Abstract
Hypothesis/introduction:
Renal fibrovascular injury often persists in chronic kidney disease patients treated with renin-angiotensin system blockers. Intriguingly, early outgrowth cell-derived factor infusion also inhibits chronic renal injury. We sought to determine whether early outgrowth cell-derived factor administration provides further renoprotection when added to renin-angiotensin system blockade.
Materials and methods:
Conditioned medium was generated by incubating rat early outgrowth cells with serum-free endothelial basal medium-2 to collect their secreted factors. Subtotal nephrectomy rats received enalapril 0.5 mg/L in drinking water or placebo, beginning 8 weeks post-surgery. Four weeks later, enalapril-treated rats received intravenous injections of either conditioned medium or control endothelial basal medium-2 for 2 weeks. Glomerular filtration rate, urinary protein excretion and renal structure were assessed 4 weeks later at 16 weeks post-surgery.
Results:
Enalapril-treated subtotal nephrectomy rats receiving control endothelial basal medium-2 injections experienced only partial renoprotection when compared to vehicle-treated subtotal nephrectomy rats. In contrast, conditioned medium infusion, when administered in addition to enalapril, attenuated the progression of renal dysfunction in subtotal nephrectomy rats, improving glomerular filtration rate and reducing proteinuria without affecting blood pressure.
Conclusions:
Early outgrowth cell-derived factors exert additive renoprotective effects on top of angiotensin-converting enzyme inhibitor therapy in experimental chronic kidney disease, providing the rationale for clinical trials of early outgrowth cell-based therapies for chronic kidney disease.
Keywords
Introduction
Blockade of the renin-angiotensin system (RAS) with either an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB) slows the progression of both non-diabetic and diabetic kidney disease.1,2 Accordingly, their use is indicated in almost all forms of proteinuric chronic kidney disease (CKD). Unfortunately, however, the protection that they afford is incomplete with reductions of 20–30% in the commonly used clinical trial composite endpoint of doubling of serum creatinine, prevention of end-stage renal disease and death.1 -3
The discovery, almost two decades ago, that certain bone marrow-derived cells can assist in tissue repair 4 has led to a broad range of animal and human studies, focusing in particular on the potential of these cells as a new therapeutic strategy for cardiovascular disease. More recently, cell therapy has also been shown to attenuate the structural and functional manifestations of disease in the setting of CKD, ostensibly through their secretion of soluble proangiogenic and antifibrotic factors.5-7 Intriguingly, the conditioned medium (CM) in which the cells had been grown and which contains these factors, was as effective as the cells themselves when administered intravenously, raising the potential for a cell-free approach that obviates many of the difficulties and adverse effects that may be associated with cell therapy.8,9
As patients with CKD often present with advanced disease, we sought to mimic the clinical context by studying animals with advanced disease and declining glomerular filtration rate (GFR). Similarly, given the established though incomplete effects of RAS blockade, we wanted to examine the effects of additional treatment when proteinuria had already been reduced by approximately one third, as in the trials that examined the blood pressure-independent effects of ARBs in individuals with progressive kidney disease.1,3 Finally, given its importance as the key indicator of renal prognosis, the primary aim of the study was to assess whether the secretory products of bone marrow-derived cells, when added to an ACE inhibitor, could improve GFR in an animal model that experiences a progressive decline in filtration rate, wherein the absence of intervention leads to uraemic death.
Methods
SNX animal model
Fischer 344 (F344) rats (Charles River, Montreal, Quebec, Canada) of 8 weeks of age were randomly assigned to one-step subtotal nephrectomy (SNX) (n=33) or sham surgery (n=6), as previously described.5,10 Briefly, animals were anaesthetised with inhaled 2% isofluorane. The right kidney was removed by subcapsular nephrectomy and infarction of approximately two thirds of the left kidney was achieved by selective ligation of two out of the three or four branches of the renal artery. Sham surgery consisted of laparotomy and manipulation of both kidneys before wound closure. All animal studies were approved by the St Michael’s Hospital animal ethics committee.
