Abstract
Objective:
The study aimed to evaluate the effects of oral administration of irbesartan in adriamycin-induced nephropathy considering laboratory changes, kidney histology, and expression of proteins related to slit diaphragm and cytoskeleton of the podocyte.
Methods:
The animals were divided into control, model, methylprednisolone (MP), and irbesartan groups. The 24-hour urinary protein and biochemical indicators were determined, and renal pathological changes were observed. The mRNA and protein expression of nephrin, podocin, CD2-associated protein (CD2AP), and desmin in the kidney tissue were analyzed.
Results:
The urinary protein excretion levels in the MP and irbesartan groups were lower than those in the model group (
Conclusions:
For rats with adriamycin-induced nephropathy, irbesartan could significantly reduce proteinuria. As a possible mechanism, irbesartan may improve the slit diaphragm protein of the glomerular podocyte and stabilize the cytoskeleton of the podocyte.
Introduction
Studies have found that the renin–angiotensin–aldosterone system (RAS) is closely related to the occurrence of proteinuria.1,2 Inhibition of angiotensin-converting enzyme (ACE) or blocking of angiotensin II (AngII) can reduce proteinuria and protect renal function. 3 It has been shown that podocytes play an important role in maintaining the normal structure and function of the glomerular filtration barrier. 4 The slit diaphragm in the foot processes of podocytes is the most important component comprising the integrated glomerular filtration membrane. 5 If the slit diaphragm is destroyed, small protein molecules will be filtered off, causing proteinuria. 6 The cytoskeleton structure of podocytes is the basis for the normal function of podocytes, which maintain the normal glomerular filtration rate by self-adjusting and adapting to external changes. 7 Under pathological conditions, the cytoskeleton structure of podocytes is not able to adaptively change, resulting in podocyte damage and severe proteinuria. 8 As reported in the literature, morphological changes in podocytes and damage to the slit diaphragm are involved in the pathogenesis of diabetic nephropathy, and RAS blockade may protect the podocytes in diabetic nephropathy and maintain the normal function of the slit diaphragm.9,10 This study used the adriamycin nephropathy animal model, which might induce glomerular permeability changes and proteinuria as observed in human primary nephrotic syndrome.11,12 The study aimed to evaluate the effects of oral administration of irbesartan in adriamycin-induced nephropathy considering laboratory changes, kidney histology and expression of proteins related to slit diaphragm and cytoskeleton of the podocyte.
Materials and methods
Materials
Experimental animals
Fifty healthy and specific-pathogen-free male Wistar rats, weighing 180–200 g, were purchased from Beijing Vital River Laboratory Animal Technology Co. Ltd, China under license number SCK (Beijing) 2012-0001. The rats were adaptively reared in a clean animal room of the Experimental Animal Center in the People’s Liberation Army General Hospital for one week, with free access to water and a normal diet. All operations in the animal experiments were performed in accordance with the protocols and related recommendations for experimental animals specified by the Laboratory Animal Commission’s Liberation Army General Hospital.
Drugs
Methylprednisolone (MP) was purchased from Pfizer, Italy. Adriamycin hydrochloride was purchased from Sigma, USA, and irbesartan was purchased from Sanofi Pharma, France.
Equipment and reagents
Nephrin primary antibody, podocin primary antibody, CD2-associated protein (CD2AP) primary antibody, and desmin primary antibody were purchased from Abcam, UK. Goat anti-rabbit antibody was purchased from Beyotime Biotechnology Corporation, China, and β-actin antibody was purchased from Santa Cruz Biotechnology, Inc., USA. Trizol reagent was purchased from Ambion Corporation, China. Nephrin, podocin, CD2AP, and desmin Taqman were purchased from ABI, USA. An EasyScript First-Strand cDNA Synthesis SuperMix kit was purchased from TransGen Biotech, China.
Electrophoresis apparatus (DYCP-40) was purchased from the Beijing Liuyi Instrument factory. A Hitachi H-600 transmission electron microscope was obtained from Hitachi. A Mindary BS480 analyzer and a COBAS701 biochemical analyzer were used. An ABI 7900HT high-throughput real-time fluorescence quantitative real-time PCR (qPCR) system (Applied Biosystems, USA) was used in this study.
