Abstract
Hypothesis/Introduction:
Polymorphisms of the angiotensin converting enzyme (ACE) gene can interfere with exercise-induced acute blood pressure (BP) reduction. This cross-over study investigated the acute effect of a single walk on BP and tested whether polymorphisms of the ACE gene might explain the variation in BP responses.
Materials and methods:
Thirty-four healthy medicated individuals were randomized to one control and one walking session at 60–75% of heart rate reserve. Subjects left the laboratory wearing an ambulatory BP monitor until waking the next morning.
Results:
Overall, systolic BP was somewhat lower following the walking session (p=.06), which could be attributed to a consistently lower systolic BP for 5 h after exercise (p-interaction<.04) compared with control rest. Similarly, II/ID individuals had a lower systolic BP (p-interaction=.02) and diastolic BP (p-interaction<.01) for 5 h after walking compared with control rest. Among DD individuals, a single walk did not induce a reduction in BP (p-interaction>.05).
Conclusions:
Our results showed that postexercise hypotension can occur after a walk at moderate intensity in carriers of the I allele; we were not able to demonstrate this in DD individuals. Our results suggest that genetic variation in the ACE gene might affect the BP response to exercise, although more research is needed to confirm these findings.
Keywords
Introduction
Hypertension is a major risk factor for cardiovascular and cerebrovascular diseases and mortality,1–4 affecting approximately 1 billion individuals worldwide. 5 It is estimated that this number will escalate to more than 1.56 billion by the year 2025, 6 putting a tremendous burden on our health care systems.6–9 Hypertension is also a complex disease influenced by an interaction between environmental and genetic factors. 10 Regarding these environmental factors, lifestyle changes, including physical activity, play a primordial role in the prevention and treatment of high blood pressure (BP).11,12 There is substantial evidence supporting the beneficial effect of aerobic endurance training on resting and ambulatory BP (ABP).13–18 In addition, it has also been demonstrated that one single bout of endurance exercise may cause a transient reduction in BP for prolonged periods of time.11,19–23 This phenomenon has been called postexercise hypotension (PEH)11,21,22 and may vary in magnitude and duration depending on the exercise characteristics.16,24,25 However, it is also widely known that not all individuals respond in the same way to such an acute bout of exercise or to exercise training, which might partly be explained by genetic variations. 26 The HEalth, RIsk factors, exercise Training And Genetics (HERITAGE) family study concluded that genetic factors explain up to 17% of the training-induced reduction in systolic BP (SBP). 27
The renin-angiotensin-aldosterone system (RAAS) is an important BP-regulating mechanism, with angiotensin II (Ang II) being an important and powerful vasoconstrictor, increasing renal sodium and fluid reabsorption by aldosterone release, and consequently resulting in an increase in BP. 10 Given its role in BP regulation, it is reasonable to assume that genetic variations in the RAAS influence BP and its response to exercise. Indeed, earlier studies already demonstrated that individuals with the deletion polymorphism (DD) of the angiotensin converting enzyme (ACE) gene have higher circulating ACE levels compared with individuals with the insertion polymorphism (II),28–30 resulting in a higher BP and an increased risk for hypertension in DD homozygotes.31–34 Less is known about the role of the RAAS system and its polymorphisms in BP reduction following a single exercise session. It is generally accepted that in most subjects, PEH is due to a persistent drop in vascular resistance, which is not completely offset by increases in cardiac output. 35 Although the mechanisms of the vasodilatation underlying this PEH largely remain to be elucidated, changes in the sympathetic nervous system and nitric oxide (NO) have all been implicated,35–38 with studies demonstrating an association between RAAS and these mechanisms.39,40 Similarly, ACE polymorphisms have also been shown to be related to NO release postexercise, with carriers of the I allele presenting greater NO bioavailability after a single bout of aerobic exercise compared with DD individuals, and consequently larger PEH. 40 Finally, exercise also triggers an increase in release of Ang (1–7) as well as ACE2, which are vasodilators, leading to greater NO production and resulting in a reduction in BP. 36 To the best of our knowledge, only a few studies have directly investigated the effects of ACE polymorphisms on post-aerobic exercise hypotension.19,26,40,41 Blanchard et al. 26 observed a lower SBP in DD individuals compared with individuals with an I allele after light-intensity exercise (40% VO2max) but not following moderate intensity exercise (60% VO2max). On the other hand, Santana et al. 40 showed a significantly larger decrease in SBP over the course of 1 h after a maximal incremental cycle test and a single bout of exercise at 90% of anaerobic threshold in II/ID individuals compared with DD.
