Abstract

The interplay between the renin-angiotensin-aldosterone system (RAAS) and kidneys in patients with heart failure (HF) can lead to highly complex and challenging clinical scenarios. Among these scenarios, worsening renal function (WRF) and/or hydro-electrolytic alterations (hyperkalemia in particular) often raise clinicians’ awareness leading to downstream decisions (e.g. change in medications and/or its dosages), which can have consequences for the patients. 1 It should, nonetheless, be noted that WRF (a major determinant of adverse outcomes in HF patients) may be of multifactorial origin and the RAAS inhibitors (RAASi) are one of its multiple causes. This is thought to occur because RAAS activation in HF leads to glomerular efferent arteriolar vasoconstriction that preserves glomerular filtration (in the face of a fall in glomerular perfusion pressure caused by HF). By reducing this neurohormonal activation, RAAS blockers can reduce the glomerular perfusion pressure (reducing systemic arterial and glomerular afferent arteriolar pressure while also preventing the compensatory efferent arteriolar constriction) and cause WRF. 2 However, used chronically, RAASi treatment may attenuate renal function deterioration, suggesting an initial creatinine rise may not be clinically relevant in certain populations. 3 Increment in serum creatinine/WRF occurring in the setting of decongestion or titration of neurohormonal antagonists is also commonly encountered, primarily as a result of the effects of loop diuretics and/or of RAASi on renal hemodynamics. Recent data showed that when HF medications (i.e. angiotensin-converting enzyme inhibitors/angiotensin receptor blockers, mineralocorticoid receptor antagonists (MRAs), β-blockers and diuretics) were held for 48 hours, serum creatinine decreased but natriuretic peptides and cardiac volumes increased. 4 These data support the concept that serum creatinine increases and WRF should not be evaluated in isolation but rather considered in the context of the whole clinical scenario: not all increases in serum creatinine adversely affect prognosis. For example, a meta-analysis showed that WRF in patients with HF with reduced ejection fraction (HFrEF) randomized to an RAASi therapy was associated with lower mortality than WRF on placebo. 5 It should be emphasized that RAASi have convincing evidence of benefit on prolonging survival and reducing morbidity in patients with HFrEF, and both United States and European guidelines give a Class I, Level of Evidence A recommendation for their use. In contrast, WRF in patients with HF with preserved ejection fraction (HFpEF) was associated in increased events regardless of the use of RAASi. This is because, to date, the HFpEF trials failed to demonstrate a clear morbidity and mortality improvement. 5 Despite this, a particularity should be noted with spironolactone in the TOPCAT (Spironolactone for Heart Failure with Preserved Ejection Fraction) trial. 