Abstract
Introduction:
The purpose of this study was to compare the effects of benazepril and losartan on endothelial function and vascular stiffness, in patients with diabetes mellitus and hypertension.
Materials and methods:
We included hypertensive diabetic patients with an office systolic blood pressure (BP) ⩾ 130 mmHg and/or diastolic BP ⩾ 80 mmHg. Patients were rolled over to amlodipine for 6 weeks, then we performed C-reactive protein assays, BP measurement and vascular tests; next, patients were randomized to benazepril or losartan. The tests were repeated after 12 weeks.
Results:
We randomized 14 patients to benazepril and 16 to losartan. There were no differences in systolic (139 versus 134 mmHg, p = 0.618) and diastolic (82 versus 80 mmHg, p = 0.950) BP at the end of the study. C-reactive protein values were lower in the benazepril group (0.38 versus 0.42 mg/dl, p = 0.020). There was a slightly higher flow-mediated vasodilation (FMD) response in the benazepril group (45% increase, p = 0.057) than in the losartan group (19% increase, p = 0.132). Both central systolic BP (129 versus 123 mmHg, p = 0.934) and carotid-femoral pulse wave velocity (cfPWV) (8.5 versus 8.5 m/s, p = 0.280) were the same between groups.
Conclusions:
Hypertensive diabetic patients using benazepril had a greater reduction in C-reactive protein, and a slight improvement in FMD, than those taking losartan.
Keywords
Introduction
Hypertension and Type 2 diabetes mellitus are associated with an increased risk for cardiovascular disease, mainly due to atherosclerotic vascular changes.1–4 Endothelial dysfunction and increased arterial stiffness are considered markers of subclinical atherosclerosis, and are related to a worse prognosis.5,6 Angiotensin II plays a central role in both conditions, as it promotes an increased formation of oxygen-reactive species, reduced bioavailability of nitric oxide, vascular cell apoptosis, matrix deposition, low-density lipoprotein cholesterol oxidation and release of pro-inflammatory cytokines.7–9
The angiotensin-converting enzyme inhibitors (ACEi) and the angiotensin II receptor blockers (ARB) are the most commonly used drugs that limit the effects of angiotensin II. Albeit most guidelines recommend both classes interchangeably,10,11 their mechanisms of action are not the same. ACEi, but not ARB, lead to increased levels of bradykinin and angiotensin-(1–7), both with anti-atherogenic and vasodilator properties. 12 Three recent meta-analyses with hypertensive patients review the effects of ACEi and ARB on endothelial function, as assessed by flow-mediated vasodilation (FMD)13–15; another meta-analysis reviews their effects on arterial stiffness, as assessed by pulse wave velocity (PWV) and central pressure waveforms. Although both ACEi and ARB were better than placebo, their results were the same, when compared among themselves; however, all these meta-analyses included very few trials with diabetic patients.
The primary objective of this study was to compare the effects of benazepril and losartan, which are an ACEi and an ARB, respectively, on endothelial function and vascular stiffness in patients with Type 2 diabetes mellitus and hypertension.
Materials and methods
Patient selection
Patients were recruited from our outpatient hypertension clinic. Inclusion criteria were: age between 40–70 years old, previous diagnosis of Type 2 diabetes mellitus and hypertension, and office systolic blood pressure (BP) ⩾ 130 mmHg and/or diastolic BP ⩾ 80 mmHg. We excluded patients with secondary hypertension, resistant hyper-tension, insulin therapy, Stage 4 or 5 of chronic renal disease, previous myocardial infarction, cerebral vascular accident, history of transient ischemic attack, heart failure Stage C or D, atrial fibrillation, malignant ventricular arrhythmias, symptomatic peripheral arterial disease, retinopathy with visual loss, nephrotic syndrome and symptomatic neuro-pathy. All patients accepted by written informed consent. The hospital ethics committee approved the trial (no. 01539612.6.0000.5259) and it was registered in ‘Clinical Trials’ under identifier NCT01603940.
Study design
The study was a randomized, open label, clinical trial with two treatment arms (Figure 1). In Visit 1, all patients were rolled over for amlodipine 5 mg once a day (q.d.). After 6 weeks (Visit 2), we conducted BP measurements, and tests to evaluate vascular stiffness and endothelial function. Venous blood was sampled for the biochemistry and C-reactive protein (CRP) assays.