Enalapril treatment and CM infusion
The study design for the administration of enalapril and CM following subtotal nephrectomy is shown in Figure 1 wherein three groups of SNX animals were studied: vehicle only, enalapril followed by unconditioned medium and enalapril followed by CM.

Study design. Sx: surgery; CM: conditioned medium; EBM-2: endothelial basal medium-2; Wks: weeks.
To allow the animals to develop advanced disease before intervening, animals were randomly assigned to receive enalapril 0.5 mg/L in drinking water or vehicle beginning 8 weeks following SNX surgery. The animals remained on this treatment for a further 8 weeks, until the end of the study (Figure 1). We chose an enalapril dose of 0.5 mg/L because previous studies have demonstrated that this regimen exerts modest renoprotection, akin to the average human response to ACE inhibition.11,12 Four weeks following the initiation of ACE inhibitor therapy, enalapril-treated rats were randomly assigned to receive thrice weekly intravenous injections of 10× concentrated cell-free CM (n=11) as described below, or unconditioned medium (endothelial basal medium-2 (EBM-2); Lonza, Walkersville, MD, USA) (n=13) for 2 weeks (Figure 1). These animals were followed for a further 2 weeks after the final injection of CM, that is, for a total of 4 weeks from the first injection. The controls for this study consisted of sham animals (n=6) and SNX animals treated with vehicle (n=9).
Isolation of early outgrowth cells and cell culture
Early outgrowth cells (EOCs) were cultured as previously described. 13 Briefly, bone marrow cells were flushed from the femora and tibiae of 4-week-old male F344 rats with sterile phosphate-buffered saline. The collected cells were plated in differential endothelial cell culture medium (EGM-2; Lonza) on human fibronectin-coated tissue culture flasks and incubated at 37°C with 5% carbon dioxide for 7–10 days to produce EOCs.
CM generation
EOC CM was generated by incubating subconfluent 10-day cultured EOCs with serum-free EBM-2 (Lonza) for 24 hours after washing of the EOCs to remove any residual serum. 14 Cell free EOC CM and EBM-2 were concentrated 10-fold using a 10 kDa cut off centrifuge filtration column (Millipore, Billerica, MA, USA), followed by filtration using a 0.45 µm filter (Millipore). The 0.5 mL aliquots of concentrated CM and EBM-2 were stored at –80°C until injection.
Renal functional parameters
Body weight was determined weekly
Pre-surgery, and 4, 8, 12 and 16 weeks post-surgery, animals were housed in metabolic cages to collect 24-hour urine samples. Systolic blood pressure was measured in conscious rats using an occlusive tail-cuff plethysmograph (Powerlab, AD Instruments, Colorado Springs, CO, USA). 15 Prior to termination, rats underwent GFR measurement using a modified FITC-inulin plasma clearance assay. 16 Briefly, rats were tail vein injected with 3.74 µL/g body weight of FITC-inulin. Tail vein blood was sampled at various time points post-FITC-inulin injection. The concentration of this agent was then assayed by its fluorescence with a Spectramax M5e microplate reader (Molecular Devices, Sunnyvale, CA, USA) with 485 nm excitation and 527 nm emission settings. GFR was calculated using the following two phase, exponential decay curve using non-linear regression statistics as previously described: 16 GFR = I/(A/α + B/β), where I is the amount of FITC-inulin injected, A and B are the y-intercept values for the two decay rates, and α and β are the decay constants for the distribution and elimination phases.
Tissue preparation and histochemistry
At the end of the study, 16 weeks post-surgery, animals were killed. Kidneys were excised and immersion fixed in 10% neutral buffered formalin, embedded in cryostat matrix (Tissue-Tek, Sakura, Kobe, Japan), and flash frozen in liquid nitrogen. Formalin-fixed kidneys were embedded in paraffin, and sectioned before staining with immunohistochemical stains as described below.
