Abstract
Introduction:
Whilst sustained lowering of brachial systolic blood pressure (Br-SBP) and central aortic systolic pressure (CASP) have been demonstrated in patients with hypertension, effects of treatment withdrawal on these parameters have not been investigated. The ASSERTIVE study previously reported more sustained control of Br-SBP with aliskiren versus telmisartan in patients with hypertension, following 7-days treatment withdrawal. In this ASSERTIVE sub-study, we hypothesised that aliskiren would similarly exert more sustained control of CASP than telmisartan during treatment withdrawal.
Methods:
We investigated the effects of treatment withdrawal on both Br-SBP and CASP following 12-weeks treatment with either aliskiren (300 mg) or telmisartan (80 mg). Br-SBP and CASP were measured at the end of treatment, and at days 2 and 7 following treatment withdrawal in 303 patients (CASP randomised set).
Results:
Of the CASP randomised set, 94 patients completed CASP measurements at all time points (CASP completer set). After 7 days of treatment withdrawal, aliskiren demonstrated lesser increases in both Br-SBP and CASP than telmisartan; Br-SBP change: −2.0±1.6 vs. +5.6±1.7 mmHg, p = 0.001; CASP change: −0.4±1.6 vs. +4.6±1.7 mmHg, p = 0.041, n = 94. Similar findings were obtained for the CASP randomised set.
Conclusions:
Following treatment withdrawal, aliskiren demonstrated more sustained control of both brachial and central SBP than telmisartan.
Keywords
Introduction
Clinical interest in the utility of non-invasive measurement of central aortic pressure (CAP) has heightened in recent years. 1 This is due, in part, to the development of simple, robust methods for evaluating this clinical parameter,2,3 and also to the emergence of data demonstrating that central pressure may be a better marker of risk for developing cardiovascular pathologies and clinical outcomes than conventional assessment of brachial pressure.4,5 Other key studies have also established the concept of differential effects of blood pressure (BP) lowering treatments on brachial and central pressure, promoting calls for the routine incorporation of central BP measurement in clinical trials. 6 More recently, the feasibility of monitoring 24-hour ambulatory central aortic pressure was reported. 7 This has further extended the potential clinical utility of central pressure monitoring.
Whilst the influence of drug treatment on both brachial and central pressure has been extensively discussed, 8 the influence of drug treatment withdrawal on seated central pressure is not yet reported. Since missed medication doses are a frequent occurrence during the treatment of elevated BP, it is important to verify the consequences on central aortic pressure in comparison to what has been described for peripheral blood pressure.9,10 It is possible that treatment withdrawal could change the relationship between brachial and central pressure, effects which might not be apparent from monitoring brachial pressure alone.
Two different strategies for lowering BP via inhibition of the renin–angiotensin–aldosterone system (RAAS) were studied. We compared the effects of withdrawal following prior treatment with either aliskiren, a direct renin inhibitor, or telmisartan, an angiotensin receptor blocker (ARB), both known for their long-lasting efficacy.11,12 The primary focus of this study was on the 7-day treatment withdrawal period, following a 12-week treatment period, evaluating the corresponding changes in brachial BP and central aortic systolic pressure (CASP), in patients with hypertension. The ASSERTIVE study previously reported more sustained control of Br-SBP with aliskiren versus telmisartan in patients with hypertension, 13 following 7-days treatment withdrawal. In this ASSERTIVE sub-study, we hypothesised that aliskiren would similarly exert more sustained control of CASP than telmisartan during treatment withdrawal. Importantly, this paper reports, for the first time, the impact of treatment withdrawal on brachial BP and CASP in a randomised clinical trial of BP lowering.
Methods
Study design
This was a sub-study embedded within the AliSkiren Study of profound antihypERtensive efficacy in hyperTensIVE patients (ASSERTIVE), the design and primary results of which have been previously published. 13 ASSERTIVE was a randomised, double-blind, double-dummy, parallel-group, multi-centre study. Following a 2-week washout and 1- to 2-week placebo run-in period, patients were randomised to 2 weeks of active treatment with either once-daily aliskiren 150 mg or telmisartan 40 mg monotherapies, and then force-titrated to double the initial dose for 10 weeks.