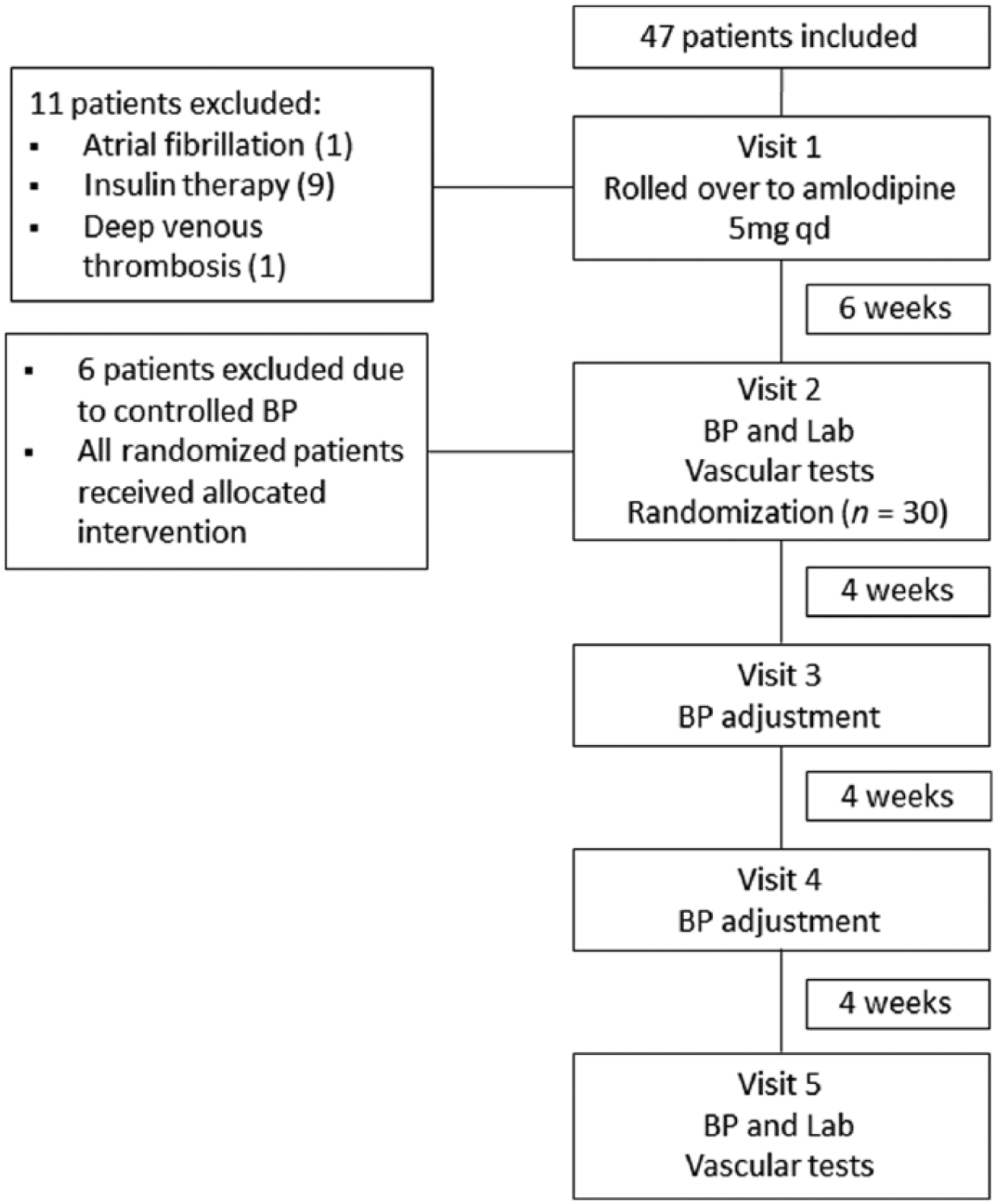
Flowchart of participants throughout the study. BP, blood pressure.