Glomerular endothelial cell density was assessed by immunostaining with the mouse anti-rat monoclonal antibody JG-12 (Bender Medsystems, Atlanta, GA, USA)), which binds to endothelial cells of blood vessels but not to lymphatics in rat kidney. 16 Glomerular capillary density was quantified as the percentage positive area for JG-12 staining in 30 randomly selected glomeruli from each rat, as previously described. 15 Glomerulosclerosis and tubulointerstitial fibrosis were assessed by examining the accumulation of type IV collagen in 30 randomly selected glomeruli and six random non-overlapping 20× fields, respectively, for each animal following immunostaining with type IV collagen polyclonal antibody (Southern Biotech, Birmingham, AL, USA). The assessment of renal structure for the above stains was done in a blinded fashion using computer assisted image analysis, as described previously. 17
Statistical analysis
All data are shown as mean ± SEM unless otherwise stated. Differences between groups were analyaed by analysis of variance with a post-hoc Fisher’s protected least significant difference test. All statistics were performed using GraphPad Prism 6.00 for Mac OS X. A change was considered statistically significant if P<0.05.
Results
Kidney function
After 4 weeks of enalapril treatment (12 weeks post-nephrectomy), SNX rats showed an approximate 30% reduction in proteinuria when compared with untreated animals (89±30 vs. 117±20 mg/day). At this stage, enalapril-treated rats were randomly assigned once more to receive either CM or unconditioned medium and studied for another 4 weeks. At the end of the study, now 16 weeks after surgery, SNX animals displayed heavy proteinuria, hypertension and a substantial reduction in GFR when compared with sham-operated animals. Two animals treated with enalapril and subsequent unconditioned medium died between 12 and 16 weeks post subtotal nephrectomy with no deaths noted within the other two groups.
When compared with untreated SNX rats, urinary protein excretion was reduced in animals that received enalapril followed by the addition of CM, while those treated with enalapril followed by unconditioned medium had an intermediate effect (Figure 2). GFR was higher in enalapril plus CM treated animals while, consistent with the original studies by Anderson et al., 18 enalapril treatment alone did not attenuate GFR loss in SNX rats (Figure 3). Blood pressure was elevated in SNX rats and unaffected by enalapril alone or in combination with either EOC CM or unconditioned medium (Figure 4).
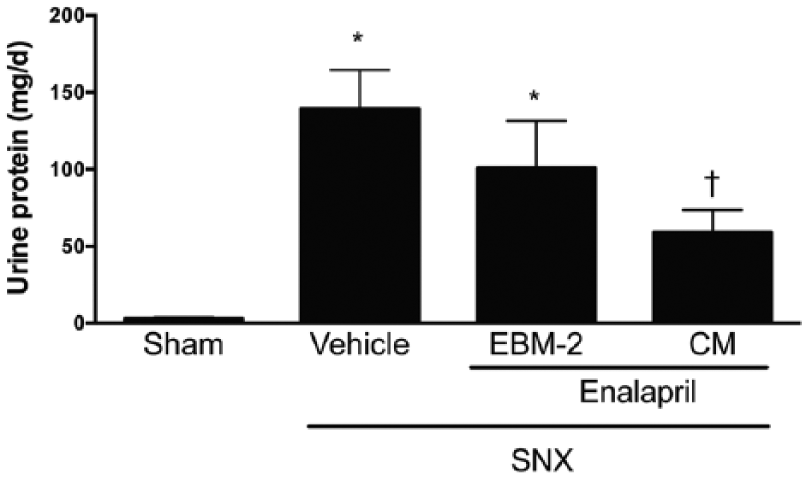
The addition of CM to enalapril provides additive anti-proteinuric protection when used as an add-on therapy for the treatment of late-stage experimental CKD. SNX animals treated with both enalapril and CM excreted significantly lower amounts of protein than vehicle-treated SNX animals. *P<0.05 vs. sham animals; †P<0.05 vs. SNX – vehicle treated animals. CM: conditioned medium; CKD: chronic kidney disease; SNX: subtotal nephrectomy.