Nevertheless, participants in these studies were unmedicated healthy patients, or had been treated with medication but undergone a washout period before enrollment in the study. However, today, 58.9% of the population are treated with BP-lowering medication. 42 Therefore, it would be of interest to see whether exercise still has some adjunctive effect in reducing BP in patients already treated with medication. Therefore, given the current limited and inconsistent results, we aimed in the present study (1) to investigate the acute effect of one single walking session at moderate intensity on PEH and (2) to test whether polymorphisms of the ACE gene contribute to the variation in PEH response in medicated patients.
Methods
Subjects
Potential participants were recruited by means of announcements in journals and magazines and on the internet. After being screened and signing the informed consent, 34 individuals (Table 1) performed one control session and one exercise session. The inclusion criteria were: healthy men or women without known cardiovascular or pulmonary disease, age ⩾40 years, non-smokers, clinically diagnosed with hypertension according to the Seventh Report of the Joint National Committee on Prevention, Detection, Evaluation and Treatment of High Blood Pressure 5 and having been treated for at least 3 months with antihypertensive medication acting on RAAS. The study was approved by the Research Ethics Committee of the State University of Londrina - Brazil (232/2013).
General characteristics of the overall study sample and by ACE polymorphism.
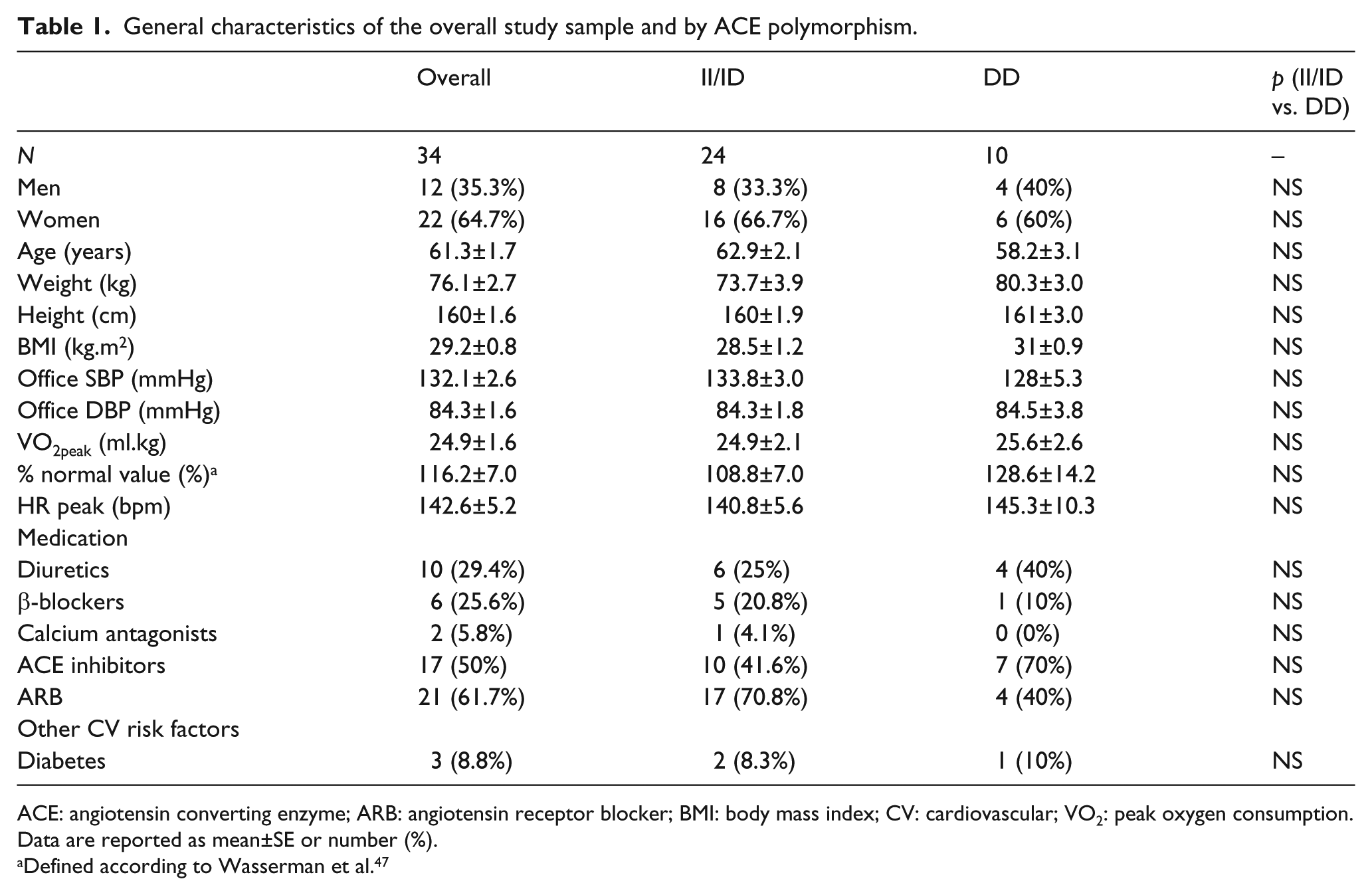
ACE: angiotensin converting enzyme; ARB: angiotensin receptor blocker; BMI: body mass index; CV: cardiovascular; VO2: peak oxygen consumption.
Data are reported as mean±SE or number (%).
Defined according to Wasserman et al. 47
Study design
This study was performed as a randomized controlled cross-over trial. During the screening visit, patients received information about the study and filled in a questionnaire about their habitual physical activity behavior, medication and medical history. In addition, anthropometrics (height and weight) and resting BP were assessed, and volunteers completed a maximal graded exercise test on a treadmill (T170, h/p/cosmos, Traunstein, Germany) until volitional exhaustion using an individualized ramp protocol. 43 The criteria for test interruption followed the recommendations of the American Heart Association. 