Abstract
Objective:
In our recent study, non-Gaussianity of heart rate variability (λ25s), an indicator of sympathetic nerve activity, did not change during two-day treatment with the angiotensin II type 1 receptor blocker (ARB) azilsartan. Coadministration of calcium channel blockers (CCBs) might affect the study results.
Methods:
In this subanalysis, 20 patients with chronic kidney disease (14 men; age 61±15 years) were divided into three groups: patients with coadministration of L-type CCB, patients without coadministration of CCB, and patients with coadministration of sympathoinhibitory (L/T- or L/T/N-type) CCB. λ25s was calculated separately in daytime and nighttime.
Results:
Daytime λ25s at baseline was higher in patients with L-type CCB coadministration (0.62±0.18, n = 5) compared with those without CCB (0.49±0.13, n = 11) and those with sympathoinhibitory CCB (0.46±0.06, n = 4). The relationship between the changes in daytime λ25s and systolic blood pressure was positive in patients with L-type CCB coadministration, whereas the relationship was inverse in the other two groups. A larger decrease in daytime λ25s was shown in patients with L-type CCB coadministration compared with those in the other two groups.
Conclusions:
CCBs, as well as diuretics, are recommended as second-line antihypertensive agents. Our results suggested that ARBs can overwhelm the activation of sympathetic nerve activity stimulated by coadministration of L-type CCBs.
Keywords
Introduction
In experimental or clinical studies, sympathetic or parasympathetic (or vagal) nerve activity was assessed by the microneurographic method for recording muscle sympathetic nerve activity (MSNA), radiolabeled norepinephrine spillover in the target organs, neuroimaging technique using 123-metaiodobenzylguanidine, and heart rate variability (HRV) obtained by 24-hour–Holter electrocardiogram (hECG).1–5 Among them, hECG has the advantages of being noninvasive and it can be carried out in an ambulatory setting. Most HRVs are known to indicate impaired cardiac vagal function,
6
whereas power of low-frequency (LF) to high-frequency (HF) ratio (LF/HF) are used as sympathetic nerve activity. The
Methods
Participants
This is a subanalysis of our study in 20 patients with hypertensive CKD (14 men and six women; age 61±15 years; body mass index (BMI): 23.0±3.8 kg/m2; office BP 150±16/87±14 mmHg (an average of two different visits)). 15 The protocol of the single-arm, open-label study has been reported in detail elsewhere. 15 The study was approved by the institutional review board of Nagoya City University Hospital (approval number: 45-12-0022, University Hospital Medical Information Network registration number: 000009549) and was conducted in accordance with the Declaration of Helsinki, under the helm of the Ministry of Health, Labour and Welfare. Briefly, inclusion criteria were (1) age ⩾16 years; (2) diagnosis of CKD based on Kidney Disease Outcomes Quality Initiative (K/DOQI) criteria; (3) office BP >130/80 mmHg, or 125/75 mmHg if proteinuria ⩾1 g/day on at least one occasion; and (4) dietary salt intake ⩽6 g/day. 15 Exclusion criteria were (1) treatment with ARBs or diuretics two months before enrollment; (2) change of antihypertensive agents in the two months before enrollment; or (3) contraindication to azilsartan (history of allergic reactions to the drug, or renal artery stenosis). 15 All participants were enrolled after providing informed consent to participate in the study. At the time of enrollment, 11 patients were taking no antihypertensive agents and nine were taking CCBs. The CCBs were amlodipine (L-type, n = 5), azelnidipine (L/T-type, n = 3) and benidipine (L/T/N-type, n = 1), respectively. The original renal disease was nephrosclerosis in all eight patients taking L-type and L/T-type CCB, and glomerulonephritis in one patient taking L/T/N-type CCB. None of these nine patients had diabetic nephropathy. Only one patient taking L/T-type CCB had a smoking habit.