At the end of the 12-week treatment period, both drugs were replaced with placebo and brachial and central pressures were measured at days 2 and 7 following treatment withdrawal. The present analysis focuses on the 7-day treatment withdrawal period. Details of the run-in and active treatment periods concerning sitting brachial and central pressure are presented in the online supplement.
Patients
The ASSERTIVE study included men or women ⩾18 years, with essential hypertension (Grades 1–2), who met both of the following criteria at randomisation: office systolic BP (SBP) ⩾140 mmHg and <180 mmHg, and 24-hour mean ambulatory SBP (MASBP) ⩾135 mmHg. Major exclusion criteria included the following: severe hypertension (mean sitting SBP (msSBP) ⩾180 mmHg and/or mean sitting DBP (msDBP) ⩾110 mmHg), secondary hypertension, and pregnant women.
The trial was conducted in accordance with the Declaration of Helsinki, the International Conference on Harmonisation Guidelines for Good Clinical Practice, and the US Code of Federal Regulations, Part 46, Protection of Human Subjects. The trial protocol was approved by independent ethics committees at all centres, and written informed consent was obtained from each patient before participating in any trial procedures. The trial is registered as EudraCT no. 2008-007831-41 and on ClinicalTrials.gov under the code NCT00865020.
Participants in this ASSERTIVE sub-study were recruited from 45 out of the 111 ASSERTIVE study centres worldwide. All participants at each centre were invited to participate (mean participation 95±15% per centre) and this comprised the CASP randomised dataset (n = 303).
Study assessments
The primary endpoint for this analysis was to compare the sustained efficacy of prior treatment with aliskiren versus telmisartan on the change in seated CASP at day 7 following treatment withdrawal. Other assessments included change in (1) seated CASP at day 2 following treatment withdrawal, (2) seated brachial BP at days 2 and 7 following treatment withdrawal, (3) brachial ambulatory BP at day 7 following treatment withdrawal and (4) evaluation of biomarkers, plasma renin activity (PRA) and aldosterone across the treatment and 7-day withdrawal periods. Study visits were performed between 07:00 and 10:00 hrs so that set-up of ambulatory blood pressure measurements could be standardised across the study.
Brachial BP measurements
Seated brachial BP was measured at the end of the 12-week treatment period (baseline for this analysis) and on days 2 and 7 following treatment withdrawal. Brachial BP was measured using a validated and automated BP monitor (Omron® HEM-705) with an appropriate cuff size, according to the European Society for Hypertension Guidelines. 15 Briefly, the cuff device was applied on the non-dominant arm or the arm with the higher reading at screening if there was a clinically relevant difference (SBP ⩾10 mmHg and/or DBP ⩾5 mmHg) between arms at the first study visit. Three sitting BP measurements were taken at 1–2-minute intervals after the patient had been seated for 5 minutes. The mean of these readings was recorded as brachial BP.
CASP measurements
Seated CASP was measured at the end of the treatment period (baseline for this analysis) and on days 2 and 7 following treatment withdrawal, using the BPro™ device (HealthSTATS, Singapore). The BPro™ device uses applanation tonometry to capture high-fidelity radial artery pulse waveforms (RAPWF) at the wrist in blocks of 10-second intervals. RAPWFs were calibrated to brachial BP measured in the same arm using the OMRON HEM-705 device, immediately prior to waveform collection. CASP was derived from the calibrated RAPWFs using a validated algorithm. 16
Ambulatory BP Monitoring
Twenty-four-hour brachial ambulatory BP monitoring (ABPM) using Spacelabs 90208 (Spacelabs Medical Inc., Redmond, Washington, USA) was performed at the end of the treatment period and on day 7 following withdrawal. ABPM sessions commenced between 7 a.m. and 10 a.m. and monitors were applied to the non-dominant arm of the patient, as previously described.13,17 Validity criteria for ABPM measurements included a minimum test duration of 24 hours, at least 70% of valid expected BP measurements during the 24 hour duration, and no more than two non-consecutive hours without readings. If the ABPM measurement failed to achieve validity criteria at randomisation, a repeat measurement was permitted within 24–72 hours, provided the patient continued to receive placebo treatment.
