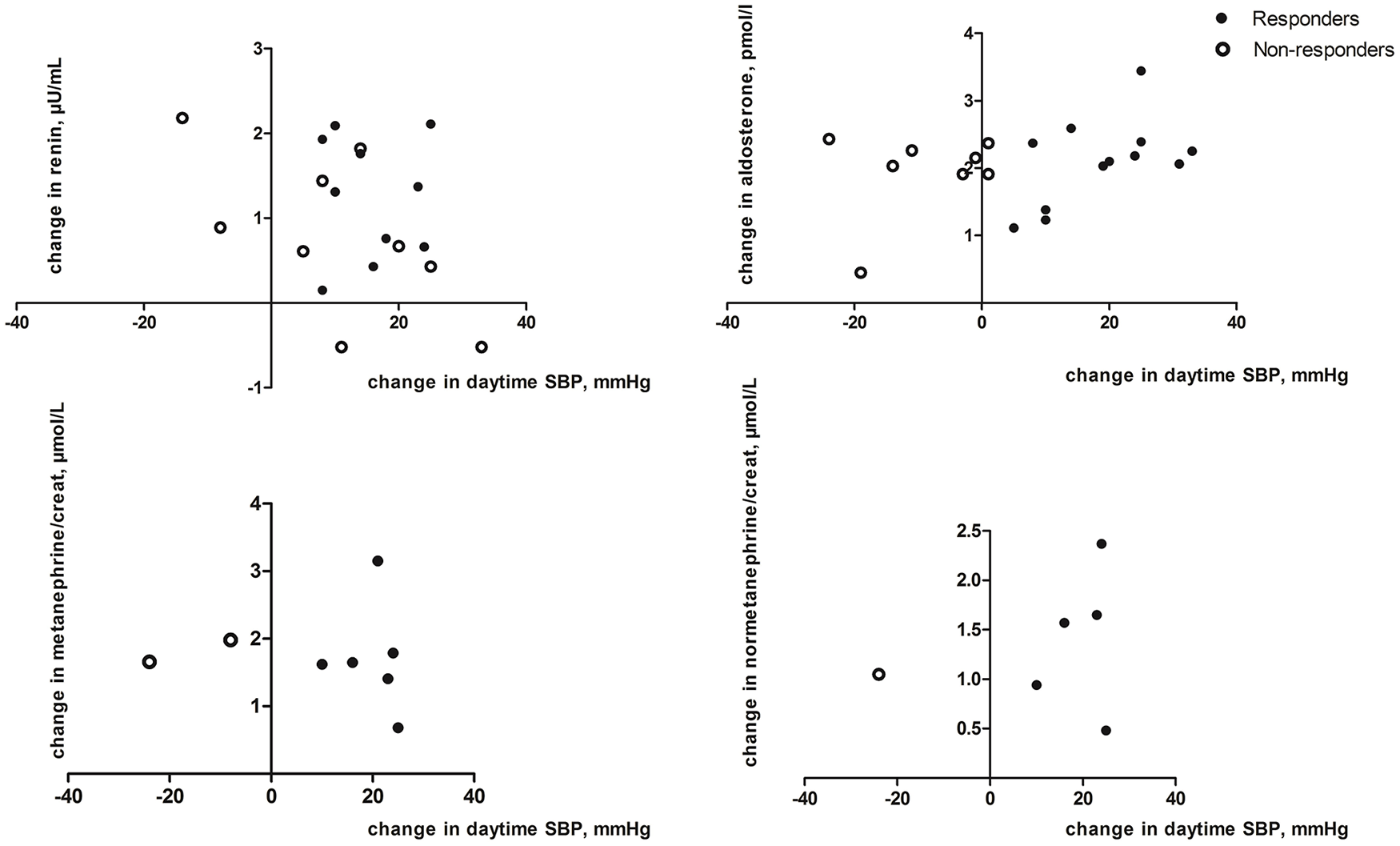
Change in plasma renin and aldosterone and endogenous catecholamines after RDN in responders versus non-responders (on a logarithmic scale).
Change in RAAS hormones and endogenous catecholamines in responders versus non-responders to RDN.
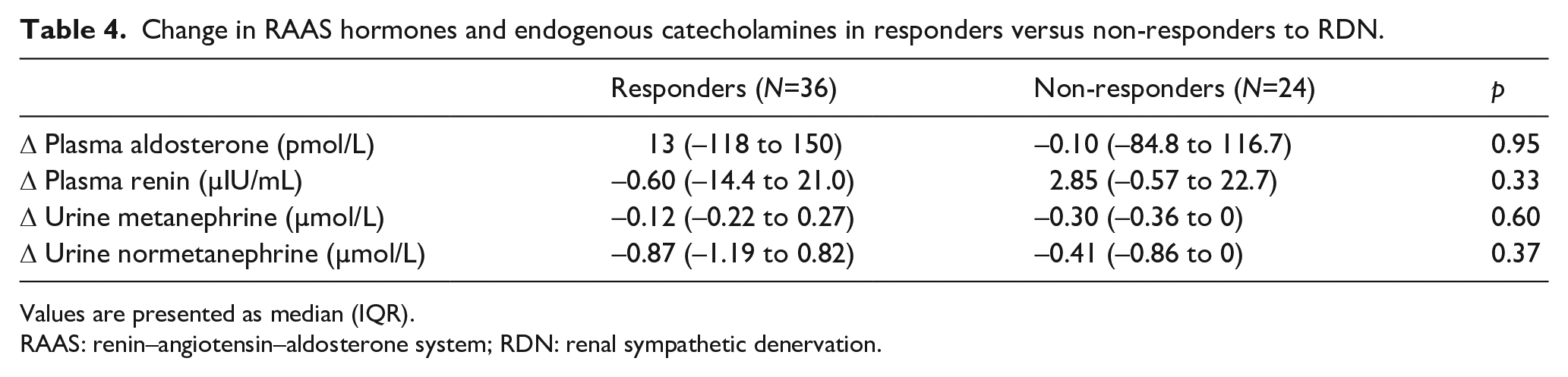
Values are presented as median (IQR).