44 The oxygen uptake was evaluated continuously by a breath by breath analysis of expired gases (QuarKCPET, Cosmed, Rome, Italy), and a polar heart rate (HR) monitor (FS1, Polar, Kempele, Finland) was used to assess the HR. Peak oxygen consumption (VO2 peak) was defined as the mean oxygen uptake of the final 30 s of the exercise test. The maximal duration of the test was between 7 and 12 min. This test was used to define the target HR for the exercise session and to evaluate the current physical fitness of the participants.
Following this visit and having signed the informed consent, participants returned to the laboratory twice, during which they completed the control and the exercise session. The randomization order was determined by a computer program (http://www.random.org/). All tests were performed in the morning (9:00–12:00) at the same time of day for each individual patient to control for diurnal variation in BP and to avoid any influence of time of medication intake. All measurements were performed by the same investigator (KG).
Exercise and control intervention
Each session started with a 10 min seated rest period, after which BP and HR were measured by means of an automatic device (Omron HEM 742 INT, Comfort, Japan). This pre-intervention BP was measured three times with a 5 min interval between measurements. Following pre-intervention measurements, each volunteer performed one of the two sessions in random order: a control session of seated rest and one walking session of 55 min, including a warming up and cooling down of 5 min each and 45 min of walking at 60–75% of HR reserve. During the supervised walking session, training intensity was controlled by a HR monitor (FT1, Polar, Kempele, Finland).
Ambulatory blood pressure monitoring
After the exercise and control sessions, individuals left the laboratory wearing an ABP device (Meditech, ABPM04, Budapest, Hungary) on the left arm until waking the next morning. The ABP monitor was programmed to measure BP every 30 min, and the first BP reading was assessed 30 min following the sessions. Patients were instructed to proceed with their normal daily activities and were asked to fill in an activity diary. Daytime ABP was defined as the average of all BPs recorded between 06:00 and 22:00; nighttime BP was defined as the mean BP of all values between 22:00 and 06:00. All ABP readings were manually checked and invalid measurements were excluded. A reading was considered invalid if it differed by more than 40 mmHg from the previous and the next reading and this could not be explained by a change in physical activity. We subsequently manually calculated hourly averages on the edited data.