Plasma renin activity and aldosterone levels
Blood samples were collected from a subset of patients prior to treatment initiation, at the end of the treatment period, and on day 7 following withdrawal. Blood samples were collected after a fast of at least 8 hours. EDTA samples were centrifuged within 5 minutes of collection and plasma was immediately frozen at −20°C for a maximum of 4 weeks, and then at −80°C at the central laboratory (Eurofins, Breda, The Netherlands), until assayed. The biomarker, PRA was assessed using RIA kits from DiaSorin (Stillwater, Minnesota, USA) and aldosterone was measured using RIA Coat-a-Count kits from Siemens (Deerfield, Illinois, USA).
Statistical analyses
In this sub-study, CASP was measured at sub-study baseline (end of active treatment) and at days 2 and 7 following treatment withdrawal. However, not all patients had valid CASP measurements at all sub-study time points. Hence, two sets of patient populations, based on the extent of availability of their CASP data, were analysed. The CASP completer dataset was obtained from participants who had valid seated CASP measurements at all time points, i.e. at the end of the treatment period (baseline for this analysis) and on days 2 and 7 following treatment withdrawal. In addition, the CASP randomised dataset was obtained from participants who had a valid seated CASP measurement at least at one of the above mentioned time points. Changes from baseline in seated brachial SBP (msSBP), CASP, and MASBP were analysed using an analysis of covariance (ANCOVA) model with treatment, study region and gender as factors and baseline mean arterial pressure as a covariate. The primary analysis focussed on changes in CASP from the end of the 12-week treatment period to days 2 and 7 of the treatment withdrawal period. Between-treatment analysis was also performed for changes in msSBP, CASP and MASBP from baseline to end of the withdrawal period.
Results
Patient disposition
Of the 1359 patients who entered the placebo run-in period of the ASSERTIVE study, 822 were randomised to the aliskiren (n = 414) and telmisartan (n = 408) treatment groups. Of these patients, 303 (aliskiren, n = 157; telmisartan, n = 146) participated in the central aortic pressure sub study (CASP randomised set), of whom 94 patients (aliskiren, n = 49; telmisartan, n = 45) had a valid CASP measurement completed at every time point across the treatment withdrawal period (CASP completer set).
Demographic and baseline characteristics