Patients with systolic BP ⩾ 130 mmHg and/or diastolic BP ⩾ 80 mmHg were randomized to benazepril 10 mg q.d. or losartan 50 mg q.d. Patients were randomized by the method of ‘randomly permuted blocks’. The randomization was not stratified and was done by an independent investigator. Patients were evaluated at 4-week intervals to adjust BP (Visit 3 and Visit 4). We titrated the benazepril to 20 mg q.d. and losartan to 100 mg q.d., for those patients with BP ⩾ 130/80 mmHg. At Visit 4, hydrochlorothiazide 25 mg q.d. was added, if necessary, to control BP. In Visit 5 (at 12 weeks) study patients had repeats of the vascular tests, laboratory assays and BP measurement. Hypoglycemic drugs dosages were rigorously maintained throughout the study. We checked adherence each visit, by tablet counting. We considered patients had good compliance when they had taken more than 80% of expected drug doses for the study period.
Blood pressure measurement
Office BP was obtained using an electronic device (model HEM-705CP, Omron Healthcare, Lake Forest, IL, USA) and an appropriate-sized cuff. Before measurement, patients were seated for 30 min and refrained from smoking and caffeine ingestion. Three readings were done 1 min apart and the average of these measurements was defined as the patient BP.
Assays
Fasting venous blood was collected to measure total cholesterol (TC), triglycerides (TG), high-density lipoprotein cholesterol (HDL-C), glucose, glycated hemoglobin (by high-performance liquid chromatography (HPLC)), creatinine (by an enzymatic method) and uric acid. The low-density lipoprotein cholesterol (LDL-C) level was calculated by the Friedewald formula. The glomerular filtration rate was estimated (eGFR) by modified Modification of Diet in Renal Disease (MDRD) study equation (Equation 1):
We also measured CRP by nephelometry (BN II, Siemens AG, Munich, Germany), and morning urinary spot albumin and creatinine by nephelometry (Immage, Beckman Coulter, Fullerton, CA, USA). The lower detection limit for CRP was 0.02 mg/dL. Values for microalbuminuria were considered normal, if up to 30 mg/g creatinine.
Vascular tests
All vascular tests were performed in the morning, in the supine position, after a minimum of 10 min rest in a quiet, temperature-controlled room. Patients were requested to fast; and to abstain from tea, coffee, alcohol and smoking for 12 h. The study and other morning medication were to be taken 1–2 hours before measurements. The vascular equipment operator was blinded to the patients’ treatment.
Brachial flow-mediated vasodilation
Imaging studies were performed according to recent guidelines.16,17 Endothelial-dependent (flow-mediated) vasodilation was measured using a 10 MHz high-resolution ultrasound (Vivid 3, General Electric Healthcare, Milwaukee, WI, USA). Vessel diameters were analyzed on frozen longitudinal images over the right brachial artery, 2–5 cm above the antecubital fossa. Three measurements were taken for three cardiac cycles at the end of diastole (R wave on the electrocardiogram (EKG)), and then the mean was calculated. Ischemia was induced by inflating the cuff 50 mmHg above the patient’s systolic BP, for 5 min. The maximal vessel diameter was recorded at 30, 60 and 90 seconds after cuff release; and we expressed the percentage of change over to basal diameter as FMD.
Pulse wave velocity
Pulse waves were obtained using a Complior SP device (Alam Medical, Vincennes, France), with transducers placed in the regions of the right carotid artery, right radial artery (crPWV), and right femoral artery (cfPWV). 18 Two measurements were made: The mean was used for analysis. When the difference was > 0.5 m/s, a third measurement was made: That median was then considered for analysis.
Determination of central aortic pressure
Pulse waves of the right radial artery were obtained using an applanation tonometer (model SPC-301-Millar Instruments, Houston, TX, USA), calibrated according to the brachial artery pressure. Pulse wave analysis was performed to obtain central aortic pressures and other hemodynamic parameters, using the SphygmoCor system (Atcor Medical, Sydney, Australia). The system’s software calculates the central systolic BP, central pulse pressure (PP), augmentation pressure (AP) and both augmentation index (AIx) and AIx adjusted to heart rate (HR) of 75 beats per minute (AIx@HR75). AIx represents the percentage of central pulse pressure caused by the early return of the reflected wave in the aortic root and it is calculated using a validated mathematical transfer function. Two measurements were made, and when differences were > 10%, a third measurement was made. The mean of the measurements was used for analysis.
