Abstract
Introduction:
Binge eating disorder (BED) is associated with dopaminergic activation as food reward, resulting in metabolism-related disorders. Stimulation of angiotensin type 2 (AT2) receptor is reported to inhibit dopamine synthesis. We investigated the possible roles of AT2 receptor-mediated dopamine regulation in the pathogenesis of BED.
Materials and methods:
Male C57BL/6 mice, type 2 diabetic (KKAy) mice and AT2 receptor-null (AT2KO) mice at eight weeks old were treated with AT2 receptor agonist, compound 21 (C21) or saline for two weeks. Mice were subjected to fasting for two days followed by re-feeding for seven days.
Results:
Treatment with C21 attenuated the rebound proportion of body weight, food intake and water intake in KKAy mice, but not in C57BL/6 and AT2KO mice. Dopamine concentration in the striatum was further increased by fasting in KKAy and AT2KO mice. Administration of C21 significantly attenuated this fasting-induced increase in dopamine level only in KKAy mice. Dopamine receptor D1, D2 expression in the substantia nigra were markedly lower in KKAy mice compared with C57BL/6 mice, while administration of C21 increased their expression in KKAy mice.
Conclusions:
Our study suggests that AT2 receptor stimulation may be a new therapeutic approach to improve eating disorder associated with dopamine resistance.
Keywords
Introduction
Binge eating disorder (BED) is a serious condition characterized by uncontrollable eating, and results in weight gain and related health problems, such as metabolic syndrome, 1 obesity2,3 and diabetes.4, 5 Central dopamine-related mechanisms are involved in the motivational aspects of eating and food choices.6,7 Comparison of the genotypes of obese individuals between those with BED and without BED demonstrated that an increase in dopamine D2 receptors (DRD2s) could contribute to BED pathogenesis. 8 Moreover, Parkinson’s disease (PD) patients treated with a dopamine agonist showed a higher rate of BED compared with PD patients without dopamine agonist treatment. 9 On the other hand, it is reported that striatal DRD2 availability was significantly lower in obese individuals than in controls. 10 Moreover, striatal DRD2 was down-regulated in obese rats, and knockdown of striatal DRD2 enhanced the development of addiction-like reward deficits and the onset of compulsive food seeking in rats. 11 Low brain dopamine activity in obese subjects could predispose them to excessive food intake with impairment of dopaminergic pathways that regulate neuronal systems associated with reward sensitivity and incentive motivation.12, 13 These results indicate that impairment of dopaminergic pathways, such as excess dopamine signaling or “dopamine resistance” in which the neuronal system fails to respond to normal levels of dopamine, may lead to uncontrollable food intake, and modulate the pathology of obesity and diabetes.
The renin-angiotensin system (RAS) has been reported to be involved in dopamine release in the striatum via the angiotensin type 1 (AT1) receptor. 14 For example, when angiotensin II was introduced by microdialysis into the striatum, the level of dopamine increased to three times baseline. 15 On the other hand, stimulation of the angiotensin type 2 (AT2) receptor, which has been expected to play a protective role in RAS.16,17 by a direct agonist, compound 21 (C21), decreased dopamine synthesis in the rat striatum. 18 Moreover, a previous in vitro study also demonstrated that stimulation of the AT2 receptor reduced catecholamine biosynthesis via a decrease in cGMP level in adrenal medullary cells. 19 However, it is not known whether AT2 receptor-induced dopamine reduction may regulate BED in diabetic mice and thereby regulate body weight. Accordingly, we hypothesized that AT2 receptor signaling may affect BED and excess body weight gain involving prevention of excess dopamine synthesis in diabetic mice. To explore a new therapeutic option for obesity and diabetes with BED, we investigated whether administration of C21 in obese type 2 diabetic mice could prevent excess food intake and rebound weight gain after short-term food deprivation.
Materials and methods
This study was performed in accordance with the National Institutes of Health guidelines for the use of experimental animals. All animal studies were reviewed and approved by the Animal Studies Committee of Ehime University.