Blood sampling and ACE genotyping
Blood samples (5 ml) were collected from the brachial artery during the screening visit and prepared by an independent biochemical laboratory (The Novaes Center for Clinical Analysis), after which they were stored at −80ºC. Genotyping was performed by the polymerase chain reaction (PCR) method 45 at the Laboratory of Biomechanical and Molecular Biology (University of São Paulo). DNA was extracted from whole blood and the ACE gene (II/ID and DD) was determined by means of a two-primer system (I/D: forward: 5ʹ CTGGAGACCACTTCCCATCCTTTCT 3ʹ and reverse: 5ʹ GATGTGGCCATCACATTCGTCAGAT 3ʹ; and I: forward: 5ʹ TGGGACCAGACCGCCCCGCCACTAC 3ʹ and reverse: 5ʹ TCGCCAGCCCTCCATGCCCATAA 3ʹ) with 12.5µl of each primer. Additionally, a solution was prepared containing 0.3 µl MgCl2, 0.2 µl dNTP mix, 1 unit of Taq polymerase, 10 µl of PCR buffer 10× free Mg2+ and 6.3 µl of H2O RNAase free. The DNA was amplified for 30 cycles with denaturation at 94°C for 1 min, annealing at 58°C for 1 min, and extension at 72°C for 2 min using a thermal cycler (MJ Research, PTC-200, Peltier Thermal Cycler, USA). The PCR product is a 190 bp fragment in the absence of the insertion and a 490 bp fragment in the presence of the insertion. A third fragment with an intermediate molecular weight is present in PCR from heterozygotes, and likely corresponds to a heteroduplex DNA fragment. 46 Amplification products were visualized after electrophoresis in 1% agarose gels with loading buffer (Figure 1).

Agarose gel for ACE polymorphism gene identification.
Statistical analysis
Statistical analyses were performed by means of the Statistical Package for the Social Sciences (SPSS 17.0, Chicago, USA). The data are reported as mean and standard error for continuous data and number (%) for dichotomous data. The Shapiro–Wilk test was used to check the normality, followed by Mauchly’s and Greenshouse–Geiser for the sphericity. Independent Student’s t-tests were used to compare pre-intervention data across the different polymorphism groups, and dependent Student’s t-tests were used to compare pre-intervention BP between sessions. Categorical variables were compared by means of χ2 tests. Two-way analysis of variance (ANOVA) with repeated measures was performed to compare ABP (SBP and diastolic blood pressure (DBP)) over time and between the two experimental sessions. When applicable, the least significant difference (LSD) post-hoc test was performed. BP data are reported for 5 h because this is the time period over which PEH was sustained following the exercise session. To test whether the observed genotype frequencies were in Hardy–Weinberg equilibrium, a χ2 test with one degree of freedom was used. Statistical significance was established as p<.05 (one-tailed).
Results
Subject characteristics
The general characteristics of the subjects are presented in Table 1. Overall, participants (35.3% men, mean age 61 years (range 40 to 79)) were overweight or obese individuals with controlled (n=20) or uncontrolled (n=14) hypertension and above average physical fitness for individuals of their age. 47 Baseline characteristics were similar for the two genotype groups. ACE allele and genotype frequencies in our population did not differ significantly from Hardy–Weinberg expectations (p allele frequency = .49; q allele frequency = .51).
Blood pressure response
Overall study sample
As shown in Table 2, pre-intervention BP was not different between the sessions (p>.05). Following the walking session, no differences could be observed for average daytime, nighttime or 24h ABP (p>.05) compared with a non-exercise control session. The hourly mean BP for 5 h following the interventions can be seen in Figure 2. Over the course of 5 h, one single walk induced a significantly more pronounced reduction in BP compared with control for both SBP (p-interaction=.04) and DBP (p-interaction=.001). Further, when we calculated the average change over the 5 h period, Figure 3 shows that the average decrease in DBP (−6.5±1.5mmHg) was significantly larger (p=.01) compared with control rest (−3.1±1.5mmHg), and there was a borderline non-significant (p=.06) larger reduction in SBP (−5.7±2.6mmHg) following exercise compared with a non-exercise control session (−1.3±2.6mmHg).