Methods
Preparation of the animal model
Rat modeling was initiated after adaptive rearing with free access to water and diet for seven days. From a group of 50 rats, 10 were randomly selected for the control group, and the remaining 40 rats were administered a single injection of doxorubicin hydrochloride (5 mg/kg) to prepare the adriamycin-induced nephropathy model according to the protocol of Sun et al. 13 The rats in the control group were administered an intravenous injection of saline (5 ml/kg). Two weeks after the modeling, 24-hour urinary protein excretion was determined, and 24-hour urinary protein excretion greater than 50 mg was indicative of successful modeling. The modeling was successful in 34 rats, of which 30 rats were randomly selected and divided into three groups of 10 rats: model, MP, and irbesartan groups.
Drug intervention program
Gavage was performed for the rats in the four groups for 12 weeks after successful modeling. The control and model groups received saline at 5 ml/kg/day. The MP group received MP dissolved in saline at 20 mg/kg/day according to method of Ren et al. 14 The irbesartan group received irbesartan dissolved in saline at 50 mg/kg/day according to method of Dai et al. 15
Detection of the indicators
Tests of urine and blood biochemistry
The 24-hour urine excretion and urine volume of the rats was collected before modeling and at the end of the 4th, 8th, and 12th weeks after intervention. Determination of 24-hour protein was by the biuret method. At the end of the 12th week after intervention, the rats were sacrificed, and inferior vena cava blood was collected to detect the serum creatinine (Scr), blood urea nitrogen (BUN), albumin (ALB), total cholesterol (CHO), triglyceride (TG), and uric acid (UA) levels using a COBAS701 biochemical analyzer.
Renal pathology examined by optical microscopy
After the kidney tissue of the rats was conventionally embedded in paraffin, sectioning and trimming were performed parallel to the minor axis of the kidney crossing the renal hilum, with a slice thickness of 4 µm. The sectioned slice containing the entire kidney was selected for periodic acid-Schiff staining, and pathological changes in the renal tissue were observed using an optical microscope. The standards of renal pathology score evaluation were: (a) glomerulosclerosis score, 0 (normal glomerulus), 1 (mesangial expansion or hardened area of less than 25%), 2 (hardened area of 25%~50%), 3 (hardened area of 50%~75%), 4 (hardened area of more than 75%). Each specimen was placed under the light microscope observation at ×400, using 50 glomeruli to compute the average as the glomerulosclerosis score; (b) renal tubular integral, according to the extent of disease (renal tubular atrophy, protein casts, interstitial inflammation and fibrosis), respectively to scores between 0~3: 0 (no tubulointerstitial damage), 1 (lesion range of less than 25%), 2 (lesion extent of 25%~50%), 3 (lesion range of more than 50%). Each specimen was observed using 10 fields of view at 100× magnification under the light microscope and the average number of renal interstitial tubular lesions was obtained.
Electron microscopy
The renal cortex was treated with phosphate buffer containing 3% glutaraldehyde and 0.22 mmol/l sucrose (pH 7.2) and fixed with 1% osmium tetroxide. Samples were then dehydrated using an ethanol gradient followed by epoxy resin embedding. A Hitachi H-600 transmission electron microscope was used to observe pathological changes in the renal ultrastructure.
Real-time PCR quantitative analysis
The total RNA was extracted from the renal cortex using the TRIzol method. The concentration of RNA was determined by an absorbance at 260 nm and the purity of the RNA was evaluated by measuring the A260/A280 ratio. The reverse transcription system included 2×ES Reaction Mix (10 μl), Enzyme Mix (1 μl), oligo dT primer (1 μl), RNA template, and RNase-free water to a final volume of 20 μl. The reaction conditions were as follows: 37°C for 30 min, and then 85°C for 5 min. Real-time PCR was performed using a Taqman probe. Real-time quantitative PCR was performed in a total volume of 20 μl including cDNA sample (4 μl), Master Mix (10 μl), distilled deionized water (5 μl), and Taqman probe (1 μl). The real-time quantitative PCR used ABI 7900HT (Applied Biosystems, USA) and the cycling program was set at one cycle of predenaturation at 93°C for 2 min, and then 40 cycles at 93°C for 30 s, 60°C for 1 min, 72°C for 30 s, followed by one cycle at 72°C for 5 min. All real-time quantitative PCRs were performed in strict accordance with the guidelines of Minimum Information for Publication of Quantitative Real-Time PCR Experiments (MIQE). Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) was used as an internal reference.