Animals and treatment
Eight-week-old C57BL/6 mice, type 2 diabetes model (KKAy) mice, and AT2 receptor-null (AT2KO) mice were used in the following experiments. C57BL/6 and KKAy mice were purchased from CLEA Japan (Tokyo, Japan). AT2KO mice were provided by Hein et al. 20 Mice were kept in a room in which lighting was controlled (12 h on, 12 h off) and temperature was kept at 25°C. They were given a standard diet (MF, Oriental Yeast, Tokyo, Japan) and water ad libitum. Mice were subjected to intraperitoneal (i.p.) injection of saline (vehicle-treated group) or compound 21 (C21; provided by Vicore Pharma, Gothenburg, Sweden) (10 µg/kg/day) (C21-treated group). Systolic blood pressure was monitored in a conscious state by the tail-cuff method (BP-98A-L, Softron Co. Ltd, Tokyo, Japan), as described previously. 21
Y-maze test
The Y-maze test was performed as previously described. 22 The three arms were each 400 mm in length and 150 mm in height, and separated by angles of 120º (Brain Science Idea Co. Ltd, Osaka, Japan). Each mouse was placed in the center of the apparatus and allowed to explore the maze for 8 min. Arm choices were manually recorded. The occurrence of three consecutive choices of three different arms was counted as a correct choice. The ratio of correct choices was determined by (“number of alternations”)/(“total number of arm visits”–2) ×100.
Effect of fasting followed by re-feeding on weight
After two weeks of treatment with saline or C21, the mice were fasted for two days with free access to drinking water. Water intake, food intake and body weight were measured every 24 h. After two days of fasting, the mice were fed for seven days. Rebound weight gain was estimated by the rebound proportion defined as (“daily body weight”–“body weight after fasting”)/(“body weight before fasting”–“body weight after fasting”)×100. The study protocol is shown in Figure 1.
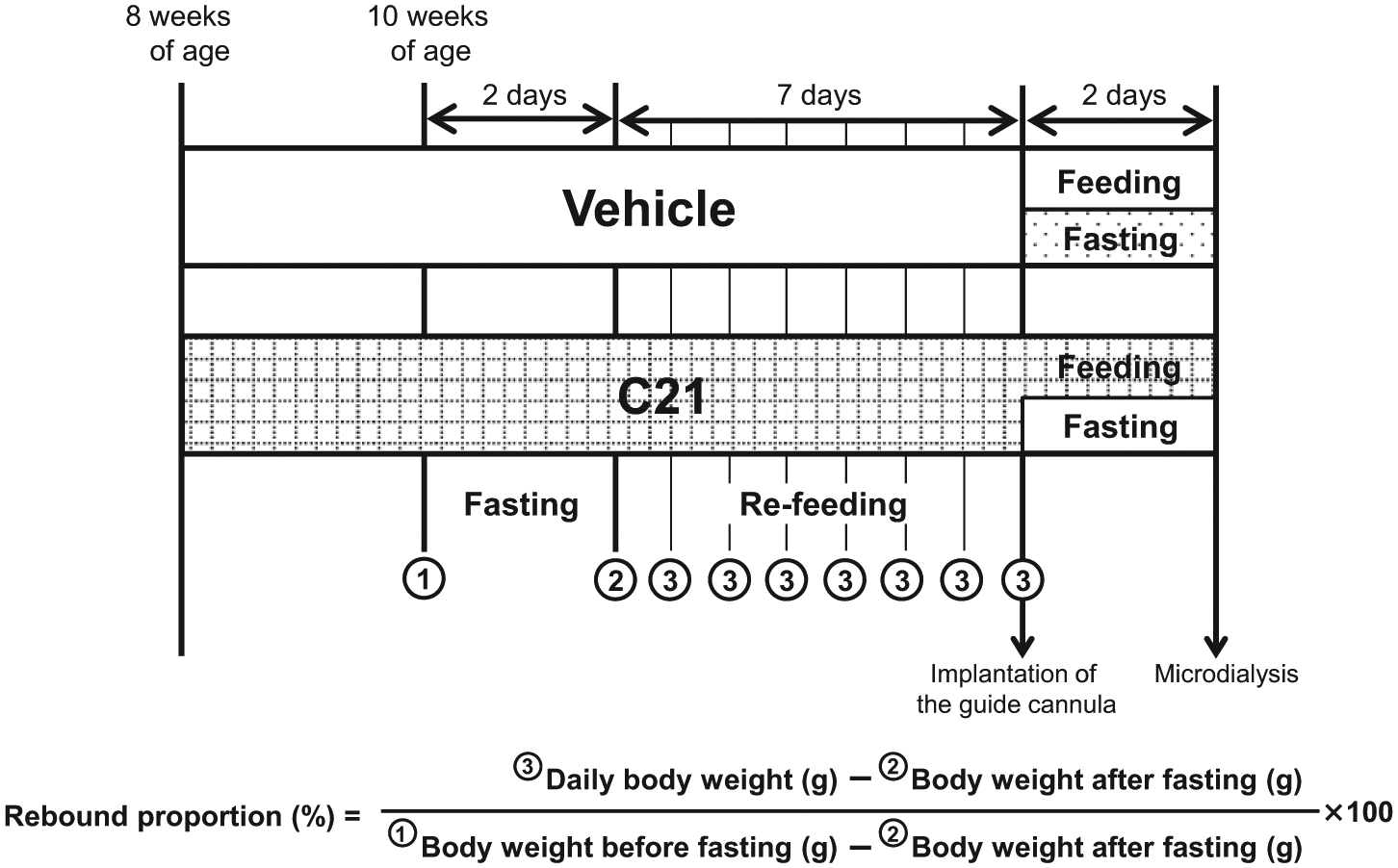
Study protocol. C21: compound 21.