Demographic and baseline characteristics of the sub-study population were generally comparable between the aliskiren and telmisartan groups at randomisation for the CASP randomised and CASP completer sets (Table 1).
Patient demographics and baseline characteristics by treatment (CASP completer set (n = 94) and CASP randomised set (n = 303)).
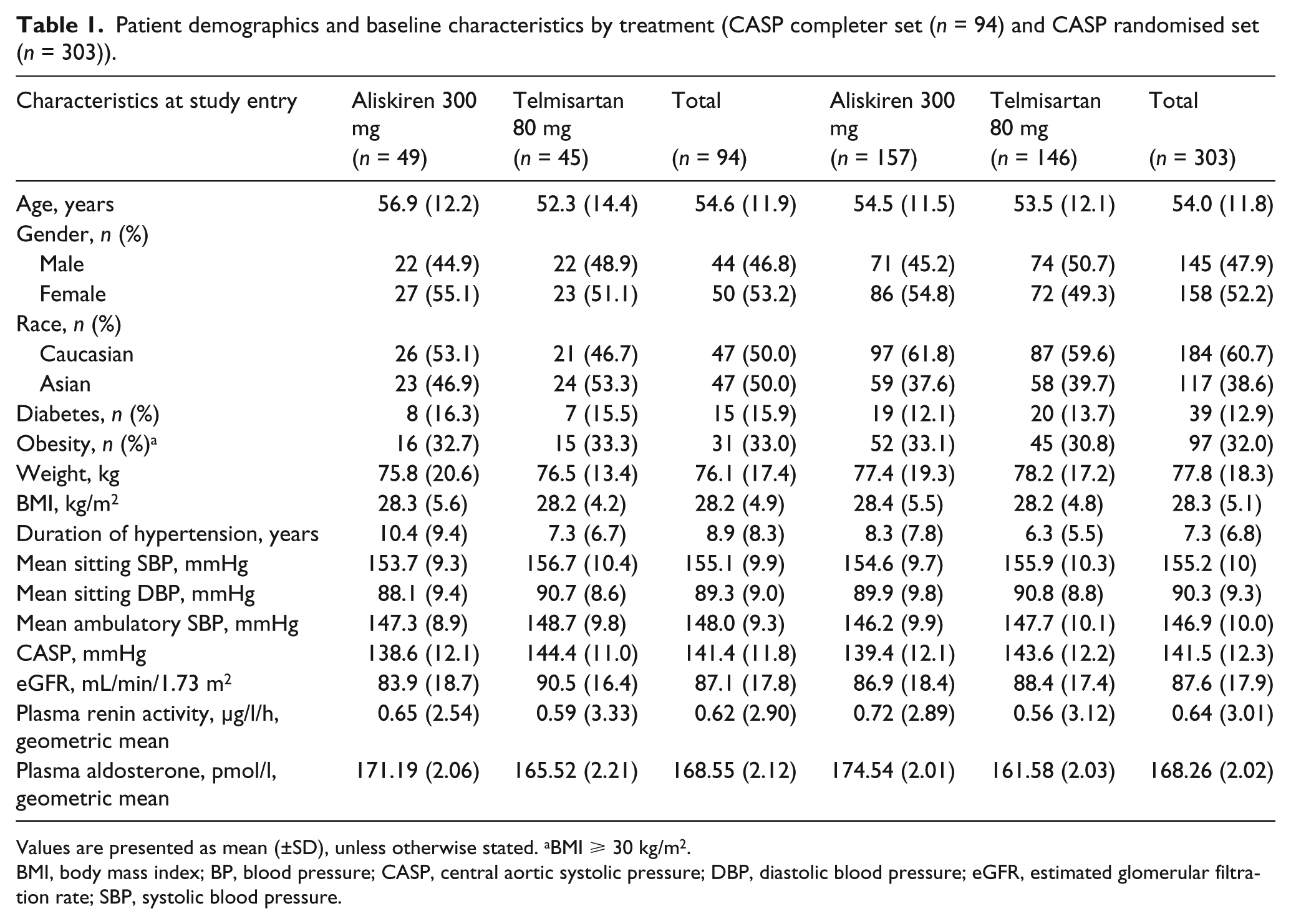
Values are presented as mean (±SD), unless otherwise stated. aBMI ⩾ 30 kg/m2.
BMI, body mass index; BP, blood pressure; CASP, central aortic systolic pressure; DBP, diastolic blood pressure; eGFR, estimated glomerular filtration rate; SBP, systolic blood pressure.
Efficacy
Changes in seated brachial SBP
In the CASP randomised set, msSBP was similar between both treatment groups at baseline for the treatment withdrawal period, i.e. at the end of the treatment period (aliskiren, 138.8±13.8 mmHg versus telmisartan, 139.4±15.9 mmHg, as shown in Figure 1(a)). At day 2 of treatment withdrawal, change in msSBP was −0.8±0.9 mmHg with aliskiren and +2.1±0.9 mmHg with telmisartan, with a significant least squares mean (LSM) between-treatment difference of −2.9±1.3 mmHg (95% confidence interval (CI): −5.4, −0.4; p = 0.026) in favour of aliskiren. Across the 7-day withdrawal period, msSBP remained almost unchanged with aliskiren (+1.6±1.0 mmHg), but increased with telmisartan (+3.2±1.0 mmHg). However, LSM change between-treatment difference (−1.6±1.5 mmHg; 95% CI: −4.5, 1.3) was not statistically significant (p = 0.275).

(a) Mean msSBP, (b) mean CASP, (c) LSM change in CASP and (d) mean MASBP from end of treatment to end of withdrawal (day 7 of withdrawal) for the CASP randomised set.
At baseline for the treatment withdrawal period, mean sitting brachial SBP (msSBP) was similar between treatment groups in the CASP completer set (aliskiren, 139.6±12.4 mmHg versus telmisartan, 140.2±14.9 mmHg) (Figure 2(a)). At day 2 of treatment withdrawal, msSBP changed little (−2.1±1.6 mmHg) with aliskiren, but increased with telmisartan (+4.1±1.6 mmHg). LSM change between-treatment difference (−6.3±2.3 mmHg; 95% CI: −10.8, −1.8) was statistically significant in favour of aliskiren (p = 0.006). Similarly, across the 7-day withdrawal period, msSBP remained almost unchanged with aliskiren (−2.0±1.6 mmHg), but increased with telmisartan (+5.6±1.7 mmHg). LSM change between-treatment difference (−7.5±2.3 mmHg; 95% CI: −12.1, −3.0) was also statistically significant in favour of aliskiren (p = 0.001).