Study protocol
The study was performed under hospitalization. Patients ate a diet containing 6 g/day of salt; and they were asked to wake up at 06:00 and to start bed rest at 21:00. On the day when ambulatory blood pressure monitoring (ABPM) was finished, we confirmed whether they actually had done so. Measurements were made before initiation of ARB treatment (baseline) and two days after the start of oral administration of the ARB azilsartan (acute phase). Azilsartan was administered once in the morning (average of two office systolic BP (SBP) readings in two visits: ⩾160 mmHg, 40 mg/day (n = 2); ⩾125 and <160 mmHg, 20 mg/day (n = 12); and <125 mmHg, 10 mg/day (n = 6)). At baseline and in the acute phase of ARB treatment, 24-hour ABPM (monitored every 30 minutes with a validated automatic device, model TM-2425; A&D) and hECG (recorded with a portable recorder, RAC-3103, Nihon Koden) were conducted on the same day, and the data were calculated separately in the daytime (06:00–21:00) and nighttime (21:00–06:00). Real-time monitoring of the BP and HR was not performed.
Non-Gaussianity of HRV index λ25s
We used λ25s as an indicator of sympathetic nerve activity. The rationale and mathematical description of the non-Gaussianity index λ25s have been reported in detail elsewhere.9,10,14,15 In brief, using an ECG scanner (DSC-3300, Nihon Koden), hECG signals were digitized at 125 Hz and 12 bits. QRS complexes were labeled automatically and experienced technicians edited all possible errors. Recorded data were not considered valid for analysis if analyzable recording time was <23.5 hours, or if ventricular and supraventricular ectopic beats were >10% of all recorded beats. Obtained normal-to-normal R-R interval data were used for the analysis. λ25s can detect intermittency of the HR increment and can indicate probabilities of a volcanic HR deviation of departure from each SD level.9,10,14,15 Control data for the HRV analysis were obtained from age-, gender-, and BMI-matched same number of people, including individuals who underwent 24-hour ambulatory ECG for evaluation of chest discomfort without medication with antihypertensive agents, but were proven not to have cardiac and kidney diseases or hypertension, or healthy volunteers (n = 20). 15 For ethical reasons, the ARB was not started in these 20 individuals.
Statistical analysis
Results are expressed as mean±SD. Differences in parameters between the baseline and corresponding control, and between before and during ARB treatment, were examined by Student t-test for paired samples or by Wilcoxon signed-rank test, according to the data distribution, which was tested using a Kolmogorov–Smirnov test. Correlations among quantitative variables were evaluated by the least-squares method. Relationships between the changes in the variables were analyzed by linear regression through the origin. We divided the participants into three categories: patients who had taken L-type CCB before the initiation of ARB treatment (i.e. coadministration), patients without coadministration of CCB, and patients with coadministration of sympathoinhibitory CCB (L/T- or L/T/N-type CCB). The significance of differences in each variable among the three groups was tested by one-way analysis of variance, followed by a post hoc Fisher protected least significant difference test to interpret which means are significantly different from each other. A value of p < 0.05 was considered to be significant. Statistical analyses were performed using SPSS Statistics 22 (IBM Corp).
Results
Baseline
At baseline, the 24-hour λ25s in CKD patients was higher compared with that in the control individuals (0.51±0.15 vs 0.41±0.08, p = 0.007). In 20 patients with CKD, daytime λ25s was higher than nighttime λ25s (0.51±0.14 vs 0.38±0.20, p = 0.003). Twenty-four–hour λ25s in CKD patients who had taken L-type CCB before the initiation of ARB treatment (i.e. coadministration of CCB and ARB) (n = 5), in patients without coadministration of CCB (n = 11), and in patients with coadministration of sympathoinhibitory CCB (L/T- or L/T/N-type CCB) (n = 4), were 0.61±0.20, 0.48±0.13, and 0.44±0.06 (p = 0.2), respectively.