SBP and DBP in the exercise and control sessions for pre-intervention, daytime, nighttime and 24 h monitoring for 34 participants.

Data are reported as mean ± SE.
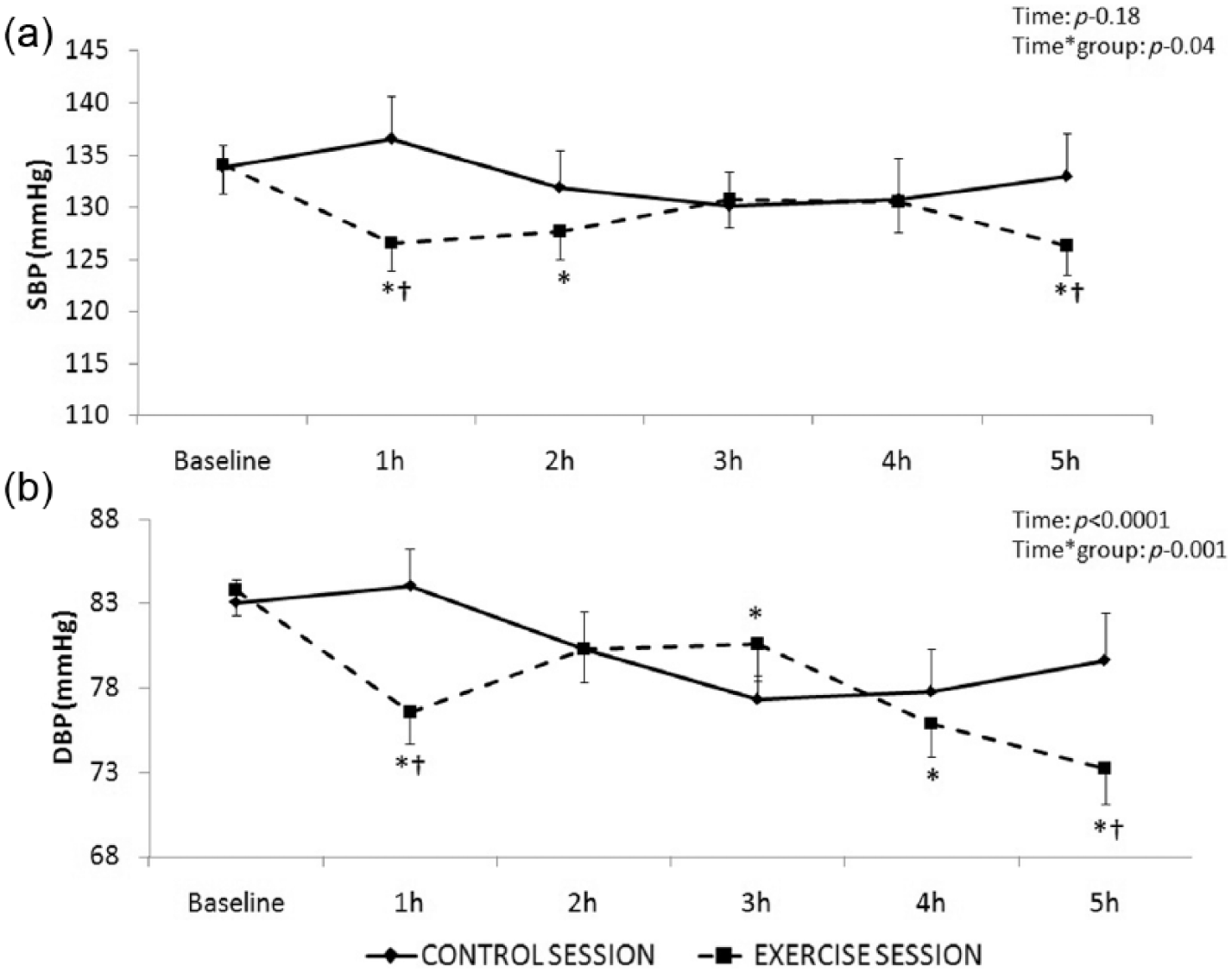
SBP (a) and DBP (b) at hourly intervals over 5 h for both control and exercise sessions in the overall study sample (N=34).