Microdialysis
The study protocol for sample preparation is shown in Figure 1. Mice were anesthetized with sodium pentobarbital (84.5 mg/kg, i.p.), and a guide cannula (AG-4, Eicom Co. Ltd, Kyoto, Japan) was stereotaxically implanted in the striatum (AP +0.5 mm, ML +2.0 mm, DV –2.5 mm, from the bregma and skull) as a dialysis probe(A-I-4-02, Eicom Co. Ltd, Kyoto, Japan), as reported. 22 The guide cannula was cemented in place with dental selfcuring resin (Quick Resin, Shofu Inc., Kyoto, Japan), and the animal was kept warm and allowed to recover from anesthesia. Two days after surgery, microdialysis samples (20 µl) were collected every 20 min (at the 1 µl/min flow rate) for 3 h. We selected the seventh (120–140 min), eighth (140–160 min) and ninth (160–180 min) samples for dopamine assay to increase sample stability. Then, three samples were injected into a high-performance liquid chromatography column (AC-ENZYM II, Eicom Co. Ltd, Kyoto, Japan) for detection of dopamine. Values were assessed as an average of these three samples. We checked the site of microdialysis probe inserted into the striatum with Hematoxylin & Eosin (H&E) staining of the brain after the experiments.
Expression of dopamine receptors and dopamine active transporter
Expression of dopamine receptor D1 (DRD1), dopamine receptor D2 (DRD2) and dopamine transporter (DAT) were evaluated by immunohistochemical staining. Formalin-fixed, paraffin-embedded sections were prepared using brain samples taken after microdialysis sampling. Endogenous peroxidase and nonspecific binding of the antibody were blocked with 3% hydrogen peroxide and 5% goat serum in antibody diluent (Dako Japan, Tokyo, Japan). Then, the sections were incubated overnight at 4oC with the primary antibody, anti-DRD1, diluted 1:100 (Sigma-Aldrich, St Louis, Missouri, USA), anti-DRD2, diluted 1:100, (EMD Millipore Corp., Billerica, Massachusetts, USA), or anti-DAT, diluted 1:10,000, (Sigma-Aldrich, St Louis, Missouri, USA). Subsequently, tissue sections were treated with the secondary antibody, MAX-PO(R) (Nichirei Bioscience Inc., Tokyo, Japan). Antibody binding was visualized by 3,3’-diaminobenzidine (DAB) staining using a detection kit, ENVISION (Dako Japan, Tokyo, Japan), and all sections were counterstained with hematoxylin. Positive area of DRD1, DRD2 and DAT was evaluated visually by brown stained regions of five different fields in tissue slice from 2–4 mice in each group in per field at 400×magnification. The microscope fields were selected at random in each tissue slice.
Statistical analysis
All values are expressed as mean±standard deviation (SD) in the text and figures. Data were evaluated by one-way analysis of variance (ANOVA). If a statistically significant effect was found, post-hoc analysis was performed with the Tukey-Kramer test to detect the difference between the groups. Values of p<0.05 were considered statistically significant.