Twenty-four–hour λ25s in the three groups was augmented compared with their corresponding controls, but these trends were not significant. Twenty-four–hour λ25s of CKD patients (vs corresponding controls) in patients with coadministration of L-type CCB, without CCB, and with coadministration of sympathoinhibitory CCB, were 0.61±0.20 (vs 0.44±0.13, p = 0.1), 0.48±0.13 (vs 0.40±0.05, p = 0.04), and 0.44±0.06 (vs 0.41±0.06, p = 0.5), respectively.
Table 1 shows daytime and nighttime λ25s in the three groups. Though not significant, daytime λ25s at baseline was higher in patients with coadministration of L-type CCB (0.62±0.18), compared with the patients without CCB treatment (0.49±0.13) and those with coadministration of L/T- or L/T/N-type CCB (0.46±0.06) (Table 1). Nighttime λ25s was comparable in these three groups (coadministration of L-type CCB, 0.39±0.29; without CCB treatment, 0.37±0.19; coadministration of L/T- or L/T/N-type CCB, 0.39±0.10, Table 1).
Daytime and nighttime λ25s in patients with preceding treatment (i.e. coadministration) of L-type CCB, those without CCB, and those with L/T- or L/T/N-type CCB at before and during ARB treatment.

λ25s: non-Gaussianity of heart rate variability index; ARB: angiotensin II type 1 receptor blocker; CCB: calcium channel blocker.
Control, values of corresponding control subject; Change, change from baseline to during ARB treatment.
Difference was significant between corresponding control subject and baseline values (Student t-test).
Difference was significant between baseline and ARB treatment (Student t-test).
Differences were significant among coadministration of L-type CCB, without CCB and L/T- or L/T/N-type CCB groups (one-way analysis of variance).
Effect of ARB
In 20 patients, change in 24-hour, daytime and nighttime λ25s did not show a significant relationship with the change in 24-hour, daytime and nighttime SBP, diastolic BP (DBP) and mean arterial pressure (MAP).
The opposite trend for the change in daytime λ25s between the patients taking L-type CCB and those in the other two groups is noteworthy. In patients who received ARB as an additional treatment to the preceding treatment with L-type CCB, the change in daytime λ25s exhibited a positive relationship with the changes in daytime SBP (r2 = 0.46), daytime DBP (r2 = 0.65) and MAP (r2 = 0.57). To the contrary, inverse relationships between change in daytime λ25s and changes in daytime SBP were noted in the patients without coadministration of CCB (r2 = 0.10) and in those with coadministration of L/T- or L/T/N-CCB (r2 = 0.31), respectively. Inverse relationships between change in nighttime λ25s and change in nighttime SBP were found in all three groups. Based on these findings, we secondly focused on the change in daytime λ25s.
As mentioned above, daytime λ25s was higher in patients with coadministration of L-type CCB, compared with the patients without CCB treatment and those with coadministration of L/T- or L/T/N-type CCB. SBP at baseline in these three groups was 134±11, 143±21 and 156±19 mmHg, respectively (p = 0.2). The reduction in daytime SBP was larger in the L-type CCB coadministration group compared with the other two groups; changes in daytime SBP in patients with L-type CCB coadministration, patients without CCB, and those with sympathoinhibitory CCB coadministration, were ‒13±12, ‒6±6 and ‒7±13 mmHg, respectively. However, a larger decrease in daytime λ25s was shown in patients with the L-type CCB coadministration compared with those in the other two groups (Table1, Figure 1). Accordingly, during the acute phase of ARB treatment, no difference in daytime λ25s was seen among the three groups (0.55±0.20, 0.53±0.16 and 0.43±0.05, respectively, p = 0.5) (Table 1, Figure 1). For reference, changes in 24-hour λ25s in the three groups were ‒0.06±0.09, 0.03±0.07 and ‒0.01±0.009, respectively (p = 0.07); and changes in nighttime λ25s in the three groups were 0.04±0.09, 0.001±0.07 and 0.03±0.02, respectively (p = 0.5).