The addition of CM to enalapril ameliorates GFR. While low dose enalapril did not attenuate the reduced GFR observed in late-stage SNX animals, the combination of enalapril and CM significantly attenuated GFR loss. *P<0.05 vs. sham animals; †P<0.05 vs. SNX – vehicle treated animals; #P<0.05 vs. SNX – enalapril/EBM-2 treated animals. CM: conditioned medium; GFR: glomerular filtration rate; SNX: subtotal nephrectomy; EBM-2: endothelial basal medium-2.

Neither low dose enalapril, nor the combination of enalapril and CM, significantly lower systolic blood pressure when used to treat late stage experimental CKD. Blood pressure was measured by conscious tail cuff plethysmography. *P<0.05 vs. sham animals. CM: conditioned medium; CKD: chronic kidney disease.
Kidney structure
SNX rats developed marked glomerulosclerosis as indicated by extensive deposition of type IV collagen within the glomerulus. These fibrotic changes were accompanied by glomerular capillary rarefaction as evidenced by the loss of glomerular endothelial JG-12 immunostaining. When compared with untreated animals, those that received enalapril followed by CM showed substantial attenuation in type IV collagen accumulation and glomerular endothelial cell loss. Rats treated with enalapril followed by unconditioned medium had effects on glomerular fibrosis (Figure 5) and capillary rarefaction (Figure 6) that were intermediate between those that had received vehicle only and those that had been treated with enalapril and CM.

The addition of CM as an add-on therapy to low dose enalapril attenuates glomerulosclerosis when used in the treatment of late stage experimental CKD. Kidney sections were immunolabelled with a type IV collagen antibody. (a) Sham; (b) SNX – vehicle; (c) SNX – enalapril/EBM-2; (d) SNX – enalapril/CM; (e) glomerular collagen type-IV positivity. Original magnification 400×. Quantification is presented following sampling of 30 randomly selected glomeruli cut through the macula densa per kidney section. *P<0.05 vs. sham animals; †P<0.05 vs. SNX – vehicle treated animals; #P<0.05 vs. SNX – enalapril/EBM-2 treated animals. CM: conditioned medium; CKD: chronic kidney disease; SNX: subtotal nephrectomy; EBM-2: endothelial basal medium-2.

The addition of CM as an add-on therapy to low dose enalapril preserves glomerular endothelial cell density when used for the treatment of late stage experimental CKD. Kidney sections were immunolabelled with JG-12, an antibody that binds to glomerular endothelial cells. (a) Sham; (b) SNX – vehicle; (c) SNX – enalapril/EBM-2; (d) SNX – enalapril/CM; (e) glomerular JG-12 positivity. Original magnification 400×. Quantification is presented following sampling of 30 randomly selected glomeruli cut through the macula densa per kidney section. *P<0.05 vs. sham animals; †P<0.05 vs. SNX – vehicle treated animals. CM: conditioned medium; CKD: chronic kidney disease; SNX: subtotal nephrectomy; EBM-2: endothelial basal medium-2.
Discussion
The use of agents that block the RAS in conjunction with control of hypertension are key factors in reducing the progression of CKD. While effective, the majority of patients treated with these agents continue to experience a decline in GFR such that the need for additional agents is readily apparent. However, in contrast to the clinical setting, most preclinical studies examine new therapies as single agents. Moreover, unlike in humans with CKD, these new agents are typically administered as a preventive strategy to animal models that develop proteinuria but frequently do not experience progressive GFR loss. Here we show that culture medium conditioned with the secretory products of bone marrow-derived cells, when given as add-on therapy to an ACE inhibitor to animals late in the course of their kidney disease, improves GFR, reduces proteinuria and attenuates the fibrosis and capillary loss that characterise CKD.
In general, therapeutic interventions seem to work best early in the course of a disease, before pathological changes become severe and only slowly, if at all, reversible. Regrettably, most patients with CKD present late in the course of their disease, at a time when substantial GFR loss has already occurred. Such patients will invariably receive treatment with an ACE inhibitor or ARB, so that the efficacy of any new treatment would need to be assessed as an addition rather than an alternative to blockade of the RAS. This preclinical study sought to mimic that scenario.