Mean SBP and DBP change at 5 h monitoring following exercise or control session in the overall study sample (N=34).
ACE polymorphisms (II/ID and DD)
Figure 4((a)–(d)) shows BP at baseline and after exercise and control sessions over the course of 5 h by ACE genotype groups. Among the 24 participants with the ACE I allele (i.e. II/ID), we observed a significant time effect for both SBP (p-time<.05) and DBP (p-time<.001) and a significant treatment effect (p-interaction SBP=.02; p-interaction DBP=.002) (Figure 4(a) and (b)). As demonstrated in Figure 5(a), the exercise-induced decrease in DBP (6.3±1.9 mmHg) was significantly larger compared with the non-exercise control session (−2.9±1.9 mmHg) (p=.01 for II/ID). However, as can be seen in Figure 4(c) and (d) and Figure 5(b), participants homozygous for the D allele of the ACE gene showed no significant change in BP following the interventions (p-time >.05 for both; p-interaction > .05 for both).

SBP and DBP at hourly intervals over 5h for both control and exercise sessions in the II/ID group ((a) and (b), N=24) and the DD group ((c) and (d), N=10).
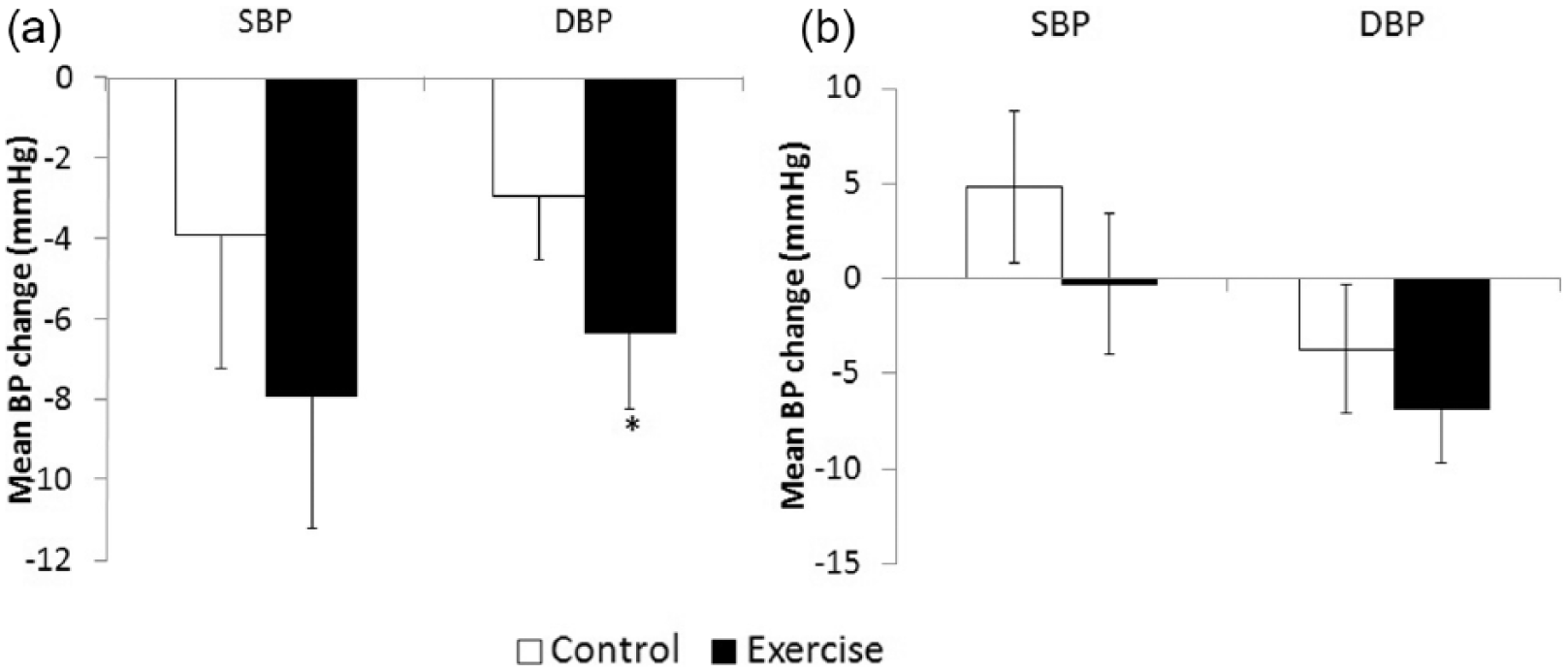
Mean SBP and DBP change at 5 h monitoring following exercise or control session in the II/ID group ((a), N=24) and the DD group ((b), N=10) (overall study sample N=34).