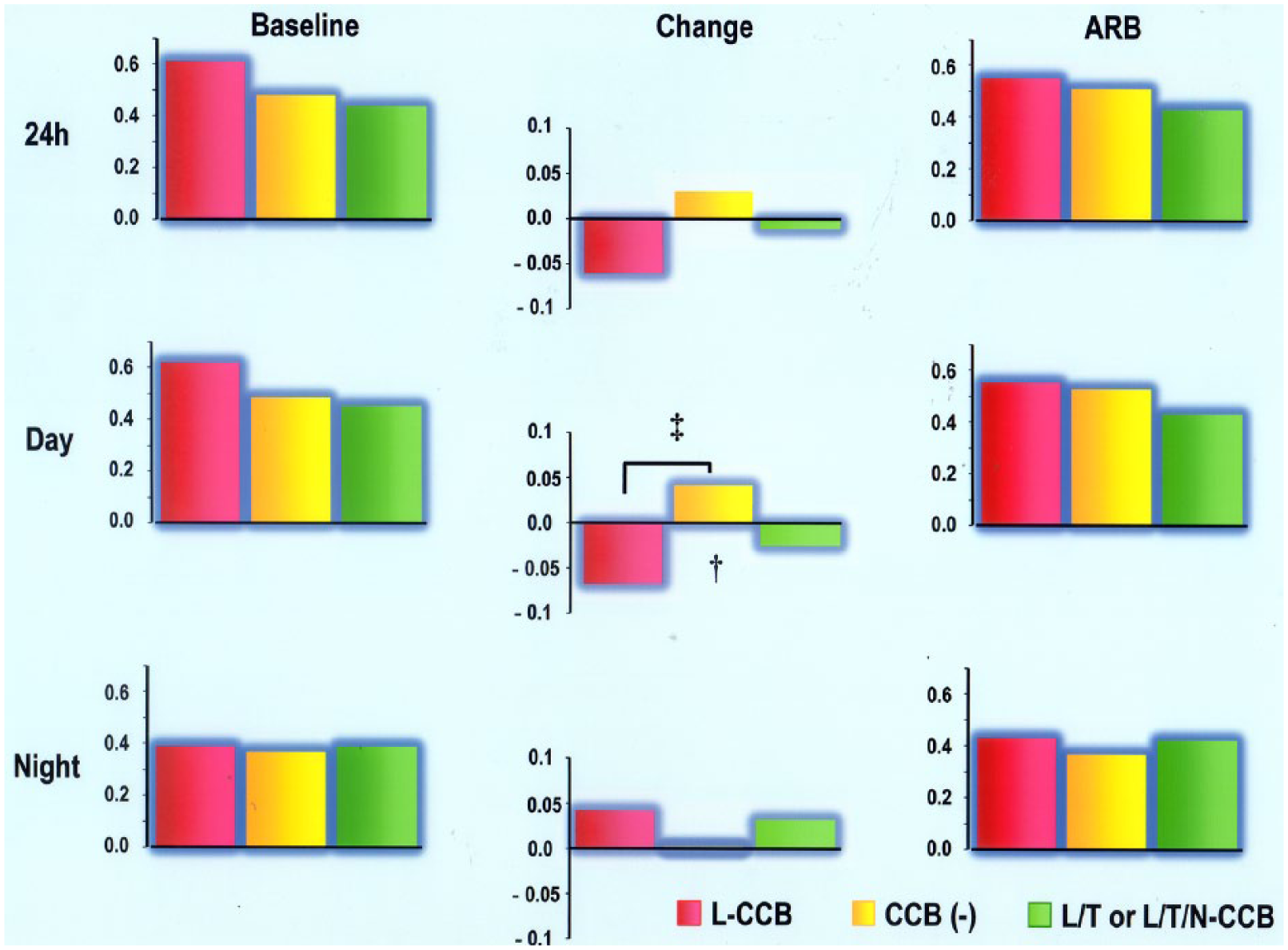
Twenty-four–hour (24h)-, daytime- and nighttime λ25s at baseline and during ARB treatment, as well as change by ARB treatment.