Western blot analysis
Western blot assays were used to analyze the protein expression of nephrin, podocin, CD2PA, and desmin. Total protein was extracted from the renal cortical tissue using radio-immunoprecipitation assay (RIPA) protein extraction buffer (containing protease inhibitor phenylmethanesulfonyl fluoride (PMSF)), and the protein concentration was determined using the bicinchoninic acid (BCA) method. Total protein (100 μg) was loaded into each well for sodium dodecyl sulfate polyacrylamide gel electropheresis (SDS-PAGE) electrophoresis. Proteins were then transferred onto a polyvinylidene fluoride (PVDF) membrane using a wet method at a constant current (100 mA), followed by incubation with nephrin (1:2500), podocin (1:2000) CD2PA (1:1000), desmin (1:500), and β-actin (1:1000) primary antibodies overnight at 4°C. After washing with TBST, the membrane was incubated with secondary antibody at room temperature for 60 min, followed by washing in Tris-Buffered Saline and Tween 20 (TBST). After gradient exposure in the dark, the results were analyzed using ImageJ software. β-Actin was used as an internal reference.
Statistical methods
SPSS 17.0 software was used for statistical analysis. The measurement data are represented as
Results
Changes in body weight
Compared with the control group, the body weight of the rats in the model group increased at a slower rate and was significantly decreased after the intervention for 12 weeks, which was statistically significant (

Comparisons of body weight for all groups. The double asterisk sign designates significant differences. **
Quantitation of 24-hour urinary protein and urine volume
The 24-hour urinary protein excretion prior to modeling of the rats in all groups showed the same baseline level. At two weeks after modeling, with the exception of the control group, the levels of urinary protein excretion of rats in all other groups were increased. At four weeks after drug intervention, the level of urinary protein excretion of the rats in the MP group was lower than that of the model group (
Comparisons of 24-hour urinary protein for all groups (mg/24 h, mean±standard deviation (SD)).
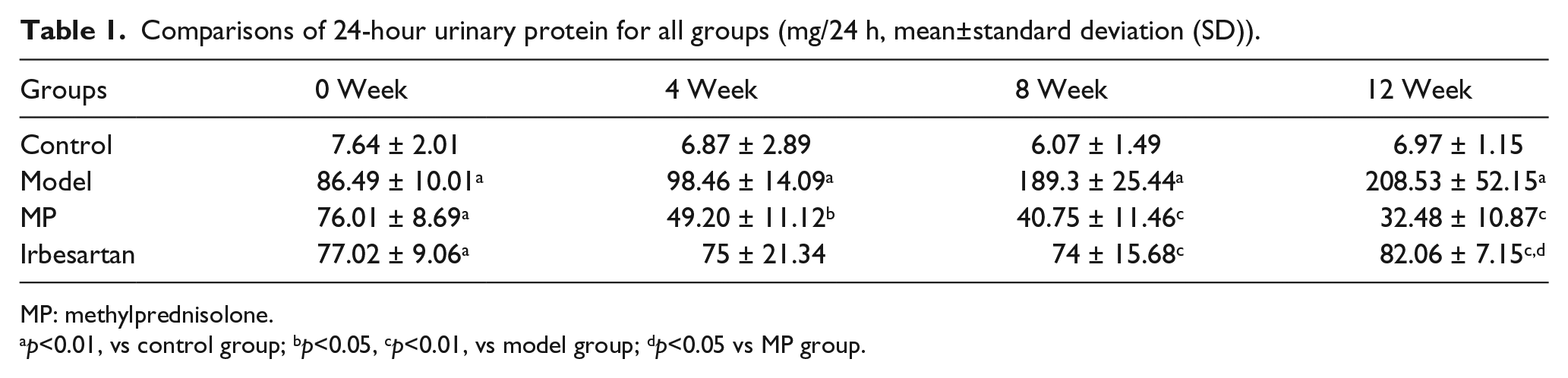
MP: methylprednisolone.