Discussion
The purpose of this study was (1) to assess the acute effect of a single walk at moderate intensity on PEH and (2) to examine the effects of the ACE polymorphisms on the PEH response to this walk in 34 hypertensive medicated men and women. Our results demonstrate that a 45 min walk at moderate intensity already results in BP reductions that last for a 5 h period compared with a non-exercise control session. Our results further show that carriers of the I allele experience this PEH following a single walk, whereas a reduction in BP is not observed among D homozygotes.
The acute effects of aerobic endurance exercise on BP have been widely studied among different populations48–52 and using a wide variety of exercise protocols.25,53,54 Previously, Guidry et al. 55 suggested that PEH following aerobic endurance exercise is a low-threshold phenomenon. They reported that the immediate BP-lowering effects of short-duration (15 min), lower-intensity exercise (40% VO2peak) are comparable to those of higher amounts of exercise (30 min at 60% VO2peak). 55 In line with this, Mach et al. 56 reported that PEH occurred after any duration of moderate-intensity endurance exercise in the laboratory. In our study, we used moderate-intensity walking, which could easily be incorporated into daily life and can safely be performed by any individual. Our results are in line with previous studies that have already demonstrated that PEH is a low-intensity threshold phenomenon. Namely, for 5 h after the walking session, both SBP (−5.7±2.6 mmHg)and DBP (−6.5±1.5 mmHg) were lower compared with a non-exercise control session, suggesting that a simple brisk walk, which is accessible to a large proportion of hypertensive patients worldwide, might already result in significant health benefits. As is well known, the primary recommendation for the treatment of high BP starts with lifestyle modification; however, if the BP is not sufficiently lowered, treatment with antihypertensive medication is necessary. 5 Previous studies have reported that BP control at normal levels is difficult, with only 25–62% of patients being properly controlled;13,57–59 consequently, most hypertensive patients receive antihypertensive medication. In Brazil, 76% of patients are using drug therapy. 60 In view of the importance of external validity to contribute to strategies for BP control in hypertensive patients, we included medicated patients. To our knowledge, this is the first study to investigate the acute effects of exercise on BP in patients treated with ACE inhibitors or angiotensin receptor blockers (ARBs). Previously, Ciolac et al. 13 investigated a single aerobic exercise effect on ABP in long-term-treated hypertensive patients. They demonstrated for the first time that a single session of aerobic exercise reduced ABP in long-term-treated patients. In line with this, Melo et al. 61 showed reductions in BP over the course of 10 h following a single bout of resistance exercise at low intensity in women treated with captopril.