(a) Mean msSBP, (b) mean CASP, (c) LSM change in CASP and (d) mean MASBP from end of treatment to end of withdrawal (day 7 of withdrawal) for the CASP completer set.
Changes in seated CASP
At baseline for the withdrawal period, mean seated CASP values were 129.9±16.2 mmHg versus 127.6±14.9 mmHg for aliskiren versus telmisartan, respectively, in the CASP randomised set (Figure 1(b)). Two days following withdrawal, change in mean CASP was −1.9±1.7 mmHg with aliskiren and +0.4±1.7 mmHg with telmisartan, with LSM change between-treatment difference of −2.4±2.4 mmHg (95% CI: −7.1, 2.3; p = 0.324). Mean change in CASP from baseline following the 7-day withdrawal period was −0.7±1.2 mmHg for aliskiren. In contrast, an increase in CASP of +5.5±1.3 mmHg was observed upon telmisartan withdrawal. The LSM change between-treatment difference in CASP from baseline to day 7 of withdrawal was −6.2±1.8 mmHg (95% CI: −9.7, −2.7), which was a statistically significant and potentially clinically relevant difference in favour of aliskiren (p < 0.0001) (Figure 1(c)). It is interesting to note that despite a higher baseline CASP value in the aliskiren compared to the telmisartan group, mean CASP decreased following aliskiren withdrawal while it increased upon telmisartan withdrawal across the 7-day withdrawal period in the CASP randomised set.
At baseline for the treatment withdrawal period, mean seated CASP values were very similar between both treatment groups in the CASP completer set (aliskiren, 129.4±15.3 mmHg; telmisartan, 129.3±15.4 mmHg), as shown in Figure 2(b). Two days following withdrawal, mean CASP remained unchanged with aliskiren (−0.8±2.5 mmHg) and increased with telmisartan (+2.1±2.6 mmHg), the LSM change between-treatment difference being −2.9±3.7 mmHg (95% CI: −10.2, 4.4; p = 0.436). Mean change in CASP from baseline following the 7-day withdrawal period was −0.4±1.6 mmHg for aliskiren. In contrast, an increase in CASP of +4.6±1.7 mmHg was observed with telmisartan withdrawal. The LSM change between-treatment difference in CASP from baseline to day 7 of withdrawal was −4.9±2.4 mmHg (95% CI: −9.6, −0.2), which was a statistically significant and potentially clinically relevant difference in favour of aliskiren (p = 0.041) (Figure 2(c)).
Changes in 24-hour ABPM
At baseline for the withdrawal period, MASBP was 133.3±11.6 mmHg and 132.7±12.2 mmHg for aliskiren and telmisartan, respectively, in the CASP randomised set (Figure 1(d)). Across the 7-day withdrawal period, MASBP changed little with aliskiren (+1.9±0.7 mmHg). In contrast, an increase in MASBP was seen 7 days following telmisartan withdrawal (+6.3±0.7 mmHg), with a significant between-treatment difference (−4.4±1.0 mmHg; 95% CI: −6.4, −2.5; p < 0.0001), in favour of aliskiren.
At baseline for the treatment withdrawal period, MASBP was 135.2±10.5 mmHg and 136.6±11.8 mmHg for aliskiren and telmisartan, respectively, in the CASP completer set (Figure 2(d)). Across the 7-day withdrawal period, MASBP changed little with aliskiren (+1.2±1.1 mmHg). In contrast, an increase in MASBP was seen 7 days following telmisartan withdrawal (+5.5±1.2 mmHg), with a significant between-treatment difference (−4.3±1.7 mmHg; 95% CI: −7.5, −1.0; p = 0.010), in favour of aliskiren.
Changes in PRA and aldosterone levels
Plasma biomarkers were analysed as an indication of treatment response. Accordingly, the response of biomarkers from end of the treatment period to end of the withdrawal period is presented.