With a prolonged but defined natural history and pathophysiology, the SNX rat provides a well-characterised model of progressive non-immune mediated glomerulosclerosis that ultimately leads to renal failure and death. 19 As such, this model permits the assessment of both preventive and late intervention strategies. In the present study, we explored the efficacy of CM as a potential add-on therapy to RAS blockade. Consistent with the response to ACE inhibition in humans with CKD,1,3,20 we first induced a 30–50% reduction in proteinuria as might be seen in the clinical setting, while concomitantly providing renal protection but incompletely so. The amount of administered drug was based on previously published dose–response relationships. 11 This low dose of enalapril that was used further permitted the assessment of ACE inhibition without the confounding effects of blood pressure reduction.
Despite two decades of research, the mechanisms by which bone marrow-derived cells induce their beneficial effects remain incompletely understood. While opinion favours a paracrine mechanism of action, 21 the demonstration of tissue repair at distances that are substantially greater than what would conventionally be considered paracrine 22 suggests that the issue may be more complex. From a therapeutic development perspective, the potency of the bone marrow-derived cells’ secretory products effects is notable whereby dilutions of almost 1:2000 were required before the IC50 was reached. 23 Furthermore, while these secretory products have a limited duration of action in their treatment of preclinical CKD, repeated administration of these factors has been shown to provide prolonged structural and functional preservation of the kidney. 24 These findings, along with their ability to replicate the effects of the cells themselves,22,23 raise the possibility of a cell free regenerative therapy that could be given repeatedly, throughout the course of CKD, to augment the effects of RAS blockade.
Following reduction in proteinuria with ACE inhibition, SNX rats were then administered the CM in which bone marrow-derived EOCs had been cultured. Like CKD in humans, the SNX rat develops progressive glomerulosclerosis and capillary rarefaction, enabling the assessment of kidney structure as well as function. With previous studies having shown that CM exerts both proangiogenic and antifibrotic effects, 23 we examined the impact of intervention on the extent of glomerulosclerosis and capillary endothelial cell loss, showing that both were substantially attenuated by a combination of CM and ACE inhibition, while ACE inhibition alone had an intermediate effect. Similar results were obtained for proteinuria and GFR in which the combination of enalapril and CM provided better renoprotection.
While the present study attempted to mimic the human CKD setting, the similarities are incomplete. To reduce proteinuria by approximately 30%, as observed in the clinical trial setting,1,3 SNX animals required only very low doses of the ACE inhibitor, enalapril. It is certainly possible that had we used higher doses, we may have seen additional renoprotective effects in the animal model. And while humans do respond to higher than usual doses of RAS blockade with further reductions in proteinuria, these are notably not accompanied by improvements in GFR, 25 the primary outcome of this study. It is also likely that doses of enalapril higher than the one used in the present study would have reduced blood pressure, the control of which is a key factor in CKD management. We sought, however, to reduce proteinuria by only 30% and to do this independently of the confounding effects of blood pressure reduction.
Conclusions
In summary, in a scenario that is somewhat reminiscent of the clinical CKD context, the administration of cell free CM derived from early outgrowth bone marrow cells, when added as a late intervention to ACE inhibition, further attenuated the structural and functional manifestations of the disease.
Footnotes
Acknowledgements
The authors would like to thanks Mrs Christine Kuliszewski, Ms Bailey Stead and Ms Melissa Mitchell for their technical support of these animal studies. The authors would also like to express their gratitude to Suzanne Advani for her technical assistance and guidance throughout the operation of these studies.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These data were presented at the 2011 Annual Scientific Meeting of the American Society of Nephrology, and was supported by grants from the CIHR (CCT 83030, MOP 97912) and Physicians’ Services Incorporated Foundation (PSI 09-42). Dr Yuen is currently sponsored by a KRESCENT new investigator and Canadian Diabetes Association clinician scientist award. David Kepecs was supported by an Ontario graduate scholarship. Dr Kim Connelly is supported by a CIHR new investigator award. Dr Richard Gilbert is the Canada research chair in diabetes complications and this research was supported in part by the Canada Research Chair Program.