Results
Effect of C21 on spontaneous alternation behavior and activity
Administration of C21 at a dose of 10 µg/kg/day did not affect systolic blood pressure, as previously reported (Table 1). 23 Spontaneous alternation behavior (Figure 2(a)) and spontaneous activity (Figure 2(b)) determined by the Y-maze test were not different between vehicle- and C21-treated groups in C57BL/6 mice, indicating that treatment with C21 did not influence the willingness to explore new environments. However, spontaneous alternation behavior and spontaneous activity in diabetic KKAy mice were significantly impaired in comparison with those in C57BL/6 mice, and were improved by the administration of C21.
Systolic blood pressure after treatment in C57BL/6 mice, type 2 diabetic (KKAy) mice and AT2 receptor-null (AT2KO) mice.
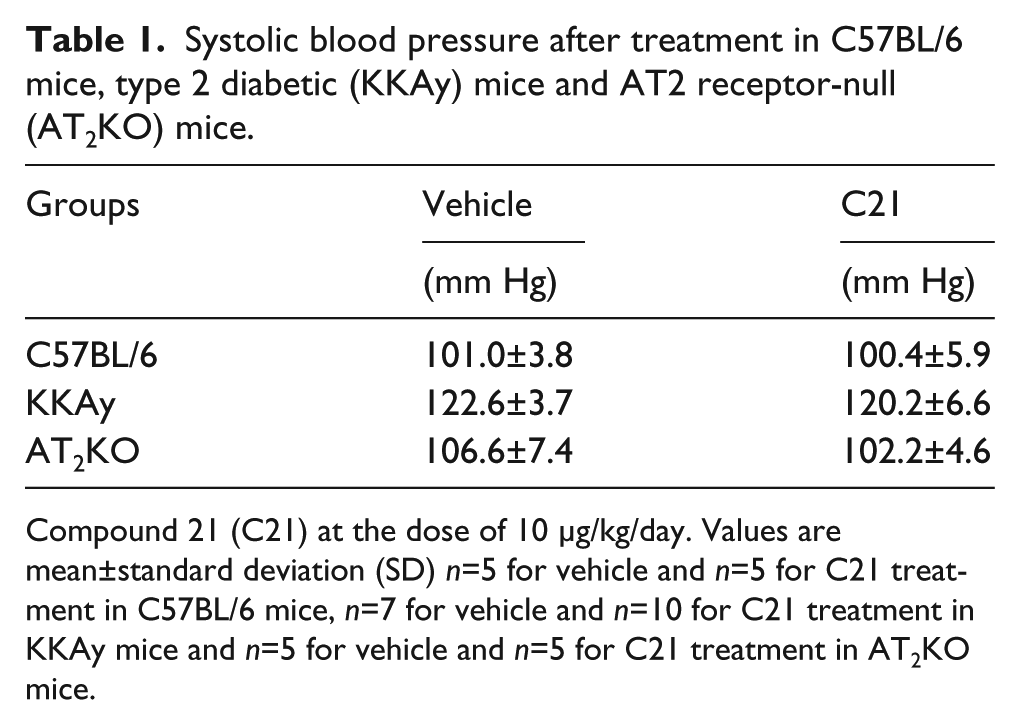
Compound 21 (C21) at the dose of 10 μg/kg/day. Values are mean±standard deviation (SD) n=5 for vehicle and n=5 for C21 treatment in C57BL/6 mice, n=7 for vehicle and n=10 for C21 treatment in KKAy mice and n=5 for vehicle and n=5 for C21 treatment in AT2KO mice.