At four weeks after drug intervention, the level of urine volume of the rats in the model group was lower than that of the control group (
Comparisons of urine volume for all groups (ml, mean±standard deviation (SD)).

MP: methylprednisolone.
Serum ALB, CHO, TG, BUN, Scr, and UA levels at the end of the 12th week
Compared with the control group, the levels of CHO (
Comparisons of serum albumin (ALB), serum total cholesterol (CHOL), serum triglycerides (TRIG), serum creatinine (Scr), blood urea nitrogen (BUN), uric acid (UA) for all groups (mean±standard deviation (SD).

ALB: serum albumin; CHOL: serm total cholesterol; MP: methylprednisolone; TRIG: serum triglycerides; Scr: serum creatinine BUN: blood urea nitrogen; UA: uric acid.
The pathomorphological changes in the renal tissue
The kidney of the rats in the normal control group showed no pathological changes. Mesangial cells and matrix showed a mild increase in focal and segmental parts, but necrosis, crescents, and sclerosis were not observed in the kidney in the model group. The renal tubular epithelial cells showed multifocal granular and vacuolar degeneration but did not show any significant tubular atrophy. The cells occasionally showed protein casts. The renal interstitium showed inflammatory cell infiltration simultaneously with a small amount of fibrous tissue hyperplasia. Compared with the model group, the MP and the irbesartan groups all showed an improvement in the pathological changes in the renal tissue and reduction in the inflammatory cell infiltration in the renal interstitium (Figure 2.)

Renal tissues viewed under a light microscope (periodic acid–Schiff (PAS) stain, ×400). (a) control group, no pathological changes; (b) model group, mesangial cells and matrix showed a mild increase, protein casts, the renal interstitium showed inflammatory cell infiltration simultaneously with a small amount of fibrous tissue hyperplasia; (c) methylprednisolone (MP) group, an improvement in the pathological changes in the renal tissue compared with the model group; (d) irbesartan group, an improvement in the pathological changes in the renal tissue compared with the model group.
The level of glomerulosclerosis score and renal tubular integral of the rats in the model group was higher than that of the control group (
Comparisons of renal pathology score for all groups.
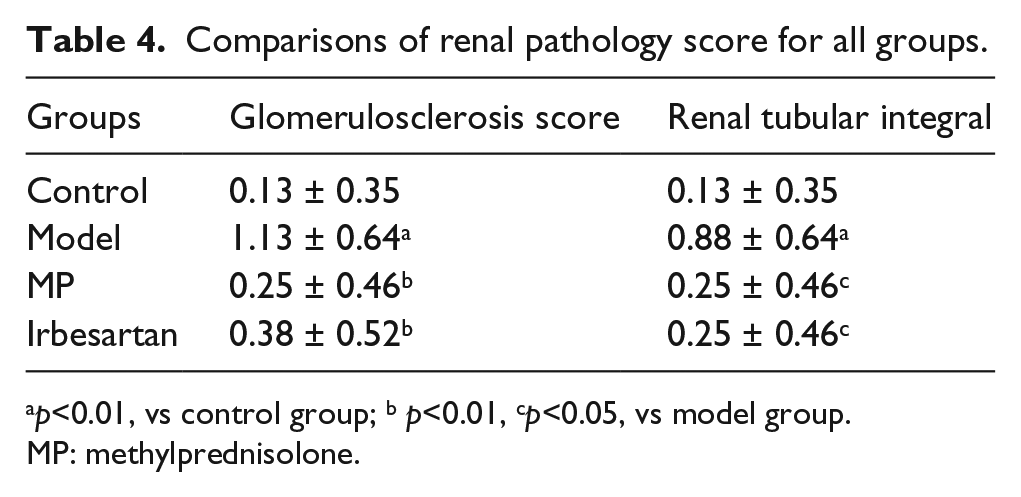
MP: methylprednisolone.