Discussion
ARBs are known to inhibit central and peripheral sympathetic nerve activity. 16 However, an original study of the present subanalysis failed to demonstrate the sympathoinhibitory effect of the ARB azilsartan. 15 We speculated that sympathoinhibitory effect of azilsartan was partially offset by reflex activation of sympathetic nerve activity caused by rapid BP reduction at the initial phase of treatment. The present subanalysis demonstrates inverse relationships between changes in daytime λ25s and the changes in daytime BPs in patients with coadministration of L/T- or L/t/N-CCB and in patients without coadministration of CCB, supporting this speculation. By contrast, in patients with preceding L-type CCB treatment, the change in daytime λ25s exhibited a positive relationship with the changes in daytime BPs. The discrepancy reminds us of the association of the treatment with CCBs and sympathetic nerve activity. Among CCBs, there remains controversy as to whether administration of L-type dihydropyridine CCB, including amlodipine, can induce a reflex sympathetic stimulation. 17 Meanwhile, L/T-type, L/N-type and L/T/N-CCBs are known to suppress sympathetic nerve activity.11–13,18–20 The opposite trend for the change in daytime λ25s between patients taking L-type CCB and others is noteworthy. In the present study, patients with coadministration of L-type-CCB exhibited higher daytime λ25s, but their daytime λ25s decreased to a greater extent during the acute phase of ARB treatment (Figure 1). Combination treatment of ARB and CCBs raises concerns about reflex activation of the sympathetic nerve. However, our study results suggest azilsartan can overwhelm the activation of sympathetic nerve activity stimulated by coadministration of L-type CCBs.
CCBs, as well as diuretics, are recommended as second-line antihypertensive agents. As to renoprotection, in an extension study of African American Study of Kidney Disease and Hypertension, treatment with the L-type CCB amlodipine reduces progression of chronic kidney disease less effectively than angiotensin-converting-enzyme inhibitors (ACEIs) in patients with proteinuria. 21 In turn, the Avoiding Cardiovascular Events through Combination Therapy in Patients Living with Systolic Hypertension (ACCOMPLISH) trial 22 showed that combination treatment with the ACEI benazepril plus the L-type CCB amlodipine prevents progression of CKD (doubling of serum creatinine concentration or end-stage renal disease) more effectively than does benazepril plus a diuretic agent (hydrochlorothiazide) in patients without overt proteinuria (only 5.1% of the participants had albuminuria). In this way, the renoprotective effect of antihypertensive agents differed demending on the presence/absence of proteinuria, which is the indicator of glomerular hypertension, of the study population. As to all-cause mortalities, there is a little concern about a reflex sympathetic stimulation regarding treatment with L-type dihydropyridine CCB. 17 However, our study demonstrated that treatment with ARB can overwhelm the activation of sympathetic nerve activity by coadministration of L-CCB. An increase in λ25s exclusively resulted in higher rates of cardiac mortality, independent of clinical risk factors and other HRVs, in patients with chronic heart failure 9 or with a history of acute myocardial infarction. 10
Our study clarified that combination therapy with ARB and L-type CCB can decrease daytime λ25s in patients with elevated daytime λ25s before the initiation of ARB. The limitation of our study is the considerably small number of each of the three groups to make widespread conclusions. We could not clarify the difference in the λ25s between L/T-CCB and L/T/N-CCB. Another limitation of our study is that we cannot explain the reason why azilsartan can achieve the larger BP reduction with larger suppression of sympathetic nerve activity (λ25s) in patients whose BP was lowered but whose daytime λ25s was elevated by preceding L-type CCB treatment.
In conclusion, azilsartan can overwhelm the activation of sympathetic nerve activity by coadministration of L-type CCBs. Further studies are needed to investigate whether the λ25s can be suppressed during the chronic phase of combination treatment of ARB plus any type of CCB (i.e. L-, L/T-, or L/N, L/T/N-CCB), and whether the reduction in λ25s by combination treatment of ARB and CCBs can be associated with the improvement of prognosis in renal endpoint or cardiac mortality.
Footnotes
Declaration of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