(a) Spontaneous alternation behavior and(b) spontaneous activity determined by Y-maze test in C57BL/6 and type 2 diabetic (KKAy) mice with or without compound 21 (C21) treatment. Mice were treated as described in the Methods section. □ shows vehicle and ■ shows C21 treatment (10 μg/kg/day). Data are expressed as mean±standard deviation (SD). n=5 for vehicle-treated and n=5 for compound 21-treated C57BL/6 mice; n=7 for vehicle-treated and n=10 for compound 21-treated KKAy mice. *p<0.05, **p<0.01 vs vehicle-treated C57BL/6 mice,
Effect of C21 on rebound weight gain induced by fasting
There were no significant differences in body weight, food intake and water intake between the vehicle- and C21-treated groups both in C57BL/6 and KKAy mice under a fed condition (data not shown). We next examined the effects of C21 on rebound body weight gain after fasting followed by re-feeding. Mice were subjected to fasting for two days followed by re-feeding for seven days. There was no significant difference in body weight loss by two days of fasting between C57BL/6 and KKAy mice, and treatment with C21 did not affect body weight (Figure 3(a)). There was no significant difference in the rebound proportion of body weight between the vehicle- and C21-treated groups after re-feeding in C57BL/6 mice (Figure 3(b)). Interestingly, in KKAy mice, a significant decrease in the rebound proportion of body weight was observed in the C21-treated group from day 6 of re-feeding (Figure 3(c)). Moreover, food intake and water intake were attenuated by C21 treatment in KKAy mice (Figure 3(d) and 3(e)).

Effect of fasting with or without compound 21 (C21) treatment on body weight loss by (a) fasting, and the rebound proportion of body weight in (b) C57BL/6 mice and (c) type 2 diabetic (KKAy) mice after two days of fasting followed by re-feeding with or without C21 treatment. Rebound weight gain was defined by the rebound proportion as (“daily body weight”–“body weight after fasting”)/(“body weight before fasting”–“body weight after fasting”)×100. Total amount of (d) food intake and (e) water intake in C57BL/6 and KKAy mice for seven days after two days of fasting with or without compound 21 treatment. Mice were treated as described in the Methods section. □ shows vehicle and ■ shows C21 treatment (10 μg/kg/day). Data are expressed as mean±standard deviation (SD) n=5 for vehicle-treated and n=5 for C21-treated C57BL/6 mice; n=4 for vehicle-treated and n=7 for compound 21-treated KKAy mice. *p<0.05, **p<0.01 vs the same condition in C57BL/6 mice.
Effect of C21 on dopamine concentration in the striatum, and expression of DRD1, DRD2 and DAT in the substantia nigra