Observation of the pathological changes in renal ultrastructure of the rats in each group by transmission electron microscopy
In the control group, the glomerular basement membrane (GBM) was smooth and even with no thickening, and the foot processes were clear, intact and orderly arranged with no fusion. In the model group, the GBM showed occasional thickening, with diffusive fusion and a disappearance of the foot processes. These findings were significantly different from those of the control group, indicating that the modeling was successful. The foot processes of the rats in the MP group were clear, similar to the normal control group. The GBM of the rats in the irbesartan group was mostly smooth and even, with significantly reduced foot process fusion (Figure 3)

Morphological changes in kidney podocytes under electron microscopy (×6000). (a) control group, foot processes were clear; (b) model group, fusion and a disappearance of the foot processes; (c) methylprednisolone (MP) group, the foot processes were clear, similar to the normal control group; (d) irbesartan group, with significantly reduced foot process fusion.
The expression of nephrin, podocin, CD2AP, and desmin RNA in renal tissues
The genes encoding nephrin and podocin are two important functional genes of podocytes, and these genes have been confirmed to have an important role in the occurrence of proteinuria. At 12 weeks after the intervention, the expression levels of nephrin and podocin mRNA in the renal tissues of the rats in the model group were significantly lower than those of the control group (
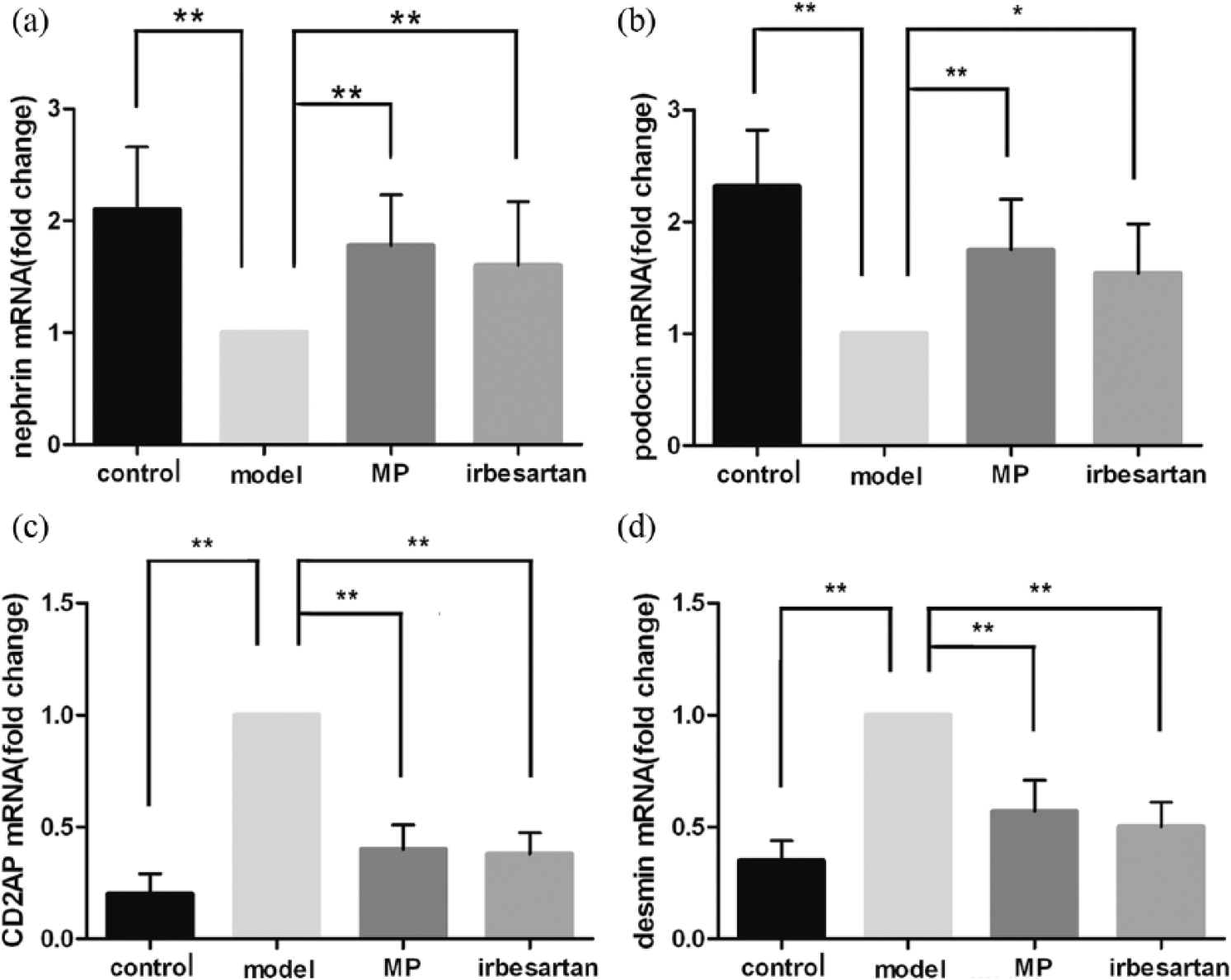
Bar graphs representing the nephrin, podocin, CD2-associated protein (CD2AP) and desmin mRNA levels in the kidney tissue. Asterisks designate significant differences. *
The expression of nephrin, podocin, CD2AP, and desmin proteins in renal tissues
The expression level of nephrin protein in renal tissue at 12 weeks after modeling in the model group was significantly lower than that of the control group (

Western blot analysis for nephrin, podocin, CD2-associated protein (CD2AP), desmin and β-actin protein in renal tissue.