Statistical analysis
Data are presented as median (interquartile range (IQR)) for numerical variables and frequency distribution for categorical variables, unless otherwise specified. The Mann-Whitney and Fisher’s exact tests were used to compare, respectively, the numerical and categorical variables between groups; and the Wilcoxon signed-rank test was used to compare variables before and after treatment. We used previous studies of FMD in similar populations to estimate sample size. 19 Assuming an alpha error of 5% and a standard deviation (SD) of 3.5%, we had 14 patients in each group with 80% power to detect a 4% difference in FMD between groups, by the end of the study. We used Statistica 12 (Statsoft, Tulsa, OK, USA) for database management and statistical analysis.
Results
Study cohort
A total of 47 patients were included and 30 of these were randomized to treatment arms (Figure 1): 14 to the benazepril group and 16 to the losartan group. The baseline demographics and clinical characteristics of study patients are presented in Table 1.
Baseline demographics and clinical characteristics.

Values are median (IQR) unless otherwise specified.
BMI: body mass index; CRP: C-reactive protein; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; HgbA1c: glycated hemoglobin; IQR: interquartile range; LDL: low-density lipoprotein; UACR, urinary albumin-to-creatinine ratio.
Clinical and laboratory parameters
The benazepril group presented non-significant higher values of systolic (p = 0.193) and diastolic BP (p = 0.152) at baseline (Table 2). There were no statistical differences between the benazepril and losartan groups, both in systolic (139 versus 134 mmHg, p = 0.618) and diastolic (82 versus 80 mmHg, p = 0.950) BP, at the end of the study. In addition, the number of patients who achieved a BP < 130/80 mmHg was the same between groups (14.3% versus 12.5%, p = 0.916). The percentage of patients requiring increased doses of study medication and thiazide association was the same, in both groups (57% versus 50% and 17% versus 19%, p > 0.05). Total cholesterol, TG, HDL-C, LDL-C, eGFR, glucose, glycated hemoglobin, creatinine, uric acid and microalbuminuria were also statistically similar between groups. Although the initial values of CRP were similar between the groups (Table 1), CRP values were lower in the benazepril group at the end of the study (Figure 2).
Effects of benazepril and losartan on blood pressure and vascular tests.

Values are median (IQR), unless otherwise specified.
AIx: augmentation index; AIx@HR75: augmentation index adjusted for heart rate of 75 beats per minute; AP: augmentation pressure; BP: blood pressure; cfPWV: carotid-femoral pulse wave velocity; crPWV: carotid-radial pulse wave velocity; FMD: flow-mediated vasodilation; IQR: interquartile range; p 1: p from comparison of the benazepril group, before and after treatment; p 2: p from the comparison of the losartan group, before and after treatment; p 3: p from the comparison between the benazepril and losartan group, at the end of the study; PP: pulse pressure; SBP: systolic blood pressure.
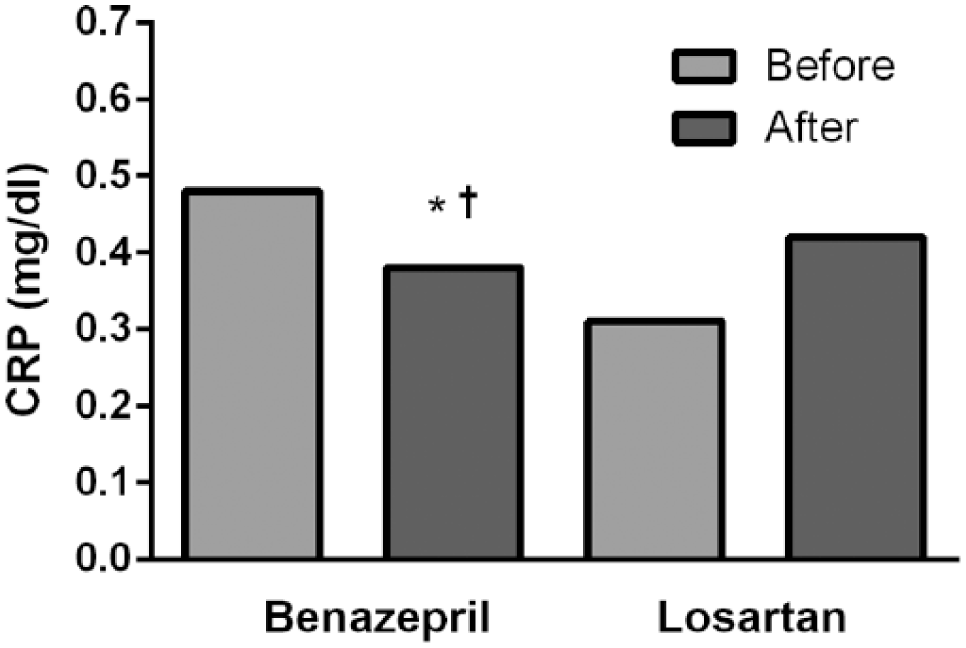
C-reactive protein concentration before and after 12-week treatment with benazepril and losartan. Data are presented as median.