6 In TOPCAT only patients from the Americas had characteristics compatible with HFpEF and adequate blood levels of the drug metabolites.7,8 Hence, when considering this HFpEF population, spironolactone improved outcomes regardless of the baseline estimated glomerular filtration rate; however, more adverse events, including WRF, were observed in patients with impaired renal function in whom close laboratory monitoring is recommended. 9 In concordance, the updated American Heart Association/American College of Cardiology/Heart Failure Society of America (AHA/ACC/HFSA) guidelines give spironolactone a class IIb indication for HFpEF. 10
Along with WRF, one of the major concerns clinicians have by using RAASi is hyperkalemia, often overlooking hypokalemia. 11 In fact, both hypo- and hyperkalemia have been associated with higher risk for cardiovascular and all-cause mortality, 12 suggesting that serum potassium should be maintained within a relatively narrow range (4–5 mmol/L) 11 . To add the complexity of the RAAS-kidney interactions in HF, patients with impaired renal function taking RAASi have higher odds of hyperkalemia, drug intolerance and discontinuation, but (in the case of spironolactone) the treatment benefit is maintained in patients with renal impairment. 9 Despite this, a recent observational study including all Stockholm citizens initiating MRA therapy during 2007–2010 assessed the 1-year incidence of clinical hyperkalemia, and quantified drug prescription changes after an episode of hyperkalemia. After hyperkalemia, 47% discontinued MRA and only 10% reduced the prescribed dose. Strikingly, when MRA was discontinued, most patients (76%) were not reintroduced to therapy during the subsequent year. 13
Post hoc analyses of major randomized controlled trials suggest that patients with renal impairment should not be withdrawn from therapy; on the contrary, the most robust evidence suggests these patients should have an adequate biological monitoring, medical surveillance and drug-dose adaptations when needed. 14 This is indeed a key point: observational studies have repeatedly shown that RAASi are underused, sub-optimally dosed and inadequately monitored, and that these (mal)practices are associated with worse outcomes. 1 This ‘real-world’ inaction is not due to lack of information or guidance. In fact, current guidelines provide specific recommendations on how to handle RAASi in the setting of WRF and/or hyperkalemia and recommend close monitoring of serum potassium and renal function during periods of RAASi titration.15,16 Monitoring should continue until the patient is stable and at regular intervals thereafter. Down-titration of doses or discontinuation of RAASi may be attempted temporarily for persistent hyperkalemia, hypotension, or WRF. Up-titration and re-initiation should be attempted as soon as possible and is