In the CASP randomised set, PRA levels increased during 12 weeks of treatment with telmisartan, while it decreased in patients receiving aliskiren (Figure 3(a)). Seven days following treatment withdrawal, PRA returned towards baseline with both aliskiren and telmisartan in the CASP randomised set. Similarly, in the CASP completer set, 12 weeks of treatment was associated with an increase in PRA for patients receiving telmisartan, whilst PRA was suppressed in patients receiving aliskiren. Seven days following treatment withdrawal, PRA returned towards baseline values with both treatments in the CASP completer set (Figure 4(a)).
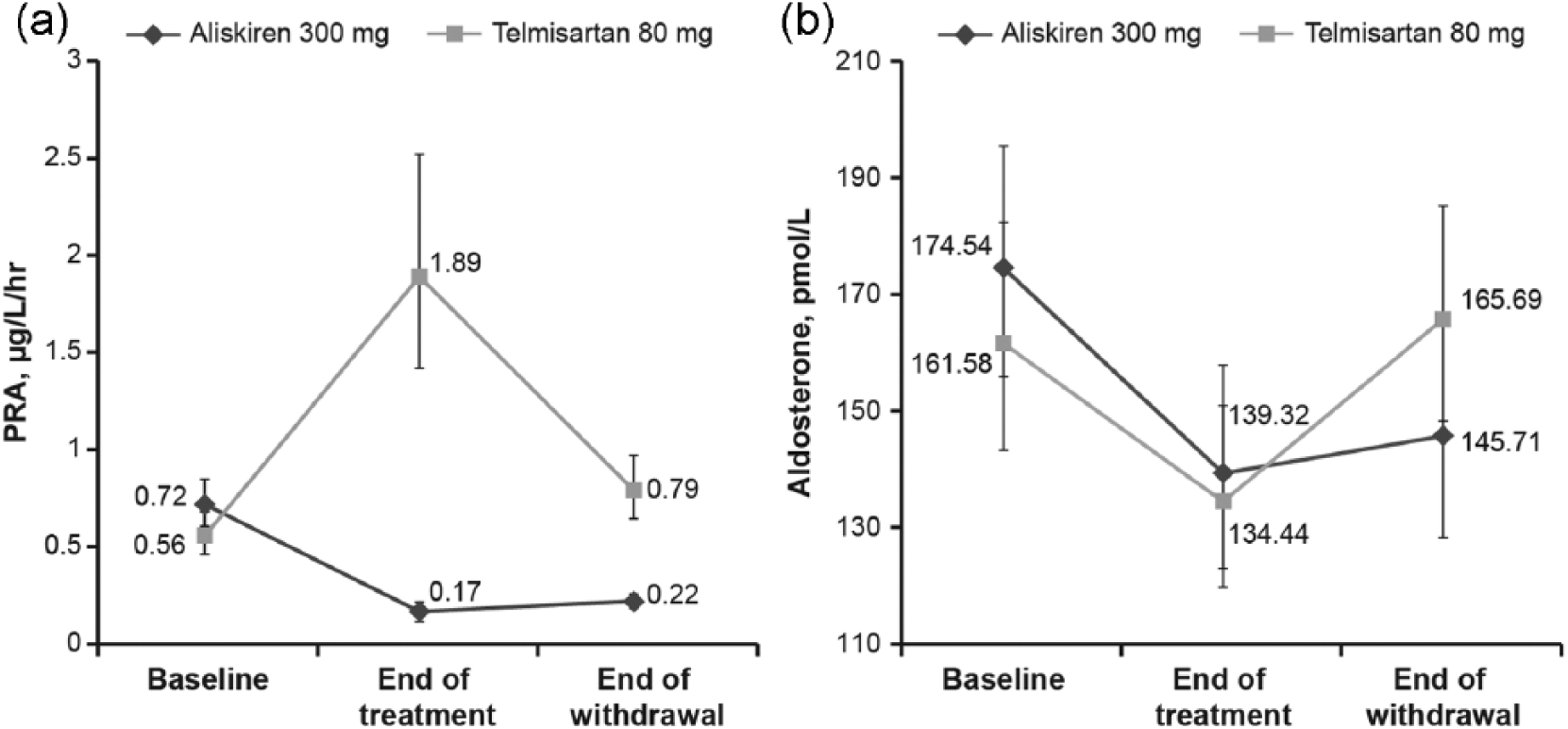
Mean changes in (a) plasma renin activity (PRA) and (b) plasma aldosterone from end of treatment to end of withdrawal (day 7 of withdrawal) for the CASP randomised set. Results are expressed as geometric means and error bars indicate 95% CI.