Dopamine concentration in the striatum was significantly higher in KKAy mice than in C57BL/6 mice, and fasting further increased dopamine concentration in KKAy mice but not in C57BL/6 mice (Figure 4). Administration of C21 significantly attenuated the fasting-induced increase in dopamine concentration in KKAy mice (Figure 4). Dopamine receptor D1 (DRD1) expression in the substantia nigra determined by immunostaining was markedly lower in KKAy mice than in C57BL/6 mice, while administration of C21 increased DRD1 expression in KKAy mice (Figure 5(a)). The expression of DRD2 (Figure 5(b)) was also lower in KKAy mice than in C57BL/6 mice, and these reductions were also blunted by administration of C21. Moreover, the expression of DAT tended to be decreased in KKAy mice compared with C57BL/6 mice; however, we had not a reliable comparison in its staining due to the difficulty of quantification (Figure 5(c)).

Dopamine level in the striatum of C57BL/6 and type 2 diabetic (KKAy) mice with or without fasting in vehicle-treated and compound 21 (C21)-treated groups. Mice were treated as described in ‘Methods’. Data are expressed as mean± standard deviation (SD) (n=2 to 4 for each group). *p<0.05 vs vehicle-treated group without fasting,
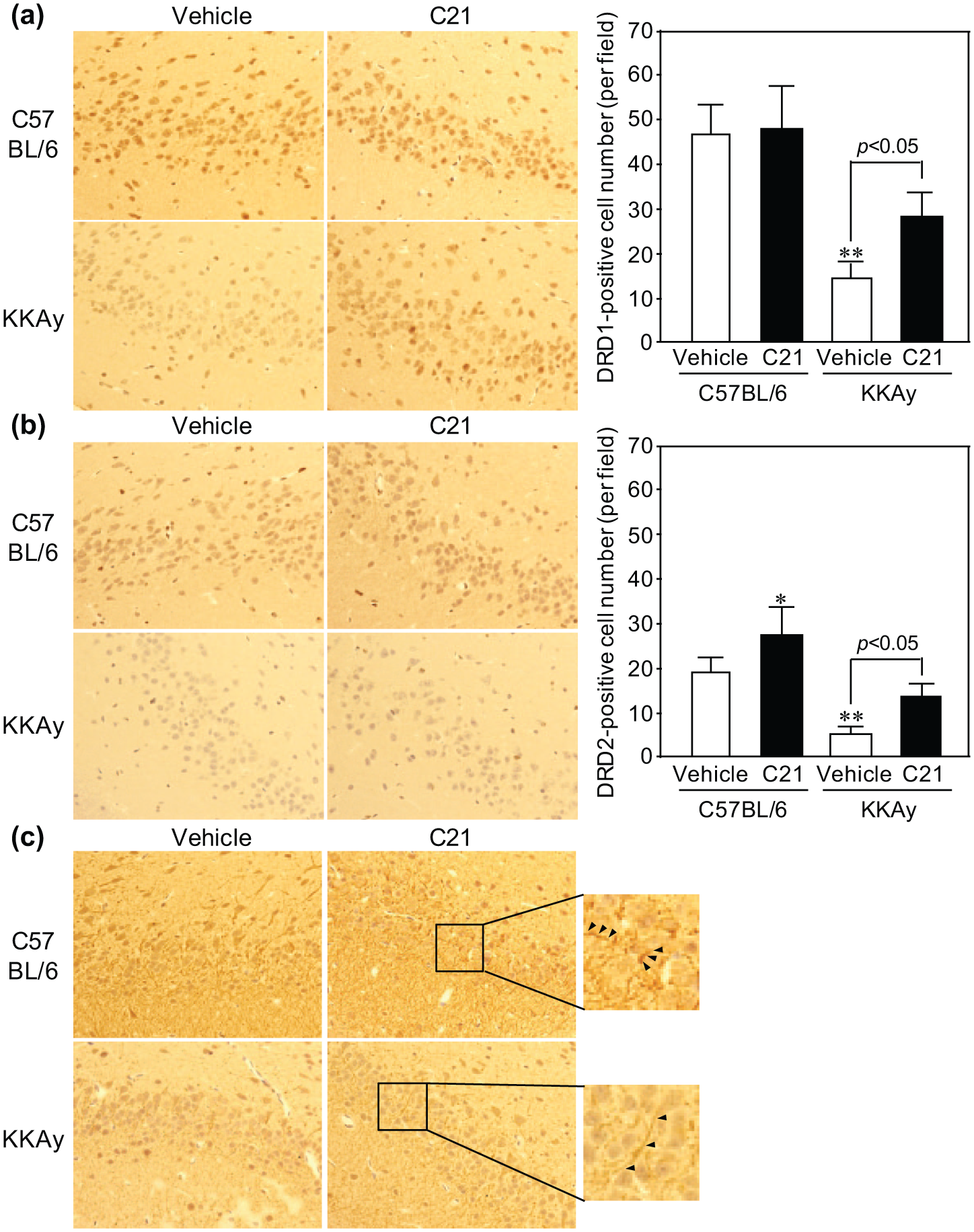
Immunohistochemical staining of (a) dopamine receptor D1 (DRD1), (b) dopamine receptor D2 (DRD2) and (c) dopamine transporter (DAT) in the substantia nigra. Mice were treated as described in the Methods section. Original magnification×400. Data are expressed as mean±standard deviation (SD) (n=5 for each group). *p<0.05, **p<0.01 vs vehicle-treated group in C57BL/6 mice. Arrows indicate representative positive regions of DAT. AT2KO: angiotensin type 2 receptor-null mice; C21: compound 21.
Effect of AT2 receptor deletion on fasting-induced rebound weight
To further confirm the roles of the AT2 receptor in BED, we also investigated the effect of deletion of the AT2 receptor on BED using AT2KO mice. The body weight of AT2KO mice quickly returned to that before fasting, even two days after re-feeding (24.6±1.2 g (before fasting), 20.1±1.2 g (after fasting) and 24.7±1.4 g (at two days)). Rebound proportion of body weight in AT2KO mice was 20–50% higher than that in C57BL/6 mice (e.g. 84% in C57BL/6 mice and 101% in AT2KO mice at two days). Moreover, body weight three days after re-feeding reached 20% more than that before fasting. This increase was not inhibited by C21 (Figure 6(a)). In AT2KO mice, both food intake (Figure 6(b)) and water intake (Figure 6(c)) were significantly increased compared with those in C57BL/6 mice.