feasible in most cases. A dietary counselling regarding potassium and concomitant medication reappraisal (e.g. potassium supplements, non-steroidal anti-inflammatory drugs [NSAIDs], loop diuretic doses, with the latter adapted to the patient’s volemic status) is warranted. The use of potassium binders may facilitate up-titration in patients prone to hyperkalemia (although whether this may prevent cardiovascular outcome occurrence needs to be demonstrated in a dedicated outcome trial). Moreover, several biomarkers of renal injury are being evaluated for their utility to detect kidney injury and to differentiate ‘real’ kidney injury from functional decline without injury. In this regard, novel ‘tubular dysfunction markers’ (e.g. neutrophil gelatinase–associated lipocalin, kidney injury molecule-1) are promising, but further validation is needed before adoption in clinical practice. 17
One should also note that fibrosis is a major pathway leading to renal function decline throughout life, and that anti-fibrotic agents (e.g. MRAs) may mitigate the progression of kidney disease. 1
In our opinion, concerns about WRF and hyperkalemia are probably excessive in some cases and should probably not be used as a justification for suboptimal use of life-prolonging therapies.
1
These concerns should instead be positively converted into a firm commitment to perform a mindful biological monitoring, aimed at retrieving in the real-life setting the best benefit of RAASi observed in clinical trials. Such monitoring is even more compelling in patients with renal impairment. In Buddhism, ‘Samsara’ refers to the repeated cycle of birth, mundane existence (unsatisfactory and painful) and death. By analogy, stopping RAASi in patients with HF may lead to a repeated cycle of WRF, clinical deterioration and death. Unlike Buddhism, where only a few are liberated through meditation, in HF one only needs to continue the RAAS inhibitors (
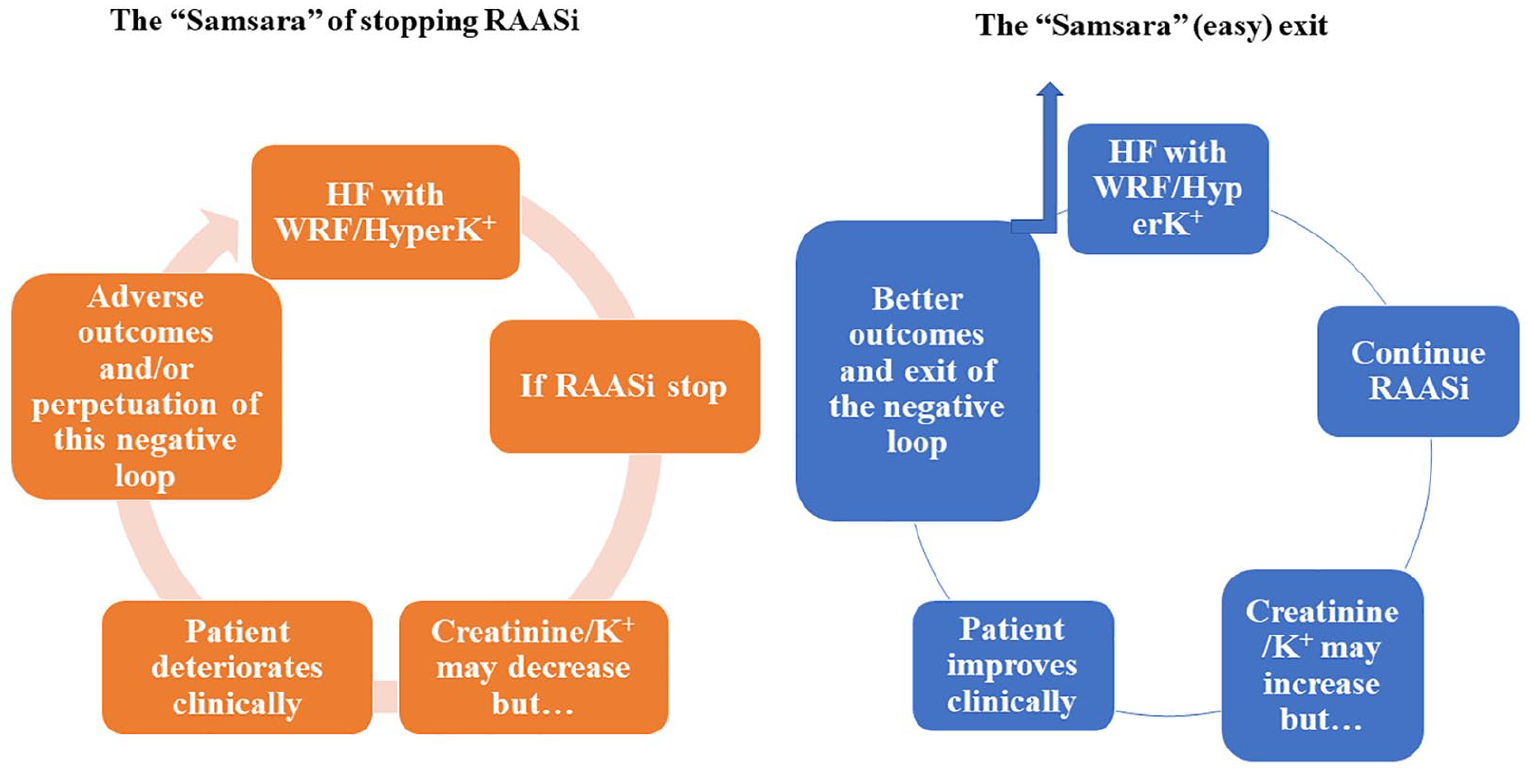
Harmful effects of stopping RAASi and beneficial effects on continuing them.
Footnotes
Acknowledgements
JPF, PR and FZ are supported by the French National Research Agency Fighting Heart Failure (ANR-15-RHU-0004), by the French PIA project ‘Lorraine Université d’Excellence’ GEENAGE (ANR-15-IDEX-04-LUE) programs.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: FZ received personal fees for Steering Committee membership from Janssen, Bayer, Pfizer, Novartis, Boston Scientific, Resmed, Takeda, General Electric and Boehringer Ingelheim, as well as consultancy fees from Amgen, CVRx, Quantum Genomics, Relypsa, ZS Pharma, AstraZeneca and GSK. He is the founder of Cardiovascular Clinical Trialists and CardioRenal. PR received personal fees (consulting) from Fresenius, Idorsia, Novartis, NovoNordisk, Relypsa, Stealth Peptides, Vifor; lecture fees from Bayer and CVRx; cofounder of CardioRenal. JPF received consulting fees from Novartis.