Mean changes in (a) plasma renin activity (PRA) and (b) plasma aldosterone from end of treatment to end of withdrawal (day 7 of withdrawal) for the CASP completer set. Results are expressed as geometric means and error bars indicate 95% CI.
In the CASP randomised set, 12 weeks of treatment was associated with a decrease in plasma aldosterone levels for both treatments. Plasma aldosterone increased during the 7-day withdrawal period for both treatments (Figure 3(b)). The increase in aldosterone was greater following withdrawal of telmisartan compared to aliskiren, which was statistically significant (p = 0.013). In the CASP completer set, 12 weeks of treatment was associated with a marked reduction in plasma aldosterone levels for both treatments. Plasma aldosterone increased during the 7-day withdrawal period for both treatments (Figure 4(b)). There was a greater increase in aldosterone following withdrawal of telmisartan compared to aliskiren, which did not achieve significance (p = 0.11). However, the clinical implications of differences in effect on RAAS components are not known.
Discussion
This study investigated the effects of simulating a period of non-adherence to BP-lowering medication on the sustainability of BP control by withdrawing treatment over a 7-day period. Two different treatments inhibiting the RAAS were used for BP lowering, and the effects of treatment withdrawal on both brachial pressure and CASP are reported here. The study demonstrates sustained control of both brachial and central BP even after 7 days of aliskiren withdrawal in the CASP completer and randomised sets. In contrast, both brachial and central BP increased gradually by 4-5 mmHg over the same time period following withdrawal of telmisartan, indicating a gradual loss of BP control over the withdrawal period. This is the first reported data on the effects of treatment withdrawal on both brachial and central aortic pressure and extends the previously reported observations of differential effects of treatment withdrawal on brachial BP and 24-hour ambulatory brachial BP in the ASSERTIVE study.13,18
The primary focus of this study was a comparison between treatments for the change in seated CASP following treatment withdrawal, i.e. from the end of the 12-week treatment period to day 2 and to the end of the 7-day treatment withdrawal period in the CASP completer patients set. To complement the results from the CASP completer patients set, we also report changes in seated CASP following treatment withdrawal in the CASP randomised set. The primary endpoint showed statistical significance in favour of aliskiren over telmisartan in the longevity of CASP control, with little change in CASP during the 7-day period following aliskiren withdrawal in both CASP completer and randomised sets. Moreover, data from the present study illustrate that the pattern of change for brachial BP and CASP was broadly similar following withdrawal of either treatment. From this data it would seem that conventional monitoring of either brachial or central pressure is sufficient to determine the haemodynamic consequences of treatment withdrawal as these changes occur in parallel during this period. This concept is also supported by the pattern of change for ambulatory brachial BP across the treatment withdrawal period, which was similar to that seen for both seated brachial BP and CASP in the CASP completer as well as in the CASP randomised sets.
With regard to the longevity of BP lowering across the treatment withdrawal period, aliskiren and telmisartan are both regarded as long-acting drugs from their respective classes. As reported previously, both drugs were very effective in decreasing brachial BP in the overall population.19,20 In the present study, both treatments reduced BP to the same level following 12 weeks of treatment, such that the baseline brachial and central pressures for the withdrawal period did not differ between treatment arms. Moreover, brachial and central pressures remained proportional across the withdrawal period for both treatments, i.e. there was no evidence of differential effects between the treatments.
Other studies investigating the influence of simulated non-adherence have assessed effects only on brachial BP and have used shorter treatment withdrawal time periods, typically 24–48 hours. Depending on the treatment regimen investigated, these studies have generally demonstrated short-term maintenance, or partial loss of BP control with increases of 4-5 mmHg occurring over a short period of time. 19 Data from the present study demonstrate consistency with previous findings over a 2-day treatment withdrawal period, and extend the same over a 7-day withdrawal period, demonstrating loss in both brachial and central BP control upon telmisartan withdrawal but little change upon aliskiren withdrawal. These data demonstrate the sustained duration of action of aliskiren on seated brachial BP, CASP and brachial ambulatory BP across the prolonged treatment withdrawal period of 7 days.