Bar graphs representing nephrin, podocin, CD2-associated protein (CD2AP) and desmin expressions relative densities to β-actin. Asterisks designate significant differences. *
Discussion
In recent years, there has been progress in the study of AngII receptor blockers (ARBs) in protecting podocyte injury in diabetic nephropathy.16,17 RAS activation can lead to lesions in diabetic podocytes, which would directly mediate changes in the expression and distribution of podocyte-associated proteins by AngII. AngII can also promote the apoptosis of podocytes and damage the charge barrier of the GBM. Therefore, RAS blockers can promote the repair of the podocyte damage and reduce the level of urinary protein excretion, thereby delaying the progression of diabetic nephropathy. 18 These findings suggest that RAS blockers have a protective effect on the podocytes in diabetic nephropathy. AngII signaling and its reactivation induces apoptosis in podocytes through control of the Hippo pathway. 19 AngII induces podocyte dysmotility via the protein kinase G-mediated downregulation of transient receptor potential canonical 6 (TRPC6) activity. 20 However, whether they have a protective effect on primary nephrotic syndromes, such as focal segmental glomerulosclerosis (FSGS), which is the nephrotic syndrome characterized with podocyte injury, has not been widely reported. In this study, a classic model of adriamycin-induced nephropathy, with clinical pathological changes similar to minimal change nephritic syndrome in humans, 21 was applied to investigate the therapeutic effect and mechanism of irbesartan on rats with adriamycin-induced nephropathy, with MP as a positive control.
The results showed that in the model group, urinary protein excretion was significantly increased, serum ALB was decreased, and blood lipids were elevated, which is consistent with the manifestations of nephrotic syndrome. Observation using electron microscopy revealed an obvious fusion of the foot processes in the glomerular podocytes, suggesting ideal modeling.
The results after intervention showed that compared with the model group, treatment with irbesartan for eight weeks could significantly reduce the 24-hour urinary protein excretion and elevated the serum ALB in the rats, whereas treatment for 12 weeks was more effective, with CHO and TG levels similar to normal levels. The observation of renal tissues by electron microscopy showed that the partial fusion of the foot processes in the podocytes in the treatment group was less than that of the rats in the model group, indicating that irbesartan had a protective effect on the podocytes of the rats with adriamycin-induced nephropathy. However, the effects of reducing urinary protein excretion and protecting the podocytes were slightly weaker than those in the MP group.