Effect on endothelial function
Baseline brachial artery diameter was similar, between groups (4.75 versus 4.50 mm, p = 0.325). In the benazepril group, there was a higher FMD response (45% increase) after 12 weeks (6.05 to 8.80%, p = 0.057), although this did not reach statistical significance. In the losartan group, the FMD response was smaller (19% increase) after 12 weeks (7.90 to 9.40%, p = 0.132), as shown in Table 2.
Effect on pulse wave velocity and central aortic pressure
The benazepril group presented non-significant higher baseline values of cfPWV (p = 0.394) and central systolic BP (p = 0.119). The other baseline variables were very similar between the groups (Table 2). The cfPWV and crPWV values were not significantly different in the ACEi and ARB groups, before and after treatment (8.5 versus 8.5 m/s, p = 0.280; 10.2 versus 9.5 m/s, p = 0.724). Both groups also presented similar central systolic BP (129 versus 123 mmHg, p = 0.934) and AIx@HR75 (29.5 versus 30.5%, p = 0.632), before and after treatment (Table 2).
Adverse effects
One (7.1%) patient had a cough in the benazepril group and two (12.5%) patients had edema in the losartan group, but none discontinued treatment.
Discussion
The renin-angiotensin-aldosterone system (RAAS) plays a major role in physiopathology of essential hypertension. Both ACEi and ARB inhibit RAAS, although their mechanisms of actions are different, raising concerns about whether they really are interchangeable.20,21 Important aspects are peculiar to the ACEi mechanism of action: There is an increase in the bradykinin and the angiotensin-(1–7) concentrations, and both substances have vasodilatory and antiproliferative actions. 21 On the other hand, angiotensin-1 receptor blockage by ARB causes upregulation of angiotensin-2 receptors. Some recent studies, contradicting initial reports, suggest that angiotensin-2 receptors might contribute to vascular fibrosis. 22 We designed this trial to compare the effects of benazepril and losartan, an ACEi and an ARB, respectively, on endothelial function and vascular stiffness, in patients with Type 2 diabetes mellitus and hypertension. Our findings suggested that hypertensive diabetic patients using benazepril have a greater reduction in CRP and a slight improvement in flow-mediated vasodilation than have those taking losartan. Moreover, these effects occurred despite similar BP reduction.
CRP is an important inflammatory biomarker in atherosclerosis and is related to leukocyte adhesion molecules, tissue factors and monocyte recruitment to the arterial wall. 21 Moreover, it has prognostic significance: Higher levels are related to worse cardiovascular outcomes. 23 Rosei et al. 24 compared candesartan and enalapril in patients with non-insulin-dependent diabetes mellitus. These authors find no statistical difference in CRP concentration between ACEi and ARB. Likewise, Schram et al. 25 do not observe differences when comparing candesartan and lisinopril; however, in their results, CRP concentration increases in the candesartan group and decreases in patients using lisinopril, albeit this difference is not statistically significant. Koulouris et al. 26 observe that both ramipril and telmisartan lead to a reduction in CRP, but there was no difference among these drugs.