However, not all individuals respond in the same way to exercise, for reasons that still remain to be elucidated. Forjaz et al. 48 showed that only 65% of hypertensive patients experienced a fall in ABP after one bout of exercise, indicating that 35% of the patients did not. Further, Fagard 17 concluded that exercise characteristics, such as intensity, frequency and duration, explain only 4.9% of the variance in SBP and 1.1% in DBP in response to exercise. Additionally, the HERITAGE 62 study concluded that 30% of the variance in resting BP can be explained by genetics. This all suggests that genetic variation between individuals might also help to explain part of the different responses to acute and chronic exercise.10,27
In this study, we focused on the effect of ACE polymorphisms on PEH. We observed a BP reduction after a simple walk in subjects who carry the I allele, while we were unable to show this effect in DD individuals. Others have shown that individuals carrying an I allele (II/ID) are less prone to develop hypertension, and the present results suggest that they are also more responsive to the PEH effect. This was explained by the observation of higher levels of circulating ACE in DD homozygotes compared with individuals carrying the I allele. 28 However, our results further suggest that this lower risk for hypertension might also be explained by their better responsiveness to exercise, although this should be confirmed in larger studies. Our results are in line with those of Santana et al., 40 who showed that only individuals carrying an I allele presented reductions in SBP after an incremental test and after 20 min of exercise at 90% anaerobic threshold. Earlier studies investigating the chronic effects of exercise on BP found similar results.63,64 Namely, Zhang et al. 64 reported a significant reduction in BP after 10 weeks of training only in individuals with an I allele, and not in DD homozygotes, while Hagberg et al. 63 found larger reductions in BP following exercise in II and ID genotype individuals compared with DD individuals.
On the other hand, Blanchard et al. 26 demonstrated that SBP was lower for 14 h after exercise at light but not moderate intensity in homozygous DD individuals compared with II/ID, suggesting that intensity might influence the relationship between ACE polymorphism and PEH. Finally, Taylor-Tolbert et al. 41 reported larger reductions in BP in II and DD compared with ID 24 h after a single bout of vigorous exercise. However, it is difficult to compare their results with ours, as they compared ACE homozygotes (II and DD) with ACE heterozygotes (ID).
The present study was not designed to examine possible mechanisms that could explain the different response between the ACE polymorphisms. However, it has previously been shown that the DD allele of ACE is associated with higher ACE circulating levels, resulting in a higher BP due to an increase in Ang I and Ang II, triggering vasoconstriction, water retention and sodium reabsorption, and resulting in a higher blood volume. 28 Others showed that higher activity of Ang II may blind the vasodilatation induced by bradykinin, resulting in reduced NO bioavailability. 31 Although we did not evaluate NO bioavailability, a previous study demonstrated that ID/II individuals have an increased NO release post incremental exercise test compared with DD individuals. 40 Further, Tanriverdi et al. 65 showed greater endothelium-dependent vasodilatation in II-homozygous athletes compared with DD or ID, due to increased NO bioavailability and bradykinin concentration. These mechanisms might in part explain the PEH response, taking into account that bradykinin and NO release are related to PEH.20,66
The present study has some limitations. We only evaluated insertion/deletion polymorphisms of the ACE gene, and did not include other polymorphisms or parameters of the RAAS, such as plasma renin levels, ACE plasma levels, angiotensin levels and angiotensinogen. Based on a previous review by Pescatello et al., 67 we calculated that we would need a sample size of 33 patients to detect an absolute net difference of 4.7 mmHg, assuming a standard deviation of 8.5 mmHg with a statistical power of 0.8 and alpha of 0.05. Although the number of participants in the DD group corresponds to the overall representation of this group in the general population, this lower number of participants might have resulted in a lack of power to detect changes in BP following exercise. Consequently, we lacked the power to perform a three-way ANOVA in which we could have included the polymorphism groups as a covariate. Therefore, we suggest that the results of this study should be interpreted as hypothesis generating, and further studies are needed to confirm our findings.
Conclusions
In hypertensive subjects receiving medications for the RAAS, a single walk at moderate intensity promotes a reduction in BP compared with a rest session. Overall, a single walk at moderate intensity promoted somewhat larger reductions in BP compared with a control rest session. Further, our results suggest that this PEH might be influenced by genetic factors.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: KFG is supported as a doctoral fellow by the Brazilian Agency for Support and Evaluation of Graduate Education - CAPES foundation (process 2446/14-6); VAC is supported as a postdoctoral research fellow by the Research Foundation Flanders (FWO); MDP is supported by the Brazilian Council for the Scientific and Technological Development (CNPq, process 303566/2013-2).