The occurrence of proteinuria is closely related to damage to the slit diaphragm of podocytes. The slit diaphragm consists of multiple protein molecules including podocin, nephrin, and CD2AP. 5 Nephrin is a transmembrane protein that is specifically expressed in the slit diaphragm, which is the main component of the slit diaphragm protein complex and the first discovered slit diaphragm protein. 22 Podocin is also a transmembrane protein, which is a member of the stomatin family. Podocin can interact with nephrin and the intracellular domain of CD2AP through its carboxyl terminus, thereby promoting or amplifying the signal transduction induced by nephrin.23,24 The results of this study showed that compared with the normal control group, the mRNA and protein expression levels of podocin and nephrin in the model group were significantly decreased, suggesting that podocin and nephrin play an important role in the occurrence of nephrotic syndrome and proteinuria, which is consistent with reported findings in literature. Compared with the model group, the mRNA and protein expression levels of podocin and nephrin in the irbesartan group were increased to varying degrees, and expression levels were similar to those of the MP group. These results suggest that irbesartan can stabilize the nephrin and podocin proteins in the slit diaphragm, maintain the glomerular filtration barrier, and reduce urinary protein leakage.
In recent years, the relationship between the cytoskeleton and the slit diaphragm proteins in podocytes, which play an important role in stabilizing the cell structure and preventing the occurrence of proteinuria, have drawn considerable attention. Studies have shown that CD2AP is one of the podocyte cytoskeleton-associated proteins and is involved in the construction of the cytoskeleton and signal transduction of podocytes. CD2AP connects the slit diaphragm of the podocytes and the cytoskeleton in the foot processes to play the role of an adapter. An absence of CD2AP in podocytes may weaken the start and the end of the signaling pathway mediated by tyrosine kinase receptor. 25 Desmin is a cytoskeletal intermediate filament protein, which previously often served as one of the markers of myogenic cells. Under normal circumstances, desmin can be occasionally expressed in glomerular mesangial cells. If podocyte injury is caused by certain kidney diseases, the composition of the intermediate filament protein may change. Desmin expression may be significantly up-regulated, and this change in the expression is more sensitive than the observed change in the ultrastructure of the podocytes. Therefore, abnormal expression of desmin can be used as an indicator of podocyte injury. 26 This study found that rats with adriamycin-induced nephropathy not only had extensive proteinuria but also showed up-regulated expression of the podocyte cytoskeletal protein CD2AP and intermediate filament protein desmin. After treatment with irbesartan, the mRNA and protein expression levels of CD2AP and desmin were significantly down-regulated, suggesting that the therapeutic effect of irbesartan on adriamycin-induced nephropathy is related to changes in the expression of cytoskeletal proteins and genes, which may be one of the additional mechanisms of its protective effect.
Pereira et al. have shown that a cell migration profile involving phagocytosis and cytotoxicity at different times in the progression of doxorubicin-induced nephropathy. The intense activation of monocytes is a potential role in pathogenesis of kidney damage. 27 Losartan treatment was the reduction of levels of renal transforming growth factor-β (TGF-β) in adriamycin-induced nephropathy. TGF-β can be synthesized by numerous cells including macrophages, T and B lymphocytes fibroblasts, and resident renal cells. 28 Accordingly, the angiotensin type 1 (AT1) receptor antagonist plays an important role in anti-inflammation processes. Studies suggest that, in addition to AngII, Ang-(1–7) also plays a key role in regulating renal function by acting at glomerular.29,30 Exogenous activation of the angiotensin-(1-7) Mas receptor protects from adriamycin-induced nephropathy and contributes to the beneficial effects of AT1 receptor blockade. 28
In this study, an investigation of the expression levels of podocin, nephrin, CD2AP, and desmin in rats with adriamycin-induced nephropathy confirmed that these are important proteins in maintaining the structural integrity and normal functions of podocytes. For rats with adriamycin-induced nephropathy, irbesartan could significantly reduce proteinuria, improve serum albumin levels, and lower blood lipid levels. As a possible mechanism, irbesartan may improve the slit diaphragm protein of the glomerular podocyte and stabilize the cytoskeleton of the podocyte, and thereby promote repair of the damage in the glomerular podocytes. Our research results provide experimental evidence for the clinical treatment of nephrotic syndrome using irbesartan. However, the effect and mechanism of irbesartan on signal transduction between podocytes and cytoskeletal proteins and the slit diaphragm -related proteins as well as the internal regulation await further study. Besides, the effect anti-inflammatory of irbesartan was not studied. Therefore, we will continue to investigate further.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Sciences Foundation of China (grant numbers 81273968 and 81471027), Ministerial Projects of the National Working Commission on Aging (grant number QLB2014W002), and The Four Hundred Project of 301 (grant number YS201408)