Rebound proportion of (a) body weight, (b) food intake and (c) water intake in angiotensin type 2 receptor-null (AT2KO) mice with or without compound 21 (C21) treatment. Mice were treated as described in the Methods section. □ shows vehicle and ■ shows C21 treatment (10 μg/kg/day). Rebound weight gain was defined by the rebound proportion as (“daily body weight”–“body weight after fasting”)/(“body weight before fasting”–“body weight after fasting”)×100. Data are expressed as mean±standard deviation (SD) (n=5 for each group).
Effect of C21 on dopamine concentration in the striatum in AT2KO mice
Dopamine level without fasting showed no significant difference between C57BL/6 and AT2KO mice. In a fasting condition, dopamine level was significantly increased in AT2KO mice; however, this increase was not inhibited by C21 (Figure 7).
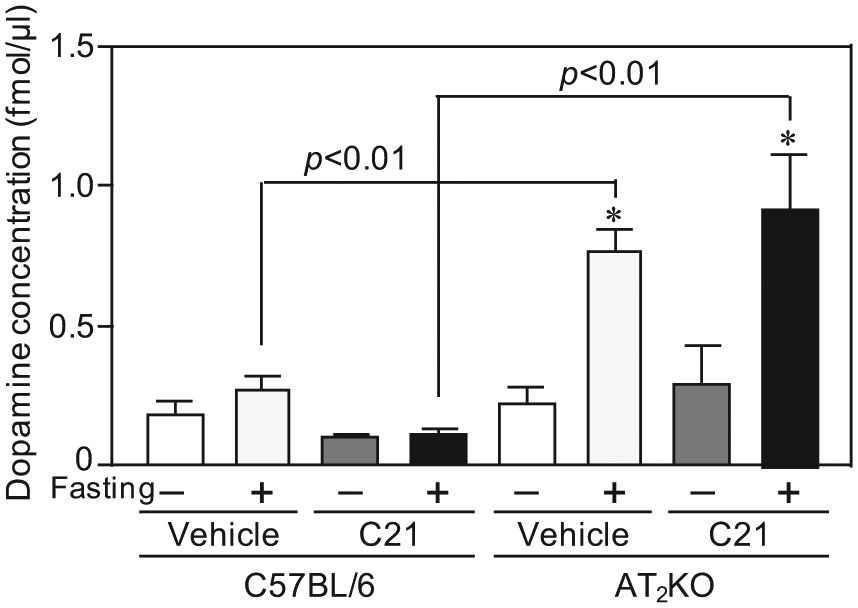
Dopamine level in the striatum of C57BL/6 and angiotensin type 2 receptor-null (AT2KO) mice with or without fasting in vehicle-treated and compound 21 (C21)-treated groups. Mice were treated as described in the Methods section. Data are expressed as mean±standard error of the mean (SEM) (n=2 to 3 for each group). Data in C57BL/6 mice are used for comparison. *p<0.01 vs vehicle-treated group without fasting in AT2KO mice.
Discussion
We demonstrated that activation of the AT2 receptor could contribute to inhibition of rebound weight gain after food deprivation with modulation of dopamine signaling. Treatment with C21 reduced dopamine concentration in the striatum and increased dopamine receptors, resulting in improvement of “dopamine resistance”. Moreover, we observed that deletion of the AT2 receptor enhanced rebound weight via excessive food- and water-intake with an increase in dopamine level of the striatum. These findings suggest that improvement of disturbed dopamine signaling via AT2 receptor stimulation may prevent overeating after fasting followed by re-feeding in diabetic mice.
Over-stimulation of the brain reward system through excessive consumption of palatable energy-dense food induces reward hyposensitivity with downregulation of DRD2. 11 Individuals with BED or obesity have been shown to have decreased reward sensitivity due to decreased DRD2 density. 24 Such hyposensitivity of the reward pathway is considered to lead obese individuals to compulsively consume palatable food to compensate reward hyposensitivity.11,12 Therefore, to overcome BED, it is crucial to regulate not only dopamine level but also dopamine receptor density. Our data demonstrated that dopamine concentration in the striatum of KKAy mice was significantly higher than that in C57BL/6 mice, while expression of dopamine receptors was reduced in KKAy mice compared with wild-type mice, indicating the presence of reward hyposensitivity in KKAy mice. AT2 receptor activation by C21 not only decreased dopamine concentration but also increased dopamine receptors in KKAy mice, resulting in attenuation of food and water intake. However, treatment with C21 did not attenuate the motivation to explore new environments, indicating that C21-induced reduced food- and water-intake was not induced by apathy.