RAAS: renin–angiotensin–aldosterone system; RDN: renal sympathetic denervation.
Discussion
In the present study, the significant reduction in ABP and office BP induced by RDN in patients with moderate to severe hypertension could not be explained by the change in endogenous catecholamines and in RAAS hormones at 6 months. In addition, no correlation was found between the response to RDN as measured using the mean daytime systolic ABP and change in both RAAS hormones and endogenous catecholamines.
In a search towards a better understanding of the pathophysiological mechanisms behind the BP-lowering effect by RDN, we attempted to search for potential differences in RAAS hormones and endogenous catecholamines as induced by RDN. While our findings are in line with several preclinical studies,10,11 previous animal data by Zhao et al. suggested that RDN might suppress an increase in plasma aldosterone in 28 dogs with progressive heart failure at 3 weeks. 15 In addition, Hong et al. demonstrated a clear suppression in aldosterone release following RDN in rats with angiotensin (Ang)-II-induced hypertension after 14–17 days. Conversely, no change was found in renin levels, which could probably be explained by the negative feedback effect of Ang-II on renin.16,17 With the latter evidence, a relation between the renal sympathetic nerves and aldosterone regulation was suggested – a process that could act independently of renin. As such, a small clinical study (N=17) found a significant change in plasma noradrenaline 6 months post RDN. However, no change was found in BP or in renin levels. 18
To the best of our knowledge, the present work represents the largest body of evidence to date on the topic, which failed to show a significant impact of RDN on RAAS hormones and endogenous catecholamine release in vivo. Based on the available evidence, it could be that endogenous catecholamines are not sensitive enough to detect a difference which would correlate with a BP decrease. 19 Furthermore, it should be noted that catecholamines are the shortest lived signalling molecules in plasma, with an initial biological half-life of approximately one to two minutes. 20
It is well known that activation of the sympathetic nervous system and stimulation of the RAAS leads to an increase in BP.21,22 Blocking the RAAS with Ang-converting enzyme (ACE) inhibitors and Ang-II type 1 receptor (AT1R) antagonists subsequently became one of the most frequent ways to treat hypertension and proved to improve cardiovascular morbidity and lower mortality significantly.23–25
ACE inhibitors, AT1R antagonists, beta-blockers and aldosterone receptor blockers are known to affect plasma renin and aldosterone levels.26,27 Therefore, in the present study, we paid specific attention to keep the confounding effect of drugs as low as possible. For this reason, we only included patients on a stable drug regimen for at least 4 weeks before blood samples were taken and RDN was performed. Second, patients in whom the antihypertensive drugs were changed during the course of the study were excluded. Furthermore, we attempted to correct for potential differences in the change of dietary sodium and/or potassium intake, which are known to affect plasma renin and aldosterone.28,29
Finally, the present work confirms the difficulty in linking RDN treatment success. Along these lines, even studies correlating RDN response to muscle sympathetic nerve traffic (MSNA) did not found a correlation. MSNA has been considered the gold standard for measuring systemic sympathetic nerve activity due to its good sensitivity and reproducibility. 30 However, other studies showed conflicting results.31,32 MSNA measurement, however, proved significantly more invasive and complex than measuring neurohormones.
Limitations
The present study has several limitations. First, most recent RDN trials identified primary end points at 2–3 months post procedure in order not to expose patients to unnecessary long periods off drugs (for trials performed in an off-med population) and in order to reduce the likelihood of the impact of changes in antihypertensive drug regimens in on-med trials in patients on predefined drug regimens.1–3 At the time this study was designed (2012), there was an overall believe that 6 months of follow-up were needed to observe the actual effect of RDN. 9 The latter explains the decision to perform RAAS and neurohormone levels at the same time point: 6 months post procedure. Future trials with assessment of RAAS and neurohormone levels at shorter-term time points are awaited, although it is almost technically not feasible to determine the real-time rate of neurohormone production. Second, two thirds of the patients in our study received an ACE inhibitor, an AT1R antagonist or a diuretic, which could have influenced our findings. The latter bias, however, was considered to be minimal due to a similar degree of interference at baseline and follow-up in a population in which no changes in drug regimen occurred. Third, we did not test for drug adherence at baseline and at 6-month follow-up. Fourth, since no sham control group was included in the present study, the potential of a placebo effect on BP could not be ruled out. Finally, procedures were performed with different devices and different procedural strategies. A total of 25/60 RDN procedures were performed with RDN systems that are no longer available. A sensitivity subgroup analyses into radiofrequency versus ultrasound catheter-based RDN did not reveal any heterogeneity in the findings on RAAS hormones and endogenous catecholamines. However, we could not perform a logistic regression due to a small sample size.
Conclusion
Despite significant reductions in ABP, RDN did not result in a significant change in endogenous catecholamines and in RAAS hormones at 6 months post procedure.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