Persistence in the BP-lowering response to aliskiren and telmisartan is consistent with the pattern of biomarker activity monitored across the treatment withdrawal period in the CASP completer and the CASP randomised sets. As expected, PRA showed a divergent response over the 12-week treatment period. As demonstrated previously, up-regulation of PRA was observed following active angiotensin receptor blockade,13,21 which results from stimulation of renin secretion in the juxtaglomerular apparatus (JGA) in response to elevated circulating angiotensin II levels. 22 In contrast, PRA was suppressed during active treatment with aliskiren, thereby reflecting inhibition of plasma renin. The extent of inhibition of PRA with aliskiren was similar to that reported previously. 21 Following treatment withdrawal, PRA returned towards baseline levels; however, the extent of this restoration appeared to differ between treatments. Thus, at the end of the 7-day withdrawal period, PRA reduced toward baseline levels in patients previously randomised to telmisartan, while there were signs of continued PRA suppression for patients previously randomised to aliskiren.
Active treatment was associated with suppression of plasma aldosterone levels, which returned to near-baseline levels at the end of the 7-day treatment withdrawal period for patients previously randomised to telmisartan. In contrast, a trend towards continued suppression of plasma aldosterone throughout the 7-day withdrawal period was evident in patients previously randomised to aliskiren. These data imply sustained inhibition of the RAAS with aliskiren withdrawal, while RAAS activity was gradually restored upon telmisartan withdrawal, and is consistent with other findings using these treatment types.7,13
Persistence in the BP lowering and aldosterone lowering effects of aliskiren over the 7-day withdrawal period is consistent with tissue localisation studies with this drug. Animal studies indicate persistent accumulation of aliskiren in the kidney. 23 A tissue distribution study in humans indicates affinity for lipophilic tissues, 11 providing a likely rational for the sustained effects of aliskiren on RAAS and BP lowering. Typical half-lives for ARBs have been reported to be between 5 (eprosartan, valsartan) and 24 (telmisartan) hours. The relatively long half-life for telmisartan is consistent with the slow restoration of BP and biomarker activity in this study.
This study has limitations that must be acknowledged. Central pressure was measured non-invasively as it was neither practical nor appropriate to use invasive measurements with frequent visits in this study population. As this population was late middle-aged and predominantly white or Asian, these findings may not be applicable to young or black patients with hypertension. As this was a sub-study embedded within the ASSERTIVE trial, and as only 94 out of 303 participants had valid central BP measurements at every time point, this study was not powered to investigate the impact of treatment withdrawal on effects beyond BP lowering, such as cardiovascular outcomes. Therefore, the clinical implications of the differences observed in BP upon treatment withdrawal are not known.
In conclusion, we report for the first time the impact of treatment withdrawal on both brachial and central aortic pressure, demonstrating that both respond proportionately, and that brachial BP is a reasonable surrogate for changes in central pressure following withdrawal of these medications. The importance of such properties with regard to cardiovascular protection needs further investigation.
Footnotes
Acknowledgements
B Williams is a National Institute for Health Research (NIHR) Senior Investigator and his research is supported by the University College London Hospitals, London, NIHR Biomedical Research Centre. The authors thank Michael Ulrich (Novartis Pharma AG) for project management and Margaret Prescott (Novartis Pharmaceuticals Corporation, East Hanover, NJ, USA) for helpful discussions on the biomarker results. We acknowledge Parvathy Ramakrishnan (Novartis, Hyderabad) for editorial support. Support from PPD for statistical programming and analysis (Winchester, UK) are gratefully acknowledged. All authors participated in the development and writing of the paper and approved the final manuscript for publication.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BW and RD have previously received research support from Novartis and have acted as a consultant for Novartis. PB, GB and FB have been involved in the design, conduct, analysis and reporting of the study; they are employees of Novartis (Novartis Pharma AG, Basel, Switzerland) and are, therefore, eligible for Novartis stock and stock options.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The ASSERTIVE study from which the data for this sub-study were derived was supported by Novartis Pharma AG, Basel, Switzerland. This sub-study analysis was performed at Novartis AG (G Bader) in collaboration with University College London.