Rodriguez-Pallares et al. demonstrated that AT2 receptor signaling increased differentiation of dopaminergic neurons from mesencephalic precursors. 25 They also reported that brain angiotensin II enhanced dopaminergic cell death via microglial activation and Nicotinamide Adenine Dinucleotide Phosphate (NADPH)-derived reactive oxygen species via AT1 receptors. 26 However, these effects of angiotensin II on dopaminergic neurons appear to be a long-term effect leading to dopaminergic degeneration. In tobacco smoking, Narayanaswamia et al. reported that crosstalk between AT1 and AT2 receptors in the striatum appeared to complicate angiotensin II modulation of the effect of nicotine on dopamine release. 27 These results indicate that the balance or interaction between AT1 and AT2 receptor signaling might play an important role in the brain dopaminergic system. Although we have focused on AT2 receptor signaling in relation to brain dopamine levels in the present study, the detailed interaction between the RAS and dopaminergic systems should be further investigated in the future.
In obese rats, mesolimbic dopamine turnover is impaired 28 and the density of DRD2 and DAT are reduced. 29 In humans, DRD2 availability evaluated by Positron Emission Tomography (PET) decreases with increasing body mass index (BMI). 10 Similar results were observed for DAT. 30 In contrast, the roles of DRD1 are controversial, while DRD2 is reported to be involved in food addiction. Trugman et al. showed that stimulation of DRD1 by endogenous dopamine contributed to basal metabolism in substantia nigra and DRD1 agonists increased glucose utilization, suggesting that stimulation of DRD1 receptors may be also tied to feeding behavior. 31 However, DRD1 expression was upregulated in brainstem of high-fat high-sugar diet-fed rats compared to chow-fed rats. Therefore, further investigation is necessary to know the meaning of an increase in expression of DRD1 chronic AT2 receptor activation in diabetic mice. Our results showed that the expression of dopamine receptors and transporter in the substantia nigra were markedly decreased in KKAy mice compared with C57BL/6 mice and these reductions were improved by administration of C21. The improvement of “dopamine resistance,” by treatment with C21 is considered to be mediated by reduction of excess brain dopamine concentration.
Feeding behavior is also regulated by other orexigenic peptides such as neuropeptide Y (NPY) and melanin-concentrating hormone (MCH), and anorexigenic peptides such as corticotropin-releasing hormone (CRH) in the hypothalamus. 32 Previously, Yamamoto et al. demonstrated that AT1 receptor signaling regulates food intake through CRH in the hypothalamus. 33 In the present study, we have only focused on dopamine level in feeding behavior, and several factors may be involved in the regulation of food intake by RAS. We are interested in the effect of combination treatment with an Angiotensin II Receptor Blocker (ARB) and C21 on BED as a future research subject. Moreover, our previous study demonstrated that treatment with C21 accompanied by Peroxisome Proliferator-activated Receptor (PPARγ) activation ameliorated insulin resistance in KKAy mice. 34 Therefore, systemic administration of C21 could provide a new therapeutic option in patients with diabetes mellitus to prevent the progression of diabetes via multiple pathways, not only improvement of glucose metabolism but also rebound of body weight with regulation of dopamine-related appetite.
Conclusions
Our results indicate that AT2 receptor signaling could contribute to the pathogenesis of BED with a decrease in dopamine levels. Our study suggests that AT2 receptor stimulation may be a new therapeutic approach to improve eating disorder associated with dopamine resistance.
Footnotes
Conflict of interest
None declared.
Funding
This study was supported by Japan Society for the Promotion of Science (JSPS) KAKENHI Grant Numbers 25293310 (MH), 25462220 (MM) and 24791506 (JI), and research grants from pharmaceutical companies: Astellas Pharma Inc., Bayer Yakuhin, Ltd, Daiichi-Sankyo Pharmaceutical Co. Ltd, Nippon Boehringer Ingelheim Co. Ltd, Novartis Pharma K. K., Shionogi & Co., Ltd, and Takeda Pharmaceutical Co. Ltd.