To our knowledge, the present study was the first one to show a greater reduction of CRP with ACEi, when compared to ARB. We suppose that this finding may be related to our sample’s higher baseline CRP values (0.48 and 0.31 mg/dL) than in other studies (from 0.13 to 0.30 mg/dL).24–26 During clinical consultation, we tried to exclude other potential causes of an elevated CRP, such as infections and rheumatic diseases. In addition, we looked for use of salicylic acid and statins, which were the same between groups. The elevated baseline CRP in both groups may have facilitated showing even small differences in the effects of ACEi and ARB.
Impaired endothelium-dependent vasodilation is a predictor of cardiovascular events, according to studies in population-based adults27,28; and it is related to atherosclerosis risk factors, such as hypertension and diabetes mellitus. 29 Three recent meta-analyses with hypertensive patients show that both ACEi and ARB improve FMD, when compared to placebo or no treatment, but there is no difference among them.13–15 Eight trials compare ACEi to ARB, but only one shows a higher FMD response with ACEi.14,30 There were no head-to-head comparisons of ACEi and ARB in diabetic patients.
Flammer et al. 19 observe that losartan is better than atenolol in improving FMD, in hypertensive diabetic patients. Yilmaz et al. 31 report that valsartan and valsartan plus amlodipine obtain higher FMD than amlodipine monotherapy, in a similar population. Three trials of ACEi versus placebo or no treatment have failed to show significant FMD improvement in mixed populations that included diabetic patients.32–34 To date, there has been no randomized trial concerning the effects of ACEi on FMD, exclusively in patients with Type 2 diabetes mellitus. Our results suggested that there might be an improvement in brachial FMD with benazepril use, an effect not observed in the losartan group.
Arterial stiffness is recognized as a predictor of increased cardiovascular risk and PWV is considered the gold standard procedure to measure it. A meta-analysis with more than 15,877 patients in 17 longitudinal studies shows that an increase in PWV by 1 m/s corresponds to a 15% increase in cardiovascular mortality. 35 Shahin et al. 36 carried out a meta-analysis to study the effects of ACEi on PWV and they conclude that these drugs reduce PWV by 1.69 m/s, when compared to placebo. Most studies are able to demonstrate that ARB reduces PWV, when compared to placebo.37–39 Moreover, the effects of ACEi and ARB on arterial stiffness seem to be independent of BP reduction 40 ; however, only four trials compare ACEi to ARB and they do not find any difference in PWV among these two drugs.37–39,41 Furthermore, none of these studies include diabetic patients. Our study was the first one to compare ACEi and ARB on PWV in diabetic patients and it showed there was no difference in this parameter.
Central pressure waveform methods analyze the interaction between arterial wall properties and the left ventricle. Some authors propose them as prognostic indexes of cardiovascular risk.42,43 As with PWV, Shahin et al. 36 observe that ACEi reduces AIx, when compared with other antihypertensive drugs. There are also reports of AIx and central BP reduction with various ARB.39,44,45 Only one trial compares ACEi to ARB: These drugs have the same effect on AIx, 39 although it did not include diabetic patients. In our study, central systolic BP, central pulse pressure, AP and AIx were also similar in both groups.
This trial had some important limitations. First, despite randomization, the ACEi group had a lower baseline FMD. We do not know if this influenced a better FMD response in this group. Second, despite statistical power calculation, our sample size was small and so these findings should be confirmed in trials including more patients, as small differences in PWV and central pressure waveform analysis could be missed. Regarding PWV, 12 weeks could also be a short time to observe antihypertensive drugs’ effect on arterial stiffness.
Conclusions
In summary, taking benazepril led to lower CRP and a slightly higher FMD response, when compared to losartan in hypertensive diabetic patients with BP not controlled by amlodipine. Most hypertension guidelines recommend ACEi or ARB as first-line therapy in diabetic patients with high BP; however, the mechanism of action of these drugs differ, and this may have laboratory and clinical consequences. Future clinical trials with a head-to-head comparison and larger samples are necessary, to elucidate whether the differences between ACEi and ARB have prognostic relevance.
Footnotes
Conflict of interest
The authors declare that there are no conflicts of interest.
Funding
This work was supported by the Brazilian agencies CNPQ (National Council for Research and Technology, grant 309316/2012-0) and FAPERJ (Foundation for Supporting Research in the State of Rio de Janeiro, grant E-26/103.309/2011).

